Highly-branched poly(N-isopropyl acrylamide)s with core–shell morphology below the lower critical solution temperature†
Richard Plenderleith,
Thomas Swift and
Stephen Rimmer*
Polymer and Biomaterials Chemistry Laboratories, Department of Chemistry, Brook Hill, University of Sheffield, Sheffield, South Yorkshire S3 7HD, UK. E-mail: s.rimmer@sheffield.ac.uk; Fax: +44 114 222 9346
First published on 3rd October 2014
Abstract
Highly-branched poly(N-isopropyl acrylamide)s in water pass through coil-to-globule transitions. Using calorimetry and the colour change of a solvatochromic dye within the polymer, we show that some compositions have biphasic core–shell morphologies, with globular cores and open coil shells. The two-phase structure is favoured by increased branching and arises because the chain ends penetrate only to a limited degree into the polymer coil.
Introduction
Stimulus responsive materials are finding increasing applications in biotechnology and medicine.1–3 In general these are water swollen or water soluble polymers that either degrade in the presence of a stimulus or respond to changes in the environment (pH, ionic strength or temperature). Recently, we have shown that highly-branched poly(N-isopropyl acrylamide) (HB-PNIPAM) with ligands at the chain ends can respond, by passing from a open solvated conformation to a more compact desolvated conformation, on binding of the ligands to cellular targets.4–6 The transition from open coil to compact globule occurs at a temperature (Tc–g) that is manifest as a lower critical solution temperature (LCST). The effect of binding of the chain-end ligands is to perturb end group solvation and reduce the Tc–g to below the application temperature (approximately 37 °C for most medical applications), at constant temperature the polymer passes from the open coil to the desolvated globular form. In earlier work we had shown that, in HB-PNIPAM with either imidazole,7,8 pyrrole9 or carboxylic acid9 end groups, the Tc–g reduces as the average degree of branching increases. In our continuing work, which aims to design polymers that respond to binding by passing through a coil-to-globule transition, we have observed that for some compositions a model that involves a biphasic morphology is more appropriate.Here we provide the first evidence for this model. The data support an hypothesis that is illustrated in Fig. 1. The inner regions of the HB-PNIPAM are in a partially desolvated-globular state, even below the main Tc–g event, but the outer segments maintain an open-solvated coil until the Tc–g is reached. As far as we are aware this is behaviour that has not been observed in conventional linear homoPNIPAM, copolymers of PNIPAM or any of the related thermally responsive polymer systems that use other monomers. In our previous work on HB-PNIPAMs4–9 we had assumed that above the LCST polar end groups, such as COOH, would be located at the surface. The inner PNIPAM domain would be in a homogeneous globular state and this could be treated as a partially swollen network. However, our more recent data suggests that this is not the case so that certain compositions adopt a core–shell morphology, above a preliminary transition point (T′c–g), which is best represented by the schematic diagram in Fig. 1.
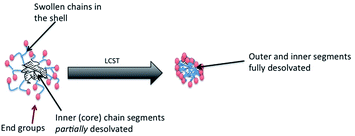 | ||
| Fig. 1 Schematic diagram showing how a highly branched polymer in aqueous medium can have a biphasic structure with a globular desolvated inner core and a more solvated outer shell. | ||
Results and discussion
HB-PNIPAM was synthesised by self-condensing reversible addition fragmentation transfer (RAFT) polymerisation, with varying ratios of NIPAM to 1, a styryl functional dithioate ester.9 Table 1 shows the monomer conversion and characterisation data for a set of HB-PNIPAM polymers obtained by a modification of our previously reported self-condensing vinyl polymerisation route.9 The aim of our work in this area was to prepare HB-PNIPAM that can be functionalised with biochemical ligands and to this end we used a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of the initiator, azobiscyanovaleric acid (ACVA), to 1.
1 ratio of the initiator, azobiscyanovaleric acid (ACVA), to 1.NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) RAFT RAFT |
Conva/% | Pyrroleb/% | Acidc/% | DBd |
|---|---|---|---|---|
| a Percentage conversations based on 1H NMR of crude reaction mixture.b The fraction of chain ends with pyrrole functionality, calculated from 1H NMR of purified product, compared to the number of aryl branch points, see ESI.c The fraction of ends groups with COOH functionality, determined following reaction with trichloroacetyl isocyanate, compared to the number of aryl branch points.d Degree of branching, reported as branch points per repeat unit determined by NMR spectroscopy. | ||||
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.9 | 50.87 | 23.43 | 0.091 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.7 | 47.6 | 11.27 | 0.059 |
25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.7 | 53.3 | 14.06 | 0.036 |
35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.1 | 50.53 | 22.22 | 0.027 |
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.6 | 50.58 | 26.50 | 0.023 |
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.7 | 49.70 | 21.24 | 0.019 |
65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.7 | 56.41 | 32.24 | 0.016 |
75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.7 | 57.65 | 21.29 | 0.013 |
85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
98.6 | 52.72 | 24.61 | 0.012 |
This is a higher concentration of initiator than would be usual in conventional RAFT polymerisations. Carrying out the polymerisations in this way provides carboxylic acid groups at a large fraction of chain ends as chains become terminated by bimolecular combinations between macro-radicals and primary ACVA radicals. The polymers were analysed by 1H NMR to find percentage conversions, functionality and degree of branching. As shown in Table 1 the conversions were above 98% for all feed compositions. As expected, decreasing the amount of 1 relative to NIPAM produced polymers with decreasing numbers of branch points per repeat unit. The chain end structures were analysed by 1H NMR, directly for the determination of the pyrrole end groups and after reaction with trichloroacetyl isocyanate for the COOH functionalities.10
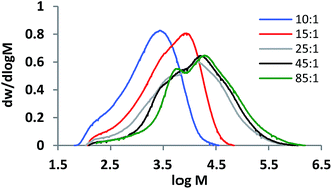
These were presented relative to the number of branch points so that in the case of zero frequency of bimolecular terminations between two polymer radicals the pyrrole and acid values will sum to 100%. Clearly, this is not the case in these data so that the data indicated a substantial fraction of chains terminated by bimolecular chain–chain radical combinations: a result that is to be expected at high conversions with high initial initiator concentrations. However, the polymers remained soluble and did not gel. Fig. 2 shows molar mass distributions derived from size exclusion chromatography (DMF, polystyrene standards). The distributions were as expected, for polymers produced by SCVP RAFT polymerisation, broad and asymmetrical. The apparent molar mass averages are also given in Table 2. However, comparison of these means should be applied with caution because they are parameters of non-similar, non-Gaussian distributions. Not withstanding this comment the dispersities (Mw/Mn) as expected are large and vary from 3.5 to 7.5.
NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
Mn | Mw | Mz | Mz+1 | D |
|---|---|---|---|---|---|
| a Data calculated from DMF GPC. | |||||
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1050 | 3350 | 6650 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
3.2 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2200 | 7650 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
3.5 |
25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
3350 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 850 850 |
103![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
236![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
7.5 |
35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
5100 | 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 250 250 |
123![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
295![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
5.7 |
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
4800 | 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
121![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 250 250 |
296![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 650 650 |
6.2 |
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
6300 | 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
115![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
280![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 750 750 |
4.8 |
65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
7350 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 750 750 |
143![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
362![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 350 350 |
4.7 |
75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
7400 | 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
142![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 750 750 |
4.6 |
85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
7977 | 45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
251![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 050 050 |
671![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
5.6 |
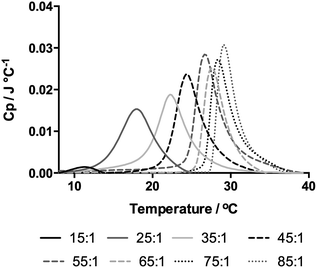
The solution behaviour of these polymers in water was investigated by measuring the cloud point by turbidimetry at 500 nm and the Tc–g was recorded by differential scanning micro-calorimetry (micro-DSC). The thermograms obtained from these polymers are shown in Fig. 3 and the Tc–g was taken as the peak of the endotherm. Also, of note is that the peaks were relatively symmetrical.
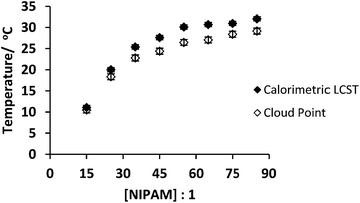
Fig. 4 shows that the LCST (cloud point) and Tc–g decreased with increased branching. Previously we have shown that this decrease in LCST with branching is due to both the incorporation of the hydrophobic aryl branching unit and increased segmental crowding.11 For the 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 sample a cloud point could not be measured as the polymer had already passed through to the globule state at 10 °C and therefore it had limited solubility. The relationship between the cloud point and branching is closely followed by the calorimetric data. The observation of cloud points for these polymers indicates aggregation of the globular structures. On the other hand the calorimetric Tc–gs were measured at the peak of the desolvation endotherm. Therefore, the absolute temperatures of the transitions appear to be slightly different. It should be noted that we had shown that not all of the HB-PNIPAM systems exhibited cloud points at the LCST.4 This will occur if the charge on the end groups is sufficient to prevent aggregation by electrostatic repulsion.
1 sample a cloud point could not be measured as the polymer had already passed through to the globule state at 10 °C and therefore it had limited solubility. The relationship between the cloud point and branching is closely followed by the calorimetric data. The observation of cloud points for these polymers indicates aggregation of the globular structures. On the other hand the calorimetric Tc–gs were measured at the peak of the desolvation endotherm. Therefore, the absolute temperatures of the transitions appear to be slightly different. It should be noted that we had shown that not all of the HB-PNIPAM systems exhibited cloud points at the LCST.4 This will occur if the charge on the end groups is sufficient to prevent aggregation by electrostatic repulsion.
The micro-DSC data were examined further and the peak areas were used to estimate the relative magnitude of the endothermic transitions. The data are presented in Fig. 5. Plotting these peak areas (q) in Fig. 5 showed (ANOVA p < 0.01) that the magnitude of the endotherm decreased as branching increased and that there was an abrupt change between the 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 15
1 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 polymers. The differences between the 15
1 polymers. The differences between the 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 polymer and all other compositions and the 85
1 polymer and all other compositions and the 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 polymer and all other compositions was significant (post hoc Tukey, p < 0.01). There were also other pairwise significant differences in the data (not shown) and there was a trend of increasing q as the degree of branching decreased. The data indicated that as the branching increased the amount of polymer and water involved in the transition decreased and importantly there was a step change between the 25
1 polymer and all other compositions was significant (post hoc Tukey, p < 0.01). There were also other pairwise significant differences in the data (not shown) and there was a trend of increasing q as the degree of branching decreased. The data indicated that as the branching increased the amount of polymer and water involved in the transition decreased and importantly there was a step change between the 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 15
1 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 polymers. Thus, there was a step decrease in the amount of material involved in the transition.
1 polymers. Thus, there was a step decrease in the amount of material involved in the transition.
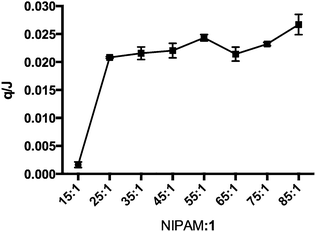 | ||
| Fig. 5 Peak areas derived from micro-DSC measurements providing the heat released at the transition. Errors bars are standard errors of the mean. | ||
The microDSC data indicated that decreasing amounts of polymer and water were involved in the transition as the degree of branching increased and there was a step change as the composition was changed from 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 15
1 to 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. These results could best be rationalised by considering that, as the degree of branching increases segmental crowding increases. This disrupts hydration so that there is a general decrease in the amount of water that solvates the segments below the Tc–g. As the degree of branching increases, the concentration of hydrophobic aryl units increases, this decreases solvation and chain swelling. However, these hydrophobic effects of the aryl branching units were to some extent offset by the presence of increased amounts of polar carboxylic acid groups at chain ends. We showed previously that branching decreases the Tc–g independently of the hydrophobic effects of the aryl branch points.7 In this work we observed that as well as this general decrease in the magnitude of the endotherm associated with the Tc–g as branching increased, there was a very large decrease as the composition was changed from NIPAM
1. These results could best be rationalised by considering that, as the degree of branching increases segmental crowding increases. This disrupts hydration so that there is a general decrease in the amount of water that solvates the segments below the Tc–g. As the degree of branching increases, the concentration of hydrophobic aryl units increases, this decreases solvation and chain swelling. However, these hydrophobic effects of the aryl branching units were to some extent offset by the presence of increased amounts of polar carboxylic acid groups at chain ends. We showed previously that branching decreases the Tc–g independently of the hydrophobic effects of the aryl branch points.7 In this work we observed that as well as this general decrease in the magnitude of the endotherm associated with the Tc–g as branching increased, there was a very large decrease as the composition was changed from NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 of 25
1 of 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 15
1 to 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.
The 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 polymer also displayed a cloud point (i.e. the solution was clear below the cloud point) and this indicated that the chains were not aggregated below the Tc–g. The most appropriate model that can be used to explain this behaviour appears to be to propose that at a composition of 15
1 polymer also displayed a cloud point (i.e. the solution was clear below the cloud point) and this indicated that the chains were not aggregated below the Tc–g. The most appropriate model that can be used to explain this behaviour appears to be to propose that at a composition of 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NIPAM
1 NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the polymers have core–shell morphology with a globular inner core and an open coil outer shell. The globular inner core would be above the Tc–g at all temperatures observed in the microDSC experiments. Whereas the swollen outer shell passed through a Tc–g as the temperature was increased. Thus, only a small fraction of the polymer chain would be involved in this Tc–g and this provides a much reduced endotherm at the transition.
1, the polymers have core–shell morphology with a globular inner core and an open coil outer shell. The globular inner core would be above the Tc–g at all temperatures observed in the microDSC experiments. Whereas the swollen outer shell passed through a Tc–g as the temperature was increased. Thus, only a small fraction of the polymer chain would be involved in this Tc–g and this provides a much reduced endotherm at the transition.
To further investigate this proposal we used a solvatochromic dye to probe the state of solvation of the series of polymers. In recent work we had included two dyes that acted as either a donor or acceptor in non-radiative energy transfer (Förster resonance energy transfer (FRET)) and we were able to show at the molecular level that binding of vancomycin chain ends on HB-PNIPAM to bacteria induced a coil-to-globule transition.12 The use of FRET is one way of examining the switch from coil-to-globule in response to a stimulus. However, it is also possible to use molecular probes that change their photophysical properties dependant on the polarity of their location. We had considered that such solvatochromic dyes could be added to the polymers in water above and below the Tc–g so that their spectra could be used to probe the state of the polymer chain: globular and relatively apolar or open coil and polar. Therefore, for a homogenous model of the phase behaviour of these polymers, in which an open solvated coil passes in one step through to an homogeneous globule, one would expect a solvatochromic dye to provide similar spectra regardless of polymer composition but also to provide different spectra above and below the Tc–g. However, this was not the case and data presented below provide further evidence of a core(globular)–shell(coil) structure in the HB-PNIPAM materials in water that maybe a general feature of similar polymers.
Nile red is a solvatochromic fluorophore that can be used to detect the polarity of the medium in which it is solvated.13 The fluorescence emission of the dye changes systematically with solvent polarity and in this work we first correlated the wavelength to the polarity of various solvents then we made the extension that the wavelength in particular solvents can be extrapolated to describe the general polarity of the dye within polymer coils/globules.
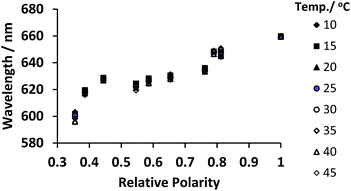
Nile red was dissolved in a variety of solvents and diluted to the concentration of 10−7 mol dm−3, heated from 10 to 45 °C and the emission spectra recorded following sample excitation at 580 nm. In most solvents the fluorescence intensity decreased as the solvent was heated, except in the case of glycerol where there was a marked increase from 10 to 45 °C. The fluorescence emission of Nile red occurs over a broad spectrum that shifts in response to both solvent polarity and temperature, so the emission peak was fitted to a Gaussian distribution to calculate the peak emission wavelength and each of these peak values in various solvents from 10–45 °C are shown in Fig. 6. The data showed that Nile red can be used to probe the polarity of a polymer's micro-environment by correlating the peak emission of the dye added to a polymer solution to the peak emission in a solvent of known polarity.14
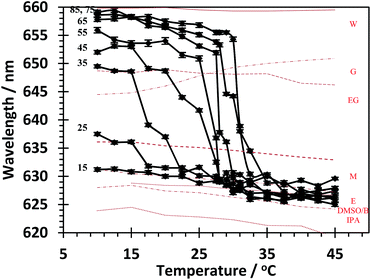
Therefore, Nile red was added to dilute polymer solutions in water to probe the relative polarity of the polymers across the temperature range and the data are shown in Fig. 7. Linear polymers were not capable of solvating Nile red below the Tc–g at these low concentrations of DMSO. Polymers with a low degree of branching showed marked decreases in the peak emission wavelength as the polymer passed through a conformational transition, and for the 85, 75 and 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 samples the wavelength decreased from 659 to 625 nm. As expected from the cloud point and calorimetric data as the degree of branching increased the conformational change occurred at lower temperatures. However, the peak emission below the Tc–g was observed at lower wavelength. Using the emission wavelength as a measure of sample polarity at 10 °C the 85, 75 and 65
1 samples the wavelength decreased from 659 to 625 nm. As expected from the cloud point and calorimetric data as the degree of branching increased the conformational change occurred at lower temperatures. However, the peak emission below the Tc–g was observed at lower wavelength. Using the emission wavelength as a measure of sample polarity at 10 °C the 85, 75 and 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 samples had a polarity only slightly lower than water (relative polarity 1).14
1 samples had a polarity only slightly lower than water (relative polarity 1).14
The sub-Tc–g peak emissions for the other samples gradually decreased as the degree of branching increased and the 35, 25 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 samples had peak emissions that had average polarity equivalents to ethylene glycol (relative polarity 0.79), methanol (0.76) and ethanol (0.65) respectively.
1 samples had peak emissions that had average polarity equivalents to ethylene glycol (relative polarity 0.79), methanol (0.76) and ethanol (0.65) respectively.
These observations suggested that, below the Tc–g, with increased branching and increasing concentration of aromatic branching points, the branched polymers provided an increasingly more hydrophobic environment in which the Nile red was solvated. Above the Tc–g all samples show a similar emission peak wavelength of approximately 628 nm, suggesting that when the PNIPAM collapses the Nile red solvated by the polymer coil has a relative polarity between that of ethanol and butanol (relative polarity 0.65) and butanol (relative polarity 0.65 and 0.58).
These data should be considered in consort with the cloud point data and calorimetric data. Both data sets, in Fig. 4, indicated transitions in all of the samples yet the Nile red data indicate minimal changes in the fluorescence spectra for the sample with 15 repeat units per branch point and a much reduced change in the sample with 25 repeat units per branch point. The minimal change in the spectrum of Nile red for the 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 composition appears to confirm the calorimetric data, which showed a much decreased endotherm at Tc–g for this polymer. Since the Nile red preferentially partitions into hydrophobic phases in the presence of more hydrophobic phases the data indicate the presence of a hydrophobic (globular phase) below the cloud point/calorimetric Tc–g reported in Fig. 4. Therefore, we propose that as the degree of branching increased the inner regions of the polymer coils become desolvated and formed a globular structure. However, the presence of the polar end groups maintained a solvated shell and it is the desolvation of this shell that produces the thermally responsive cloud point/calorimetric Tc–g for the 15
1 composition appears to confirm the calorimetric data, which showed a much decreased endotherm at Tc–g for this polymer. Since the Nile red preferentially partitions into hydrophobic phases in the presence of more hydrophobic phases the data indicate the presence of a hydrophobic (globular phase) below the cloud point/calorimetric Tc–g reported in Fig. 4. Therefore, we propose that as the degree of branching increased the inner regions of the polymer coils become desolvated and formed a globular structure. However, the presence of the polar end groups maintained a solvated shell and it is the desolvation of this shell that produces the thermally responsive cloud point/calorimetric Tc–g for the 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 25
1 and 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 polymers and to a lesser degree the 35
1 polymers and to a lesser degree the 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 polymer. Also, the data showed a gradual decrease in the peak emission wavelength as branching increased before the step change was observed between compositions of 25
1 polymer. Also, the data showed a gradual decrease in the peak emission wavelength as branching increased before the step change was observed between compositions of 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 15
1 and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Therefore, these data and the microDSC data provided evidence for the presence of an increasingly desolvated phase as the degree of branching increases.
1. Therefore, these data and the microDSC data provided evidence for the presence of an increasingly desolvated phase as the degree of branching increases.
These results have important implications for the production of stimulus responsive devices because they indicated that the end groups: (1) penetrate to a limited degree into the polymer coil and (2) that only small changes in the solvation of end groups (e.g. binding to receptors) would be required to drive these materials through a phase transition because the solvation of the shell would be dominated by the penetration of the end groups. Penetration of polar end groups (e.g. carboxylic acid groups) into the outer regions of the polymer would increase the swelling and solvation of a shell domain in the same way that copolymerising NIPAM with polar monomers, such as acrylic acid, increases the Tc–g because of increased solvation and swelling. However, the extent of the effect of the end groups will be limited to the outer shell so that the Tc–g of the inner regions of these highly branched structures would be dominated by the desolvating effects of segment crowding and hydrophobicity of the branch points.
Conclusion
We provide evidence that shows that at certain temperatures, in aqueous media, it is possible to provide stimulus responsive polymers with both globular (desolvated) and open coil (solvated) structures. These are highly branched polymers and we propose that they form core (globular)–shell (open coil) particles.Experimental
Synthesis of 4-vinylbenzyl pyrrolecarbodithioate
Pyrrole (100 ml) was distilled. NaH (11.98 g, 0.497 mol) was added to a 3 neck flask under N2, with stirring, as a suspension in DMF (240 ml) and the flask allowed to purge with N2. Pyrrole (20 g, 0.298 mol) in DMF (40 ml) was added dropwise to the reaction over 30 min at 0 °C. The solution was stirred at room temperature for 30 minutes. Carbon disulphide (22 g, 0.298 mol) in DMF (40 ml) was added dropwise over 10 minutes at 0 °C. The solution was stirred at room temperature for 30 minutes. 4-vinylbenzyl chloride (45.48 g, 0.289 mol) in DMF (40 ml) was added to the dropwise over 20 minutes at 0 °C with stirring. The solution was stirred at room temperature for 30 minutes. 400 ml of deionised water was added to the reaction mixture and the product was extracted with 4 × 500 ml of diethyl ether. The organic layers were combined dried with magnesium sulphate and evaporated yielding a dark coloured liquid which was purified by silica column chromatography in hexane to yield 51.78 g of a yellow solid.Expected results: carbon 64.9%, hydrogen 5.1%, nitrogen 5.4%, sulphur, 24.7%.
Actual results: carbon 65.3%, hydrogen 5.1%, nitrogen 5.3%, sulphur 22.41%.
1H NMR (400 MHz, CDCl3) (ppm): δ 4.61 (2H, s, RCH2Ar), δ 5.81 (1H, m, RC = CH), δ 6.35 (1H, m, RC = CH), δ 6.76 (2H, m, Ar), δ 7.18 (2H, m, Ar), δ 7.45 (2H, d, Ar), δ 7.59 (2H, d, Ar).
Synthesis of pyrrolecarbodithioate ended HB-PNIPAM
N-Isopropyl acrylamide was recrystallized from 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 hexane
60 hexane![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) toluene. It was dissolved in dioxane (10 ml) with 4-vinylbenzyl pyrrolecarbodithioate and ACVA in the quantities shown in Table 3. The solution was transferred to a glass amopoule, 3 cycles of freeze–pump–thaw were carried out. The ampoule was flame sealed and reacted at 60 °C for 48 hours. The viscous solution was precipitated into diethyl ether and dried under vacuum. Precipitation was repeated three times to give an off white solid.
toluene. It was dissolved in dioxane (10 ml) with 4-vinylbenzyl pyrrolecarbodithioate and ACVA in the quantities shown in Table 3. The solution was transferred to a glass amopoule, 3 cycles of freeze–pump–thaw were carried out. The ampoule was flame sealed and reacted at 60 °C for 48 hours. The viscous solution was precipitated into diethyl ether and dried under vacuum. Precipitation was repeated three times to give an off white solid.
NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
NIPAM moles | NIPAM mass/g | 1 moles | 1 mass/g | ACVA moles |
|---|---|---|---|---|---|
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 2.655 | 0.687 | 2.655 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 1.770 | 0.458 | 1.770 |
25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 1.062 | 0.275 | 1.062 |
35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 0.759 | 0.197 | 0.759 |
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 0.590 | 0.153 | 0.590 |
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 0.483 | 0.123 | 0.483 |
65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 0.410 | 0.106 | 0.410 |
75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 0.354 | 0.092 | 0.354 |
85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
2.655 | 3.000 | 0.312 | 0.081 | 0.312 |
1H NMR (400 MHz, CDCl3) (ppm): δ 0.9–1.3(6H, s, –N(CH3)2), δ 1.4–1.8 (2H, br m, –CH2–CH–Ar–), δ 1.9–2.2(2H, br m, –CH2–CH–CO–NH–) and (1H, br m, CH2–CH–CONH–), δ 3.7(H2O-polymer bound), δ 4.0 (1H, br s, (CH3)2CH–), δ 6.3 (H2, br s, N-pyrrole–H), δ 6.6–7.2 (br m, –Ar–), δ 7.65 (2H, br s, N-pyrrole–H).
Micro DSC
Polymer samples were dissolved in H2O at 5 mg ml−1 and stored at 5 °C for 24 hours prior to use to ensure complete dissolution. Samples were run on a Microcal inc VP-DSC over the temperature range 5–60 °C with a heating rate of 1.5 °C per minute and cooling rate of 1 °C per minute. The Tc–g was taken as the temperature at the thermogram peak maximum.Cloud point
Polymer samples were dissolved in H2O at 5 mg ml−1 and stored at 5 °C for 24 hours prior to use to ensure complete dissolution. Samples were run on a Perkin Elmer UV spectrometer over the temperature range 10–60 °C with a heating rate of 1.5 °C per minute. The turbidity of the solution was measured by the absorbance at 500 nm. The LCST was taken as the onset of the increase in turbidity.Nile red solutions
A stock solution of Nile red was made up by adding 2 mg of Nile red to 25 cm3 solvents. This was diluted down to 10−7 M concentration. Measurements were made in 99% water by diluting the DMSO stock solution down to 10−7 M by further addition of water, this was necessary due to the insolubility of Nile red in water.Labelling polymer solution with Nile red
A stock solution of Nile red was made up by adding 2 mg of Nile red to 25 cm3 on DMSO. The HB-PNIPAM polymers (11 mg) were dissolved in ultrapure water (7 ml). 100 μl of the Nile red stock solution was added to the polymer solution and mixed well. The Nile red polymer solution was then further diluted 1 part in 2 parts ultrapure water.Notes and references
- Y. Guan and Y. Zhang, PNIPAM microgels for biomedical applications: from dispersed particles to 3D assemblies, Soft Matter, 2011, 7, 6375–6384 RSC.
- B. R. Saunders, et al. Microgels: from responsive polymer colloids to biomaterials, Adv. Colloid Interface Sci., 2009, 147–148, 251–262 CrossRef CAS PubMed.
- Z. M. O. Rzaev, S. Dinçer and E. Pişkin, Functional copolymers of N-isopropylacrylamide for bioengineering applications, Prog. Polym. Sci., 2007, 32, 534–595 CrossRef CAS PubMed.
- J. Shepherd, et al. Binding Bacteria to Highly Branched Poly(N-isopropyl acrylamide) Modified with Vancomycin Induces the Coil-to-Globule Transition, J. Am. Chem. Soc., 2010, 132, 1736–1737, DOI:10.1021/ja907466y.
- P. Sarker, et al. Highly Branched Polymers with Polymyxin End Groups Responsive to Pseudomonas aeruginosa, Biomacromolecules, 2010, 12, 1–5, DOI:10.1021/bm100922j.
- J. Shepherd, et al. Hyperbranched poly(NIPAM) polymers modified with antibiotics for the reduction of bacterial burden in infected human tissue engineered skin, Biomaterials, 2011, 32, 258–267 CrossRef CAS PubMed.
- S. Carter, B. Hunt and S. Rimmer, Highly Branched Poly(N-isopropylacrylamide)s with Imidazole End Groups Prepared by Radical Polymerization in the Presence of a Styryl Monomer Containing a Dithioester Group, Macromolecules, 2005, 38, 4595–4603, DOI:10.1021/ma047742n.
- S. Carter, S. Rimmer, A. Sturdy and M. Webb, Highly Branched Stimuli Responsive Poly[(N-isopropyl acrylamide)-co-(1,2-propandiol-3-methacrylate)]s with Protein Binding Functionality, Macromol. Biosci., 2005, 5, 373–378, DOI:10.1002/mabi.200400218.
- S. Rimmer, S. Carter, R. Rutkaite, J. W. Haycock and L. Swanson, Highly branched poly-(N-isopropylacrylamide)s with arginine-glycine-aspartic acid (RGD)- or COOH-chain ends that form sub-micron stimulus-responsive particles above the critical solution temperature, Soft Matter, 2007, 3, 971–973, 10.1039/b705188c.
- A. Postma, et al. A simple method for determining protic end-groups of synthetic polymers by 1H NMR spectroscopy, Polymer, 2006, 47, 1899–1911 CrossRef CAS PubMed.
- R. Xie, Y. Li and L.-Y. Chu, Preparation of thermo-responsive gating membranes with controllable response temperature, J. Membr. Sci., 2007, 289, 76–85 CrossRef CAS PubMed.
- L. Swanson, et al. Fluorescence resonance energy transfer confirms the bacterial-induced conformational transition in highly-branched poly(N-isopropyl acrylamide) with vancomycin end groups on binding to Staphylococcus aureus, Soft Matter, 2014, 10, 5824–5835 RSC.
- P. G. Jessop, D. A. Jessop, D. Fu and L. Phan, Solvatochromic parameters for solvents of interest in green chemistry, Green Chem., 2012, 14, 1245–1259, 10.1039/c2gc16670d.
- C. Reichardt & T. Welton, Solvents and solvent effects in organic chemistry, John Wiley & Sons, 2011 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra10076j |
| This journal is © The Royal Society of Chemistry 2014 |