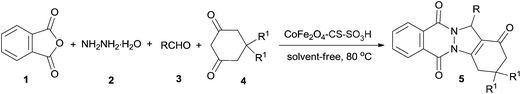
A highly efficient and recyclable cobalt ferrite chitosan sulfonic acid magnetic nanoparticle for one-pot, four-component synthesis of 2H-indazolo[2,1-b]phthalazine-triones†
Xiao-Na Zhao,
Guo-Fei Hu,
Meng Tang,
Tai-Tai Shi,
Xiao-Li Guo,
Tao-Tao Li and
Zhan-Hui Zhang*
College of Chemistry and Material Science, Hebei Normal University, Shijiazhuang 050024, China. E-mail: zhanhui@126.com; Fax: +86 31180787431
First published on 2nd October 2014
Abstract
A highly efficient magnetic CoFe2O4 chitosan sulfonic acid nanoparticle (CoFe2O4-CS-SO3H) was prepared and applied for one-pot, four-component synthesis of 2H-indazolo[2,1-b]phthalazine-triones by condensation of phthalic anhydride, hydrazinium hydroxide, 1,3-cyclohexanedione and aldehydes under solvent-free conditions at 80 °C. The magnetic nanocatalyst could be readily recovered by applying an external magnet and recycled several times without significant loss of its catalytic activity.
Introduction
Heterogeneous catalysts have gained much importance in recent years due to environmental and economic considerations. These heterogeneous catalysts are advantageous over homogeneous catalysts as they can be recovered from the reaction mixture through filtration or centrifugation and reused after activation, thereby making the process economically viable.1 Compared to other supports such as silica, alumina, activated carbon, metal oxides, zeolites, clays, polymers, magnetic nanoparticles (MNPs) are arguably the most extensively investigated and emerged as excellent and ideal supports with significant industrial potential due to their extraordinary properties such as large specific surface area, readily dispersed in reaction solution, and easy functionalization with various groups.2 Their super-paramagnetic character makes them be effective and easily recovered from the reaction system using an external permanent magnet, which eliminates the necessity of tedious filtration, centrifugation or membrane separation steps and enhances the product purity. Thus, a wide range of the magnetically recyclable nanocatalysts with excellent catalytic activities have been developed and applied in versatile organic reactions.3 Among the various MNPs as the core magnetic supports, cobalt ferrite is one of the most versatile magnetic materials as it has moderate saturation magnetization, high chemical stability and mechanical strength and can be prepared by simple methods.4A wide range of organic reactions depend on the use of protic acids as catalysts. However, difficulties in the separation of the catalyst from the product and generation of enormous quantities of waste have been limited the applications of these liquid acids. There are many reports on the preparation and applications of solid-supported acid.5 Recently, chitosan (CS) as one kind of the widely used bio-based polymers has been attarcting more and more attention.6 It is natural, non-toxic, biodegradable, hydrophilic and reproducible molecule, and widely used in many industrial and manufacturing processes. Chitosan has also been used as effective catalyst in organic synthesis, however, it can not be separated easily from water due to its low density.7 It is the alkaline deacetylated product of chitin and comprises a great quantity of hydroxyl and amino groups, which make it be attached on magnetic nanoparticles ions through chelate mechanism without using any linkers.8 Thus, combining the attractive properties of magnetic CoFe2O4 and chitosan, the development of novel, highly active and reusable immobilized catalysts by further functionalization is therefore an interesting challenge.
Due to growing concerns over the adverse influence of organic solvents on the environment, solvent-free organic reactions have also attracted tremendous attention. Meanwhile, the construction of structurally diverse and complex molecular from simple and readily available starting materials while combining economic aspects with environmental ones is especially important in modern synthetic organic and medicinal chemistry. Multicomponent reactions (MCRs) involving domino processes have emerged as powerful tools to reach this near ideal goal. Such transformations reduce the consumption of catalyst, solvent, time, labor, and energy, thereby minimizing waste compared to the corresponding series of individual reactions.9 One of these MCRs is the preparation of indazolo[2,1-b]phthalazine-trione derivatives. These compounds have been shown to possess a broad spectrum of biological activities.10 They have proved to be promising luminescence materials and fluorescence probes.11 The synthesis of indazolo[2,1-b]phthalazine-triones can be carried out by three-component condensation of phthalhydrazide, dimedone, and aromatic aldehydes12–18 or four-component reaction of phthalic anhydride, hydrazinium hydroxide, dimedone and aromatic aldehydes in the presence of various catalysts such as sulfuric acid-modified PEG-6000 (PEG-OSO3H),19 Ce(SO4)2·4H2O,20 ZnO nanoparticles,21 cellulose–SO3H,22 starch sulfate,23 ionic liquid N,N,N,N-tetramethylguanidinium acetate [TMG][Ac],24 1-butyl-3-methylimidazolium bromide ([Bmim]Br),25 and 2-pyrrolidonium hydrogensulfate ([Hnhp][HSO4]).26 Despite being effective, some of these methods involve the use of transition metal as catalyst, extended reaction times, unsatisfactory yields of the products, or require additional instruments such as ultra sound. Therefore, the development of novel, more efficient, cheaper, and more easily recovered catalysts for this four-component reaction remains highly desirable.
Considering the above subjects and in continuation of our efforts toward the design of magnetic nanocatalysts27 and sustainable synthesis development,28 we wish to report here a new type of magnetic CoFe2O4 chitosan sulfonic acid nanoparticle (CoFe2O4-CS-SO3H) as a powerful catalyst for the green and efficient synthesis of indazolo[2,1-b]phthalazine-triones by one-pot, four-component reaction of phthalic anhydride, hydrazinium hydroxide, 1,3-cyclohexanedione and aldehydes (Scheme 1).
Results and discussion
Preparation and characterization of CoFe2O4-CS-SO3H
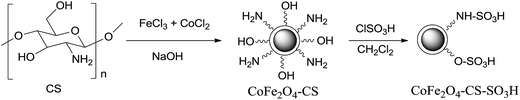
The magnetic CoFe2O4 chitosan sulfonic acid nanoparticle (CoFe2O4-CS-SO3H) was prepared by two steps as presented in Scheme 2. The first step is the preparation of magnetic CoFe2O4-chitosan nanoparticles by one-pot synthesis method. Chitosan was firstly dissolved in 3% acetic acid solution, then aqueous solution of FeCl3·6H2O and CoCl2·6H2O was added. The mixture was stirred strongly for 2 h, and then NaOH solution was added. The formed CoFe2O4 has weak interaction with amino and hydroxyl groups in chitosan. Then, the CoFe2O4-CS served as supported for the immobilization of SO3H group by the reaction of amino and hydroxyl groups with chlorosulfonic acid in CH2Cl2 led to sulfonic acid-functionalized magnetic CoFe2O4 nanoparticles (CoFe2O4-CS-SO3H). It was found that the content of sulfonic acid was 0.50 mmol g−1 determined by acid–base titration. Also, the pH of this solid acid (10%, w/v) was measured using pH meter at 25 °C. At first 0.5 g solid sulfonic acid was dispersed in 5 mL distilled water by ultrasonic bath for 1 h, then measured and found to be 1.25.XRD analysis
The XRD pattern of the sample is shown in Fig. 1. As seen in Fig. 1, CoFe2O4-CS-SO3H show typical characteristic peaks at the 2θ values of 18.2, 29.0, 35.5, 43.6, 57.7 and 61.7 with the corresponding reflection of (111), (220), (311), (400), (511) and (440) crystal planes. The observed diffraction peaks agree with the cubic structure of CoFe2O4 (JCPDS 221086),4 suggesting that the coating process did not result in the phase change of CoFe2O4.TEM and EDX elemental analysis
The TEM images of the synthesized CoFe2O4-CS-SO3H are shown in Fig. 2. The images confirmed the formation of single-phase CoFe2O4 nanoparticles, with spherical morphology. The average nanoparticle diameter of CoFe2O4-CS-SO3H was estimated to be ca. 50–60 nm based on the TEM image, which is also in accordance with the result calculated by the Scherrer formula. The presence of C, O, Fe, Co and S atoms was observed in the EDX spectrum (Fig. 3).FT-IR analysis
Fig. 4 shows the Fourier transform infrared (FTIR) spectra of CoFe2O4 (a), CoFe2O4-CS (b) and CoFe2O4-CS-SO3H (c). The Co–O and Fe–O stretching vibration near 593 cm−1 was observed in Fig. 4(a)–(c). The significant features observed for Fig. 4(b) are the appearance of the peaks at 1074, 1300, 1606, 2891, 3427 cm−1 corresponding to C–OH, C–N, N–H, C–H, and OH stretching models of the chitosan molecules. The three new bands appeared in Fig. 4(c) at 650, 1090 and 1160 cm−1 corresponding to the O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S
S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O asymmetric and symmetric stretching vibrations and S–O stretching vibration of the sulfonic acid groups.
O asymmetric and symmetric stretching vibrations and S–O stretching vibration of the sulfonic acid groups.
The magnetic properties
The magnetic properties of the samples CoFe2O4 and CoFe2O4-CS-SO3H were measured at room temperature using a vibrating sample magnetometry (VSM). As shown in Fig. 5, the value of magnetic saturation (Msat) for CoFe2O4-CS-SO3H is about 41.5 emu g−1, which is lower than that of bare CoFe2O4 nanoparticles (ca. 49.6 emu g−1). The reason should be ascribed to the existence of CS-SO3H hybrid material shells on CoFe2O4 cores. Even with this reduction in the saturation magnetization, the prepared catalyst could still be efficiently separated with the help of an external magnetic force.Catalytic activity of CoFe2O4-CS-SO3H
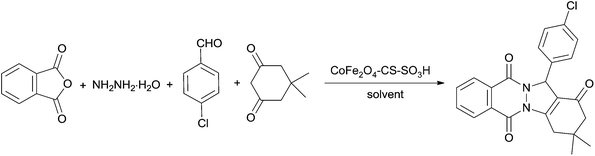
In order to evaluate the catalytic reactivity of, CoFe2O4-CS-SO3H one-pot four-component reaction of phthalic anhydride, hydrazinium hydroxide, 5,5-dimethyl 1,3-cyclohexanedione and 4-chlorobenzaldehyde was chosen the model reaction for our investigation, and the results are displayed in Table 1. In the absence of the catalyst, only a trace amount of the desired product was observed even after 1 h (Table 1, entry 1), which demonstrated that catalyst plays an important role in this reaction. Several reaction conditions were tested in order to identify the optimized conditions. Firstly, the influence of different solvents on the reaction was examined, and a good yield was obtained with EtOH (Table 1, entry 5). Toluene, CH3CN, AcOEt, H2O, PEG 400, glycerol, [bmin]BF4, and [bmin]PF6 were found to be inferior. To our delight, when the reaction was carried out under solvent-free condition at 80 °C, the yield could be improved to 95%. Other experimental parameters such as temperature and amount of catalyst were also optimized. It was observed that only 5% yield of product was obtained at room temperature. The yield of the desired product was greatly improved with the increasing of temperature. The best yield was observed when the reaction was performed at 80 °C. Increasing or decreasing the temperature from 80 °C led to a decrease in the yield (Table 1, entries 13 and 15). With respect to the catalyst loading, 0.5 mol% (10 mg) was found to be optimal. When the loading of catalyst was lowered to 0.4 mol%, the reaction would proceed incompletely (Table 1, entry 17). Any further addition of catalyst had no positive effect on the overall yield of product. Therefore, on the basis of the above experiments, the most suitable reaction conditions for this one-pot process included CoFe2O4-CS-SO3H (0.5 mol%, 10 mg) as the catalyst under solvent-free conditions at 80 °C (Table 1, entry 14).| Entry | Catalyst (mol%) | Solvent | Temp. (°C) | Time (min) | Yieldb (%) |
|---|---|---|---|---|---|
| a Experimental conditions: phthalic anhydride (1 mmol), hydrazinium hydroxide (1.2 mmol), 4-chlorobenzaldehyde (1 mmol) and 5,5-dimethyl 1,3-cyclohexanedione (1 mmol), solvent (5 mL).b Isolated yields. | |||||
| 1 | No | No | 80 | 60 | Trace |
| 2 | 0.5 | Toluene | 80 | 10 | 7 |
| 3 | 0.5 | CH3CN | Reflux | 10 | 33 |
| 4 | 0.5 | AcOEt | Reflux | 10 | 29 |
| 5 | 0.5 | EtOH | Reflux | 10 | 52 |
| 6 | 0.5 | H2O | 80 | 10 | 10 |
| 7 | 0.5 | PEG 400 | 80 | 10 | 18 |
| 8 | 0.5 | Glycerol | 80 | 10 | 21 |
| 9 | 0.5 | [bmin]BF4 | 80 | 10 | 31 |
| 10 | 0.5 | [bmin]PF6 | 80 | 10 | 19 |
| 11 | 0.5 | No | 25 | 10 | 6 |
| 12 | 0.5 | No | 50 | 6 | 77 |
| 13 | 0.5 | No | 60 | 6 | 82 |
| 14 | 0.5 | No | 80 | 6 | 95 |
| 15 | 0.5 | No | 100 | 6 | 90 |
| 16 | 0.25 | No | 80 | 6 | 30 |
| 17 | 0.4 | No | 80 | 6 | 60 |
| 18 | 1.0 | No | 80 | 6 | 95 |
After optimizing of the reaction conditions, the generality and scope of the reaction were investigated. As illustrated in Table 2, a variety of aromatic aldehydes bearing electron-rich and electron-poor groups at either ortho-, meta- or para-positions of the aromatic ring were smoothly converted into 2H-indazolo[2,1-b]phthalazine-triones in high to excellent yields. A wide range of synthetically useful functional groups including thioether, halide and trifluoromethyl groups remained intact during the present reaction conditions. In addition, the presence of three methoxy electron donating groups on the aromatic ring of the aldehyde performed well and afforded the desired product in 90% yield. Also, heterocyclic aromatic aldehyde such as picolinaldehyde also underwent efficient conversion into the targeted product in 86% yield. It should be noted that aliphatic aldehyde such as cyclohexanecarbaldehyde was also compatible under these reaction conditions and was successfully transformed into the desired products in 83% yield (entry 17). Replacement of 5,5-dimethyl 1,3-cyclohexanedione with 1,3-cyclohexanedione under identical conditions also produced the expected products in high yields (Table 2, entries 18–22).
| Entry | Aldehydes | R | Product | Time (min) | Yieldb (%) | MP (°C) |
|---|---|---|---|---|---|---|
| a Reaction conditions: phthalic anhydride (1 mmol), hydrazinium hydroxide (1.1 mmol), aldehydes (1 mmol), CoFe2O4@CS-SO3H (0.01 g) 80 °C.b Isolated yield. | ||||||
| 1 | PhCHO | Me | 5a | 7 | 90 | 209–210 (208–209)18 |
| 2 | 4-OMeC6H4CHO | Me | 5b | 6 | 92 | 219–220 (218–209)18 |
| 3 | 4-O(CH2)2MeC6H4CHO | Me | 5c | 7 | 91 | 159–160 |
| 4 | 4-O(CH2)4MeC6H4CHO | Me | 5d | 7 | 89 | 165–166 |
| 5 | 2,3,4-(OMe)3C6H2CHO | Me | 5e | 8 | 90 | 184–185 |
| 6 | 4-MeC6H4CHO | Me | 5f | 6 | 93 | 229–230 (227–228)18 |
| 7 | 4-SMeC6H4CHO | Me | 5g | 6 | 91 | 230–231 (229–231)14 |
| 8 | 2-FC6H4CHO | Me | 5h | 7 | 86 | 271–222 (270–272)15 |
| 9 | 4-FC6H4CHO | Me | 5i | 6 | 89 | 221–222 (220–222)18 |
| 10 | 2-ClC6H4CHO | Me | 5j | 8 | 90 | 269–270 (268–270)18 |
| 11 | 3-ClC6H4CHO | Me | 5k | 7 | 91 | 206–207 (204–206)18 |
| 12 | 4-ClC6H4CHO | Me | 5l | 6 | 95 | 260–262 (262–264)18 |
| 13 | 3-NO2C6H4CHO | Me | 5m | 6 | 89 | 271–272 (270–272)18 |
| 14 | 4-NO2C6H4CHO | Me | 5n | 6 | 93 | 225–226 (222–225)14 |
| 15 | 4-CF3C6H4CHO | Me | 5o | 6 | 93 | 217–218 (215–217)25 |
| 16 | 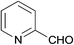 |
Me | 5p | 8 | 86 | 230–232 (229–231)15 |
| 17 | 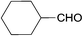 |
Me | 5q | 10 | 83 | 220–222 (221–222)18 |
| 18 | 4-OMeC6H4CHO | H | 5r | 7 | 91 | 253–254 (254–255)13 |
| 19 | 4-MeC6H4CHO | H | 5s | 7 | 94 | 243–245 (244–246)13 |
| 20 | 4-ClC6H4CHO | H | 5t | 7 | 92 | 273–275 (272–273)12 |
| 21 | 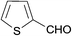 |
H | 5u | 8 | 85 | 240–242 |
| 22 | 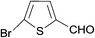 |
H | 5v | 8 | 88 | 206–208 |
Recycling of the catalyst
The magnetic property of CoFe2O4@CS-SO3H facilitates efficient recovery of the catalyst from the reaction mixture during work-up procedure. After completion of the reaction, ethyl acetate was added to the reaction mixture. The catalyst was separated by a strong external permanent magnet, washed with ethyl acetate to remove residual product, dried under vacuum, and reused directly for the next cycle. The catalyst was used over five runs, and no significant loss of catalytic activity was observed (Fig. 6). FT-IR images of fresh and recovered catalysts indicated that no significant change had occurred. This indicates that CoFe2O4@CS-SO3H was very high chemical stable and can endure high temperature condition (Fig. 4).To compare the efficiency of our catalyst with some of the previously employed catalysts, we have tabulated the results of these catalysts for the synthesis of product 5a in Table 3. As can be seen in Table 3, the results clearly indicated that our catalyst is an equally or more efficient catalyst for this four-component reaction.
| Entry | Catalyst | Reaction conditions | Time (min) | Yield (%) |
|---|---|---|---|---|
| 1 | PEG-OSO3H (8 mol%) | Solvent-free, 80 °C | 13 | 87 (ref. 19) |
| 2 | Ce(SO4)2·4H2O (10 mol%) | Solvent-free, 125 °C | 6 | 78 (ref. 20) |
| 3 | ZnO (5 mol%) | Solvent-free, 60 °C | 5 | 97 (ref. 21) |
| 4 | Cellulose–SO3H (4 mol%) | Solvent-free, 60 °C | 7 | 91 (ref. 22) |
| 5 | Starch sulfate (0.8 mol%) | Solvent-free, 80 °C | 7 | 89 (ref. 23) |
| 6 | [TMG][Ac] (10 mol%) | Solvent-free, 80 °C | 15 | 92 (ref. 24) |
| 7 | [Bmim]Br (0.5 g) | Ultrasonic irradiation | 10 | 93 (ref. 25) |
| 8 | ([Hnhp][HSO4]) (5 mol%) | Solvent-free, 80 °C | 7 | 88 (ref. 26) |
| 9 | CoFe2O4-CS-SO3H (0.5 mol%) | Solvent-free, 80 °C | 7 | 90 (This work) |
Conclusions
In summary, a nanosized magnetic CoFe2O4 chitosan sulfonic acid was prepared and characterized by XRD, TEM, SEM, EDX and IR spectroscopy. The catalyst showed highly activity for one-pot, atom-economical synthesis of 2H-indazolo[2,1-b]phthalazine-triones by four-component reaction of phthalic anhydride, hydrazinium hydroxide, 1,3-cyclohexanedione and aldehydes. This simple and environmentally benign catalysis proceeds under solvent-free conditions, requires short reaction times, and provides products in high to excellent yields. The catalyst could be readily recovered by applying an external magnet and recycled for several times without appreciable loss of its catalytic activity, which demonstrated the value of the present CoFe2O4-CS-SO3H as a green heterogeneous catalyst with potential use for industrial applications.Experimental
Materials and instruments
Chemicals were purchased from and used without further purification. The known products were characterized by comparison of their spectral and physical data with those reported in the literature. 1H NMR (500 MHz) and 13C NMR (125 MHz) spectra were recorded on a Bruker DRX-500 spectrometer in CDCl3 solution with TMS as an internal standard. X-ray power diffraction (XRD) analysis was carried out on a PANalytical X'Pert Pro X-ray diffractometer. FT-IR spectra were obtained with potassium bromide pellets on potassium bromide pellets in the range 400–4000 cm−1 with a Bruker-TENSOR 27 spectrometer. Surface morphology and particle size were studied using a Hitachi S-4800 SEM instrument. Transmission electron microscope (TEM) images was obtained using Hitachi H-7650 microscope at 80 KV for characterization of the catalyst. Elemental compositions were determined with a Hitachi S-4800 scanning electron microscope equipped with an INCA 350 energy dispersive spectrometer (SEM-EDS) presenting a 133 eV resolution at 5.9 keV. Melting points were determined using an X-4 apparatus and are uncorrected. Elemental analyses were performed by using a Vario EL III CHNOS elemental analyzer.Preparation of magnetic CoFe2O4 chitosan sulfonic acid nanoparticle (CoFe2O4-CS-SO3H)
Chitosan coated CoFe2O4 nanoparticles (CoFe2O4-CS) were prepared as follow: 1.50 g of chitosan was dissolved in 3% acetic acid solution (100 mL) in a 500 mL three-necked flask equipped with a stirrer and dropping funnel. Then, the aqueous solution of 2.70 g of FeCl3·6H2O and 1.19 g of CoCl2·6H2O (50 mL) was slowly added into the mixture. The mixture was stirred strongly for 2 h, and then NaOH (3 mol L−1) solution was added until the pH increased to 12. The reaction mixture was then continually stirred under refluxing condition for 1 h. After cooling the solution to room temperature, the chitosan coated CoFe2O4 NPs were collected using a permanent magnet and washed with water, ethanol, then dried under vacuum at 50 °C for 24 h.CoFe2O4-CS-SO3H was prepared by the reaction of CoFe2O4-CS and chlorosulfonic acid. Typically, a mixture of CoFe2O4-CS (1.2 g) was suspended in dichloromethane (5 mL) in a 100 mL round bottom flask equipped with a gas outlet tube and a dropping funnel containing a solution of chlorosulfonic acid (2 mL) in dichloromethane (15 mL). The chlorosulfonic acid solution was added drop-wise over a period of 30 min at 0 °C. After the addition was complete, the mixture was shaken for 1 h until all HCl was removed from reaction vessel. Then, the product was separated by magnetic decantation and washed with EtOH to remove unattached substrates. Finally, the obtained CoFe2O4-CS-SO3H NPs were dried under vacuum at 80 °C for 24 h.
General procedure for synthesis of amides
Aldehyde (1 mmol), 1,3-dicarbonyl compounds (1 mmol), and CoFe2O4-CS-SO3H (0.005 mol, 10 mg) was added to a mixture of hydrazinium hydroxide (1.2 mmol) and phthalic anhydride (1 mmol). The reaction mixture was heated at 80 °C. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to room temperature and ethyl acetate (5 mL) was added. The catalyst was separated by an external magnet, washed with ethyl acetate, dried and re-used for a consecutive run under the same reaction conditions. Evaporation of the solvent of the filtrate under reduced pressure gave the crude product. The pure product was obtained by column chromatography on silica gel with hexane/ethyl acetate.Some selected spectra data for non-reported products
Acknowledgements
We are grateful for the financial support for this research from the National Natural Science Foundation of China (no. 21272053).References
- (a) M. B. Gawande, S. N. Shelke, R. Zboril and R. S. Varma, Acc. Chem. Res., 2014, 47, 1338–1348 CrossRef CAS PubMed; (b) M. B. Gawande, R. K. Pandey and R. V. Jayaram, Catal. Sci. Technol., 2012, 2, 1113–1125 RSC; (c) S. Shylesh, V. Schunemann and W. R. Thiel, Angew. Chem., Int. Ed., 2010, 49, 3428–3459 CrossRef CAS PubMed.
- (a) D. Wang and D. Astruc, Chem. Rev., 2014, 114, 6949–6985 CrossRef CAS PubMed; (b) M. B. Gawande, R. Luque and R. Zboril, ChemCatChem, 2014, 6 DOI:10.1002/cctc.201402663.
- (a) R. B. N. Baig and R. S. Varma, Green Chem., 2013, 15, 398–417 RSC; (b) M. B. Gawande, P. S. Branco and R. S. Varma, Chem. Soc. Rev., 2013, 42, 3371–3393 RSC; (c) R. Mrówczyński, A. Nan and J. Liebscher, RSC Adv., 2014, 4, 5927–5952 RSC; (d) D. Wang and D. Astruc, Chem. Rev., 2014, 114, 6949–6985 CrossRef CAS PubMed; (e) A. Bazgir, G. Hosseini and R. Ghahremanzadeh, ACS Comb. Sci., 2013, 15, 530–534 CrossRef CAS PubMed; (f) A. S. Burange, S. R. Kale, R. Zboril, M. B. Gawande and R. V. Jayaram, RSC Adv., 2014, 4, 6597–6601 RSC; (g) S. R. Kale, S. S. Kahandal, M. B. Gawande and R. V. Jayaram, RSC Adv., 2013, 3, 8184–8192 RSC; (h) A. Khalafi-Nezhad, M. Nourisefat and F. Panahi, RSC Adv., 2014, 4, 22497–22500 RSC; (i) H. Moghanian, A. Mobinikhaledi, A. G. Blackman and E. Sarough-Farahani, RSC Adv., 2014, 4, 28176–28185 RSC; (j) J. M. Pérez, R. Cano and D. J. Ramón, RSC Adv., 2014, 4, 23943–23951 RSC; (k) S. Sobhani, Z. M. Falatooni and M. Honarmand, RSC Adv., 2014, 4, 15797–15806 RSC; (l) M. Tajbakhsh, M. Farhang, R. Hosseinzadeh and Y. Sarrafi, RSC Adv., 2014, 4, 23116–23124 RSC; (m) S. N. Shelke, S. R. Bankar, G. R. Mhaske, S. S. Kadam, D. K. Murade, S. B. Bhorkade, A. K. Rathi, N. Bundaleski, O. Teodoro, R. Zboril, R. S. Varma and M. B. Gawande, ACS Sustainable Chem. Eng., 2014, 2, 1699–1706 CrossRef CAS; (n) M. J. Jacinto, F. P. Silva, P. K. Kiyohara, R. Landers and L. M. Rossi, ChemCatChem, 2012, 4, 698–703 CrossRef CAS; (o) H. Zhu, X. X. Jiang, X. H. Li, C. Hou, Y. Jiang, K. Hou, R. Wang and Y. F. Li, ChemCatChem, 2013, 5, 2187–2190 CrossRef CAS; (p) A. Khalafi-Nezhad and S. Mohammadi, ACS Comb. Sci., 2013, 15, 512–518 CrossRef CAS PubMed.
- (a) J. K. Rajput and G. Kaur, Catal. Sci. Technol., 2014, 4, 142–151 RSC; (b) B. L. Li, M. Zhang, H. C. Hu, X. Du and Z. H. Zhang, New J. Chem., 2014, 38, 2435–2442 RSC; (c) P. H. Li, B. L. Li, Z. M. An, L. P. Mo, Z. S. Cui and Z. H. Zhang, Adv. Synth. Catal., 2013, 355, 2952–2959 CrossRef CAS; (d) B. L. Li, H. C. Hu, L. P. Mo and Z. H. Zhang, RSC Adv., 2014, 4, 12929–12943 RSC; (e) X. N. Zhao, H. C. Hu, F. J. Zhang and Z. H. Zhang, Appl. Catal., A, 2014, 482, 258–265 CrossRef CAS PubMed.
- (a) M. B. Gawande, R. Hosseinpour and R. Luque, Curr. Org. Synth., 2014, 11, 526–544 CrossRef CAS; (b) M. B. Gawande, A. K. Rathi, I. D. Nogueira, R. S. Varma and P. S. Branco, Green Chem., 2013, 15, 1895–1899 RSC; (c) C. S. Gill, B. A. Price and C. W. Jones, J. Catal., 2007, 251, 145–152 CrossRef CAS PubMed; (d) H. Mahmoudi and A. A. Jafari, ChemCatChem, 2013, 5, 3743–3749 CrossRef CAS.
- (a) S. K. Shukla, A. K. Mishra, O. A. Arotiba and B. B. Mamba, Int. J. Biol. Macromol., 2013, 59, 46–58 CrossRef CAS PubMed; (b) R. B. N. Baig, M. N. Nadagouda and R. S. Varma, Green Chem., 2014, 16, 2122–2127 RSC; (c) R. B. N. Baig, B. R. Vaddula, M. A. Gonzalez and R. S. Varma, RSC Adv., 2014, 4, 9103–9106 RSC; (d) C. Shen, J. Xu, W. B. Yu and P. F. Zhang, Green Chem., 2014, 16, 3007–3012 RSC.
- Y. J. He, M. S. Pei, Y. K. Du, F. Q. Yu, L. Y. Yang and W. J. Guo, RSC Adv., 2014, 4, 30352–30357 RSC.
- (a) Z. Zarnegar and J. Safari, RSC Adv., 2014, 4, 20932–20939 RSC; (b) J. H. Zhou, Z. P. Dong, H. L. Yang, Z. Q. Shi, X. C. Zhou and R. Li, Appl. Surf. Sci., 2013, 279, 360–366 CrossRef CAS PubMed; (c) A. Maleki, N. Ghamari and M. Kamalzare, RSC Adv., 2014, 4, 9416–9423 RSC.
- (a) J. T. Hou, J. W. Gao and Z. H. Zhang, Appl. Organomet. Chem., 2011, 25, 47–53 CrossRef CAS; (b) Y. L. Gu, Green Chem., 2012, 14, 2091–2128 RSC; (c) J. Yang, H. Q. Li, M. H. Li, J. J. Peng and Y. L. Gu, Adv. Synth. Catal., 2012, 354, 688–700 CrossRef CAS; (d) S. Karamthulla, S. Pal, T. Parvin and L. H. Choudhury, RSC Adv., 2014, 4, 15319–15324 RSC; (e) H. Veisi, A. A. Manesh, N. Khankhani and R. Ghorbani-Vaghei, RSC Adv., 2014, 4, 25057–25062 RSC.
- P. Salehi, D. I. MaGee, M. Dabiri, L. Torkian and J. Donahue, Mol. Diversity, 2012, 16, 231–240 CrossRef CAS PubMed.
- K. Mazaahir, C. Ritika and J. Anwar, Chin. Sci. Bull., 2012, 57, 2273–2279 CrossRef CAS.
- L. Nagarapu, R. Bantu and H. B. Mereyala, J. Heterocycl. Chem., 2009, 46, 728–731 CrossRef CAS.
- J. M. Khurana and D. Magoo, Tetrahedron Lett., 2009, 50, 7300–7303 CrossRef CAS PubMed.
- R. Ghorbani-Vaghei, R. Karimi-Nami, Z. Toghraei-Semiromi, M. Amiri and M. Ghavidel, Tetrahedron, 2011, 67, 1930–1937 CrossRef CAS PubMed.
- M. V. Reddy, G. C. S. Reddy and Y. T. Jeong, Tetrahedron, 2012, 68, 6820–6828 CrossRef PubMed.
- S. Safaei, I. Mohammadpoor-Baltork, A. R. Khosropour, M. Moghadam, S. Tangestaninejad and V. Mirkhani, Catal. Sci. Technol., 2013, 3, 2717–2722 CAS.
- G. Shukla, R. K. Verma, G. K. Verma and M. S. Singh, Tetrahedron Lett., 2011, 52, 7195–7198 CrossRef CAS PubMed.
- H. J. Wang, X. N. Zhang and Z. H. Zhang, Monatsh. Chem., 2010, 141, 425–430 CrossRef CAS.
- A. Hasaninejed, M. R. Kazerooni and A. Zare, Catal. Today, 2012, 196, 148–155 CrossRef CAS PubMed.
- E. Mosaddegh and A. Hassankhani, Tetrahedron Lett., 2011, 52, 488–490 CrossRef CAS PubMed.
- H. R. Shaterian, E. Mollashahi and A. Biabangard, J. Chem. Soc. Pak., 2013, 35, 329–332 Search PubMed.
- H. R. Shaterian and F. Rigi, Res. Chem. Intermed., 2014, 40, 1989 CrossRef CAS PubMed.
- H. R. Shaterian and F. Rigi, Starch, 2011, 63, 340–346 CrossRef CAS.
- H. Veisi, A. A. Manesh, N. Khankhani and R. Ghorbani-Vaghei, RSC Adv., 2014, 4, 25057–25062 RSC.
- M. Shekouhy and A. Hasaninejad, Ultrason. Sonochem., 2012, 19, 307–313 CrossRef CAS PubMed.
- H. R. Shaterian and M. Aghakhanizadeh, C. R. Chim., 2012, 15, 1060–1064 CrossRef CAS PubMed.
- (a) J. Deng, L. P. Mo, F. Y. Zhao, L. L. Hou, L. Yang and Z. H. Zhang, Green Chem., 2011, 13, 2576–2584 RSC; (b) Y. H. Liu, J. Deng, J. W. Gao and Z. H. Zhang, Adv. Synth. Catal., 2012, 354, 441–447 CrossRef CAS; (c) J. Deng, L. P. Mo, F. Y. Zhao, Z. H. Zhang and S. X. Liu, ACS Comb. Sci., 2012, 14, 335–341 CrossRef CAS PubMed; (d) F. P. Ma, P. H. Li, B. L. Li, L. P. Mo, N. Liu, H. J. Kang, Y. N. Liu and Z. H. Zhang, Appl. Catal., A, 2013, 457, 34–41 CrossRef CAS PubMed; (e) P. H. Li, B. L. Li, H. C. Hu, X. N. Zhao and Z. H. Zhang, Catal. Commun., 2014, 46, 118–122 CrossRef CAS PubMed; (f) J. Lu, X. T. Li, E. Q. Ma, L. P. Mo and Z. H. Zhang, ChemCatChem, 2014, 6, 2854–2859 CrossRef CAS.
- (a) X. N. Zhang, Y. X. Li and Z. H. Zhang, Tetrahedron, 2011, 67, 7426–7430 CrossRef CAS PubMed; (b) Z. H. Zhang, X. N. Zhang, L. P. Mo, Y. X. Li and F. P. Ma, Green Chem., 2012, 14, 1502–1506 RSC; (c) R. Y. Guo, Z. M. An, L. P. Mo, S. T. Yang, H. X. Liu, S. X. Wang and Z. H. Zhang, Tetrahedron, 2013, 69, 9931–9938 CrossRef CAS PubMed; (d) R. Y. Guo, P. Wang, G. D. Wang, L. P. Mo and Z. H. Zhang, Tetrahedron, 2013, 69, 2056–2061 CrossRef CAS PubMed; (e) B. L. Li, P. H. Li, X. N. Fang, C. X. Li, J. L. Sun, L. P. Mo and Z. H. Zhang, Tetrahedron, 2013, 69, 7011–7018 CrossRef CAS PubMed; (f) R. Y. Guo, Z. M. An, L. P. Mo, R. Z. Wang, H. X. Liu, S. X. Wang and Z. H. Zhang, ACS Comb. Sci., 2013, 15, 557–563 CrossRef CAS PubMed; (g) P. H. Li, F. P. Ma, P. Wang and Z. H. Zhang, Chin. J. Chem., 2013, 31, 757–763 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra09984b |
| This journal is © The Royal Society of Chemistry 2014 |