Electrospun anatase TiO2 nanorods for flexible optoelectronic devices
Abstract
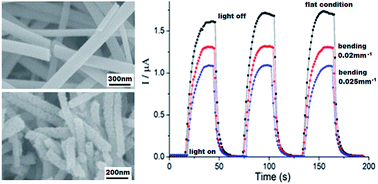
Titanium dioxide (TiO2) nanorods with anatase phase were successfully fabricated by electrospinning and followed calcination. The TiO2 nanorods were characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), energy dispersive X-ray spectroscopy (EDS) and UV-visible spectroscopy. The diameter of the TiO2 nanorods was about 60–150 nm and the length was 200 nm–2 μm. Electrical properties under bending were investigated by fixing the device to a curved surface with different curvatures, and the device showed a fast and stable resistance response to curvature changing. Photoelectric properties were studied by irradiation with different light intensities. The device exhibited a short response time (∼10 s) and a high sensitivity (∼103) which increased with the light intensity increasing. These results indicate that electrospun anatase TiO2 nanorods have potential applications in flexible photodetectors and solar cells.

 Please wait while we load your content...
Please wait while we load your content...