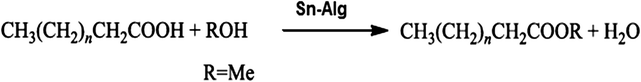
A green approach for the production of biodiesel from fatty acids of corn deodorizer distillate†
Saba Nazabc,
Huseyin Karaab,
Syed Tufail Hussain Sherazi*abd,
Abdalaziz Aljaboure and
Farah Naz Talpurc
aSelcuk University, Faculty of Science, Department of Chemistry, Campus 40275, Konya, Turkey
bNecmettin Erbakan University, Faculty of Science, Department of Biotechnology, Konya, Turkey
cDr M. A. Kazi Institute of Chemistry, University of Sindh, Jamshoro, Pakistan
dNational Centre of Excellence in Analytical Chemistry, University of Sindh, Jamshoro, Pakistan. E-mail: tufail.sherazi@yahoo.com; Fax: +92 22923431; Tel: +92 229213430
eAdvanced Technology Research and Application Center 42075, Konya, Turkey
First published on 16th September 2014
Abstract
A novel alginic acid derived tin catalyst, tin alginate (Sn–Alg), was successfully synthesized, characterized and applied for methyl esterification. Initially, the amount of catalyst, methanol to fatty acid ratio and reaction time were optimized using an oleic acid standard for esterification. The optimal reaction conditions were found to be 4% catalyst, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 oleic acid to methanol mole ratio and 2 h reaction time with 98.7% fatty acid methyl ester recovery. The capability of Sn–Alg beads to esterify the fatty acids of corn deodorizer distillate was evaluated. High recovery (97.6%) of esters was obtained after 8 cycles using reprocessed catalyst under the optimized parameters. The results of the present study indicated that based on the environmental pollution, reusability, avoiding the use of potassium or sodium hydroxides or sulphuric or phosphoric acids, and ease of catalyst separation, the solid Sn–Alg catalyst has a great potential for biodiesel production from highly free fatty acid deodorizer distillates.
12 oleic acid to methanol mole ratio and 2 h reaction time with 98.7% fatty acid methyl ester recovery. The capability of Sn–Alg beads to esterify the fatty acids of corn deodorizer distillate was evaluated. High recovery (97.6%) of esters was obtained after 8 cycles using reprocessed catalyst under the optimized parameters. The results of the present study indicated that based on the environmental pollution, reusability, avoiding the use of potassium or sodium hydroxides or sulphuric or phosphoric acids, and ease of catalyst separation, the solid Sn–Alg catalyst has a great potential for biodiesel production from highly free fatty acid deodorizer distillates.
1. Introduction
As the world begins to accept the changes towards green energy, biofuels stand at the forefront of this ideology. Biodiesel is rapidly evolving as a sustainable attractive alternative source of petroleum fuel because of its environmental friendly, renewable and green properties.1 Biodiesel is a low carbon exhaust emission neutral fuel that can be prepared by either the transesterification of triglycerides (the main component of vegetable oils or animal fats) or the esterification of free fatty acids (FFAs) with a short chain alcohol (mainly methanol).2 In recent years, biodiesel has gained progressively increasing attention as one of the most promising alternative to offset the negative environmental effects resulting from the overconsumption of non-sustainable fossil fuels.3 Currently, more than 95% of the world's biodiesel is mainly prepared from commercially available vegetable oils, such as soybean, rapeseed, sunflower and palm, which leads to food versus fuel disputes.4 However, the demand of vegetable oils for food supply has enormously increased because of the growing population, it is difficult to justify the use of these oils for biodiesel production. Furthermore, these oils could be more expensive when used as fuel.5Therefore, the contribution of non-edible plant oil and the use of waste vegetable oil is promising for biodiesel production. This is an effective way of resolving the conflict between food supply and fuel besides reducing the cost of raw material.6 In addition, substantial attention has been paid to inexpensive feedstocks such as vegetable oil, palm oil fatty acid distillates (POFAD) and vegetable oil deodorizer distillate (VODD).7,8
Deodorizer distillate (DD) is a waste produced during the deodorization process of vegetable oils. Mainly, it contains free fatty acids (FFAs; 30–60% w/w), sterols, sterol esters (10–30%, w/w), hydrocarbons (10–30%, w/w, mainly squalene), and glycerides (10–20%, w/w, mainly mono- and diglycerides), along with several other minor substances. Owing to its high content of free fatty acids, this product can be used as an excellent inexpensive source of biodiesel.9 Conventionally, homogeneous acid and basic catalysts (such as H2SO4, H3PO4, NaOH, KOH) are most frequently used for the preparation of biodiesel; however, these types of catalytic systems have several drawbacks, such as environmental pollution, generation of considerable waste water during product washing, long reaction time, difficulty in separation and purification of target product, and equipment corrosion that severely hampers the commercialization of the relevant process.7,10 In view of the difficulties faced with homogeneous catalysts, research on the development of heterogeneous catalysts was escalated in several academic and industrial laboratories.11–13 These types of catalysts offer several distinct advantages related to the simplicity for the continuous production methods over batch methods. The development of a solid catalyst with inexpensive and reusable materials has great potential in this specific research area.13
Trakarnpruk et al.14 reported biodiesel production from palm fatty acids distillate (PFAD) containing 93% of FFAs using two catalyst systems, i.e., tungstophosphoric acid and Cs-salt immobilized-silica. Relatively, tungstophoshoric acid was reported to be a good catalyst with 96.7% recovery of FAME conversion at 85 °C in a 15 h reaction time with 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio of methanol to PFAD and 15 wt% catalyst. Boey et al.15 used ferric alginate beads as a solid acid catalyst for the esterification of lauric acid with methanol using a response surface methodology for optimization. The optimized parameters (3 h reaction time, 15
1 molar ratio of methanol to PFAD and 15 wt% catalyst. Boey et al.15 used ferric alginate beads as a solid acid catalyst for the esterification of lauric acid with methanol using a response surface methodology for optimization. The optimized parameters (3 h reaction time, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol to PFAD molar ratio) were used for the esterification of PFAD, which brings about 88.8% methyl esters conversion. An alginic acid-derived aluminum-alginate solid acid catalyst was synthesized by Qiuyun et al.10 and exhibited high catalytic activity with 92.6% conversion of methyl oleate in the presence of 4% catalyst after refluxing for 3 h in methanol and acid mixed at a molar ratio of 10
1 methanol to PFAD molar ratio) were used for the esterification of PFAD, which brings about 88.8% methyl esters conversion. An alginic acid-derived aluminum-alginate solid acid catalyst was synthesized by Qiuyun et al.10 and exhibited high catalytic activity with 92.6% conversion of methyl oleate in the presence of 4% catalyst after refluxing for 3 h in methanol and acid mixed at a molar ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.
Based on the aforementioned literature information, a novel tin-alginate catalyst was prepared from a cost-effective and risk-free sodium alginate polymer, which was used for the esterification reaction of oleic acid with methanol for optimization and afterward applied for the esterification of corn deodorizer distillates (CDD). Finally, the reusability of Sn–Alg was evaluated.
2. Material and methods
2.1. Chemicals
Oleic acid (99%) was obtained from Fluka. Tin chloride (SnCl2)·2H2O and sodium alginate was supplied by Sigma-Aldrich Chemie GmbH (USA). Methanol and n-hexane were provided by Fisher Chemical. Anhydrous sodium sulfate (99%) was purchased from Sigma-Aldrich Corporation (USA). All the chemicals used were of analytical grade. The chromatographic methyl ester standard, methyl heptadecanoate was supplied by Sigma-Aldrich Chemie GmbH (USA). The deodorizer distillate samples were obtained from Zade edible oil refining industries (Konya, Turkey).2.2. Preparation of tin-alginate beads for esterification
Tin-alginate (Sn–Alg) beads were prepared according to the literature.15 About 4 g of sodium alginate was added to 200 mL of distilled water (heated to 60 °C), and stirred until a clear viscous solution was obtained. Then, the solution was allowed to cool at room temperature. The viscous solution was added drop-wise using a Pasteur pipette into a 0.1 mol L−1 solution of (SnCl2)·2H2O at room temperature (28–30 °C). The Sn–Alg beads formed were left for 2 h to equilibrate in the (SnCl2)·2H2O solution. Finally, the beads were washed and dried in an oven at 60 °C for 2 days.2.3. Characterization
The amount of Sn metal in the Sn–Alg beads was evaluated using a Perkin Elmer SCIEX inductively coupled plasma mass spectrometer (ICP-MS) with ELAN DRC-e technology (USA) using the calibration model of standard metal. The chemical composition of the prepared Sn–Alg solid acid catalyst was characterized by scanning electron microscopy (SEM) coupled with an energy dispersive X-ray (EDX) detector EVO-LS 10 (Carl Zeiss, Germany), and X-ray diffraction (XRD) on Bruker Advance D8 XRD, Germany model using CuKα radiation (α = 1.5417 Å). Functional group characterization of Sn–Alg beads was performed on a Vertex 70 (Bruker, Germany) model armed with a Platinum ATR Diamond accessory. The thermostability of the synthesized Sn–Alg catalyst was determined with a 5–9 mg sample on a SETARAM thermogravimetric analyzer (Setsys Evolution, France). The analysis temperature was ranged 25–1000 °C at a heating rate of 10 °C min−1 under oxygen atmosphere with a gas flow rate of 20 mL min−1. The acidic strength of the Sn–Alg beads was verified using 0.1 g of dried beads placed in 5 mL of anhydrous methanol and one drop of 0.1% Hammett indicator (crystal violet, bromophenol blue, methyl red and neutral red), and then left to equilibrate for 2 h. All the color changes were noted.72.4. Esterification of corn deodorizer distillate (CDD)
On the basis of the optimized results, 2.5 g of corn deodorizer distillate (CDD) was subjected to an esterification reaction at 60°C for 2 h with Sn–Alg beads. The initial acid value of CDD was determined using the AOCS Cd 3d-63 method.16 The main fatty acid composition of CDD was 16.17%, which comprised of the following: palmitic acid, 2.05%; palmitelaidic acid, 0.32%; palmitoleic acid, 5.37%; stearic acid, 30.93%; elaidic acid, 0.74%; oleic acid, 42.12%; linoleic acid, 2.17%; and linolenic acid 0.14%.2.5. Esterification reaction
An appropriate amount of oleic acid, methanol and Sn–Alg catalyst were added to a round bottom flask and refluxed under optimized conditions. For comparison, oleic acid and methanol were also refluxed without the addition of the Sn–Alg catalyst under similar conditions. Then, the small aliquots of the sample were withdrawn from the reaction mixture to determine the esterification efficiency through their acid content by titration. The conversion of FFA was calculated based on the following eqn (1).10
 | (1) |
To confirm the good reproducibility of the reaction, the esterification experiments were conducted three times.
2.6. Gas chromatographic analysis
Gas chromatography (Agilent Technologies, 7890A GC System) equipped with a flame ionization detector (FID) was used for methyl oleate determination. Standard materials and samples of CDD were also analyzed by a gas chromatography-mass spectroscopy system. GC-MS analysis for FAMEs was performed on an Agilent 6890 N gas chromatography instrument coupled with an Agilent MS-5975 inert XL mass selective detector and an Agilent autosampler 7683-B injector (Agilent Technologies, Little Fall, NY, USA). For the identification of methyl esters, the confirmation of the GC-FID results, and to identify the peaks of unknown compounds, GC-MS was used a supportive technique. The content of fatty acid methyl esters (FAME) in the samples was determined using the European regulation procedure EN 14103 with a high cyanopropyl-containing polysiloxane HP-88 GC capillary column (100 m × 0.25 mm ID × 0.25 μm film thickness) using methyl heptadecanoate as an internal standard. The FAMEs content was calculated using eqn (2).17
 | (2) |
3. Result and discussion
3.1. Characterization of the Sn–Alg catalyst
Alginate is a copolymer composed of the alternating blocks of unbranched binary copolymers of β-D-mannuronic and α-L-guluronic acid through a 1,4-linkage. When the sodium salt of alginic acid was placed into the (SnCl2)·2H2O solution, gelatin like spherical white small globules were instantly formed because of the fast cross-linking process between the alginate and tin ions. According to Ruiz et al.,18 when divalent cations ionically interact with blocks of β-D-mannuronic and α-L-guluronic acid residues, a three-dimensional network is formed, which is usually described using the “egg-box” model.19The Sn–Alg beads were washed with deionized water to remove sodium chloride that was formed as a side product. The catalyst was stabilized in the presence of alginate crosslinking, which prevented it from hydrolysis.15
The result obtained for the detection of Sn metal through ICP–MS showed that the amount of Sn was 0.266 ± 0.005 g per g of catalyst beads. The acid strength of the Sn–Alg beads lies in the range between pH 1 and pH 3. This shows that the Sn–Alg beads are strongly acidic and are most applicable for the esterification reaction.
Scanning electron microscopy images of the Sn–Alg beads are shown in ESI Fig. 1,† which shows a clear surface morphology. The Sn–Alg beads displayed a core–shell structure with a smooth surface. The mean size of the dry beads was measured to be 1270 mm.
3.2. X-ray diffraction analysis
According to the literature, sodium alginate does not have any prominent peaks for the polymers, as predictable from polysaccharides.15 The X-ray diffraction pattern of the Sn–Alg beads are represented in Fig. 1, which shows two intense peaks of Sn metal at 2θ values of around 31.5° and 45.1°angels with other several broad signals of Sn metal cross linked with the hydroxyl group of alginic acid located at 2θ values of 27.3°, 33.2°, 36.5°, 51,5°, 51.5°, and 66.2°. The XRD results were comparable to the reported values.20,21 Sn(II) forms metal carboxylate bonds with alginate molecules during the ion exchange interaction. Sn(II) ions in the SnCl2 solution are exchanged with Na ions from the sodium alginate solution. The Sn(II) ions form bonds with the carboxylate groups of alginate molecules. The chloride ion will form bonds with sodium ions, which are then washed off. In Fig. 1, labelled SnO represents the signals of Sn cross linked with the hydroxyl group of alginic acid. It is not tin oxide, and the catalytic effect is due to the Sn–Alg beads.3.3. Thermogravimetric analysis
Thermogravimetric analysis (TGA) is an analytical technique that measures the volatilization of a sample by recording the mass loss as a function of temperature. Various studies have investigated the distinctive effects of different metals on the thermal stability of the alginate polymer.19 Fig. 2 shows the distinctive TG–DTG thermogram of the Sn–Alg beads operated under oxidative conditions. The TG–DTG thermogram of the Sn–Alg beads is closely analogous to the TG–DTG curves of Fe(III) alginate and calcium alginate thermogram reported by Boey et al.15 and Kong et al.22 respectively.The weight loss began around 50–150 °C because of the dehydration of the sample. The weight loss around 149–380 °C is because of the early degradation of alginate polymer, and the succeeding weight losses of approximately 380–560 °C and 560–895 °C are because of the additional putrefaction of the alginate polymer.23 From the thermogravimetric profile of Sn–Alg, it is clear that the catalyst beads remain pristine without the decomposition at the refluxing temperature (65 °C) of methyl alcohol.
3.4. FTIR studies
FTIR analysis of the alginate standard and Sn–Alg beads are presented in Fig. 3(a) and (b), respectively. The cross linking interaction between Sn(II) and alginate was investigated by FTIR functional groups analysis. The main peaks of sodium alginate appeared at 3225, 1594, 1402, and 1024 cm−1, which were assigned to νO–H, νsC=O, νasC=O, and νC–O groups, respectively. The broad band of the hydroxyl group observed around 3225 cm−1 in the sodium alginate and Sn–Alg beads spectra can be accredited to the cumulative effect of a large number of O–H groups present in the alginate polymer. A shift of the O–H broad bands to a higher frequency from 3225 to 3360 cm−1 was also observed, which was caused by the reduced hydrogen bond strength because of the presence of Sn(II) in the sample. The distinctive absorption bands in Fig. 3a at around 1594 cm−1 and 1402 cm−1 were assigned to the symmetric (νs) and asymmetric (νas) stretching vibrations of the free carboxylate group (–COOH), respectively. Conversely, the addition of Sn(II) solution in the alginate sample resulted in a shift in the free carboxylate group (–COOH) bands at 1594 cm−1 and 1402 cm−1 shifted towards higher frequencies 1621 cm−1 and 1407 cm−1 because of a cross linking interaction of the Sn(II) ion with the carboxylate group of the alginate polymer.24,15Cozzi et al.25 reported that a strong band appeared at 1726 cm−1 because of the stretching vibration of the free carbonyl group. This band was clearly seen in the Sn–Alg beads, but it was completely absent in the sodium alginate spectrum. Comparing spectrum of alginate and Sn–Alg beads another noticeable large absorption bands around 500–700 cm−1 in Sn–Alg beads are attributed to the Sn–O bond stretching vibrations, as reported by Wu et al.26
3.5. Optimization of esterification reaction catalyzed by Sn–Alg beads
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 to 1
6 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 on the methyl oleate conversion in the presence of 4 wt% of Sn–Alg catalyst for 2 h. As shown in ESI Fig. 2a,† the FFA conversion linearly increases with increasing methanol to oil molar ratio. Le Chatelier's principle play the main role in explaining the trend of the increase in the conversion rate of methyl oleate. By increasing the amount of methanol, the tendency of equilibrium shifted towards the right side, which favors more methyl oleate formation. The highest conversion 98.5% was achieved at a 1
25 on the methyl oleate conversion in the presence of 4 wt% of Sn–Alg catalyst for 2 h. As shown in ESI Fig. 2a,† the FFA conversion linearly increases with increasing methanol to oil molar ratio. Le Chatelier's principle play the main role in explaining the trend of the increase in the conversion rate of methyl oleate. By increasing the amount of methanol, the tendency of equilibrium shifted towards the right side, which favors more methyl oleate formation. The highest conversion 98.5% was achieved at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 oleic acid/methanol molar ratio. Essentially, the increase in the methanol to fatty acid molar ratio indicates the good mixing between the catalyst and reactants, which improves the mass transfer rate, ultimately resulting in higher conversion. On the other hand, a decrease in % conversion was observed when the oleic acid/methanol molar ratio exceeded 1
12 oleic acid/methanol molar ratio. Essentially, the increase in the methanol to fatty acid molar ratio indicates the good mixing between the catalyst and reactants, which improves the mass transfer rate, ultimately resulting in higher conversion. On the other hand, a decrease in % conversion was observed when the oleic acid/methanol molar ratio exceeded 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 to 1
12 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25. This might be because of the dilution effect that was caused by the addition of larger amounts of methanol.10
25. This might be because of the dilution effect that was caused by the addition of larger amounts of methanol.103.6. FAME production from FFA content of CDD using Sn–Alg catalyst
The most effective alginic acid derived Sn–Alg catalyst after the optimization studies was examined for the conversion of waste CDD containing a large amount of FFAs (86.3 wt%) in the presence of methanol to fatty acid methyl esters that produce biodiesel through simultaneous esterification and transesterification. The initial acid CDD value was found to be 166.32 ± 2.0 mg KOH g−1. The esterification of CDD with an optimized 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol to CDD molar ratio, 4 wt% of Sn–Alg catalyst to CDD at a 65 °C for 2 h yielded more than 96.8 ± 0.8% biodiesel.
1 methanol to CDD molar ratio, 4 wt% of Sn–Alg catalyst to CDD at a 65 °C for 2 h yielded more than 96.8 ± 0.8% biodiesel.In general, biodiesel is considered to be an environmental friendly fuel that is biodegradable, free from toxic components and does not produce poisonous exhaust gases, such as sulfur oxides or aromatic compounds.28 The efficacious application of the optimized reaction conditions using Sn–Alg beads for the esterification of CDD and obtaining the tremendous production of methyl esters confirmed that the Sn–Alg beads are appropriate as a heterogeneous solid acid catalysts for converting FFA CDD to biodiesel. Owing to its antimicrobial activity, Sn–Alg catalyst has little corrosiveness activity. Moreover, the ease of separation and reuse of the solid catalyst make it more economical. This process is quite simple with negligible environmental issues.
Fig. 4a shows a typical FAME chromatogram of CDD obtained with a Sn–Alg catalyzed esterification reaction and (b) with alkali catalyst (KOH) saponification. The difference in Fig. 4a and b indicates that Sn–Alg is reactive enough to convert FFAs comparable to corrosive alkali.
 | ||
| Fig. 4 Typical FAME GC chromatograms of CDD obtained by a catalyzed esterification reaction with Sn–Alg (a); with alkali catalyst (KOH) saponification (b). | ||
Some properties of the produced biodiesel, such as viscosity, acid value, density, and cetane number, are presented in Table 1, and compared with the standard values of American Society for Testing and Materials (ASTM). All the values are in good agreement with the standard specifications of biodiesel. However, the higher cetane number was found in the prepared CDD than the ASTM standards for biodiesel that specifies good ignition quality of biofuel. The higher cetane numbers of the biodiesel can decrease the NOx emissions and contribute to the absence of aromatic compounds from the biodiesels exhaust.29
| Properties | Waste CDD | FAME of CDD | ASTM 6751-11a Values | Biodiesel standard testing method |
|---|---|---|---|---|
| Density (g cm−3) | 0.97 | 0.85 | 0.86–0.90 | ASTM D4052 |
| Viscosity mm2 s−1 at 40 °C | 36.25 | 3.12 | 1.9–6.0 | ASTM D 445 |
| Acid value (mg KOH g−1) | 166.32 | 0.5–0.7 | 0.5 max | ASTM D 664 |
| Cetane number | 38.33 | 54.18 | 47 min | ASTM D 613 |
3.7. Reusability of Sn–Alg catalyst
The stability and reusability of the catalyst are essential to reduce the cost of biodiesel production. Therefore, successive experiments were performed to inspect the stability and reusability of the Sn–Alg catalyst after collecting catalyst beads from the reaction mixture and reprocessing them for further esterification reactions. The catalyst was reused without additional pretreatments, such as washing or drying. About 97.6% biodiesel was obtained up to 8 times using the reprocessed catalyst from CDD, as clearly displayed in Fig. 5. Despite this, slight decrease in the conversion efficiency was observed after 8 cycles of the esterification reaction. According to Boey et al.,15 this diminutive effect is possibly because of the repetitive heating and agitation of the beads due to the esterification reaction. In comparison to the literature,10 the Sn–Alg catalyst is stable enough to be reprocessed several times, easily handled and recovered, and there is no regeneration step necessary to renovate the acid ability of the catalyst; hence, decreasing the cost of the catalyst production and the cost of biodiesel production. | ||
| Fig. 5 Reusability of the Sn–Alg catalyst. About 97.6% biodiesel was obtained up to 8 times using the reprocessed catalyst from CDD. | ||
4. Conclusions
A newly synthesized Sn–Alg catalyst exhibited greater catalytic activity for the esterification of large amounts of FFAs from waste material such as CDD. Around 96.8% FFAs was converted to a worthwhile FAME product under the optimal reaction conditions; 4 wt% catalyst amount, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 oleic acid to methanol mole ratio and 2 h reaction time at a fixed temperature of 65 °C. Biodiesel is considered as an environmentally friendly fuel. For the preparation of biodiesel, common homogeneous catalysts, such as KOH, NaOH, H2SO4, and H3PO4, are used. Many problems, such as environmental pollution, difficulty in separating the catalyst and purified biodiesel, and corrosion of the equipment, are associated with these catalysts. Therefore, a green approach was applied using a Sn–Alg solid catalyst to convert waste corn oil deodorizer distillate to biodiesel. The prepared Sn–Alg catalyst can be easily separated from the product by simple filtration without washing and produce a high purity product.
12 oleic acid to methanol mole ratio and 2 h reaction time at a fixed temperature of 65 °C. Biodiesel is considered as an environmentally friendly fuel. For the preparation of biodiesel, common homogeneous catalysts, such as KOH, NaOH, H2SO4, and H3PO4, are used. Many problems, such as environmental pollution, difficulty in separating the catalyst and purified biodiesel, and corrosion of the equipment, are associated with these catalysts. Therefore, a green approach was applied using a Sn–Alg solid catalyst to convert waste corn oil deodorizer distillate to biodiesel. The prepared Sn–Alg catalyst can be easily separated from the product by simple filtration without washing and produce a high purity product.
Conflict of interest
The authors declare that they don't have any conflict of interest.Acknowledgements
The authors would like to thank TÜBİTAK (the Scientific and Technological Research Council of Turkey) for financial support to Miss Saba Naz under: 2216 program and Professor Dr S. T H. Sherazi under TUBITAK 2221 Fellowship for Visiting Scientists and Scientists on Sabbatical Leave program.References
- W. N. N. Wan Omar and N. A. Saidina Amin, Biomass Bioenergy, 2011, 35, 1329–1338 CrossRef PubMed.
- Q. Shu, J. Gao, Z. Nawaz, Y. Liao, D. Wang and J. Wang, Appl. Energy, 2010, 87, 2589–2596 CrossRef CAS PubMed.
- T.-H. Đặng and B.-H. Chen, Fuel Process. Technol., 2013, 109, 7–12 CrossRef PubMed.
- M. Balat, Energy Convers. Manage., 2011, 52, 1479–1492 CrossRef CAS PubMed.
- A. B. Chhetri, M. S. Tango, S. M. Budge, K. C. Watts and M. R. Islam, Int. J. Mol. Sci., 2008, 9, 169–180 CrossRef CAS PubMed.
- M. R. Uddin, K. Ferdous, M. R. Uddin, M. R. Khan and M. Islam, Chem. Eng. Sci., 2013, 1, 22–26 CrossRef CAS PubMed.
- P.-L. Boey, S. Ganesan, G. P. Maniam, M. Khairuddean and J. Efendi, Energy Convers. Manage., 2013, 65, 392–396 CrossRef CAS PubMed.
- L. Wang, W. Du, D. Liu, L. Li and N. Dai, J. Mol. Catal. B: Enzym., 2006, 43, 29–32 CrossRef CAS PubMed.
- S. Naz, S. T. H. Sherazi, F. N. Talpur, S. A. Mahesar and H. Kara, J. AOAC Int., 2012, 95, 1570–1573 CrossRef CAS PubMed.
- Z. Qiuyun, L. Hu, Q. Wenting, L. Xiaofang, Z. Yuping, X. Wei and Y. Song, China Pet. Process. Petrochem. Technol., 2013, 15, 19–24 Search PubMed.
- X. Liu, H. He, Y. Wang and S. Zhu, Catal. Commun., 2007, 8, 1107–1111 CrossRef CAS PubMed.
- A. B. Ferreira, A. Lemos Cardoso and M. J. da Silva, ISRN Renewable Energy, 2012, 142857, DOI:10.5402/2012/142857 , 13 pages.
- C. V. McNeff, L. C. McNeff, B. Yan, D. T. Nowlan, M. Rasmussen, A. E. Gyberg, B. J. Krohn, R. L. Fedie and T. R. Hoye, Appl. Catal., A, 2008, 343, 39–48 CrossRef CAS PubMed.
- W. Trakarnpruk, Walailak Journal of Science and Technology, 2012, 9, 37–47 Search PubMed.
- P.-L. Boey, S. Ganesan, G. P. Maniam, M. Khairuddean and S.-E. Lee, Appl. Catal., A, 2012, 433, 12–17 CrossRef PubMed.
- American Oil Chemists' Society, AOCS Official Method Cd 8d-63, in Official Methods and Recommended Practices of the AOCS, ed. D. Firestone, AOCS, Champaign, USA, 1973 Search PubMed.
- F. Gasparini, J. R. d. O. Lima, Y. A. Ghani, R. R. Hatanaka, R. Sequinel, D. L. Flumignan and J. E. de Oliveira, Bioenergy Technology, World Renewable Energy Conference, Sweden, 2011, pp. 101–110 Search PubMed.
- M. Ruiz, C. Tobalina, H. Demey-Cedeño, J. A. Barron-Zambrano and A. M. Sastre, React. Funct. Polym., 2013, 73, 653–657 CrossRef CAS PubMed.
- J. Rowbotham, P. Dyer, H. Greenwell, D. Selby and M. Theodorou, Interface Focus, 2013, 3, 20120046, DOI:10.1098/rsfs.2012.0046.
- S. Sladkevich, J. Gun, P. Prikhodchenko, V. Gutkin, A. Mikhaylov, V. Novotortsev, J. Zhu, D. Yang, H. Hng and Y. Tay, Nanotechnology, 2012, 23, 485–601 CrossRef PubMed.
- J. Y. Gong, S. R. Guo, H. S. Qian, W. H. Xu and S. H. Yu, J. Mater. Chem., 2009, 19, 1037–1042 RSC.
- Q. Kong, B. Wang, Q. Ji, Y. Xia, Z. Guo and J. Yu, Chin. J. Polym. Sci., 2009, 27, 807–812 CrossRef CAS.
- J. Soares, J. Santos, G. Chierice and E. Cavalheiro, Ecletica Quim., 2004, 29, 57–64 CrossRef CAS PubMed.
- M. Al-Remawi, J. Appl. Sci., 2012, 12, 727–735 CrossRef CAS.
- D. Cozzi, P. G. Desideri, L. Lepri and G. Ciantelli, J. Chromatogr. A, 1968, 35, 396–404 CrossRef CAS.
- D. Wu, J. Zhao, L. Zhang, Q. Wu and Y. Yang, Hydrometallurgy, 2010, 101, 76–83 CrossRef CAS PubMed.
- S. Gan, H. K. Ng, C. W. Ooi, N. O. Motala and M. A. F. Ismail, Bioresour. Technol., 2010, 101, 7338–7343 CrossRef CAS PubMed.
- T. G. W. Zillillah and Z. Li, Green Chem., 2012, 14, 3077–3086 RSC.
- K. A. Antonopoulos, D. C. Rakopoulos, D. T. Hountalas and E. G. Giakoumis, Energy Convers. Manage., 2006, 47, 3272–3287 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra08108k |
| This journal is © The Royal Society of Chemistry 2014 |