DOI:
10.1039/C4RA08464K
(Paper)
RSC Adv., 2014,
4, 50739-50751
Effects of Jatropha biodiesel on the performance, emissions, and combustion of a converted common-rail diesel engine
Received
10th August 2014
, Accepted 3rd October 2014
First published on 3rd October 2014
Abstract
An experimental investigation into the effects of Jatropha biodiesel fuels on the engine performance, emissions, and combustion characteristics of a single-cylinder high-pressure common-rail diesel engine was performed under six different load operations (0.1, 0.2, 0.3, 0.4, 0.5, and 0.6 MPa). The test fuels included a conventional diesel fuel and three different blends of Jatropha biodiesel fuel (JB10, JB30, and JB50). The results revealed that the biodiesel blended fuels had a significant influence on the brake specific fuel consumption (BSFC) at all of the engine load conditions examined. In general, the use of Jatropha biodiesel blends resulted in a reduction in brake specific nitrogen oxide (BSNOx), brake specific carbon monoxide (BSCO), and smoke emissions, regardless of the load conditions. A large reduction of 20.2% in BSNOx emissions and 69.5% in smoke opacity were found for the engine when it was fuelled with the biodiesel blends. In terms of the engine combustion characteristics, a slightly shorter ignition delay (ID) and faster combustion duration were found to occur with the use of biodiesel blends under all loading operations. It was revealed that the peak apparent heat release rate (AHRR) for biodiesel blends is lower during low load operation; the AHRR was found to be comparable to that of baseline diesel during high-load operation. Finally, the vibration results demonstrated that the largest reduction, 11.3%, in the root mean square (RMS) of acceleration in comparison with the baseline diesel was obtained with JB50 at an engine load of 0.5 MPa.
1. Introduction
Energy plays a vital role in our daily life. In recent years, rapid growth in population, development, and industrialization has led to a high demand for energy worldwide. This energy is predominately derived from non-renewable sources, such as fossil fuels and coal. Unfortunately, these resources are finite and are forecast to be diminished in less than a hundred years.1 Some predictions have stated that they will be depleted in less than 45 years.2 This scenario has triggered concern worldwide over energy security and has inevitably affected and pressured many countries in the world into seeking alternative approaches to satisfy ever rising energy demands. Aside from this energy issue, the world is also currently managing global warming and air pollution. The combustion of fossil fuels in the transportation sector is the primary source of greenhouse gas and pollutant emissions. Consequently, it is clear that the world is confronted with the twin crises of fossil fuel depletion and environmental degradation.
Recently, biodiesel has been considered as a major substitute for fossil diesel worldwide.3 Biodiesel can be defined as the monoalkyl esters of long-chain fatty acids that are derived from chemical reactions (transesterification) of renewable feedstocks, such as vegetable oil or animal fats, and alcohol with a catalyst. The full life-cycle analysis, including the cultivation and production of oil, and subsequent conversion to biodiesel, revealed that the net carbon dioxide (CO2) emissions are relatively low, and the use of biodiesel appears to have a significant positive impact on rural economic potential.4
Globally, there are more than 350 oil-bearing crops identified as prospective sources for the production of biodiesel.5 In general, food crops, such as corn, sugar, and vegetable oil, have been the primary source of biodiesel fuels for transportation. These sources are considered to be the first generation of biodiesel feedstock because they were the first crops to be used in the production of biodiesel. However, the extensive use of these fuels has caused food prices to rise, food price volatility, and an accelerated expansion of agriculture in the tropics. In order to avoid possible negative consequences of this, solutions including exploiting non-edible oils are being considered. Non-edible oil resources are gaining attention worldwide because they are not suitable for human consumption, eliminating competition between fuel and food sources and reducing the rate of deforestation. Hence, they are more environmentally friendly and economically competitive with edible oils. Biodiesel production from non-edible crops is regarded as the second generation of biodiesel feedstocks. These feedstocks include Jatropha curcas, Calophyllum inophyllum, Ceiba pentandra, Karanja, Neem, Jojoba, and rubber seed, among others.6
1.1. Engine performance and exhaust emission for biodiesel blends
There is considerable research focused on engine performance and emission characteristics when using edible biodiesel, and it is only recently that greater efforts have been established investigating non-edible biodiesel fuel. Chauhan et al. performed a study that investigated the effects of biodiesel produced from non-edible oil on a diesel engine equipped with a mechanical pump-line-nozzle fuel injection system.7 The authors investigated the effect of various Jatropha biodiesel blends (5, 10, 20, and 30%) on engine performance and the exhaust emissions using an unmodified diesel engine, but without performing supporting combustion results. The experimental results indicated that the engine performance with Jatropha biodiesel and its blends were comparable to the performance of the engine when using diesel fuel. The brake thermal efficiency (BTE), HC, CO, CO2, and smoke emissions were found to be lower, while the BSFC, BSEC, and NOx emissions were higher when using Jatropha biodiesel blends in comparison with diesel. The authors suggested that biodiesel derived from non-edible oil, like Jatropha and its blends, could be used in a conventional diesel engine without any modification.
An alternative potential non-edible biodiesel, investigated by Ong et al.,8 is produced from the plant Calophyllum inophyllum. The authors investigated the performance and emissions of this biodiesel in a single-cylinder, mechanical pump-line-nozzle direct fuel injection diesel engine. The test results revealed that the use of B10 biodiesel yields an improvement in engine performance, with higher BTE and lower BSFC, in comparison with diesel fuel. Emissions, such as CO and smoke opacity, were reduced when using B10 but NOx emission were found to be somewhat increased when using B10 at all engine speeds. The used of Kapok (Ceiba pentandra) oil methyl ester as the source of non-edible biodiesel in diesel engines was explored by Vedharaj et al.9 They conducted an experiment in a single-cylinder, four-stroke, water-cooled diesel engine that was equipped with a mechanical pump-nozzle injection system. The investigation revealed that the BTE of the engine when using B25 was superior to conventional diesel by 4%. The combustion and emissions of HC, CO, NOx, and smoke for B25 were all comparable with the results produced when using diesel. In a performance and emissions test on a single-cylinder, four-stroke, water-cooled Ricardo E6 engine using Mahua (Madhuca indica) biodiesel, Raheman and Ghadge10 affirmed a reduction in smoke and CO emissions, with an increase in NOx emissions when the percentage of Mahua biodiesel blends was increased. The mean BSFC was reported to be 4.3–41.4% higher for Mahua biodiesel blends. The mean BTE for B100 was 10.1% lower than diesel fuel at full load condition.
Biodiesel fuel in engines equipped with a high-pressure common-rail direct injection (CRDI) system has recently become an interesting research topic. This is predominantly owing to the fact that a majority of the diesel engines in use today employ this technology. An et al.11 experimentally investigated pure waste cooking oil biodiesel and blends of 10, 20 and 50% in a four-stroke, four-cylinder, water-cooled, high-pressure CRDI diesel engine under various loads. As would be expected, the BSFC of the biodiesel was reported to be higher in comparison with diesel fuel. In fact, the largest increase in BSFC (28.1%) was found at a 10% load. It was observed that the cylinder pressure decreased slightly with the use of biodiesel at all engine loads. The CO emissions were found to be increased when using a higher percentage biodiesel blend ratio and a decreased engine speed. However, an opposite trend was observed at higher engine loads. Tan et al.12 investigated the used of biodiesel in a multi-cylinder, four-stroke, diesel engine that was equipped with a high-pressure common-rail fuel injection system. In this study, the authors examined the impact of biodiesel fuel solely on regulated and unregulated emissions. In comparison to the baseline diesel fuel, the addition of biodiesel demonstrated no obvious difference in NOx emission, a reduction in HC, smoke, acetaldehyde and toluene, and an increase in CO, formaldehyde and acetone.
1.2. Purpose of study
Among the topics discussed above, many previous studies into biodiesel fuel have examined the engine-out responses of a conventional mechanical pump-line-nozzle fuel injection system. With this fuel injection system, the lower compressibility and the viscosity of the biodiesel will usually lead to an advanced start of injection, resulting in higher NOx emissions.13 These effects could be eliminated by using common-rail fuel injection technology, in which fuel pressurization is independent of injection timing.14 Aside from the problem of fuel injection technology, there is an instability and fluctuation in the price of crude palm oil (edible oil) in Malaysia. To minimize dependency on consuming biodiesel fuel that is primarily sourced from crude palm oil, the biodiesel policy of the Malaysian government recommended the utilization of non-edible oils for the production of biodiesel. Jatropha curcas oil is one of the major non-edible, tree-borne feedstocks used for the large scale production of biodiesel in Malaysia and south east Asia; this is because it is well adapted to local climatic conditions and is available in surplus quantities across the region.15,16 Consequently, there is strong motivation to investigate the impact of Jatropha biodiesel blends in an engine equipped with a high-pressure common-rail injection system, and to analyse the effects of this biodiesel on engine performance, emissions, combustion, and vibration characteristics. This experiment was performed under six different brake mean effective pressure (BMEP) (0.1, 0.2, 0.3, 0.4, 0.5, and 0.6 MPa) and at a rated engine speed of 1600 rpm. Parameters including BSFC, BTE, BSCO, BSNOx, EGT, smoke opacity, peak pressure, peak of heat release, and vibration analysis were investigated and evaluated.
2. Material and methods
2.1. Test fuels and operating conditions
In the present study, fossil diesel fuel was obtained in commercial form and Jatropha oil was obtained from Indonesia. Biodiesel production was conducted via the acid-esterification and alkali-transesterification process. Table 1 contains a description of the key physicochemical properties of neat Jatropha methyl ester (JME) in comparison with ASTM and EN standards. It can be observed that the physicochemical properties of the produced biodiesel was comprehensively measured and benchmarked against the biodiesel standards based on ASTM D6751 and EN14214. It appears that all physicochemical properties of JME are sufficient to meet the ASTM and EN biodiesel standards.
Table 1 The fuel properties of neat JME
| Properties |
Unit |
Limit (ASTM D6751) |
Limit (EN 14214) |
JME |
Test method |
| Kinematic viscosity @ 40 °C |
mm2 s−1 |
1.9–6.0 |
3.5–5.0 |
4.42 |
D445 |
| Density @ 15 °C |
kg m−3 |
880 |
860–900 |
882.7 |
D127 |
| Acid number |
mg KOH g−1 |
0.5 max. |
0.5 max. |
0.37 |
D664 |
| Calorific value |
MJ kg−1 |
— |
35 |
39.98 |
D240 |
| Flash point |
°C |
130 min. |
120 min. |
178.5 |
D93 |
| Pour point |
°C |
— |
— |
4 |
D2500 |
| Cloud point |
°C |
Report |
— |
5 |
D2500 |
| Cold filter plugging point |
°C |
— |
— |
1 |
D6371 |
| Oxidation stability @ 100 °C |
Hours |
3 min. |
6 min. |
6.5 |
EN14112 |
| Cetane number |
— |
47 min. |
51 min. |
58 |
D6890 |
| Carbon |
wt% |
77 |
— |
76.8 |
D5291 |
| Hydrogen |
wt% |
12 |
— |
11.8 |
D5291 |
| Oxygen |
wt% |
11 |
— |
10.9 |
D5291 |
In this study, a total of three different methyl ester blends, JB10 (10% biodiesel, 90% petroleum diesel), JB30 (30% biodiesel, 70% petroleum diesel), and JB50 (50% biodiesel, 50% petroleum diesel) were prepared and tested. The key physicochemical properties of the diesel and biodiesel blends are listed in Table 2. As can be observed, all of the physicochemical properties of the biodiesel blends satisfy the ASTM D7467 biodiesel blend standards. Additionally, the experimental results suggested that blending with petroleum diesel substantially improved the final biodiesel blend properties. In particularly, the kinematic viscosity of the biodiesel blends was reduced as the proportion of petroleum diesel was increased in the blends. In additional, the resultant flash points for all the biodiesel blends were relatively higher in comparison with petroleum diesel and were suitable for use as a transportation fuel. However, the calorific value of all of the biodiesel blends was lower than that of petroleum diesel. Another key property that significantly influences engine performance, emissions, and combustion characteristics is the cetane number of fuel. In general, it can be observed that all biodiesel blends have a higher cetane number than petroleum diesel fuels.
Table 2 The fuel properties of diesel fuel and biodiesel blends
| Properties |
Unit |
Diesel fuel |
Biodiesel blends |
JB10 |
JB30 |
JB50 |
| Limit (ASTM D7467) |
Test method |
| Kinematic viscosity @ 40 °C |
mm2 s−1 |
3.34 |
1.9–4.1 |
D445 |
3.62 |
3.73 |
3.9 |
| Density @ 15 °C |
kg m−3 |
851.9 |
858 max. |
D127 |
858.2 |
864.2 |
871.3 |
| Acid number |
mg KOH g−1 |
0.12 |
0.3 max. |
D664 |
0.17 |
0.23 |
0.27 |
| Calorific value |
MJ kg−1 |
45.31 |
35 |
D240 |
44.84 |
43.66 |
42.57 |
| Flash point |
°C |
71.5 |
52 |
D93 |
87.5 |
96.5 |
103.5 |
| Pour point |
°C |
1 |
Not specified |
D2500 |
0 |
0 |
0 |
| Cloud point |
°C |
8 |
Not specified |
D2500 |
8 |
7 |
6 |
| Oxidation stability @ 100 °C |
Hours |
>40 |
6 |
EN14112 |
18.6 |
12.8 |
10.8 |
| Cetane number |
— |
52 |
47 min. |
D6890 |
53 |
54 |
56 |
The experiment was conducted under a constant speed of 1600 rpm and with varying BMEP (i.e. 0.1, 0.2, 0.3, 0.4, 0.5, and 0.6 MPa). These six test points were selected as the most representative of a wide variety of engine load ranges. Initially, diesel fuel was used as the baseline fuel for the basis of comparison. Following this, mixtures of diesel and methyl ester with 10, 30, and 50% volumetric proportions were tested. Consequently, a total of 24 runs experimental conditions, including baseline diesel were tested in this study. When the engine was fuelled with methyl ester blended fuels, the engine ran satisfactorily throughout the entire test, which was performed at room temperature, and had no starting difficulties. The tests were performed under steady-state conditions with a sufficiently warmed exhaust gas and water coolant temperature. To enhance the accuracy of the study, each test point was repeated twice to produce average readings. The repeatability was matched over 95% for each test. This indicates that the effects on emissions and combustion characteristics can be reliably analysed from this test system.
2.2. Experimental system
2.2.2. Engine modifications and fuel delivery system. The original injection system of the engine was disassembled and a new common-rail injection system was retrofitted as illustrated in Fig. 2. The system was based on commercially available common-rail diesel engine components. A second-generation, electronically controlled common-rail high-pressure injection system was installed to replace the original mechanical type pump-line-nozzle injection system. The fuel pump was externally driven using an electric motor running at 750 rpm to maintain the required high-pressure levels in the fuel rail and to ensure a stable line pressure with minimum fluctuation. This injection system enabled variable injection pressure and injection timing. Independent control of the injection parameters was achieved using a custom-built electronic control unit (ECU). The measured nozzle diameter was found to be approximately 0.134 mm, with five evenly spaced nozzle holes.
 |
| | Fig. 2 The retrofitted engine setup. | |
2.3. Test bed configuration and instrumentation
The ST-7.5 model 7.5 kW A.C. synchronous dynamometer was used to provide loading to the engine and to maintain the engine speed. An airflow metre turbine with 2 to 70 litres per second (L s−1) measuring range was employed to measure the intake airflow rate. A type K thermocouple was used to monitor the exhaust gas temperature. The fuel flow rate was measured with a positive displacement gear wheel flow metre (model: DOM-A05H), which interfaced with a flow rate totalizer (model: ZOD-Z3). The test system was equipped with the necessary sensors for the analysis of combustion and fuel injection timing. In-cylinder gas pressure was measured using a Kistler 6125B type pressure sensor. To mount the sensor in the engine head through a water cooling jacket, a dedicated mounting sleeve was fabricated and installed, as can be observed on the right-hand side of Fig. 1. The charge signal output of the pressure sensor was converted to a low-impedance voltage signal using a PCB model 422E53 in-line charge converter; this unit was powered using a PCB model 480E09 signal conditioner. To acquire the top dead centre (TDC) position and crank angle signal for every engine rotation, a high-precision incremental encoder with 720 pulses per revolution was used. To determine and verify the SOI timing, the injector current signal was measured with a hall effect current sensor. To simultaneously sample the cylinder pressure, injector current signal, and encoder signals, a computer equipped with a high-speed ADLINK DAQ-2010 simultaneous sampling data acquisition card, which has 14 bits resolution, 2 MS per s−1 sampling rate, and four analog input channels, was used. The acquired data were further processed and analysed with Matlab software. To eliminate cycle-to-cycle variation in each test, 100 consecutive combustion cycles (or equal to 200 crankshaft revolutions) of pressure data were collected and an averaged was calculated. To reduce noise effects, smooths data using SPAN as the number of points used to compute each element was applied to the sampled cylinder pressure data. Combustion parameters, such as peak pressure magnitude, peak pressure location, heat release rate, peak heat release rate location, and ID, were all computed using Matlab software. For the exhaust emission measurement, an AVL DICOM 4000 5-gas analyser was used to measure the concentrations of CO, CO2, and NOx. Opacity of smoke was measured using AVL DiSmoke 4000. All emissions were measured during steady-state engine operation. The measurement range and resolution of both of the instruments are provided in Table 4. The experimental set-up is illustrated in Fig. 3. The CO and NOx emissions were converted into brake specific emissions by using the following equations according to SAE J177:| | |
BSCO(g kW−1 h−1) = 0.0580 × CO(ppm) × exhaust mass flow rate(kg min−1)/brake power(kW)
| (1) |
| | |
BSNOx(g kW−1 h−1) = 0.0952 × NOx(ppm) × exhaust mass flow rate(kg min−1)/brake power(kW)
| (2) |
Table 4 Measuring components, ranges and resolution of the AVL DICOM 4000 gas analyzer and DiSmoke 4000 smoke analyzer
| Equipment |
Measurement principle |
Component |
Measurement range |
Resolution |
| Gas analyzer |
Non-dispersive infrared |
Carbon monoxide (CO) |
0–10 vol% |
0.01 vol% |
| Non-dispersive infrared |
Carbon dioxide (CO2) |
0–20 vol% |
0.1 vol% |
| Electrochemical |
Nitrogen oxides (NOx) |
0–5000 ppm |
1 ppm |
| Calculation |
Excess air ratio (λ) |
0–9999 |
0.001 |
| Smoke opacimeter |
Photodiode detector |
Opacity (%) |
0–100% |
0.10% |
 |
| | Fig. 3 Schematic diagram of the experiment setup. | |
To perform engine vibration measurements, an accelerometer (PCB model 603C01) with calibrated sensitivity to a 95 mV g−1 and 50 g measurement range was used. This rugged accelerometer is capable of performing over a wide frequency range of 0.5–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Hz. Engine vibration motion in the lateral (y) axis (or perpendicular to cylinder axis) was chosen for monitoring vibrations. To sense the magnitude of vibration in this direction, the accelerometer was mounted on the engine body with an adhesive mounting base. The output signal from the sensor was connected to a constant current single channel signal conditioner (PCB model 480C02) with unity gain. In each test, engine-block vibration signals for a total of 100 consecutive combustion cycles at 0.125° CA resolution were recorded and the averaged RMS was calculated.
000 Hz. Engine vibration motion in the lateral (y) axis (or perpendicular to cylinder axis) was chosen for monitoring vibrations. To sense the magnitude of vibration in this direction, the accelerometer was mounted on the engine body with an adhesive mounting base. The output signal from the sensor was connected to a constant current single channel signal conditioner (PCB model 480C02) with unity gain. In each test, engine-block vibration signals for a total of 100 consecutive combustion cycles at 0.125° CA resolution were recorded and the averaged RMS was calculated.
3. Calculation methods
3.1. Engine performance
The engine performance in this work was evaluated based on BSFC and BTE. The BSFC and BTE were determined and calculated according to the following equations:| |
 | (3) |
| |
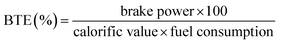 | (4) |
3.2. Combustion analysis
Heat release rate (HRR) analysis is a useful approach to assess the effects of fuel injection system, fuel type, engine design changes, and engine operating conditions on the combustion process and engine performance.17 Given the plot of AHRR versus crank angle, it is easy to identify the start of combustion (SOC) timing, the fraction of fuel burned in the premixed mode, and differences in combustion rates of fuels.18 In the present paper, fuels with different types of methyl ester and blend ratios were fuelled in an identical compression ignition engine; hence, the AHRR information is an important parameter in interpreting engine performance and exhaust emissions. In this study, the averaged in-cylinder pressure data of 100 successive cycles, acquired with a 0.125° crank angle resolution, were used to compute the AHRR. The AHRR, given by 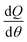 , at each crank angle was obtained from the first law of thermodynamics, and it can be calculated by the following formula:
, at each crank angle was obtained from the first law of thermodynamics, and it can be calculated by the following formula:| |
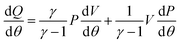 | (5) |
where, γ = specific heat ratio, P = instantaneous cylinder pressure (Pa), and V = instantaneous cylinder volume (m3).
3.3. Vibration analysis
In the present study, the engine vibrations of different types of biodiesel blends were compared with the baseline diesel engine at different engine loads. The averaged RMS of the acceleration signal was determined using the following equation:| |
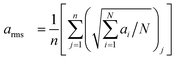 | (6) |
3.4. Statistical and equipment uncertainty analysis
In any experiment, errors and uncertainties can arise from instrument selection, condition, calibration, environment, observation, reading, and test procedure. The measurement range, accuracy, and percentage uncertainties associated with the instruments used in this experiment are listed in Table 5. Uncertainty analysis is necessary to verify the accuracy of the experiments. Percentage uncertainties of various parameters, such as BSFC, BTE, BSCO, and BSNOx were determined using the percentage uncertainties of various instruments employed in the experiment. To compute the overall percentage uncertainty due to the combined effect of the uncertainties of various variables, the principle of propagation of errors is considered and can be estimated as ±4.3%. The overall experimental uncertainty was computed as follows:
| Overall experimental uncertainty = square root of [(uncertainty of fuel flow rate)2 + (uncertainty of BSFC)2 + (uncertainty of BTE)2 + (uncertainty of BSCO)2 + (uncertainty of BSNOx)2 + (uncertainty of EGT)2 + (uncertainty of smoke)2 + (uncertainty of pressure sensor)2 + (uncertainty of crank angle encoder)2] = square root of [(2)2 + (1.95)2 + (1.74)2 + (2.22)2 + (0.73)2 + (0.15)2 + (1)2 + (1)2 + (0.03)2] = ±4.3%. |
Table 5 List of measurement accuracy and percentage uncertainties
| Measurement |
Measurement range |
Accuracy |
Measurement techniques |
% Uncertainty |
| Load |
±120 N m |
±0.1 N m |
Strain gauge type load cell |
±1 |
| Speed |
60–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm 000 rpm |
±1 rpm |
Magnetic pick up type |
±0.1 |
| Time |
— |
±0.1 s |
— |
±0.2 |
| Fuel flow measurement |
0.5–36 L h−1 |
±0.01 L h−1 |
Positive displacement gear wheel flow meter |
±2 |
| Air flow measurement |
2–70 L s−1 |
±0.04 L s−1 |
Turbine flow meter |
±0.5 |
| CO |
0–10% by vol |
±0.001% |
Non-dispersive infrared |
±1 |
| NOx |
0–5000 ppm |
±1 ppm |
Electrochemical |
±1.3 |
| Smoke |
0–100% |
±0.1% |
Photodiode detector |
±1 |
| EGT sensor |
0–1200 °C |
±0.3 °C |
Type K thermocouple |
±0.15 |
| Pressure sensor |
0–25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 kPa 000 kPa |
±12.5 kPa |
Piezoelectric crystal type |
±1 |
| Crank angle encoder |
0–12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm 000 rpm |
±0.125° |
Incremental optical encoder |
±0.03 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
| Computed |
| BSFC |
— |
±7.8 g kW−1 h−1 |
— |
±1.95 |
| BTE |
— |
±0.5% |
— |
±1.74 |
| BSCO |
— |
±0.1 g kW−1 h−1 |
— |
±2.22 |
| BSNOx |
— |
±0.1 g kW−1 h−1 |
— |
±0.73 |
4. Results and discussions
4.1. Engine performance characteristics
4.1.1. Brake specific fuel consumption. Fig. 4 illustrates the BSFC of the fuel samples tested with respect to various BMEPs. BSFC is defined as the ratio of the fuel consumption rate to the brake power output. From the results, it was observed that, at the BMEP of 0.6 MPa, baseline diesel had the lowest BSFC of 270.9 g kW−1 h−1, followed by 280.0 g kW−1 h−1, 287.6 g kW−1 h−1, and 291.2 g kW−1 h−1 for the JB10, JB30, and JB50 blends, respectively. The higher BSFC of JB50 means that more fuel was consumed to develop the same amount of power. This was expected because of the relatively low calorific value of JB50 in comparison with diesel, which was approximately 6% less than that of diesel fuel. These results are in agreement with those reported by Kivevele et al.19
 |
| | Fig. 4 BSFC with the Jatropha biodiesel blends compared with diesel fuel at various BMEP. | |
4.1.2. Brake thermal efficiency. Engine BTE is a product of two important efficiencies, namely, the mechanical efficiency and the net indicated thermal efficiency. Alternatively, it can be calculated by dividing the brake power output by the total energy input delivered to the system. Owing to the effect of various loss mechanisms, such as combustion inefficiency, exhaust blow down, heat transfer, flow, and mechanical friction, the BTE of a real operating diesel cycle is usually under 50%, and is often far lower.20 Of these loss mechanisms, the magnitude of heat transfer losses constitutes a major fraction, and it varies with the mean piston speed as well as the combustion characteristics of the fuel. The variation of BTE versus BMEP for various test fuels is recorded in Fig. 5. In general, it was observed that BTE for all tested fuels increased with an increase in BMEP. This is attributed to the twin effects of increased brake power and reduced wall heat loss at higher engine loads.7,21 Additionally, it was observed that with the addition of JME in the blend, the BTE is slightly reduced and is lower than for diesel fuel, except for JB50 at BMEP of 0.1, 0.4, and 0.5 MPa. Conversely, there is a marginal improvement of 2% in BTE with JB50 at 0.5 MPa. This may be attributed to the fact that, at higher concentrations of methyl ester blend, the early initiation of combustion and increase in peak pressures results in higher BTE.
 |
| | Fig. 5 BTE with the Jatropha biodiesel blends compared with diesel fuel at various BMEP. | |
4.2. Exhaust emissions characteristics
During combustion, CO emissions appear when the available oxygen is insufficient to fully oxidize all of the carbon in the fuel to carbon dioxide. The use of oxygenated fuel, such as methyl ester, would be expected to improve the combustion quality, especially in fuel-rich regions, consequently reducing CO emissions. The variation in BSCO emissions of the engine with different engine loads and fuel types is illustrated in Fig. 6. The results suggest that the magnitude of BSCO emissions was significantly governed by the engine load setting and biodiesel blending ratio. High fuel-borne oxygen content in biodiesel fuel plays a key role in reducing CO emissions. In general, it is observed that the reductions in BSCO emissions were obtained with the use of methyl ester in the blend. In fact, it consistently decreased with the increase in the biodiesel blending ratio. This was mainly due to the oxygen content in biodiesel promoting more complete combustion in the engine.4,22 In comparison with the diesel fuel, the BSCO emissions at a high engine load of 0.6 MPa decreased by 11.9, 30 and 49.9% for JB10, JB30, and JB50, respectively. Another observation was that the BSCO emissions at low load condition were generally higher than at high load conditions, regardless of the fuel used. This was largely owing to the better air-fuel mixing process, as a result of the higher fuel injection pressure at higher engine loads, resulting from the use of the converted common-rail system, and consequently, decreasing the BSCO emissions. An alternative explanation is that this occurred owing to the fact that the air-fuel ratio is too lean for complete combustion at low load conditions, leading to the higher BSCO emissions.23 The relative air-fuel ratio (λ) for various engine loads and fuel types is illustrated in Fig. 7. In short, λ is defined as the ratio of the actual air-fuel ratio to that of the stoichiometric air-fuel ratio required to completely burn the fuel delivered. The air-fuel ratio of the mixture affects the combustion phenomenon and the completeness of combustion, especially at the fuel lean zone. In fact, the general trend indicates that the variations in BSCO emissions were very similar to the variation in λ values. In addition, it appears that even under high load conditions (i.e., 0.6 MPa of engine load), the λ values were still above unity, indicating a lean combustion process. Additionally, the addition of methyl ester in the blend creates a slightly rich combustion process. Consequently, the ID becomes shorter, combustion duration increases, and combustion gets completed properly, leading to a further decrease in CO emissions.
 |
| | Fig. 6 BSCO with the Jatropha biodiesel blends compared with diesel fuel at various BMEP. | |
 |
| | Fig. 7 Relative air-fuel ratio with the Jatropha biodiesel blends compared with diesel fuel at various BMEP. | |
As discussed earlier, fuel injection technology in biodiesel engine has significant effects on NOx emissions. Unlike the conventional mechanical pump-line-nozzle injection system, the modified common-rail injection system in the present study eliminated the common issue of advanced injection timing owing to the relatively higher viscosity of biodiesel. Hence, the correlation of other effects on the variation of NOx emissions when using biodiesel can be analysed more comprehensibly. In automotive exhaust emissions, the formation of NOx depends on the fuel type, fuel properties, and engine operating conditions.24 In the literature, most researchers have reported an increases of NOx emissions with the use of methyl ester blended fuel.12,25,26 The explanations given are primarily based on the higher oxygen content, which results in a higher combustion temperature that promotes a thermal NOx formation pathway. However, some researchers have reported the opposite trend, with lower NOx emissions when using methyl ester blended fuel.27,28 This is in good agreement with the results obtained throughout this study. As illustrated in Fig. 8, the presence of JME decreased the BSNOx relative to baseline diesel, but it did not decrease further as the degree of JME blending increased. The largest recorded reduction in BSNOx was approximately 20.2% for the JB30 blend at BMEP of 0.2 MPa. This can be attributed to the relatively lower calorific value of the methyl ester fuels being used and, consequently, reduced HRR in the premix combustion region and lower peak combustion temperature.29 Additionally, the results also suggest that further increases in the methyl ester concentration to JB50 resulted in an increase in BSNOx across all engine loads. A similar result has been observed by Mueller et al.30 who reported that the higher cetane number of biodiesel blends relative to diesel (see Table 2) causes ignition to occur earlier in the cycle. This allows the combustion products to have a longer residence time at high temperatures, which increases NOx emissions. Another possible reason may be associated with the reduction in the heat dissipation by radiation as a consequence of the large reductions of soot emitted with the use of biodiesel (see Fig. 10), resulting in an increase in BSNOx emissions.31
 |
| | Fig. 8 Variations in BSNOx emissions with different engine loads and fuel types. | |
 |
| | Fig. 9 EGT with the Jatropha biodiesel blends compared with diesel fuel at various BMEP. | |
 |
| | Fig. 10 Smoke opacity value with the Jatropha biodiesel blends compared with diesel fuel at various BMEP. | |
Owing to the lean operation and higher expansion ratio of the diesel engine, the EGT is typically lower than a petrol engine. A higher EGT is unfavourable as this will deteriorate engine fuel economy by discharging some of the useful energy into waste exhaust thermal energy, as well as causing thermal damage to piston components. As illustrated in Fig. 9, in general, the EGT increased with an increase in the BMEP for all of the fuels tested in this study. Additionally, the presence of JME increased the EGT relative to that for baseline diesel, but it did not increase further as the degree of JME blending rose to JB50. The highest achievable EGT for JB30, JB10, and baseline diesel at a BMEP of 0.6 MPa were 494.2 °C, 489.6 °C, and 487 °C, respectively. Additionally, the EGT is lower for higher blends of JB50 because of the improved combustion provided by the biodiesel under all engine loading conditions. In fact, many researchers have also reported that the EGT is lower with the engine fuelled with biodiesel blended fuel compared to the baseline diesel.11,32–34 In general, this phenomenon is caused primarily by the lower calorific value and the existence of chemically bound oxygen in biodiesel blends, which reduces the total energy released and improves the combustion, respectively. The EGT was thereafter decreased.
Smoke is an unwanted by-product of combustion in compression ignition diesel engines, which is primarily formed through incomplete combustion of hydrocarbon fuel. In general, the smoke from the exhaust tailpipe is emitted visibly in the form of dark black smoke. The composition of smoke highly depends on the type of fuel, engine operating conditions, and carbon residue of the fuel. The emission of smoke opacity is demonstrated in Fig. 10 for different Jatropha biodiesel fuel blends. In relation to the effect of biodiesel content on the smoke opacity, it was observed that the smoke opacity generally tended to decrease as the blending ratio of biodiesel in the fuel blend increased. It was observed that the maximum reduction was 69.5% with JB50 at a BMEP of 0.5 MPa, while it was a 21.9% reduction with JB10 at a BMEP of 0.1 MPa when both were compared with their corresponding baseline diesel. The combined effects of lower impurities, higher oxygen, and lower sulphur content of methyl ester fuels are believed to be responsible for the decreased smoke opacity level.21
4.3. Combustion characteristics
To evaluate the effect of the biodiesel blending ratio on the combustion characteristics, the cylinder pressures for 100 consecutive combustion cycles were recorded and compared at various loads and at a constant engine speed of 1600 rpm. The in-cylinder pressure, AHRR and injector current traces under a BMEP of 0.1 MPa (low load) and 0.6 MPa (high load) are illustrated in Fig. 11. As can be observed, the variation of engine load and biodiesel blending ratio had the greatest effect on the combustion characteristics. Under low load conditions, the premixed combustion process dominated. Conversely, the diffusion flame combustion process dominated at high load conditions. In the case of lower engine load, the cylinder pressure profiles for all of the tested fuels were comparable with the baseline diesel. Additionally, the shift in the SOC timing was consistent with the change of biodiesel concentration in the blend. In fact, the combustion event was shifted earlier toward TDC as the biodiesel concentration was increased. A small reduction in peak pressure in the range of 0.6–1.2 bar was observed for the operation with the biodiesel blend fuels. This can be attributed to the marginal decrease in the AHRR during the premixed combustion phase. In the case of the higher engine load, more significant variations in terms of peak pressure were observed among all biodiesel blends and baseline diesel fuel. It was observed that JB50 achieved the highest peak pressure of 74.4 bar followed by JB30 (73.6 bar), JB10 (72.1 bar), and diesel (71.6 bar). The results suggest that adding biodiesel in the blend caused increases in the peak pressure and shifted the location of occurrence earlier toward the TDC point. This can be attributed to the prominent advance in SOC timing, which caused the earlier rise of the AHRR and thus increased the in-cylinder gas pressure.
 |
| | Fig. 11 In-cylinder pressure and AHRR versus crank angle for tested fuels at a BMEP of (a) 0.1 MPa and (b) 0.6 MPa. | |
Another interesting observation that can be made from the AHRR diagram is the variations in ID. Mathematically, ID is defined as the crank angle interval measured from the start of fuel injection timing to the start of combustion timing; this is typically determined from the fuel injector signal and AHRR data, respectively. As summarized in Table 6, it was found that, in general, regardless of the engine load, most of the biodiesel blends exhibited shorter ID than baseline diesel owing to their relatively higher cetane number. Similar trends of shorter IDs with biodiesel blends were also reported by Ozsezen et al.35 As illustrated in Fig. 12, lines indicating the mass fraction burned of 10% (CA10), 50% (CA50), and 90% (CA90) were marked. Empirically, 10% and 90% lines marked the start and end of the main combustion duration, respectively. The period between CA10 and CA90 was defined as the combustion duration and this is typically measured in the unit of crank angle. From the results presented in Table 6, it can be seen that the general trend that indicates a shorter combustion duration was obtained with the addition of biodiesel in the blends for all engine loads. In fact, in comparison with the corresponding baseline diesel, the JB50 blend promoted a faster combustion duration by an average of 0.125° CA and 2° CA at engine load of 0.1 MPa and 0.5 MPa, respectively. The oxygen enrichment and improved combustion process of the JB50 blend are postulated to be the reason for the shorter combustion duration. Additionally, it can be observed that the CA50 shifted according to the change in the blend of the biodiesel. Typically, CA50 was used as a parameter that affected the ensemble heat release profile, and it was applied widely in a simulation of engine performance when the Wiebe function was employed.36 In the present study, it can be observed that the CA50 timing occurred slightly earlier with biodiesel blended fuels under all operating conditions. In fact, the largest shift in CA50 was found to be an advance of 1.375° CA for JB50 in comparison with that of baseline diesel at a higher load of 0.6 MPa.
Table 6 Crank angle position corresponding to certain percent mass fraction burned for all tested fuels under various BMEPs
| BMEP (MPa) |
Fuel type |
Start of injection, SOI (°ATDC) |
Start of combustion, SOC (°ATDC) |
ID (°CA) |
Crank angle for certain percent mass fraction burned (°ATDC) |
Combustion duration (°CA) |
| 10% |
50% |
90% |
| 0.1 |
D |
−7.000 |
−0.250 |
6.750 |
3.000 |
4.875 |
17.375 |
14.375 |
| JB10 |
−7.000 |
−0.250 |
6.750 |
3.000 |
4.875 |
17.375 |
14.375 |
| JB30 |
−7.000 |
−0.375 |
6.625 |
2.750 |
4.625 |
17.125 |
14.375 |
| JB50 |
−7.000 |
−0.375 |
6.625 |
2.375 |
4.375 |
16.625 |
14.250 |
| 0.2 |
D |
−7.000 |
−0.500 |
6.500 |
2.875 |
6.000 |
20.000 |
17.125 |
| JB10 |
−7.000 |
−0.500 |
6.500 |
2.625 |
6.000 |
19.750 |
17.125 |
| JB30 |
−7.000 |
−0.500 |
6.500 |
2.625 |
6.000 |
19.500 |
16.875 |
| JB50 |
−7.000 |
−0.625 |
6.375 |
2.375 |
5.750 |
18.750 |
16.375 |
| 0.3 |
D |
−7.000 |
−0.750 |
6.250 |
2.250 |
5.750 |
20.625 |
18.375 |
| JB10 |
−7.000 |
−0.750 |
6.250 |
2.250 |
5.750 |
20.500 |
18.250 |
| JB30 |
−7.000 |
−0.750 |
6.250 |
2.125 |
5.500 |
20.375 |
18.250 |
| JB50 |
−7.000 |
−0.875 |
6.125 |
1.875 |
5.500 |
20.000 |
18.125 |
| 0.4 |
D |
−7.000 |
−0.875 |
6.125 |
2.250 |
7.250 |
23.750 |
21.500 |
| JB10 |
−7.000 |
−0.875 |
6.125 |
2.125 |
7.125 |
23.375 |
21.250 |
| JB30 |
−7.000 |
−0.875 |
6.125 |
2.125 |
7.125 |
23.375 |
21.250 |
| JB50 |
−7.000 |
−1.000 |
6.000 |
1.875 |
7.125 |
22.500 |
20.625 |
| 0.5 |
D |
−7.000 |
−1.000 |
6.000 |
2.125 |
9.000 |
26.625 |
24.500 |
| JB10 |
−7.000 |
−1.000 |
6.000 |
2.125 |
9.000 |
26.625 |
24.500 |
| JB30 |
−7.000 |
−1.000 |
6.000 |
2.125 |
8.875 |
26.250 |
24.125 |
| JB50 |
−7.000 |
−1.125 |
5.875 |
1.750 |
8.125 |
24.250 |
22.500 |
| 0.6 |
D |
−7.000 |
−1.000 |
6.000 |
2.000 |
9.375 |
27.625 |
25.625 |
| JB10 |
−7.000 |
−1.125 |
5.875 |
1.750 |
9.375 |
27.375 |
25.625 |
| JB30 |
−7.000 |
−1.250 |
5.750 |
1.500 |
8.875 |
27.000 |
25.500 |
| JB50 |
−7.000 |
−1.625 |
5.375 |
1.000 |
8.000 |
25.625 |
24.625 |
 |
| | Fig. 12 Variations in mass fraction burned for diesel and biodiesel blends at a BMEP of 0.1 MPa. | |
4.4. Vibration analysis
Vibration signals in an internal combustion engine are usually used as a diagnostic tool. They allow engine bearing to be monitored for wear and overall engine knock detection. There are many sources of vibration in an engine, including piston slap, faults in valves, knocking, burning pressure oscillation, torsional vibration, and the rotation of other engine accessories. This vibration is transmitted via a variety of paths and then ultimately radiated acoustically to the surroundings. Exposure to excess vibration can accelerate wear and tear of mechanical components and have an adverse impact on human comfort. The combustion process in a diesel engine has an effect on the engine vibration. The methyl ester fuel blends influenced the combustion process and consequently, the noise and vibration. Fig. 13 is an illustration of RMS of the vibration acceleration signal for all tested fuels, which was calculated according to eqn (6). For each test, the average RMS of the acceleration signal for 100 successive engine combustion cycles was considered. The general trend indicates that the variations in RMS of acceleration are decreased with engine load. Additionally, the results also indicate that the RMS of acceleration is affected by biodiesel fuel blends. It was observed that the JB50 blend consistently resulted in the lowest RMS of accelerations in comparison with the baseline diesel under all loading conditions. It is interesting to note that the largest reduction of 11.3% in RMS of acceleration was obtained with JB50 at engine load of 0.5 MPa in comparison with the baseline diesel.
 |
| | Fig. 13 Variations in RMS of acceleration for diesel and biodiesel blends at different engine loads. | |
5. Conclusions
In the present study, the performance, emissions and combustion characteristics of an engine fuelled with fossil diesel fuel and Jatropha biodiesel blends were investigated at engine loads of 0.1, 0.2, 0.3, 0.4, 0.5, and 0.6 MPa. The following main conclusions can be drawn from this investigation.
A prominent increase in BSFC was observed at all load conditions when biodiesel fuel was used. Additionally, there was a marginal improvement of 2% in BTE with the JB50 blend at an engine load of 0.5 MPa. In terms of exhaust emissions, it was observed that the engine load had the greatest effect on BSCO emissions. In general, the BSCO emissions decreased with the increasing biodiesel blend ratio and engine load. Additionally, it was found that, in general, the BSNOx emissions decreased with increases in engine load and biodiesel blending ratio. The presence of JME decreased the BSNOx relative to the baseline diesel, but the BSNOx did not decrease further as the degree of JME blending increased. The largest reduction in BSNOx recorded was approximately 20.2% for the JB30 blend at a BMEP of 0.2 MPa. It is worth noting that smoke emissions from the Jatropha biodiesel blends were lower than baseline diesel across all the engine loading conditions. It was observed that the maximum reduction, in comparison with the corresponding baseline diesel, was 69.5% with JB50 at a BMEP of 0.5 MPa, while a 21.9% reduction was found with JB10 at a BMEP of 0.1 MPa.
In terms of the combustion characteristics, it was found that, at a lower engine load of 0.1 MPa, most of the biodiesel blends have lower peak pressure in the range of 0.6–1.2 bar in comparison with the baseline diesel. In the case of higher engine loads, more significant variations and higher peak combustion pressures were observed among all biodiesel blends and baseline diesel fuel. In fact, the location of occurrence is shifted earlier towards the TDC point. Furthermore, it was observed that, in general, the peak AHRR for biodiesel blends is lower at low load operation and is comparable at high load operation in comparison with baseline diesel. In addition, a slightly shorter ID and faster combustion duration were also revealed with the use of biodiesel blends across the engine load operations.
For vibration analysis, the results indicated that the RMS of acceleration was affected by biodiesel fuel blends. It was observed that the largest reduction of 11.3% in the RMS of acceleration was obtained with JB50 at an engine load of 0.5 MPa in comparison with the baseline diesel.
Nomenclature and symbol
| AHRR | Apparent heat release rate |
| ASTM | American society for testing and materials |
| ATDC | After top dead centre |
| B10 | 10% biodiesel + 90% diesel fuel |
| B100 | 100% biodiesel fuel |
| BMEP | Brake mean effective pressure |
| BP | Brake power |
| BSCO | Brake specific carbon monoxide |
| BSFC | Brake specific fuel consumption |
| BSNOx | Brake specific nitrogen oxides |
| BTDC | Before top dead centre |
| BTE | Brake thermal efficiency |
| CA | Crank angle |
| CA10 | Burn point of 10% |
| CA50 | Burn point of 50% |
| CA90 | Burn point of 90% |
| CI | Compression ignition |
| CIB | Calophyllum inophyllum |
| CO | Carbon monoxide |
| CO2 | Carbon dioxide |
| D | Fossil diesel |
| DI | Direct injection |
| ECU | Electronic control unit |
| EGT | Exhaust gas temperature |
| GC-FID | Gas chromatograph-flame ionization detector |
| FC | Fuel consumption |
| HC | Hydrocarbons |
| HPCR | High pressure common-rail |
| HRR | Heat release rate |
| ID | Ignition delay |
| IDI | Indirect injection |
| JB | Jatropha biodiesel |
| JB10 | 10% Jatropha biodiesel + 90% diesel fuel |
| JB30 | 30% Jatropha biodiesel + 70% diesel fuel |
| JB50 | 50% Jatropha biodiesel + 50% diesel fuel |
| JME | Jatropha methyl ester |
| KB | Karanja biodiesel |
| λ | Relative air-fuel ratio (Lambda) |
| MPLN | Mechanical pump-line-nozzle |
| NA | Naturally aspirated |
| NOx | Nitrogen oxides |
| PB | Polanga biodiesel |
| ppm | Part per million |
| rpm | Revolution per minute |
| RMS | Root mean square |
| TC | Turbocharged |
| TDC | Top dead centre |
| THC | Total hydrocarbon |
Acknowledgements
The authors would like to acknowledge the Ministry of Higher Education (MOHE) of Malaysia and University of Malaya for financial support through UMRG (grant number RG145-12AET), HIR grant (UM.C/HIR/MOHE/ENG/07), and Postgraduate Research Grant (PPP) (grant number PG035-2012B).
References
- Y. Sharma and B. Singh, Renewable Sustainable Energy Rev., 2009, 13, 1646–1651 CrossRef CAS PubMed.
- A. Ahmad, N. Yasin, C. Derek and J. Lim, Renewable Sustainable Energy Rev., 2011, 15, 584–593 CrossRef CAS PubMed.
- T. Ganapathy, R. P. Gakkhar and K. Murugesan, Appl. Energy, 2011, 88, 4376–4386 CrossRef CAS PubMed.
- C. Carraretto, A. Macor, A. Mirandola, A. Stoppato and S. Tonon, Energy, 2004, 29, 2195–2211 CrossRef CAS PubMed.
- A. Atabani, A. Silitonga, I. A. Badruddin, T. Mahlia, H. Masjuki and S. Mekhilef, Renewable Sustainable Energy Rev., 2012, 16, 2070–2093 CrossRef PubMed.
- A. Demirbas, Energy Convers. Manage., 2009, 50, 14–34 CrossRef CAS PubMed.
- B. S. Chauhan, N. Kumar and H. M. Cho, Energy, 2012, 37, 616–622 CrossRef CAS PubMed.
- H. C. Ong, H. H. Masjuki, T. M. I. Mahlia, A. S. Silitonga, W. T. Chong and K. Y. Leong, Energy Convers. Manage., 2014, 81, 30–40 CrossRef CAS PubMed.
- S. Vedharaj, R. Vallinayagam, W. M. Yang, S. K. Chou, K. J. E. Chua and P. S. Lee, Energy Convers. Manage., 2013, 75, 773–779 CrossRef CAS PubMed.
- H. Raheman and S. V. Ghadge, Fuel, 2007, 86, 2568–2573 CrossRef CAS PubMed.
- H. An, W. Yang, S. Chou and K. Chua, Appl. Energy, 2012, 99, 363–371 CrossRef CAS PubMed.
- P.-q. Tan, Z.-y. Hu, D.-m. Lou and Z.-j. Li, Energy, 2012, 39, 356–362 CrossRef CAS PubMed.
- M. Lapuerta, O. Armas and J. Rodríguez-Fernández, Prog. Energy Combust. Sci., 2008, 34, 198–223 CrossRef CAS PubMed.
- D. M. Leahey, B. C. Jones, J. W. Gilligan, L. P. Brown, L. J. Hamilton, C. E. Gutteridge, J. S. Cowart and P. A. Caton, SAE International, 2007-01-4011, 2007 Search PubMed.
- S. Lim and L. K. Teong, Renewable Sustainable Energy Rev., 2010, 14, 938–954 CrossRef CAS PubMed.
- M. Mofijur, H. H. Masjuki, M. A. Kalam, M. A. Hazrat, A. M. Liaquat, M. Shahabuddin and M. Varman, Renewable Sustainable Energy Rev., 2012, 16, 5007–5020 CrossRef PubMed.
- J. Ghojel and D. Honnery, Appl. Therm. Eng., 2005, 25, 2072–2085 CrossRef CAS PubMed.
- M. Canakci, A. N. Ozsezen and A. Turkcan, Biomass Bioenergy, 2009, 33, 760–767 CrossRef CAS PubMed.
- T. T. Kivevele, L. Kristóf, Á. Bereczky and M. M. Mbarawa, Fuel, 2011, 90, 2782–2789 CrossRef CAS PubMed.
- J. B. Heywood, Internal Combustion Engine Fundamentals, McGraw-Hill, New York, 1988 Search PubMed.
- M. Canakci, A. N. Ozsezen, E. Arcaklioglu and A. Erdil, Expert Syst. Appl., 2009, 36, 9268–9280 CrossRef PubMed.
- E. Buyukkaya, S. Benli, S. Karaaslan and M. Guru, Energy Convers. Manage., 2013, 69, 41–48 CrossRef CAS PubMed.
- H. G. How, H. H. Masjuki, M. A. Kalam and Y. H. Teoh, Energy, 2014, 69, 749–759 CrossRef CAS PubMed.
- J. P. Szybist, A. L. Boehman, J. D. Taylor and R. L. McCormick, Fuel Process. Technol., 2005, 86, 1109–1126 CrossRef CAS PubMed.
- P. Kwanchareon, A. Luengnaruemitchai and S. Jai-In, Fuel, 2007, 86, 1053–1061 CrossRef CAS PubMed.
- S. Saravanan, G. Nagarajan, G. Lakshmi Narayana Rao and S. Sampath, Energy, 2010, 35, 94–100 CrossRef CAS PubMed.
- M. Dorado, E. Ballesteros, J. Arnal, J. Gomez and F. Lopez, Fuel, 2003, 82, 1311–1315 CrossRef CAS.
- C. L. Peterson and D. L. Reece, SAE International, 961114, 1996 Search PubMed.
- J. Huang, Y. Wang, J.-b. Qin and A. P. Roskilly, Fuel Process. Technol., 2010, 91, 1761–1767 CrossRef CAS PubMed.
- C. J. Mueller, A. L. Boehman and G. C. Martin, SAE Int. J. Fuels Lubr., 2009, 2, 789–816 CAS.
- A. Cheng, A. Upatnieks and C. Mueller, Int. J. Engine Res., 2006, 7, 297–318 CrossRef CAS.
- C. Enweremadu and H. Rutto, Renewable Sustainable Energy Rev., 2010, 14, 2863–2873 CrossRef CAS PubMed.
- C.-Y. Lin and R.-J. Li, Fuel Process. Technol., 2009, 90, 883–888 CrossRef CAS PubMed.
- O. Özener, L. Yüksek, A. T. Ergenç and M. Özkan, Fuel, 2014, 115, 875–883 CrossRef PubMed.
- A. N. Ozsezen and M. Canakci, Energy Convers. Manage., 2011, 52, 108–116 CrossRef CAS PubMed.
- X. Wang, Y. Ge, L. Yu and X. Feng, Fuel, 2013, 107, 852–858 CrossRef CAS PubMed.
|
| This journal is © The Royal Society of Chemistry 2014 |
Click here to see how this site uses Cookies. View our privacy policy here. ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Hz. Engine vibration motion in the lateral (y) axis (or perpendicular to cylinder axis) was chosen for monitoring vibrations. To sense the magnitude of vibration in this direction, the accelerometer was mounted on the engine body with an adhesive mounting base. The output signal from the sensor was connected to a constant current single channel signal conditioner (PCB model 480C02) with unity gain. In each test, engine-block vibration signals for a total of 100 consecutive combustion cycles at 0.125° CA resolution were recorded and the averaged RMS was calculated.
000 Hz. Engine vibration motion in the lateral (y) axis (or perpendicular to cylinder axis) was chosen for monitoring vibrations. To sense the magnitude of vibration in this direction, the accelerometer was mounted on the engine body with an adhesive mounting base. The output signal from the sensor was connected to a constant current single channel signal conditioner (PCB model 480C02) with unity gain. In each test, engine-block vibration signals for a total of 100 consecutive combustion cycles at 0.125° CA resolution were recorded and the averaged RMS was calculated.
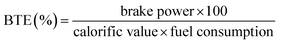
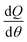 , at each crank angle was obtained from the first law of thermodynamics, and it can be calculated by the following formula:
, at each crank angle was obtained from the first law of thermodynamics, and it can be calculated by the following formula: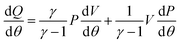
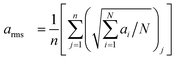
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm
000 rpm![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 kPa
000 kPa![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm
000 rpm![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)













