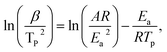Porous ZnO and ZnO–NiO composite nano/microspheres: synthesis, catalytic and biosensor properties†
Ji-Min Yangab,
Wei Zhangb,
Qing Liua and
Wei-Yin Sun*a
aCoordination Chemistry Institute, State Key Laboratory of Coordination Chemistry, School of Chemistry and Chemical Engineering, Nanjing National Laboratory of Microstructures, Nanjing University, Nanjing 210093, China. E-mail: sunwy@nju.edu.cn; Fax: +86 25 83314502
bSchool of Chemistry & Chemical Engineering, Linyi University, Linyi 276005, China
First published on 24th September 2014
Abstract
Porous ZnO and ZnO–NiO composite nano/microspheres have been successfully synthesized by the calcination of non-doped and Ni(II)-doped precursors in air, respectively. The catalytic effect was investigated for porous ZnO and ZnO–NiO composite nano/microspheres for the thermal decomposition of ammonium perchlorate (AP). The ZnO–NiO composite nano/microspheres showed remarkable catalytic effect for the thermal decomposition of AP. The decomposition temperature was decreased by 144.5 °C, and the apparent activation energy was significantly decreased to 117.8 kJ mol−1, which is considerably lower than 159.7 kJ mol−1 for pure AP. The catalytic capacity of ZnO–NiO composite nanostructures is higher than that of most of the materials reported to date. As a result, porous ZnO–NiO composite nano/microstructures could be a promising candidate material for an AP-based propellant. In addition, using the as-prepared porous ZnO microspheres, we have successfully prepared a novel, ultrahigh resolution electrochemical impedance DNA biosensor for the enhanced detection of the PML/RARA fusion gene in acute promyelocytic leukemia with a detection limit of 2.2 × 10−13 mol L−1; therefore, it can act as a biosensing materials for the recognition of DNA hybridization.
Introduction
In the past decade, the fabrication of metal–organic frameworks (MOFs) with various nanostructures have attracted considerable scientific interest, in particular that of the coordination chemists on account of their novel framework structures, topologies and interesting properties.1–5 By the thermal decomposition of MOFs nanostructures, the resulting nanoscale metal oxides, metal oxide/amorphous carbon, or metal/graphene have been obtained.6–9 In the form of novel nanostructures, these materials display functional properties such as effective catalysis, gas storage capacities and antibacterial activities.7,8,10Zinc oxide (ZnO), which is an n-type semiconductor with a wide band gap and a biosafe and biocompatible material,11–13 has attracted a significant research interest due to its unique properties and wide applications in transparent electronics, piezoelectric devices, chemical and biological sensing, and catalytic reactions.14–19 To date, various methods have been used for the preparation of nano/microscale ZnO with diverse shapes including nanorods,20,21 nanowires,22 nanobelts,23 nanorings,24 nanosheets,25 nanospheres26 and tetrapods.27 Such ZnO nanostructures show interesting optoelectronic and catalytic properties. In addition, nickel oxide (NiO) is an important p-type semiconductor, which has been considered to be one of the promising materials in supercapacitors,28,29 gas sensors,30,31 catalysis32 and chemical and biological sensors.33,34
The nanosized composite materials have attracted considerable attention due to their enhanced electronic, magnetic, catalytic, optical, and thermal properties as compared with the noncomposite ones.35 As typical examples, the gas sensing properties of the ZnO–NiO composite for H2S gas detection have been greatly improved,36 CaO–Fe3O4 composite nanospheres show enhanced catalytic performance,37 and CuO–Fe2O3 composite nanoparticles can accelerate the catalytic decomposition of ammonium perchlorate (AP).38 The individual chemical and physical properties of NiO or ZnO nanomaterials have been extensively demonstrated, although there has been less study on the preparation of ZnO–NiO composite nanostructures as a catalyst for the thermal decomposition of AP.
To date, various techniques have been reported for the detection of DNA such as optical, acoustic, gravimetric, and electronic approaches.39 However, there are limitations in these approaches, such as time-consumption, poor precision and high expense. Recently, metal oxide nanostructure-based biosensors for DNA detection exhibit powerful capability to convert the hybridization event into an analytical signal, and these have received considerable attention due to their simplicity, portability, sensitivity, and selectivity.40 Such DNA biosensors contain high specificity to target sequences in the presence of non-complementary strands and show potential for the detection of diseases and genetic disorders as well as environmental and food safety monitoring.16,40,41 The immobilization of DNA on a given matrix is a crucial step for fabricating an electrochemical DNA biosensor. Various matrices such as metal oxide nanomaterials, composite nanomaterials and noble metal nanoparticles have been used as the support for probe immobilization.42–47 Recently, nanosized metal oxides and composite nanomaterials have been used in various biosensor developments because of their high surface area, chemical inertness, thermal stability, non-toxicity and good biocompatibility.16,42–47 For example, a novel electrochemical impedance DNA biosensor for acute promyelocytic leukemia was developed by immobilizing a PML/RARA related 18-mer oligonucleotides ssDNA sequence on FePt-CNTs-GCE or FePt-GO-GCE or carbon ionic liquid electrode modified with nanosized ZnO.16,47 Wang et al. also reported a DNA biosensor based on ZnO nanowires, multiwalled carbon nanotubes (MWCNTs) and gold nanoparticles for the detection of sequence-specific target DNA.48 However, porous ZnO nano/microspheres as biosensing platforms for the detection of DNA hybridization have rarely been reported to date.
In this work, we have successfully synthesized porous ZnO and ZnO–NiO composite nano/microspheres by the calcination of the precursors in air. Compared with the sole ZnO nano/microcrystals, the ZnO–NiO composite nano/microstructures exhibit high catalytic activity for the decomposition of AP. In addition, we present the advantages of incorporating porous ZnO and carbon ionic liquid electrode (CILE) for the sensitive detection of PML/RARA fusion gene in acute promyelocytic leukemia.
Experimental
Materials and chemicals
Zinc(II) nitrate hexahydrate [Zn(NO3)2·6H2O, 99%], nickel(II) nitrate hexahydrate [Ni(NO3)2·6H2O, 98%], ammonium perchlorate (AP, 98%), acetic acid (HAc, 99.5%) N,N-dimethylformamide (DMF, 99.5%) and ethanol (C2H5OH, 99.7%) were obtained from Nanjing Chemical Reagent Co. Ltd., and 2-aminoterephthalic acid (H2N-BDC, 98%) was purchased from Tokyo Chemical Industry Co. Ltd. All chemicals were analytically pure and were used as received without further purification.Synthesis of precursor
Zinc-based coordination polymer particles designated as Zn-CPPs were prepared by modifying the previously reported procedures.49,50 Zn(NO3)2·6H2O (18.2 mg, 0.06 mmol), H2N-BDC (5.4 mg, 0.03 mmol) and 10 μL HAc were dissolved in 30 mL DMF at room temperature. The mixture was transferred into a 43 mL Teflon-lined autoclave, which was sealed and maintained at 120 °C for 4 h. The reaction vessel was then removed from the oven and allowed to cool to room temperature. The products were separated via centrifugation at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 minutes, then 4 mL of DMF was added to a 7 mL centrifuge tube including about 10 mg of the isolated products, and the products were then washed by ultrasonication under ultrasonic irradiation at a frequency of 40 kHz and an ultrasonic power output of 300 W (KH-300DE, Kunshan Hechuang Ultrasonics, China) for 5 minutes and isolated by centrifugation as mentioned above. The DMF washing was repeated for three times. The products were dried in a vacuum at 60 °C for 4 h. The precursor of nickel-doped zinc-based coordination polymer particles designated as Ni(II)-doped Zn-CPPs was synthesized by the same procedure that was used for the preparation of Zn-CPPs, except that Zn(NO3)2·6H2O (9.1 mg, 0.03 mmol) along with Ni(NO3)2·6H2O (8.9 mg, 0.03 mmol) was used. The chemical composition of the prepared Ni(II)-doped Zn-CPPs nano/microcrystals was determined by ICP-OES (inductively coupled plasma optical emission spectrometer).
000 rpm for 5 minutes, then 4 mL of DMF was added to a 7 mL centrifuge tube including about 10 mg of the isolated products, and the products were then washed by ultrasonication under ultrasonic irradiation at a frequency of 40 kHz and an ultrasonic power output of 300 W (KH-300DE, Kunshan Hechuang Ultrasonics, China) for 5 minutes and isolated by centrifugation as mentioned above. The DMF washing was repeated for three times. The products were dried in a vacuum at 60 °C for 4 h. The precursor of nickel-doped zinc-based coordination polymer particles designated as Ni(II)-doped Zn-CPPs was synthesized by the same procedure that was used for the preparation of Zn-CPPs, except that Zn(NO3)2·6H2O (9.1 mg, 0.03 mmol) along with Ni(NO3)2·6H2O (8.9 mg, 0.03 mmol) was used. The chemical composition of the prepared Ni(II)-doped Zn-CPPs nano/microcrystals was determined by ICP-OES (inductively coupled plasma optical emission spectrometer).
Synthesis of nanocrystals
The as-obtained precursors were calcined in air at 500 °C with a heating rate of 2 °C min−1 (DRZ-3D, Longkou Electric Cooker Factory, China) to produce ZnO and ZnO–NiO composite nano/microspheres, respectively. The as-obtained ZnO–NiO composite nanocrystals comprise about 25% Ni(II) and 75% Zn(II) (ICP-OES).Immobilization and hybridization of DNA
CILE, ZnO/CILE, and immobilization and hybridization of the DNA probe were prepared by modifying the previously reported procedures (see ESI†).16,45Characterization
The products were characterized by powder X-ray diffraction (PXRD) on a Bruker D8 Advance X-ray diffractometer with Cu Kα radiation (λ = 1.5418 Å) at room temperature. Field emission scanning electron microscopy (SEM) images and energy dispersive spectrometry (EDS) of the products were obtained on a field emission scanning electron micro-analyser (Hitachi S-4800), employing an accelerating voltage of 5 or 20 kV. Transmission electron microscopy (TEM) images were obtained on a JEM-2100 high resolution transmission microscope, employing an accelerating voltage of 200 kV. Inductively coupled plasma optical emission spectrometer (ICP-OES) was measured on Optima 5300DV. FT-IR spectra were recorded in the range of 400–4000 cm−1 on a Bruker Vector22 FT-IR spectrophotometer using KBr pellets. Nitrogen sorption experiments at 77 K were carried out on a Belsorp-max volumetric gas sorption instrument. Surface areas were determined by the BET equation. Ultrahigh purity N2 was used in the adsorption study. Prior to the measurement of nitrogen sorption, the samples were activated under vacuum at 100 °C for about 10 h.Catalytic measurements
To test the catalytic effect of ZnO and ZnO–NiO composite nano/microcrystals on the thermal decomposition of AP, a mixture of AP and catalytic particles was carefully ground for 10 min and was detected by a differential scanning calorimeter (DSC) and thermogravimetric analysis (TGA) on a Mettler-Toledo thermal analyzer under the nitrogen atmosphere in the temperature range from 30 to 500 °C with the different heating rate of 5, 10, 15 and 20 °C min−1, respectively. The mass percentage of catalytic particles to AP in the mixture was 2%.Electrochemical measurements
Electrochemical impedance spectroscopy (EIS) measurements of 1.0 mmol L−1 K3Fe(CN)6 and 1.0 mmol L−1 K4Fe(CN)6 were performed on an electrochemical analyzer (EC500, Wuhan Gaoss Union Company, China) with 0.1 mol L−1 KCl as the supporting electrolyte. A conventional three-electrode system was used with a modified CILE as the working electrode, a saturated calomel electrode as the reference electrode and a Pt wire as the counter electrode. The AC voltage frequencies ranged from 10 kHz to 0.1 Hz.Results and discussion
The simple route for the fabrication of ZnO–NiO composite nanostructures is shown in Scheme 1. The MOF precursor was readily obtained by mixing Zn(NO3)2·6H2O, Ni(NO3)2·6H2O, H2N-BDC and HAc in DMF. Fig. 1 shows typical field emission SEM images of the precursors. Without the addition of Ni(NO3)2·6H2O, the results shown in Fig. 1a and b indicate that Zn-CPPs is composed of uniform microspheres with diameters of about 600 nm. Further observation reveals that the Ni(II) doping results in significant change in the size of the precursor microspheres. The sizes of Ni(II)-doped particle microspheres are obviously smaller than those of the Zn-CPPs, which may be related to the effect of Ni(II) doping on the precursor structure. The average diameter of the Ni(II)-doped Zn-CPPs is around 300 nm (Fig. 1c and d). Fig. S1 (ESI†) shows the PXRD patterns of the precursors. Both of the as-obtained products are amorphous. The FT-IR spectral data and EDS analysis are shown in Fig. S2 and S3 (ESI†), respectively. The characteristic bands around 1581 and 1394 cm−1 are the stretching vibrations of νas(–COO−) and νs(–COO−) of the carboxylate in H2N-BDC2−, and the peaks of Ni, Zn, C, N and O are easily found in the EDS, indicating that the precursors might be amorphous coordination polymer nano/microstructures.The ZnO and ZnO–NiO composite nano/microspheres were prepared by the calcination of the corresponding precursor at 500 °C in air. The PXRD patterns of the calcinated products are presented in Fig. 2. In pure ZnO (Fig. 2a), all the peaks are in good accordance with the standard spectrum of ZnO (JCPDS 36-1451), which means that the precursor was completely transformed into the ZnO nano/microspheres. In ZnO–NiO composites (Fig. 2b), several weak additional diffraction peaks appeared, which correspond to NiO (JCPDS 47-1049). From the PXRD patterns, it is clear that the precursors have been completely converted to ZnO–NiO composites. Fig. 3 shows the morphology of the as-obtained products, and it can be clearly seen that the original shape has been maintained after calcination at 500 °C for 1 h in air. The average diameter of ZnO and ZnO–NiO composite nano/microspheres are around 300 and 100 nm, respectively (Fig. 3a–d). Fig. 4a shows a representative TEM image of the ZnO–NiO composite nanospheres. From high-magnification TEM images (Fig. 4b), it can be clearly seen that a large number of cracks and pores exist on the surface of the as-obtained products. The high resolution TEM images are depicted in Fig. 4c and d. The interplanar distances of 0.210 nm and 0.247 nm are in good agreement with the (200) plane of cubic NiO and (101) plane of the hexagonal structure of ZnO, respectively. Fig. 5 shows the EDS mappings of ZnO–NiO composites. The SEM images correspond to the images of the EDS mapping of Ni(II) and Zn(II). The distributions of Ni(II) are consistent with those of Zn(II) in the mapping, suggesting that the Ni(II) elements are well dispersed in the ZnO crystals. To investigate the porous structure of ZnO and ZnO–NiO composite nano/microspheres, nitrogen sorption measurements were performed. Nitrogen adsorption–desorption isotherms of the porous ZnO and ZnO–NiO composite nano/microstructures are shown in Fig. S4 (ESI†), and Fig. S5 (ESI†) exhibits the corresponding Barrett–Joyner–Halenda (BJH) pore size distribution plots, indicating that all of the products contain mesopores. The BET surface areas and pore volumes of the products are 17.62 m2 g−1 and 63.87 mm3 g−1 and 18.30 m2 g−1 and 190.01 mm3 g−1 for ZnO and ZnO–NiO nano/microspheres, respectively. The results are consistent with the observation of mesopores in the HRTEM image (Fig. 4d).
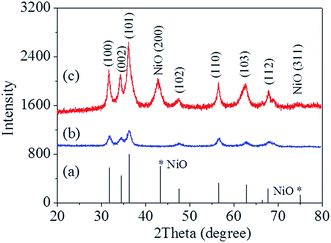 | ||
| Fig. 2 Standard spectrum of ZnO and NiO (labeled with *) (a); PXRD patterns of (b) ZnO and (c) ZnO–NiO composite nano/microspheres. | ||
 | ||
| Fig. 5 EDS mapping of ZnO–NiO composites. SEM images (left) and the corresponding elemental distributions of Ni(II) (middle) and Zn(II) (right). | ||
AP is the key component of composite solid propellants because its thermal decomposition characteristics directly influence the combustion behavior of the propellant. More specifically, the performance of solid propellants is closely associated with the reaction rate, activation energy and thermal decomposition temperature of AP. Generally speaking, the lower is the pyrolysis temperature, the higher is the combustion rate.51,52 Some important factors such as the type of the catalysts and the heating rate are crucial to the final decomposition temperature. Herein, we investigated the influence of the catalytic activity of ZnO and ZnO–NiO composite nano/microspheres and the heating rate on the thermal decomposition of AP.
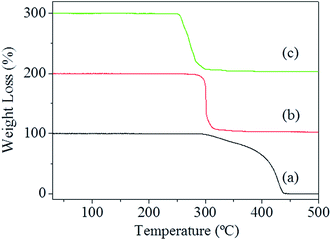
Fig. 6 shows the DSC curves of both pure AP and the mixtures of AP with the catalytic particles at a 2% mass basis with a heating rate of 10 °C min−1. From Fig. 6a, it can be seen that the first endothermic peak at 240–250 °C is due to the crystal transformation of AP from orthorhombic to cubic phase, the second exothermic peak at 310–340 °C is ascribed to the partial decomposition of AP, and the third exothermic peak around 420–440 °C corresponds to the thermal decomposition of AP.7,19,52 When porous ZnO and ZnO–NiO composite particles were added to AP, all the samples have similar endothermic peaks at around 242.8 °C, indicating that the particles have little effect on the crystallographic transition temperature of AP. However, in the relatively high temperature region, the samples containing catalytic particles have dramatic changes in the exothermic peaks of AP decomposition. When the as-obtained catalytic particles are added to AP, the original exothermic peak of pure AP at 432.3 °C disappeared and fused into one exothermic peak. The exothermic peak temperature was 302.2 °C and 287.8 °C for pure ZnO and ZnO–NiO composites, respectively. The exothermic temperature decreases by about 130.1 °C and 144.5 °C, respectively, for the above two samples compared to the pure AP. The catalytic capacity of ZnO–NiO composite nanostructures is higher than that of the most materials reported to date.7,19,38,52 Furthermore, the TGA curves of pure AP and mixture of AP with the catalytic particles are shown in Fig. 7. In the range from 30 to 500 °C, two weight loss steps are observed for pure AP (Fig. 7a), whereas for the mixture of AP with the catalytic particles (Fig. 7b and c) only one weight loss step is observed, which is in agreement with the DSC results.
To further study the catalytic effect of samples on the thermal decomposition kinetic parameters of AP, both pure AP and the mixtures of AP with ZnO or ZnO–NiO composite nano/microspheres were investigated by TGA and DSC with different heating rates from 5 to 20 °C min−1, and the results are shown in Fig. S6 and S7 (ESI†). The decomposition temperature of AP with the samples containing catalytic particles is dependent on the heating rate, and a slight increase of the exothermic temperature was accompanied with the increase of heating rate. Apparent activation energy (Ea) was calculated from the exothermic peak temperature dependence on the heating rate using the Kissinger correlation:
 | ||
| Fig. 8 Dependence of ln(β/Tp2) on 1/(RTp) for AP and the mixtures of AP with different additives. Scattered points are experimental data and lines denote the linear fitting results. | ||
EIS with [Fe(CN)6]3−/4− as indicator has been extensively employed as an effective tool for the electrochemical detection of DNA hybridization.57 In the EIS, the semicircle part corresponds to electron-transfer limited process and its diameter is equal to the electron transfer resistance Ret, which controls the electron transfer kinetics of the redox probe at the electrode interface.58 The selectivity of the DNA biosensor was investigated by using the ssDNA probe to hybridize with different DNA sequences. After the hybridization of the ssDNA probe, the changes of the Nyquist diagram of 1.0 mmol L−1 [Fe(CN)6]3−/4− are shown in Fig. 9. The curve a is the Nyquist diagram at the probe ssDNA/ZnO/CILE. After the ssDNA probe was hybridized with the cDNA, the change of the Nyquist diagram is shown in curve b. There is a significant increase in the Ret value, indicating that hybrids (dsDNA) were formed at the electrode. The increase of the Ret value was very little after the hybridization of the ssDNA probe with the ncDNA (curve c), suggesting that the hybridization reaction was not accomplished. When the ssDNA probe was hybridized with the single-base mismatched DNA (curve d) or double-base mismatched DNA (curve e), the Ret value also increased, which was considerably smaller than that obtained from the hybridization with the cDNA (curve b); therefore, the single-base mismatched and double-base mismatched DNA could also be sensed via comparing the change of the Ret value. The results demonstrated that this DNA biosensor exhibited fine selectivity for the hybridization detection. However, ZnO–NiO composite nanoparticles are not used as the support for probe immobilization because NiO phytotoxicity leads to DNA damage.
The difference between the Ret values of 1.0 mmol L−1 [Fe(CN)6]3−/4− at the ssDNA/ZnO/CPE and that at the hybridization modified electrode (dsDNA/ZnO/CILE) was labelled as ΔRet, which was used as the detection signal for the quantitative analysis of the PML/RARA fusion gene target sequence. The concentration of the PML/RARA fusion gene target sequence in the hybridization solution was varied from 1.0 × 10−12 to 1.0 × 10−8 mol L−1, and the Nyquist diagrams are shown in Fig. 10. The ΔRet value between before and after hybridization was linear with the logarithm of concentrations of the PML/RARA fusion gene target sequence from 1.0 × 10−12 to 1.0 × 10−8 mol L−1, and a detection limit of 2.2 × 10−13 mol L−1 was estimated using 3σ rule (where σ was the relative standard deviation of 11 parallel measurements of the blank solution). The results indicate that the label-free strategy based on this ZnO/CILE platform exhibited high sensitivity for the electrochemical detection of DNA hybridization.
Conclusion
In this work, porous ZnO and ZnO–NiO composite nano/microspheres have been synthesized by a simple precursor method. Further investigations show that the as-obtained ZnO and ZnO–NiO composite nano/microspheres have good abilities to decrease the decomposition temperature of AP, which make them useful for applications in AP-based composite solid rocket propellants. In addition, EIS measurements demonstrate that the DNA biosensor based on porous ZnO functionalized CILE has a wide detection range with a low detection limit for DNA hybridization. The developed DNA biosensors are expected to have further application in the detection of the PML-RARA fusion gene in real samples in future work.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant no. 21331002 and 21205057).References
- N. L. Rosi, J. Eckert, M. Eddaoudi, D. T. Vodak, J. Kim, M. O'Keeffe and O. M. Yaghi, Science, 2003, 300, 1127 CrossRef CAS PubMed.
- T. K. Maji, R. Matsuda and S. Kitagawa, Nat. Mater., 2007, 6, 142 CrossRef CAS PubMed.
- H. Guo, Y. Zhu, S. Wang, S. Su, L. Zhou and H. Zhang, Chem. Mater., 2012, 24, 444 CrossRef CAS.
- K. M. L. Taylor-Pashow, J. D. Rocca, Z. G. Xie, S. Tran and W. B. Lin, J. Am. Chem. Soc., 2009, 131, 14261 CrossRef CAS PubMed.
- D. Zacher, O. Shekhah, C. Wöll and R. A. Fischer, Chem. Soc. Rev., 2009, 38, 1418 RSC.
- F. Gao, H. Pang, S. P. Xu and Q. Y. Lu, Chem. Commun., 2009, 3571 RSC.
- L. N. Jin, Q. Liu and W. Y. Sun, CrystEngComm, 2012, 14, 7721 RSC.
- S. J. Yang, T. Kim, J. H. Im, Y. S. Kim, K. Lee, H. Jung and C. R. Park, Chem. Mater., 2012, 24, 464 CrossRef CAS.
- Z. W. Xu, H. J. Li, W. Li, G. X. Cao, Q. L. Zhang, K. Z. Li, Q. G. Fu and J. Wang, Chem. Commun., 2011, 47, 1166 RSC.
- D. Du, M. H. Wang, Y. H. Qin and Y. H. Lin, J. Mater. Chem., 2010, 20, 1532 RSC.
- A. McLaren, T. Valdes-Solis, G. Q. Li and S. C. Tsang, J. Am. Chem. Soc., 2009, 131, 12540 CrossRef CAS PubMed.
- D. J. Gargas, M. E. Toimil-Molares and P. D. Yang, J. Am. Chem. Soc., 2009, 131, 2125 CrossRef CAS PubMed.
- K. Sun, W. Wei, Y. Ding, Y. Jing, Z. L. Wang and D. L. Wang, Chem. Commun., 2011, 47, 7776 RSC.
- X. D. Wang, J. Zhou, J. H. Song, J. Liu, N. S. Xu and Z. L. Wang, Nano Lett., 2006, 6, 2768 CrossRef CAS PubMed.
- Z. L. Wang and J. H. Song, Science, 2006, 312, 242 CrossRef CAS PubMed.
- W. Zhang, X. W. Zheng and K. Jiao, Sens. Actuators, B, 2012, 162, 396 CrossRef CAS PubMed.
- M. Ahmad, C. F. Pan, Z. X. Luo and J. Zhu, J. Phys. Chem. C, 2010, 114, 9308 CAS.
- G. Tang, Y. Wen, A. Pang, D. Zeng, Y. Zhang, S. Tian, B. Shan and C. Xie, CrystEngComm, 2014, 16, 570 RSC.
- J. Yin, F. Gao, C. Wei and Q. Lu, Sci. Rep., 2014, 4, 1 Search PubMed.
- Z. J. Gu, M. P. Paranthaman, J. Xu and Z. W. Pan, ACS Nano, 2009, 3, 273 CrossRef CAS PubMed.
- M. H. Kumar, N. Yantara, S. Dharani, M. Graetzel, S. Mhaisalkar, P. P. Boix and N. Mathews, Chem. Commun., 2013, 49, 11089 RSC.
- S. Xu, Y. Ding, Y. Wei, H. Fang, Y. Shen, A. K. Sood, D. L. Polla and Z. L. Wang, J. Am. Chem. Soc., 2009, 131, 6670 CrossRef CAS PubMed.
- P. X. Gao, Y. Ding, W. Mai, W. L. Hughes, C. Lao and Z. L. Wang, Science, 2005, 309, 1700 CrossRef CAS PubMed.
- X. Y. Kong, Y. Ding, R. Yang and Z. L. Wang, Science, 2004, 303, 1348 CrossRef CAS PubMed.
- E. S. Jang, J. H. Won, Y. W. Kim, X. Chen and J. H. Choy, CrystEngComm, 2010, 12, 3467 RSC.
- C. Zhu, B. Lu, Q. Su, E. Xie and W. Lan, Nanoscale, 2012, 4, 3060 RSC.
- H. Q. Yan, R. R. He, J. Pham and P. D. Yang, Adv. Mater., 2003, 15, 402 CrossRef CAS.
- G. Q. Zhang, L. Yu, H. E. Hostera and X. W. Lou, Nanoscale, 2013, 5, 877 RSC.
- S. Ding, T. Zhu, J. S. Chen, Z. Wang, C. Yuan and X. W. Lou, J. Mater. Chem., 2011, 21, 6602 RSC.
- J. Wang, L. M. Wei, L. Y. Zhang, J. Zhang, H. Wei, C. H. Jiang and Y. F. Zhang, J. Mater. Chem., 2012, 22, 20038 RSC.
- Y. Ren, W. K. Chim, S. Y. Chiam, J. Q. Huang, C. Pi and J. S. Pan, Adv. Funct. Mater., 2010, 20, 3336 CrossRef CAS.
- J. Wang, C. Wei, H. Pang, F. Gao, J. Yin, L. Guan and Q. Y. Lu, Catal. Commun., 2011, 12, 1031 CrossRef CAS PubMed.
- X. Song, L. Gao and S. Mathur, J. Phys. Chem. C, 2011, 115, 21730 CAS.
- X. F. Chu, X. Zhu, Y. Dong, T. Chen, M. Ye and W. Sun, J. Electroanal. Chem., 2012, 676, 20 CrossRef CAS PubMed.
- N. G. Cho, H. S. Woo, J. H. Lee and I. D. Kim, Chem. Commun., 2011, 47, 11300 RSC.
- L. Xu, R. Zheng, S. Liu, J. Song, J. Chen, B. Dong and H. W. Song, Inorg. Chem., 2012, 51, 7733 CrossRef CAS PubMed.
- X. Ma, X. Feng, J. Guo, H. Cao, X. Suo, H. Sun and M. Zheng, Appl. Catal., B, 2014, 147, 666 CrossRef CAS PubMed.
- H. Liu, Q. Jiao, Y. Zhao, H. Li, C. Sun, X. Li and H. Wu, Mater. Lett., 2010, 64, 1698 CrossRef CAS PubMed.
- D. K. Prusty and A. Herrmann, J. Am. Chem. Soc., 2010, 132, 12197 CrossRef CAS PubMed.
- Y. B. Hahn, R. Ahmadwa and N. Tripathy, Chem. Commun., 2012, 48, 10369 RSC.
- A. Sassolas, B. D. Leca-Bouvier and L. J. Blum, Chem. Rev., 2008, 108, 109 CrossRef CAS PubMed.
- A. Kaushik, P. R. Solanki, A. A. Ansari, B. D. Malhotra and S. Ahmad, Biochem. Eng. J., 2009, 46, 132 CrossRef CAS PubMed.
- S. Zuo, L. Zhang, H. H. Yuan, M. B. Lan, G. A. Lawrance and G. Wei, Bioelectrochemistry, 2009, 74, 223 CrossRef CAS PubMed.
- H. Zhu, J. Wang and G. Xu, Cryst. Growth Des., 2009, 9, 633 CAS.
- W. Zhang, Sens. Actuators, B, 2013, 176, 386 CrossRef CAS PubMed.
- F. Li, Y. Feng, P. J. Dong and B. Tang, Biosens. Bioelectron., 2010, 25, 2084 CrossRef CAS PubMed.
- L. B. Wang, X. W. Zheng, W. Zhang, X. Quan, Q. Hu, W. Wu, P. Zong and M. Wu, RSC Adv., 2013, 3, 9042 RSC.
- J. Wang, S. Li and Y. Zhang, Electrochim. Acta, 2010, 55, 4436 CrossRef CAS PubMed.
- D. Britt, C. Lee, F. J. Uribe-Romo, H. Furukawa and O. M. Yaghi, Inorg. Chem., 2010, 49, 6387 CrossRef CAS PubMed.
- A. R. Millward and O. M. Yaghi, J. Am. Chem. Soc., 2005, 127, 17998 CrossRef CAS PubMed.
- R. P. Fitzgerald and M. Q. Brewster, Combust. Flame, 2004, 3, 313 CrossRef PubMed.
- N. Li, Z. Geng, M. Cao, L. Ren, X. Zhao, B. Liu, Y. Tian and C. W. Hu, Carbon, 2013, 54, 124 CrossRef CAS PubMed.
- Z. Zhou, S. Tian, D. Zeng, G. Tang and C. Xie, J. Alloys Compd., 2012, 513, 213 CrossRef CAS PubMed.
- N. Li, M. H. Cao, Q. Y. Wu and C. W. Hu, CrystEngComm, 2012, 14, 428 RSC.
- J. Lu, J. Zhu, Z. S. Wang, J. L. Cao and X. F. Zhou, Ceram. Int., 2014, 40, 1489 CrossRef CAS PubMed.
- B. Varghese, M. Reddy, Y. Zhu, C. Lit, T. C. Hoong, G. V. S. Rao, B. V. R. Chowdari, A. Wee, C. Lim and C. Sow, Chem. Mater., 2008, 20, 3360 CrossRef CAS.
- S. Mohan, P. Nigam, S. Kundu and R. Prakash, Analyst, 2010, 135, 2887 RSC.
- A. Kaushik, P. R. Solanki, A. A. Ansari, B. D. Malhotra and S. Ahmad, Biochem. Eng. J., 2009, 46, 132 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Immobilization and hybridization of DNA, XRD, FT-IR, EDS, sorption isotherms for N2, pore size distribution, DSC and TGA data. See DOI: 10.1039/c4ra08736d |
| This journal is © The Royal Society of Chemistry 2014 |