A novel ion-imprinted electrode prepared by in situ polymerization for detection of platinum
Huiping Bai,
Chunqiong Wang,
Kainan Zhang,
Zhongtao Ding and
Qiue Cao*
School of Chemical Science and Technology, Key Laboratory of Medicinal Chemistry for Nature Resource, Ministry of Education, Yunnan University, Kunming 650091, China. E-mail: qecao@ynu.edu.cn; Fax: +86-871-65033679; Tel: +86-871-65033718
First published on 3rd November 2014
Abstract
To develop a convenient method for sensitive and selective determination of platinum in complicated matrices, an ion-imprinted electrode was studied by in situ polymerization of Pt(IV) ion-imprinted membranes (Pt(IV)-IIMs) on the surface of a glassy carbon electrode (GCE). After the experimental parameters for the preparation of Pt(IV)-IIMs such as functional monomer, molar ratio of template, monomer and cross-linking agent together with extraction condition were optimized, an electrode with good regeneration, high stability and specific recognition to Pt(IV) was obtained by modifying GCE with Pt(IV)-IIMs prepared in acetonitrile using allylurea (NAU) as functional monomer, ethylene glycol dimethacrylate (EGDMA) as cross-linking agent, azobisisobutyronitrile (AIBN) as initiator under the molar ratio of template (H2PtCl6), NAU and EGDMA as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40. The resulting electrode named as Pt(IV)-IIMs/GCE exhibits high response sensitivity to Pt(IV) in phosphate buffer (pH 5.29). The calibration graph for the determination of Pt(IV) by Pt(IV)-IIMs/GCE is linear in the range of 2.0 × 10−8 ∼ 2.5 × 10−4 mol L−1 with a detection limit of 4.0 × 10−9 mol L−1. No metal ions tested at a concentration 25 times higher than that of Pt(IV) interfered in the determination. The electrode was successfully applied to determine platinum in catalyst and plant samples with a relative standard deviation (RSD) of less than 3.0% (n = 5) and recoveries in the range of 97.8–103.6%.
40. The resulting electrode named as Pt(IV)-IIMs/GCE exhibits high response sensitivity to Pt(IV) in phosphate buffer (pH 5.29). The calibration graph for the determination of Pt(IV) by Pt(IV)-IIMs/GCE is linear in the range of 2.0 × 10−8 ∼ 2.5 × 10−4 mol L−1 with a detection limit of 4.0 × 10−9 mol L−1. No metal ions tested at a concentration 25 times higher than that of Pt(IV) interfered in the determination. The electrode was successfully applied to determine platinum in catalyst and plant samples with a relative standard deviation (RSD) of less than 3.0% (n = 5) and recoveries in the range of 97.8–103.6%.
1 Introduction
Ion imprinting has been widely recognized as a powerful technique for the construction of materials containing binding sites that can recognize metal ions as the given target. The most attractive aspect of this technique is the use of a metal ion or its complex formed with a suitable ligand as template to assemble functional monomers around them into complementary orientations.1,2 After ion imprinting polymerization, the template is removed from the polymeric matrix by leaching with mineral acid to leave cavities named as ‘‘imprinted sites’’ in the polymeric matrix that are complementary in shape and size of the template metal ion. As a result, the obtained ion-imprinted materials, including ion-imprinted polymers (IIPs) and ion-imprinted membranes (IIMs), possess high selective recognition to the template metal ions. The specific binding behaviors for the target ions together with its other advantages over the biological recognition element, liked stability, reusability, simplicity and low cost in preparation, make IIPs/IIMs become ideal candidates as recognition elements for electrodes,3–5 and several electrodes based on IIPs/IIMs were constructed for the determination of metal ions, including Zn(II),6 Cd(II),6–8 Cu(II),8,9 Pb(II),10–12 Hg(II)13,14 and U(VI).15 To the best of our knowledge, there is a few studies have been published with respect to the preparation of Pt(IV) ion-imprinted polymers,16,17 but no report related to the electrode modified by ion imprinted layer for Pt(IV).Platinum is a valuable and scarce element that occurs naturally with other platinum metals only at about 0.001 μg mL−1 in the crust of the earth. It together with rhodium and palladium plays a decisive role in alloys and catalysts especially in the performance of exhaust systems to reduce the emission of gaseous pollutants by vehicles.18 Although the benefits of car catalysts are indisputable, highly toxic elements including Pt, Rh and Pd are released into atmosphere, which is found to increase the levels of contamination in environmental matrices, especially in soils, plants and road sediments19 as well as in airborne particles,20 and further imperil human health together with increasing the environmental risk. The development of analytical methods for the determination of trace amounts of platinum in environmental samples, biological matrices and secondary resources is important not only for the pollution control of the environment but also as a first step in the development of techniques for recovery of platinum from waste materials.21,22
So far, a series of methods have been developed for the determination of platinum, liked spectrophotometric methods,23,24 atomic absorption spectrometry (AAS),16,25 inductively coupled plasma atomic emission spectrometry (ICP-AES),26,27 inductively coupled plasma mass spectrometry (ICP-MS)28,22 and high performance liquid chromatography (HPLC).29 However, direct determination of platinum in complicated matrices especially at trace levels by these methods is often restricted owing to interferences caused by matrix elements and in some cases unsufficient sensitivity. Therefore, coupling of a separation/preconcentration procedure and elimination of interfering species prior to detection are necessary.16,22–29 Electrochemical methods are considered as the most favorable techniques for the determination of metal ions because of their high sensitivity in addition to low costs, ease of operation and portability. And several voltammetric methods have been reported for the determination of platinum.30–32 But such methods often use hanging mercury drop electrode as working electrode and demand a stringent process of sample pretreatment because the technique is susceptible to interferences. I. Švancara et al.33 developed a stripping voltammetric determination of platinum by a carbon paste electrode modified with cationic surfactants. Also, the detection limit (LOD) of this method is up to 7 × 10−7 mol L−1.
Considering that good selectivity and high sensitivity were obtained nearly in all of the studies with respect to the electrodes based on IIPs/IIMs,6–15 a Pt(IV) ion-imprinted electrode was investigated for simple, convenient, sensitive and selective determination of platinum by in situ polymerization of Pt(IV) ion-imprinted membrane on a glassy carbon electrode (GCE). For this purpose, a new functional monomer named as allylurea (NAU), which can coordinate with Pt(IV) using two nitrogen atoms as donor atoms, was selected for the first time to prepare Pt(IV) ion-imprinted membrane according to the Lewis theory of acids and bases (HSAB theory), and the experimental parameters together with the analytical application and selectivity behavior of the electrode were investigated in detail.
2 Experimental
2.1 Apparatus and instrumentation
Cyclic voltammetric (CV) and amperometric measurements were carried out with a CHI 660D electrochemical workstation (Shanghai CH Instruments Co., China). A three electrode cell (10 mL) with an ion-imprinted membrane modified glassy carbon electrode as working electrode, a saturated calomel electrode (SCE) as reference electrode and a platinum wire electrode as counter electrode was used. All potentials were measured and reported versus SCE. Prior to modification, the glassy carbon electrode (GCE, 3 mm diameter) was firstly polished with 0.5 μm and 50 nm alumina slurry, successively rinsed thoroughly with absolute alcohol and water in ultrasonic bath, and then dried in air.2.2 Reagents and chemicals
H2PtCl6, PdCl2, Na3RhCl6 and Na2IrCl6 were obtained from Kunming Institute of Precious Metals (Yunnan, China). Allylurea (NAU), 2-acetamidoacrylic acid (AAA), 2-(allylthiol)nicotinic acid (ANA) and acrylamide (AM) were purchased from Sigma Aldrich (USA). Ethylene glycol dimethacrylate (EGDMA) was purchased from Suzhou Anli Chemical Factory (Jiangsu, China) and distilled under vacuum to remove the stabilizers prior to use. Azobisisobutyronitrile (AIBN) was purchased from Shanghai Reagent Factory (Shanghai, China) and purified by recrystallization from ethanol before used. The other reagents and solvents were purchased from Tianjin Kemiou Chemical Reagent Co., Ltd. (Tianjin, China).The stock solution (1.0 × 10−2 mol L−1) of Pt(IV), Pd(II), Rh(III) and Ir(IV) were prepared by dissolving appropriate amount of H2PtCl6, PdCl2, Na3RhCl6 and Na2IrCl6 in 5.0 mL of 2.0 mol L−1 HCl and then diluted to 100 mL with de-ionized water, respectively. The stock solution (1.0 × 10−2 mol L−1) of the other metal ions used in this work was prepared with their nitrate or chloride. The working standard solution was obtained by diluting the stock solution with de-ionized water. The supporting electrolyte was phosphate buffer solution (pH 5.29), which was prepared with KH2PO4·2H2O and Na2HPO4·12H2O.
The reagents and solvents used without special illustration were of analytical reagent grade and used without further purification. De-ionized water was produced by a Millipore water system composed of Milli-RO 60 and Milli-Q SP.
2.3 Preparation of sample solution
2.4 Preparation of the imprinted and non-imprinted film modified electrodes
The Pt(IV) ion-imprinted and non-imprinted film modified electrodes were prepared by in situ polymerization technique. After 0.0125 mmol of H2PtCl6 and 0.05 mmol of NAU were added into 0.5 mL of acetonitrile and dissolved by ultrasonic at room temperature, 0.75 mmol of EGDMA and 0.04 mmol of AIBN were added. The polymerization mixture was purged with N2 gas for 10 min to remove molecular oxygen. Then, 4 μL of the above solution was dropped uniformly onto the surface of glassy carbon electrode, followed by covering a piece of glass. The electrode was heated by infrared lamp for 40 min, and a layer of transparent polymer membrane was formed on the electrode surface. After that, the electrode was firstly suspended in de-ionized water until the glass covered on the electrode dropped, and then immersed in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) acetic acid/methanol for 3 h to remove Pt(IV) from the ion-imprinted membrane. A Pt(IV) ion-imprinted membrane modified GCE (Pt(IV)-IIM/GCE) was obtained. Non-imprinted membrane modified GCE (NIM/GCE) was prepared under the same conditions in the absence of H2PtCl6. All of the electrodes are stored in water when not in use.
1 (v/v) acetic acid/methanol for 3 h to remove Pt(IV) from the ion-imprinted membrane. A Pt(IV) ion-imprinted membrane modified GCE (Pt(IV)-IIM/GCE) was obtained. Non-imprinted membrane modified GCE (NIM/GCE) was prepared under the same conditions in the absence of H2PtCl6. All of the electrodes are stored in water when not in use.
2.5 Electroanalytical measurements
The electrochemical characteristics of the modified electrode were characterized by cyclic voltammetric and amperometric measurements. Cyclic voltammetric measurements were carried out in 10 mL of 1/15 mol L−1 phosphate buffer solution (pH 5.29) at room temperature from −0.8 V to +0.8 V with a scan rate of 100 mV s−1. The amperometric detection was based on the change in the current response (Δi) before and after different concentrations of Pt(IV) was added in phosphate buffer solution and the potential for the determination of Pt(IV) was −0.6 V.After each experimental run, the electrode was washed by immersing in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) acetic acid/methanol for 1 h to remove the impurities in the electrode surface. The electrode is reusable after this cleaning procedure.
1 (v/v) acetic acid/methanol for 1 h to remove the impurities in the electrode surface. The electrode is reusable after this cleaning procedure.
3 Results and discussion
3.1 Optimization of the conditions for preparation of Pt(IV)-IIM/GCE
Considering that the quantity of recognition sites in the imprinted materials is a direct function of both the extent of the monomer–template interactions and the degree of cross-linking in the polymerization,34 functional monomer together with the molar ratio of template/monomer/crosslinking agent for the preparation of Pt(IV) ion-imprinted membrane were optimized. For this purpose, a series of electrodes were constructed by in situ polymerization of Pt(IV) ion-imprinted membrane with different combinations on GCE, and the current response of these electrodes were determined by amperometric measurements at the applied potential of −0.6 V in phosphate buffer (pH 5.29) containing 2.0 × 10−6 mol L−1 Pt(IV).The results listed in Table 1 point out that the film does not form on the surface of GCE when ANA is used as functional monomer, and the maximum response sensitivity is obtained with the electrode prepared using NAU as functional monomer. Therefore, NAU was selected as functional monomer for the preparation of Pt(IV)-IIM/GCE.
| Sensors | Polymerization conditionsa | Current responseb (μA) | ||
|---|---|---|---|---|
| Functional monomers | Monomer added (mmol) | EGDMA added (mmol) | ||
| a All of the Pt(IV)-IIM/GCE sensors were prepared with H2PtCl6 (0.0125 mmol) as template, acetonitrile (0.5 mL) as porogen and AIBN (0.04 mmol) as initiator.b The current response were obtained by amperometric measurements at applied potential of −0.6 V in phosphate buffer solution (pH 5.29) containing 2.0 × 10−6 mol L−1 Pt(IV).c The film can not form on GCE when ANA as functional monomer. | ||||
| Pt(IV)-IIM/GCE1 | ANA | 0.05 | 0.5 | c |
| Pt(IV)-IIM/GCE2 | AAA | 0.05 | 0.5 | 12 |
| Pt(IV)-IIM/GCE3 | AM | 0.05 | 0.5 | 16 |
| Pt(IV)-IIM/GCE4 | NAU | 0.05 | 0.5 | 178 |
| Pt(IV)-IIM/GCE5 | NAU | 0.05 | 0.25 | 10 |
| Pt(IV)-IIM/GCE6 | NAU | 0.05 | 0.75 | 82 |
| Pt(IV)-IIM/GCE7 | NAU | 0.05 | 1.00 | 44 |
| Pt(IV)-IIM/GCE8 | NAU | 0.025 | 0.5 | 80 |
| Pt(IV)-IIM/GCE9 | NAU | 0.075 | 0.5 | 42 |
| Pt(IV)-IIM/GCE10 | NAU | 0.10 | 0.5 | 10 |
As expected, the amount of functional monomer (NAU) and crosslinking agent (EGDMA) added in polymerization produced remarkably influence on the response sensitivity of the resulting electrodes. Among all of the electrodes prepared using NAU as functional monomer, the electrode (Pt(IV)-IIM/GCE4 in Table 1) obtained by modifying GCE with Pt(IV) ion-imprinted membrane prepared with the molar ratio of Pt(IV), NAU and EGDMA as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 shows the maximum response sensitivity to Pt(IV), and the other electrodes prepared with more or less NAU and EGDMA possess relatively weak current response. The molar ratio of Pt(IV), NAU and EGDMA for the preparation of Pt(IV)-IIM/GCE was selected as 1
40 shows the maximum response sensitivity to Pt(IV), and the other electrodes prepared with more or less NAU and EGDMA possess relatively weak current response. The molar ratio of Pt(IV), NAU and EGDMA for the preparation of Pt(IV)-IIM/GCE was selected as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40.
40.
It is very important for the sensitivity, selectivity and reproducibility of the imprinted electrode to extract the template from the imprinted film completely, so the extraction solvent and extraction time need to be optimized. 1.0 mol L−1 HCl, 1.0 mol L−1 HNO3 and the mixture of acetic acid/methanol with different volume ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, 1
3, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 1
2, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 2
1, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 3
1, 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were used as extraction solvent to clear the template Pt(IV) from the imprinted film coated on GCE. The results indicate that the stability and service life of the imprinted membrane is unsatisfactory by soaking in 1.0 mol L−1 HCl, 1.0 mol L−1 HNO3 and the mixture of acetic acid/methanol with the volume ratio of 2
1) were used as extraction solvent to clear the template Pt(IV) from the imprinted film coated on GCE. The results indicate that the stability and service life of the imprinted membrane is unsatisfactory by soaking in 1.0 mol L−1 HCl, 1.0 mol L−1 HNO3 and the mixture of acetic acid/methanol with the volume ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 3
1 and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and Pt(IV) is difficult to remove by using 1
1, and Pt(IV) is difficult to remove by using 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and 1
3 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (v/v) acetic acid/methanol as extraction solvent. When the mixture of acetic acid/methanol with the volume ratio of 1
2 (v/v) acetic acid/methanol as extraction solvent. When the mixture of acetic acid/methanol with the volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is selected as extraction solvent, Pt(IV) can be removed from the imprinted membrane within 3 h (Fig. 1), and the membrane can maintain integrity and stability under such conditions. Therefore, 1
1 is selected as extraction solvent, Pt(IV) can be removed from the imprinted membrane within 3 h (Fig. 1), and the membrane can maintain integrity and stability under such conditions. Therefore, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) acetic acid/methanol and 3 h were chosen as the optimum eluent and soaking time for template removal, respectively.
1 (v/v) acetic acid/methanol and 3 h were chosen as the optimum eluent and soaking time for template removal, respectively.
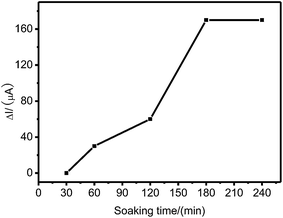 | ||
| Fig. 1 Effect of soaking time on the current response of Pt(IV)-IIM/GCE to 2.0 × 10−6 mol L−1 Pt(IV) in phosphate buffer (pH 5.29) at the applied potential of −0.6 V. | ||
3.2 Characterization of the imprinted film coated on GCE
The resulting Pt(IV) ion-imprinted film obtained by in situ polymerization were characterized by Fourier transform infrared spectroscopy (FT-IR), scanning electron microscopy (SEM) and X-ray photoelectron spectroscopy (XPS). The FT-IR spectra of the non-imprinted film, imprinted film before and after removal of Pt(IV) were recorded using KBr pellet method (Fig. 2). By comparing the IR spectra of these resins, it is observed that there is almost no change in the absorption peak of carbonyl group (vC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) located at about 1728 cm−1, pointing out that the carbonyl group in NAU does not participate in coordination with Pt(IV). After Pt(IV) was removed from the ion-imprinted film, the absorption peaks of N–H (vN–H) and C–N (vC–N) shift from 3525 cm−1 and 1449 cm−1 to 3542 cm−1 and 1458 cm−1, respectively. These results confirm that NAU can bind with Pt(IV) using two nitrogen atoms as donor atoms. Based on the Lewis theory of acids and bases (HSAB theory), Pt(IV) is a soft acid. Compared to oxygen atom (hard base), it is more easily combined with nitrogen atom (medial base). The IR spectra of imprinted film after removal of Pt(IV) and the non-imprinted film are very similar, suggesting that the two kinds of films possess a same chemical structure and the leaching process does not affect the polymeric network.
O) located at about 1728 cm−1, pointing out that the carbonyl group in NAU does not participate in coordination with Pt(IV). After Pt(IV) was removed from the ion-imprinted film, the absorption peaks of N–H (vN–H) and C–N (vC–N) shift from 3525 cm−1 and 1449 cm−1 to 3542 cm−1 and 1458 cm−1, respectively. These results confirm that NAU can bind with Pt(IV) using two nitrogen atoms as donor atoms. Based on the Lewis theory of acids and bases (HSAB theory), Pt(IV) is a soft acid. Compared to oxygen atom (hard base), it is more easily combined with nitrogen atom (medial base). The IR spectra of imprinted film after removal of Pt(IV) and the non-imprinted film are very similar, suggesting that the two kinds of films possess a same chemical structure and the leaching process does not affect the polymeric network.
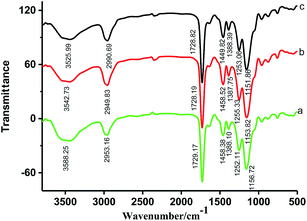 | ||
| Fig. 2 FT-IR spectra of non-imprinted film (a), the imprinted film (b) after and (c) before removal of Pt(IV) by KBr pellet method. | ||
The morphologies of non-imprinted film, imprinted film before and after removal of Pt(IV) under SEM are showed in Fig. 3. It is clearly that there is great difference in the SEM images among the three kinds of film formed on the surface of GCE. Pt(IV) ion-imprinted membrane after removal of template Pt(IV) (b) is very rough, and Pt(IV) ion-imprinted membrane before removal of Pt(IV) (a) together with the non-imprinted membrane (c) both are smooth and homogeneous and no cracks can be observed. It is probably that the roughness of imprinted membrane after template removal is beneficial for improving the rebinding efficiency and enhancing the response sensitivity of the electrode. To verify this speculation, the elemental composition of imprinted film before and after removal of Pt(IV) as well as rebinding Pt(IV) were determined by XPS measurement. In XPS spectra (Fig. 4), the Pt signals of Pt (4f) and Pt (4d) observed in the 0–400 binding energy in Fig. 4a are not found anymore in Fig. 4b, but emerge again in Fig. 4c. Those results indicate that Pt(IV) were completely removed during the extraction process, and can rebind to the imprinted film in the process of electrochemical determination. To further confirm that the roughness of imprinted membrane is beneficial for improving the rebinding efficiency, the content of Pt in the imprinted and non-imprinted membrane after rebinding Pt(IV) was determined quantitatively by atomic emission spectrometry. The mass percentage content of Pt was found correspondingly as 0.76% and 0.11%, suggesting that the rebinding efficiency of imprinted membrane is better than that of non-imprinted membrane.
 | ||
| Fig. 3 Scanning electron micrograph of the imprinted film (a) before and (b) after removal of Pt(IV), and (c) non-imprinted film. | ||
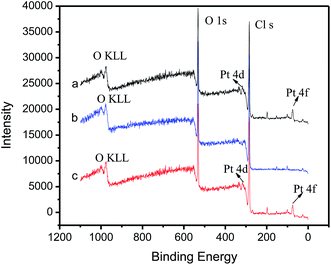 | ||
| Fig. 4 XPS analysis of Pt(IV) ion-imprinted film (a) before and (b) after removal of Pt(IV) and (c) after rebinding Pt(IV). | ||
3.3 Electrochemical characterization of the imprinted film modified electrode
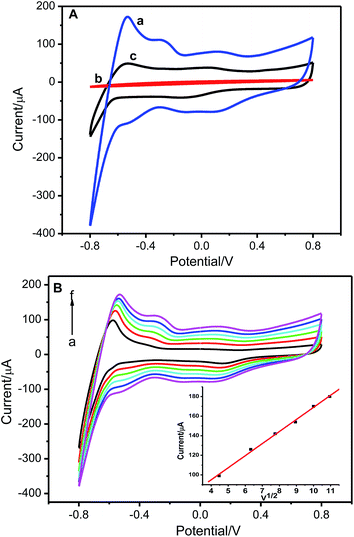
The electrochemical property of the resulting Pt(IV)-IIM/GCE was investigated by cyclic voltammetry in 10 mL of phosphate buffer solution (pH 5.29) containing 2.0 × 10−6 mol L−1 Pt(IV). It can be seen from the results shown in Fig. 5A that the peak current increases remarkably after GCE (curve b) is modified with Pt(IV) ion-imprinted film (curve a). These results demonstrate that the specific recognition sites existed in the ion-imprinted film can enhance amperometric response to Pt(IV). As comparison, the non-imprinted electrode (NIM/GCE) exhibits only a small signal in the potential range from −0.8 to 0.8 V (curve c). This small response is presumably attributed to the non-specific adsorption of Pt(IV) by the non-imprinted film. Fig. 5B is the typical cyclic voltammograms of Pt(IV)-IIM/GCE in phosphate buffer solution (pH 5.29) containing 2.0 × 10−6 mol L−1 Pt(IV) at the scan rate varied from 20 to 120 mV s−1. It is clearly that the peak currents are proportional to the square root of the scan rate, suggesting the electrode reaction is a typical diffusion-controlled electrochemical behavior.3.4 Optimization of the determination conditions
In order to improve the sensitivity of the ion-imprinted electrode, determination conditions liked the composition and pH of electrolyte solution and applied potential were optimized.The amperometric responses of the resulting Pt(IV)-IIM/GCE to 2.0 × 10−6 mol L−1 Pt(IV) were examined in 0.1 mol L−1 citric acid buffer solution (pH 5.29), 0.2 mol L−1 acetic acid buffer solution (pH 5.29) and 1/15 mol L−1 phosphate buffer solution (pH 5.29) to optimize the composition of electrolyte solution. The results suggest that the Pt(IV)-IIM/GCE obtained its maximum current response in phosphate buffer solution. Fig. 6 shows the effects of the pH value of phosphate buffer on the current response of Pt(IV)-IIM/GCE to 2.0 × 10−6 mol L−1 Pt(IV). As can be seen, the maximum current response of Pt(IV)-IIM/GCE is obtained in phosphate buffer with pH of 5.29, and all of the other buffers with pH more or less than 5.29 produce a decrease in the peak current. In strongly acidic media, the binding capacity of Pt(IV) is hindered because of the protonation of the active sites in the imprinted film. Again, the decrease in binding capacity of pHs greater than 5.29 is due to the competition between rebinding by formation of the complexes of Pt(IV) with NAU anchored to the imprinted film with the formation of hydroxide of Pt(IV). Therefore, phosphate buffer with pH of 5.29 was used for the determination of Pt(IV) by Pt(IV)-IIM/GCE.
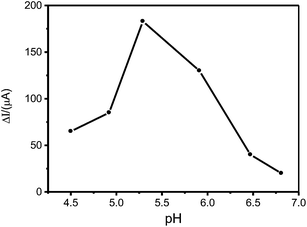 | ||
| Fig. 6 Effect of the pH value of phosphate buffer on the current response of Pt(IV)-IIM/GCE to 2.0 × 10−6 mol L−1 Pt(IV) at the applied potential of −0.6 V. | ||
The effect of applied potential on the electrode response was studied in phosphate buffer (pH 5.29) containing 2.0 × 10−6 mol L−1 Pt(IV). As can be seen from Fig. 7, the response current of the electrode increases with decreasing the potential from 0.4 to −0.6 mV, and maintains its maximum value in the potential range of −0.6 to −0.8 mV. To avoid interference at high negative applied potential, a potential of −0.6 V (vs. SCE) was selected as the applied potential for amperometric measurements.
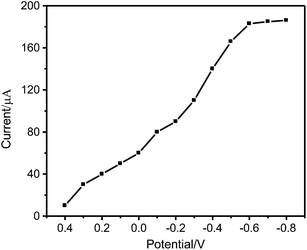 | ||
| Fig. 7 Effect of the applied potential on the current response of Pt(IV)-IIM/GCE to 2.0 × 10−6 mol L−1 Pt(IV) in phosphate buffer (pH 5.29). | ||
3.5 Amperometric i–t curve and calibration curve for determination of Pt(IV)
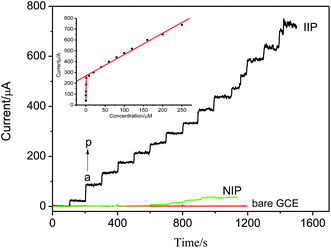
The affinity of the resulting Pt(IV)-IIM/GCE, NIM/GCE and bare GCE to Pt(IV) in the concentration range over 2.0 × 10−8 ∼ 2.5 × 10−4 mol L−1 were investigated by amperometric i–t curve at the potential of −0.6 V. As shown in Fig. 8, the amperometric currents of bare GCE is almost not varied with the concentration of Pt(IV), and the sensitivity of Pt(IV)-IIM/GCE is significantly higher than that of NIM/GCE. The mechanism of facilitated transport and lack of specific binding sites can be applied to explain the great difference between the amperometric current of Pt(IV)-IIM/GCE and NIM/GCE.The current response of Pt(IV)-IIM/GCE increased with increasing of the concentration of Pt(IV), and the relationship between the response currents and concentrations of Pt(IV) shown as inset in Fig. 8. It can be seen that the calibration curve for the electrochemical determination of Pt(IV) is composed of two distinct straight lines, suggesting that there are two types of binding sites, which can be named as high affinity binding sites and low affinity binding sites, existed in the Pt(IV)-imprinted membrane covered on GCE. This result is consistent with many studies of imprinted polymers.35 The linear regression equations in the Pt(IV) concentration range over 0.02–1.0 μmol L−1 and 1.0–250.0 μmol L−1 are obtained as Δi (μA) = 35.25 + 104.07c (μmol L−1) (r = 0.9958) and Δi (μA) = 168.65 + 1.97c (μmol L−1) (r = 0.9965), respectively. The limit of detection of the electrode, which is defined as the lowest concentration of Pt(IV) producing a peak current 3 times higher than the standard deviation of 9 blank measurements, is found to be 4.0 × 10−9 mol L−1.
3.6 Selectivity, repeatability and stability of Pt(IV)-IIM/GCE
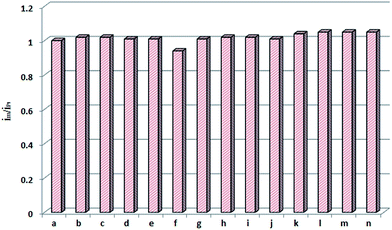
The selectivity of Pt(IV)-IIM/GCE to Pt(IV) was evaluated by testing its current responses to 2.0 × 10−6 mol L−1 Pt(IV) in the presence of 5.0 × 10−5 mol L−1 of Zn2+, Co2+, Mg2+, Ni2+, Cu2+, Mn2+, Fe3+, Cd2+, Cr3+, Pb2+, Pd2+, Rh3+ and Ir4+, respectively. And the ratio of the response current (im/iPt) of the electrode to 2.0 × 10−6 mol L−1 Pt(IV) in the present (im) and absence (iPt) of interfering ions was calculated. The results shown in Fig. 9 indicate that the im/iPt in all of the case is in the range of 0.97–1.05, suggesting that at least 25-fold excess of the metal ions tested in this paper have no effect on the determination of Pt(IV).To investigate the reproducibility of the electrochemical method, three Pt(IV)-IIM/GCE were prepared under the identical experimental conditions and used for the determination of 2.0 × 10−6 mol L−1 Pt(IV). The calculated RSD of the peak currents obtained with different electrodes is 2.4%. The repeatability and stability of the electrode were also investigated for 2.0 × 10−6 mol L−1 Pt(IV). The electrode can retain more than 90% of its original response after used at least 100 times or stored in water at room temperature for 80 day. The RSD of the peak currents of the same electrode after continuous using for 5 times is 2.1%. These results suggest that the electrode can be regenerated and possesses excellent storage stability.
3.7 Sample analysis
Two real samples, including an automotive catalyst material and a plant sample, were analyzed by the resulting Pt(IV)-IIM/GCE to evaluate the precision and accuracy of the proposed electrochemical method. The results presented in Table 2 suggest that the recoveries of Pt(IV), which was obtained via the standard addition method, are in the range of 97.8–103.6%, and the reproducibility of the procedure, expressed as RSD, is less than 3.0%. To further verify the accuracy of the quantitative results obtained by Pt(IV)-IIM/GCE, the concentrations of platinum in sample solutions were also quantified by AAS method. Platinum does not be determined in plant sample solution by AAS and the concentration of platinum in catalyst sample solution is 6.50 × 10−6 mol L−1. It can be seen that the quantitative results of platinum obtained by Pt(IV)-IIM/GCE are in agreement with that obtained by AAS, suggesting that the resulting Pt(IV)-IIM/GCE may be a feasible tool for the determination of Pt(IV) in the complicated samples.4 Conclusion
In this work, a new electrode for the determination of Pt(IV) at trace levels was constructed based on the application of ion-imprinted membrane as a novel material for modifying glass carbon electrode. The ion-imprinted membrane coated on the surface of glass carbon electrode can not only act as the selective inducing agent but also play the role of a pre-concentrator. As a result, the developed electrode exhibits good selectivity and high sensitivity. The detection limit of this electrode is lower than most of the methods reported for the determination of Pt(IV).23,24,26–30,33 The electrochemical method proposed based on the resulting Pt(IV)-IIM/GCE can be used for the direct determination of platinum at trace levels in complicated matrices.Acknowledgements
This work was financially supported by the Natural Science Foundation of China (no. 20965009) and the Doctoral Fund of Ministry of Education of China (20125301110005).Notes and references
- H. R. Rajabi, M. Roushani and M. Shamsipur, J. Electroanal. Chem., 2013, 693, 16 CrossRef CAS PubMed.
- T. Takeuchi, N. Murase, H. Maki, T. Mukawa and H. Shinmori, Org. Biomol. Chem., 2006, 4, 565 CAS.
- G. Mustafa and P. A. Lieberzeit, RSC Adv., 2014, 4, 12723 RSC.
- M. Shamsipur, J. Fasihi and K. Ashtari, Anal. Chem., 2007, 79, 7116 CrossRef CAS PubMed.
- H. Zhou, G. L. Xu, A. H. Zhu, Z. Zhao, C. C. Ren, L. L. Nie and X. W. Kan, RSC Adv., 2012, 2, 7803 RSC.
- J. Tan, H. F. Wang and X. P. Yan, Biosens. Bioelectron., 2009, 24, 3316 CrossRef CAS PubMed.
- T. Alizadeh, M. R. Ganjali, P. Nourozi, M. Zare and M. Hoseini, J. Electroanal. Chem., 2011, 657, 98 CrossRef CAS PubMed.
- B. B. Prasad, D. Jauhari and A. Verma, Talanta, 2014, 120, 398 CrossRef PubMed.
- S. Z. Bajwa, R. Dumler and P. A. Lieberzeit, Sens. Actuators, B, 2014, 192, 522 CrossRef CAS PubMed.
- Z. H. Wang, Y. X. Qin, C. Wang, L. J. Sun, X. L. Lu and X. Q. Lu, Appl. Surf. Sci., 2012, 258, 2017 CrossRef CAS PubMed.
- M. K. Bojdi, M. H. Mashhadizadeh, M. Behbahani, A. Farahani, S. S. H. Davarani and A. Bagheri, Electrochim. Acta, 2014, 136, 59 CrossRef CAS PubMed.
- A. Bahrami, A. Besharati-Seidani, A. Abbaspour and M. Shamsipur, Electrochim. Acta, 2014, 118, 92 CrossRef CAS PubMed.
- T. Alizadeha, M. R. Ganjali and M. Zare, Anal. Chim. Acta, 2011, 689, 52 CrossRef PubMed.
- H. R. Rajabi, M. Roushani and M. Shamsipur, J. Electroanal. Chem., 2013, 693, 16 CrossRef CAS PubMed.
- P. Metilda, K. Prasad, R. Kala, J. M. Gladis, T. Prasada Raoa and G. R. K. Naidu, Anal. Chim. Acta, 2007, 582, 147 CrossRef CAS PubMed.
- B. Leśniewska, M. Kosińska, B. Godlewska-Żyłkiewicz and E. Zambrzycka, Microchim. Acta, 2011, 175, 273 CrossRef.
- J. Yang and K. Dukjoon, Ind. Eng. Chem. Res., 2014, 53, 13340 Search PubMed.
- J. Chen, Metallurgical Chemistry of the platinum metals, Science press, Beijing, 2008 Search PubMed.
- M. Motelica-Heino, S. Rauch, G. M. Morrison and O. F. X. Donard, Anal. Chim. Acta, 2001, 436, 233 CrossRef CAS.
- A. N. Anthemidis, D. G. Themelis and J. A. Stratis, Talanta, 2001, 54, 37 CrossRef CAS.
- C. Font'as, V. Salvad'o and M. Hidalgo, Ind. Eng. Chem. Res., 2002, 41, 1616 CrossRef.
- M. L. Alonso Castillo, A. Garc de Torres, E. Vereda Alonso, M. T. Siles Cordero and J. M. Cano Pavo, Talanta, 2012, 99, 853 CrossRef CAS PubMed.
- E. A. Moawed, I. Ishaq, A. Abdul-Rahman and M. F. El-Shahat, Talanta, 2014, 121, 113 CrossRef CAS PubMed.
- D. L. Ma, J. Wang, G. S. Ding, K. B. Ma and J. H. Wang, Chin. J. Anal. Lab., 2011, 30, 15 CAS.
- T. Revathi Reddy, N. N. Meeravali and A. V. R. Reddy, Anal. Methods, 2013, 5, 2343 RSC.
- N. M. Lopo de Araújo, S. L. C. Ferrrira, H. C. Santos, D. Santiago de Jesus and M. A. Bezerra, Anal. Methods, 2012, 4, 508 RSC.
- L. F. Yang, Y. Chen and D. Q. Ma, Phys. Test. Chem. Anal., Part B, 2011, 47, 339 CAS.
- W. H. Hsu, S. J. Jiang and A. C. Sahayam, Anal. Chim. Acta, 2013, 794, 15 CrossRef CAS PubMed.
- S. N. Lanjwani, R. K. Zhu, M. Y. Khuhawar and Z. F. Ding, J. Pharm. Biomed. Anal., 2006, 40, 833 CrossRef CAS PubMed.
- S. Orecchio, D. Amorello and C. Carollo, Microchem. J., 2012, 100, 72 CrossRef CAS PubMed.
- A. A. Dalvi, A. K. Satpati and M. M. Palrecha, Talanta, 2008, 75, 1382 CrossRef CAS PubMed.
- O. Nygren, G. T. Vaughan, T. M. Florence, G. M. P. Mprrison, I. M. Warner and L. S. Dale, Anal. Chem., 1990, 62, 1637 CrossRef CAS.
- I. Švancara, M. Galík and K. Vytřas, Talanta, 2007, 72, 512 CrossRef PubMed.
- V. Pichon and F. Chapuis-Hugon, Anal. Chim. Acta, 2008, 622, 48 CrossRef CAS PubMed.
- J. Matsui, Y. Miyoshi, O. Doblhoff-Dier and T. Takeuchi, Anal. Chem., 1995, 67, 4404 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2014 |