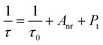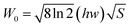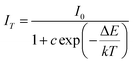DOI:
10.1039/C4RA09793A
(Paper)
RSC Adv., 2014,
4, 46313-46318
Synthesis and near-infrared luminescence of La3GaGe5O16:Cr3+ phosphors
Received
4th September 2014
, Accepted 16th September 2014
First published on 16th September 2014
Abstract
A novel Cr3+-doped La3GaGe5O16 phosphor was synthesized by a solid-state reaction, and the phase formation and microstructure, near-infrared (NIR) photoluminescence (PL) properties and PL thermal stability were investigated in detail. The excitation spectrum of La3GaGe5O16:Cr3+ at 270, 415 and 573 nm corresponded to three spin-allowed Cr3+ d–d intra-transitions of 4A2–4T1 (4P), 4A2–4T1 (4F), and 4A2–4T2 (4F), respectively, among which the main red emission peak observed at 700 nm is identified, which is due to the 2E–4A2 transition from Cr3+ ions. It is further proved that the dipole–dipole interactions result in the concentration quenching of Cr3+ in La3GaGe5O16:Cr3+ phosphors, and the energy-transfer distance at the quenching concentration was around 26.90 Å. Moreover, thermal quenching luminescence results reveal that La3GaGe5O16:Cr3+ exhibits good thermal stability. The above results indicate that La3GaGe5O16:Cr3+ has potential practical applications in luminescence solar concentrators with broad-band absorption.
1. Introduction
Recently, solar cells have attracted much attention because they are promising devices for inexpensive and large-scale solar energy conversion. However, the efficiency in the commercial Si solar cells is just 15%, which means that most of sunlight energy is lost.1 One of the main energy loss in the conversion of solar energy to electricity is attributed to the so-called spectral mismatch between incident solar photon energies and the energy gap (Eg) of the cells.2 As we know, the main energy of the solar emission dispersed on the earth surface is in visible light region, but Eg of the Si cell is suitable to the absorption in far-red and near-infrared regions. Consequently, luminescent solar concentrator (LSC) was developed that can collect solar photons and transfer them to the photons matching Eg of the Si solar cells. Moreover, when luminescent materials with efficient UV-Vis absorption and deep-red and near-infrared emissions are applied in LSCs, the efficiency of Si cells would be significantly enhanced.3 Therefore, studies on new phosphors with deep-red and near-infrared emissions are highly desirable.
Interest in Cr3+ activated inorganic materials is widespread because of its deep red colors and bright photoluminescence. Cr3+ is the most stable state of chromium element because of its narrow-band emissions (usually near 700 nm) due to the spin-forbidden 2E–4A2 transition, or a broadband emission (650–1600 nm) due to the spin-allowed 4T2–4A2 transition, which strongly depends on the crystal-field environment of the host lattices.4–6 Therefore Cr3+ can be used as a luminescent dopant with red emission in LSCs. This dopant also has its specific use in modern technologies, for example, tunable solid-state lasers, high temperature sensors, and high-pressure calibrants.7–9 Recently, various Cr3+ doped host materials has been widely investigated by several research groups and its emission in various compounds like Y3Al5O12,10 MgAl2O4,11 Cs2NaScCl6 (ref. 12) and many others have also been studied.
As for the development of Cr3+-activated optical materials with tunable red and NIR emission, gallogermanates were usually used as the host materials since Cr3+ ions can substitute for Ga3+ ions in distorted octahedral sites. Moreover, suitable host crystal-field strength around Cr3+ will help to achieve intense deep red emission.13 As we know, the reported Cr3+-doped gallogermanates include M3Ga2Ge4O14:Cr3+ (M = Sr or Ca),14 La3Ga5GeO14:Cr3+,13 and Zn3Ga2Ge2O10:Cr3+.5 In 1998, G. Adiwidjaja firstly reported the new gallogermanate of lanthanum compound, La3GaGe5O16, as a single crystal form and revealed its chemical composition and detailed crystal structure.15 However, to the best of our knowledge, there are no further reports on this compound and their potential application as optical materials hosts except for our recent work.16 In this paper, we have found that Cr3+-doped lanthanide gallogermanate garnet (La3GaGe5O16:Cr3+) phosphors fabricated by a solid-state reaction method can exhibit remarkable red emission peak at 700 nm. And the energy transfer mechanism, photoluminescence decay curves and the temperature dependent PL behaviour have also been investigated.
2. Experimental
2.1 Materials and synthesis
A series of rare earth-doped La3Ga1−xGe5O16:xCr3+ phosphors were synthesized by high temperature solid-state reaction. The starting materials were analytical reagent grade La2O3 (99.995%), Ga2O3 (99.9%), GeO2 (99.9%) and Cr2O3 (99.9%). Firstly, the starting materials were thoroughly mixed in an agate mortar according to the given stoichiometric amounts. Then, the mixture was put into an alumina crucible and calcined in a muffle furnace at 1250 °C for 5 h in air. After the samples were cooled to room temperature, the phosphors were finally obtained.
2.2 Characterization methods
The phase structures of the as-prepared samples were carried out on a SHIMADZU model XRD-6000 diffractometer using Cu Kα radiation (λ = 0.15406 nm) at 40 kV and 30 mA. Microstructure observation was observed using a scanning electron microscopy (SEM) (HITACHI, S-3400). Room temperature excitation and emission spectra were measured on a fluorescence spectrophotometer (F-4600, HITACHI, Japan) with a photomultiplier tube operating at 400 V, and a 150 W Xe lamp used as the excitation lamp. The temperature-dependence luminescence properties were measured on the same spectrophotometer, which was combined with a self-made heating attachment and a computer-controlled electric furnace. The decay curves were recorded on a spectrofluorometer (HORIBA, JOBIN YVON FL3-21), and the 460 nm pulse laser radiation (nano-LED) was used as the excitation source.
3. Results and discussion
3.1 Phase formation and microstructure
The diffraction pattern is usually used to identify the crystal structure and the phase purity of the sample. Fig. 1(a) shows the XRD patterns of as-prepared La3GaGe5O16, La3Ga0.97Ge5O16:0.03Cr3+ and La3Ga0.85Ge5O16:0.15Cr3+ phosphors. From Fig. 1(a), we can know that all of the diffraction peaks are in good agreement with the reported triclinic phase of La3GaGe5O16 (JCPDS 89-0211) and no detectable impurity phase is observed, which indicates that single-phased La3GaGe5O16 powder can be obtained by the solid-state method. Furthermore, based on the consideration of the similar effective ionic radius and valence of cations, Cr3+ ions prefer to occupy the Ga3+ sites in the host. SEM image of the selected La3Ga0.97Ge5O16:0.03Cr3+ phosphor is presented in Fig. 1(b), from which the phosphor particles were well dispersed, and their mean size was about 3–8 μm, and the uniform distribution and small particles verified that the as-prepared phosphors can be potential in the future application.
 |
| | Fig. 1 (a) XRD patterns of as-prepared La3GaGe5O16, La3Ga0.97Ge5O16:0.03Cr3+, and La3Ga0.85Ge5O16:0.15Cr3+ phosphors, the standard data for La3GaGe5O16 (JCPDS card no. 89-0211) is shown as a reference. (b) SEM image of the selected La3Ga0.97Ge5O16:0.03Cr3+ phosphor. | |
3.2 Luminescence properties and the corresponding mechanism
Cr3+ belong to 3d3 electronic configurations and the splitting of the energy levels in octahedral symmetry can be expressed by well-known Tanabe–Sugano energy level diagram as shown in Fig. 2(a). The d3 configuration gives rise to two quartet terms and 4F and 4P with 4F term lying lower to 4P as per Hund's rule. In addition to these quartet terms, there are several doublet terms. The 2G term splits into 2E level, and the 4A2, 4T2, and 4T1 levels come from the 4F term.17,18 Moreover, the luminescence and excitation spectra depend on the relative positions between 4T2 and 2E levels, which is shown in Fig. 2(b) as a configurational coordinate diagram. When Cr3+ ions locate in the intermediate field sites, 4T2 is above the 2E level, resulting in the R lines emission (assigned to 2E–4A2 transition). And when Cr3+ ions locate in the weak field sites, 4T2 is under the 2E level, which causes the band emission (assigned to 4T2–4A2 transition). Therefore, Fig. 3 shows the normalized excitation and emission spectra of La3Ga0.97Ge5O16:0.03Cr3+ at room temperature, which can be used to understand the corresponding transitions of Cr3+. Accordingly, Fig. 4 shows schematic energy levels of Cr3+, the corresponding excitation spectrum and electron transitions for the Cr3+-doped La3GaGe5O16 phosphor is also given based on the measurement results in Fig. 3. It is found that, under excitation at 415 nm, Cr3+-doped La3GaGe5O16 phosphor exhibits a broadening 2E–4A2 emission peaking at 700 nm that superimposes on a broad emission band extending from 600 to 900 nm. The broadening of the 2E–4A2 emission is probably caused by the electron–phonon coupling because the phonon energy of the Cr3+ dopant matches well with that of the gallate-containing host,19 whereas the broadband emission may be ascribed to the 4T2–4A2 transitions from some disordered Cr3+ ions in the gallogermanate system.20 The excitation spectrum monitored at 700 nm covers a very broad spectral region (from 200 to 650 nm) consists of three main excitation bands originating from the inner transitions of Cr3+, including the 270 nm band originating from the 4A2–4T1 (4P) transition, the 415 nm band originating from the 4A2–4T1 (4F) transition and 573 nm band originating from the 4A2–4T2 (4F) transition.21 Based on the above PLE spectrum for La3Ga0.97Ge5O16:0.03Cr3+, it proves that the obtained sample has excellent n-UV and visible excitation properties, which in turn means that this kind of phosphor has the potential as a promising NIR-emitting phosphor system for LSCs with broad-band absorption.
 |
| | Fig. 2 (a) Tanabe–Sugano diagram for Cr3+ in La3Ga0.97Ge5O16. (b) Mechanistic configurational coordinate diagram illustrating different emission channels. The solid and dashed arrows represent the emissions from the Cr3+ ions located in the intermediate and weak field sites, respectively. | |
 |
| | Fig. 3 Normalized excitation and emission spectra of La3Ga0.97Ge5O16:0.03Cr3+ phosphor at room temperature. | |
 |
| | Fig. 4 Schematic energy levels of Cr3+, the corresponding excitation spectrum and electron transitions for the Cr3+-doped La3GaGe5O16 phosphors. | |
Fig. 5 presents the emission (λex = 415 nm) spectra of the La3Ga1−xGe5O16:xCr3+ phosphors with different concentrations of Cr3+ (ranging from 0.01 to 0.15) at room temperature. As is shown in Fig. 5, this series of samples all produce a strong and broad-band NIR emission with a peak at around 700 nm, and the emission spectra have no obvious changes in the spectral configuration except for the emission intensity. Furthermore, the inset shows the Cr3+ content dependent PL intensities, and Cr3+ concentration (x) in La3Ga1−xGe5O16:xCr3+ reaches a saturation point at x = 0.03. After that, the PL intensity begins to decrease with increasing Cr3+ concentration due to the concentration quenching effect.
 |
| | Fig. 5 PL spectra for the La3Ga1−xGe5O16:xCr3+ (x = 0.01, 0.03, 0.05, 0.08, 0.10 and 0.15) phosphors, and the inset shows Cr3+ concentration dependence of PL intensity. | |
It is accepted that energy quenching can be ascribed on the energy transfer between Cr3+ ions followed by energy transfer to traps or quenching sites. Therefore, in order to further confirm the process of energy transfer between Cr3+ ions in the La3GaGe5O16:Cr3+ phosphor, the interaction type between sensitizers or between the sensitizer and activator can be calculated by the following equation:22
| |
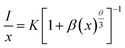 | (1) |
in this equation
x is the activator concentration which is not less than the critical concentration,
I/
x is the emission intensity (
I) per activator concentration (
x),
K and
β are constants for the same excitation condition for a given host crystal, and
θ is an indication of electric multipolar character. It is previously reported that
θ = 3 for the energy transfer among the nearest-neighbor ions, as
θ = 6, 8 and 10 corresponds to dipole–dipole (d–d), dipole–quadrupole (d–q), and quadrupole–quadrupole (q–q) interactions, respectively. To get a correct
θ value, the dependence of lg(
I/
x) on lg(
x) is plotted, and it yield a straight line with a slope equal to −
θ/3. As shown in
Fig. 6, the slope is −1.93. The value of
θ can be calculated as 5.79, which is close to 6 that means the quenching is dipole–dipole interactions in La
3GaGe
5O
16:Cr
3+ phosphors.
 |
| | Fig. 6 The fitting curve of log(I/x) vs. log(x) in La3Ga1−xGe5O16:xCr3+ phosphors. | |
The critical distance of energy transfer Rc was calculated by using the concentration quenching method. Rc can be calculated using the relation given by Blasse:23
| |
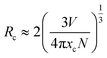 | (2) |
here
N is the number of molecules in the unit cell,
V is the unit cell volume and
xc is the critical concentration. For La
3GaGe
5O
16 host, the crystallographic parameters are
N = 2,
V = 611.81 Å
3 and
xc is 0.03. According to the above equation, the critical distance of energy transfer is estimated to be about 26.90 Å.
In order to further understand the concentration quenching in detail, the decay curves for the La3Ga1−xGe5O16:xCr3+ (x = 0.01, 0.03, 0.05, 0.08 and 0.15) phosphors excited at 460 nm and monitored at 700 nm are measured and depicted in Fig. 7. As shown in Fig. 7, it is found that the decay curves are well fitted with a second order exponential equation:24
| |
I(t) = A1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ1) + A2 exp(−t/τ1) + A2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ2) exp(−t/τ2)
| (3) |
where,
I is the luminescence intensity,
A1 and
A2 are constants,
t is the time,
τ1 and
τ2 are rapid and slow lifetime values for exponential components, respectively. Based on the
eqn (3), and we can obtain the
A1,
A2,
τ1 and
τ2 values based on the fitting of the decay curves. Therefore, the effective lifetime constant (
τ*) can be calculated as:
25| | |
τ* = (A1τ12 + A2τ22)/(A1τ1 + A2τ2)
| (4) |
 |
| | Fig. 7 Room temperature decay curves of La3Ga1−xGe5O16:xCr3+ phosphors with different Cr3+ contents x = 0.01, 0.03, 0.05, 0.08 and 0.15. | |
The effective decay time (τ*) were calculated to be 75.19, 69.66, 60.18, 47.65 and 40.29 μs for La3Ga1−xGe5O16:xCr3+ with x = 0.01, 0.03, 0.05, 0.08 and 0.15, respectively. The obtained result shows that the measured lifetime τ of Cr3+ emission decreases faster with higher doping Cr3+ concentration. The measured lifetime is also related to the total relaxation rate by:26,27
| |
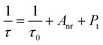 | (5) |
where
τ0 is the radiative lifetime,
Anr is the nonradiative rate due to multiphonon relaxation,
Pt is the energy transfer rate between Cr
3+ ions, and
τ is equivalent to
τ*. With increasing Cr
3+ concentration, the distance between Cr
3+ ions decreases. Therefore, both the energy transfer rate between Cr
3+–Cr
3+ and the probability of energy transfer to luminescent killer sites increased. Consequently, the lifetimes are shortened as increasing Cr
3+ concentration.
28
3.3 Thermal stability behavior
The thermal stability of phosphor is one of important parameters for the possible application. Fig. 8 shows the dependence of PL spectra of La3Ga0.99Ge5O16:0.01Cr3+, La3Ga0.97Ge5O16:0.03Cr3+ and La3Ga0.85Ge5O16:0.15Cr3+ on temperature excited by 415 nm light. The relative emission intensities of all phosphors decrease with increasing temperature in the range of 25 °C to 300 °C. We observed a decay of 12.9% for La3Ga0.99Ge5O16:0.01Cr3+, 20.5% for La3Ga0.97Ge5O16:0.03Cr3+, and 32.6% for La3Ga0.85Ge5O16:0.15Cr3+ at 100 °C. The thermal quenching of emission intensity can be explained by several mechanisms. A widely accepted mechanism is the electronic transition through the intersection between the ground and excited states. In other words this mechanism is described as a large displacement between the ground and excited state in the configuration coordinate diagram.29 Moreover, the full-width at half-maximum (FWHM) of the PL spectra of all phosphors increases with increasing temperature, which can be described by using the Boltzmann distribution according to the following equation:30| |
 | (6) |
| |
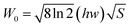 | (7) |
where W0 is the FWHM at 0 K, hw is the energy of the lattice vibration that interacts with the electronic transitions, S is the Huang–Rhys–Pekar parameter, and k is the Boltzmann constant. With the increasing of temperature, the bond length between luminescence center and its coordinated ions increase which cripple the intensity of crystal field and the interaction between electron and phonon play as the leading role, consequently the FWHM of the emission line is broadened.
 |
| | Fig. 8 Temperature-dependent PL spectra of typical La3Ga0.97Ge5O16:0.03Cr3+. The inset shows the variation of the temperature dependent relative emission intensities for La3Ga1−xGe5O16:xCr3+ (x = 0.01, 0.03 and 0.15). | |
To better understand the temperature dependence of photoluminescence, the activation energy was calculated using the Arrhenius equation:31,32
| |
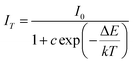 | (8) |
where
I0 is the initial PL intensity of the phosphor at room temperature,
IT is the PL intensity at different temperatures,
c is a constant, Δ
E is the activation energy for thermal quenching, and
k is Boltzmann constant (8.62 × 10
−5 eV). According to the equation, the plot of ln[(
I0/
IT) − 1]
vs. 1/
kT yields a straight line, and the activation energy Δ
E is obtained from the slop of the plot. As shown in
Fig. 9, Δ
E is calculated to be 0.309, 0.236 and 0.234 eV for the La
3Ga
0.99Ge
5O
16:0.01Cr
3+, La
3Ga
0.97Ge
5O
16:0.03Cr
3+ and La
3Ga
0.85Ge
5O
16:0.15Cr
3+ phosphors, respectively. From the results, we can find that the samples with low Cr
3+ concentration has larger Δ
E values corresponding to better thermal stability properties, which means that the sample with high Cr
3+ concentration possible possess more de-activation channels originating from the enhanced interaction among the increasing Cr
3+ ions.
 |
| | Fig. 9 A ln[(I0/IT) − 1] vs. 1/KBT fitting plot and the corresponding activation energy derived from the thermal quenching data of La3Ga1−xGe5O16:xCr3+ (x = 0.01, 0.03 and 0.15). | |
4. Conclusion
In summary, a novel near-infrared (NIR) phosphor, La3GaGe5O16:Cr3+ was synthesized via the solid-state reaction. The La3GaGe5O16:Cr3+ phosphor exhibited NIR emission peaked at 700 nm under the excitation of 415 nm UV radiation. The optimal doping concentration of Cr3+ is 3 mol%, and the concentration quenching mechanism is determined to be dipole–dipole interaction. Moreover, the temperature dependence of luminescence shows that the thermal quenching behavior is related to the Cr3+ doping concentration and the emission intensity of Cr3+ decrease with the increase of temperature and Cr3+ will decay faster with higher doping concentration. Preliminary studies showed that La3GaGe5O16:Cr3+ has potential practical applications in luminescent solar concentrator with broad-band absorption and emission.
Acknowledgements
The present work was supported by the National Natural Science Foundations of China (Grant no. 51002146, 51272242), Natural Science Foundations of Beijing (2132050), the Program for New Century Excellent Talents in University of Ministry of Education of China (NCET-12-0950), Beijing Nova Program (Z131103000413047), Beijing Youth Excellent Talent Program (YETP0635) and the Funds of the State Key Laboratory of New Ceramics and Fine Processing, Tsinghua University (KF201306).
References
- D. Timmerman, I. Izeddin, P. Stallinga, I. N. Yassievich and T. Gregorkiewicz, Nat. Photonics, 2008, 2, 105 CrossRef CAS.
- B. S. Richards, Sol. Cells, 2006, 90, 2329 CrossRef CAS.
- L. M. Shao and X. P. Jing, J. Lumin., 2011, 131, 1216 CrossRef CAS.
- B. Qiao, Z. L. Tang, Z. T. Zhang and L. Chen, Mater. Lett., 2007, 61, 401 CrossRef CAS.
- Z. W. Pan, Y. Y. Lu and F. Liu, Nat. Mater., 2012, 11, 58 CrossRef CAS.
- Y. Li, S. F. Zhou, Y. Y. Li, K. Sharafudeen, Z. J. Ma, G. P. Dong, M. Y. Peng and J. R. Qiu, J. Mater. Chem. C, 2014, 2, 2657 RSC.
- V. Singh, R. P. S. Chakradhar, J. L. Rao and J. J. Zhu, Mater. Chem. Phys., 2008, 111, 143 CrossRef CAS.
- P. R. Wamsley and K. L. Bray, J. Lumin., 1994, 59, 11 CrossRef CAS.
- K. T. V. Grattan, R. K. Selli and A. W. Palmer, Rev. Sci. Instrum., 1988, 59, 1328 CrossRef CAS.
- S. M. Kaczmarek, W. Chen and G. Boulon, Cryst. Res. Technol., 2006, 41, 41 CrossRef CAS.
- V. Singh, R. P. S. Chakradhar, J. L. Rao and D. K. Kim, J. Lumin., 2009, 129, 130 CrossRef CAS.
- O. S. Wenger, R. Valiente and H. U. Gudel, J. Chem. Phys., 2001, 115, 3819 CrossRef CAS.
- W. Z. Yan, F. Liu, Y. Y. Lu, X. J. Wang, M. Yin and Z. W. Pan, Opt. Express, 2010, 18, 20215 CrossRef CAS PubMed.
- P. I. Macfarlane, T. P. J. Han, B. Henderson and A. A. Kaminskii, Opt. Mater., 1994, 3, 15 CrossRef CAS.
- G. Adiwidjaja, M. Bröker, C. Claus, K. Friese, K. H. Klaska, O. Jarchow, M. Ruks and I. Wozniak, Z. Kristallogr., 1998, 213, 223 CAS.
- J. Zhou and Z. G. Xia, J. Mater. Chem. C, 2014, 2, 6978 RSC.
- M. N. Sanz-Ortiz, F. Rodríguez, I. Hernández, R. Valiente and S. Kück, Phys. Rev. B: Condens. Matter Mater. Phys., 2010, 81, 045114 CrossRef.
- I. Hernández, F. Rodríguez and A. Tressaud, Inorg. Chem., 2008, 47, 10288 CrossRef PubMed.
- P. D. Rack, J. J. Peterson, M. D. Potter and W. J. Park, J. Mater. Res., 2001, 16, 1429 CrossRef CAS.
- M. Grinberg, Opt. Mater., 2002, 9, 37 CrossRef.
- I. M. Peter, H. Brian, H. Keith and G. Marek, J. Phys.: Condens. Matter, 1996, 8, 3933 CrossRef.
- G. Blasse, Phys. Lett., 1968, 28, 444 CrossRef CAS.
- G. Blasse, J. Solid State Chem., 1986, 62, 207 CrossRef CAS.
- C. H. Huang and T. M. Chen, J. Phys. Chem. C, 2011, 115, 2349 CAS.
- S. Murakami, M. Herren, D. Rau and M. Morita, Inorg. Chim. Acta, 2000, 300, 1014 CrossRef.
- B. Henderson and G. F. Imbusch, Optical Spectroscopy of Inorganic Solids, Clarendon, Oxford, 1989, p. 151 Search PubMed.
- D. Wang and N. Kodama, J. Solid State Chem., 2009, 182, 2219 CrossRef CAS.
- D. Y. Wang, C. H. Huang, Y. C. Wu and T. M. Chen, J. Mater. Chem., 2011, 21, 10818 RSC.
- G. H. Munoz, C. L. de la Cruz, A. F. Munoz and J. O. Rubio, J. Mater. Sci. Lett., 1988, 7, 1310 CrossRef CAS.
- P. Dorenbos, J. Lumin., 2003, 104, 239 CrossRef CAS.
- Z. G. Xia, R. S. Liu, K. W. Huang and V. Droid, J. Mater. Chem., 2012, 22, 15183 RSC.
- J. Zhou, Z. G. Xia, M. X. Yang and K. Shen, J. Mater. Chem., 2012, 22, 21935 RSC.
|
| This journal is © The Royal Society of Chemistry 2014 |
Click here to see how this site uses Cookies. View our privacy policy here. 


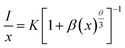
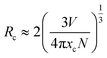
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ1) + A2
exp(−t/τ1) + A2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ2)
exp(−t/τ2)