Structural evolution of rayon-based carbon fibers induced by doping boron†
Huiqi Wang*ab,
Tao Hana,
Jinhua Yangb,
Zechao Taob,
Quangui Guo*b,
Zhanjun Liub,
Zhihai Fengc and
Lang Liub
aInstitute of Inorganic Non-metallic Materials, School of Materials Science and Engineering, North University of China, Taiyuan 030051, China. E-mail: hqiwang@163.com; Fax: +86-351-4083952; Tel: +86-351-4084106
bKey Laboratory of Carbon Materials, Institute of Coal Chemistry, Chinese Academy of Sciences, Taiyuan 030001, China. E-mail: qgguo@sxicc.ac.cn
cNational Key Laboratory of Advanced Functional Composites Materials, Aerospace Research Institute of Material and Processing Technology, Beijing 100076, China
First published on 3rd November 2014
Abstract
In the present work, we provide a systematic analysis of the structural evolution of rayon-based carbon fibers (RCFs) induced by doping boron using scanning electron microscope (SEM), transmission electron microscope (TEM), X-ray diffraction (XRD) and Raman spectroscopy. For the first time, boron-doped RCFs with tunable amounts of boron were fabricated by exposing the RCFs to a vapor of boron by the decomposition of boron carbide (B4C). SEM and XRD results indicate that at the higher temperatures the strong erosion of boron vapor not only changed the original structure of RCFs, but also produced some flaws. Interestingly, when the temperature of doping boron is higher than 2200 °C, the graphite basal planes of RCFs are perpendicular to the fiber axis. Raman spectra also confirmed the presence of disorders and flaws in graphitic layers because of the displacement and solid solution of boron in the carbon lattice. Further, the chemical environment of boron species was ascertained by 11B nuclear magnetic resonance, indicating that boron atoms exist in three chemical environments, including the substitutional boron (BC3), boron clusters and B4C. Moreover, the TGA data indicated that doping boron greatly improved the oxidation inhibition of RCFs, and is superior to increasing the heat treatment temperature for improving the oxidation resistance. Such a systematic analysis of the structural evolution and oxidation resistance of RCFs induced by doping boron thus provides industrial potential for preparing RCFs with higher oxidation resistance at up to 800 °C.
1. Introduction
As compared with other kinds of carbon fibers (polyacrylonitrile-based, pitch-based), rayon-based carbon fibers (RCFs) are a promising material used as a missile protective coating, a chemical pump in the body and medical treatments due to its light weight, high purity, high strain, superior thermal stability and low thermal conductivity.1,2 However, the high cost and low strength and modulus prevent it being applied widely.3 To extend its applicable scope, it is imperative to improve the mechanical properties and to lower the production cost.There are some works3–9 suggesting that heat-treatment temperature (HTT) above 2800 °C can effectively improve the mechanical properties of carbon fibers, which is ascribed to an increase of the preferred orientation and dimension of crystallite.10,11 However, such HTT incurs high cost in mass production. Fortunately, it has been well established that doping boron can help graphitizable carbon obtain a high degree of graphitization under a lower graphitization temperature,12,13 and it is likely to tailor the electronic properties of carbon fibers by replacing some of the carbon atoms with boron at the trigonal sites.14 Moreover, boron can be uniformly distributed in the bulk of carbon materials, and thus it is a well-known efficient catalyst for the homogeneous continuous graphitization of the entire carbon materials to avoid the formation of separated distinct carbon components.15 On the other hand, previous studies have shown that doping boron atoms into carbon fibers could obviously improve their oxidation resistance.16,17 Oxidation resistance of boron-doped RCFs is of particular importance for its use in advanced composites. Therefore, doping boron at high temperatures provides a facile route for fabricating a high-performance and low cost RCFs. Most of the documented routes of doping boron indicate that boron atoms are difficult to disperse uniformly in the carbon materials, and the boron concentration in boron-doped carbon materials was not readily controlled.16,18–20 However, in our previous work,13 we have confirmed that boron doping into polyacrylonitrile-based carbon fibers by exposing the carbon fibers in a vapor of boron by the decomposition of B4C at high temperatures, can achieve not only a uniform boron distribution, but also the controllability of boron concentration. To the best of our knowledge, until now, boron-doped RCFs have not appeared yet in the literature. Thus, an understanding of the structural evolution of boron-doped RCFs on different boron concentrations and HTT is desirable for preparing the RCFs at low cost with improved properties.
Herein, boron-doped RCFs, for the first time, was preparing by exposing the RCFs to a vapor of boron by the decomposition of the B4C at high temperatures. A systematic analysis of the structural evolution of boron-doped RCFs on the different boron concentrations and HTT was carried out. In addition, this contribution is also aimed at evaluating the effects of boron on the microstructure and oxidation resistance of the RCFs.
2. Experimental
2.1 Materials and process
Rayon-based CFs was supplied by Institute of Coal Chemistry, Chinese Academy of Sciences, China (tensile strength 0.6 GPa, tensile modulus 35 GPa, density 1.40 g m−3, and diameter 7 μm). The as-received RCFs were firstly de-resinated using acetone to remove the resin coated onto fibers during manufacture. Boron doping was achieved by our previously reported process.13 Typically, boron-doped RCFs were prepared by exposing RCFs to a vapor of boron at high temperatures in a sealed B4C/graphite crucible (BGC). A picture of the BGC was shown in Fig. S1.† Details of fabrication of BGC can be found in ref. 13. Further, the RCF tow was carefully fixed onto a high purity graphite rod to ensure the fiber length was constant during the graphitization. The prepared samples were placed in the holes of the BGCs. For each batch, 0.7 g RCFs were loaded into holes. The crucibles were sealed with a threaded-cap and then placed in a medium frequency induction furnace (Tongxin Electric Heating Apparatus Co., Ltd, China), which was sealed and evacuated. It was then pressurized to 0.05 MPa with argon (high purity, 99.99% Ar). This process was repeated at least 3 times to ensure the negligible oxygen content within the furnace prior to heating. Argon was passed through the furnace at a rate of 100 mL min−1, while maintaining the argon pressure within the furnace at 0.05 MPa. This condition was maintained during the entire heat-treatment process. An average heating rate of 10 °C min−1 was used to reach 2200, 2400 and 2600 °C, at which the sample remained for 50 min, followed by a 10 h cool-down to room temperature.2.2 Characterization
The morphologies and microstructures of the boron-doped RCFs were observed by field-emission scanning electron microscope (Hitachi S-4800, FE-SEM) and field-emission transmission electron microscope (FEI Technai G2 F20, HR-TEM, accelerating voltage 200 kV). The select area electron diffraction (SAED) patterns were also carried out. The effects of boron on the structural properties were evaluated by X-ray diffraction (XRD, Rigaku D/max-rB with Cu Kα radiation, λ = 0.15406 nm) and Raman spectroscopy at surface-section of boron-doped RCFs (Jobin-Yvon, LabRam HR800, 1 mW) with an Ar-ion laser excitation beam wavelength of 532 nm. XRD measurement was used for determining the primary structural parameters of graphite crystal: d002 and Lc. The crystalline parameters of the boron-doped RCFs were calculated by the formulas in ref. 21. The boron concentration was determined using X-ray photoelectron spectroscopy (XPS; Thermo ESCALAB 250, 1486.6 eV Al-Kα radiation). Boron nuclear magnetic resonance (11B-NMR) was performed on a Bruker AVANCE III 400 WB spectrometer using a magnetic field of 9.39 T at 297 K. All 11B-MAS chemical shifts are referenced to the resonances of boron nitride (BN), 99.5% (metals basis) standard (δ = 0.00).Oxidation resistance of various boron-doped RCFs was measured by the thermal gravimetric analyzer (TGA, NETZSCH TGA 204F1). 5 mm long fiber specimens were heated form 25 to 1200 °C with a heating rate of 5 °C min−1 in dry air.
3. Results and discussion
3.1 Morphology analysis by SEM
In this work, the graphitized boron-doped RCFs were obtained by exposing fibers to a boron vapor at high temperatures. It was denoted as BX-T, where B stands for the B4C, X is the concentration of B4C in a BGC, and T is the HTT. The morphologies of the synthesized boron-doped RCFs were observed by FE-SEM (Fig. 1 and 2). Fig. 1(a)–(d) show surfaces of boron-undoped and boron-doped RCFs. It can be seen that surfaces of the boron-doped RCFs were more ordered than those of the boron-undoped RCFs due to the catalytic graphitization of carbons by boron, but that some voids appeared at the surfaces of the boron-doped CFs due to the erosion of boron vapor.22 As the total of boron-doping increased, the RCF surface was much rougher with the coarsening grooves and folds, and cavity features become clear along the fiber axis. Surface of the boron-undoped RCFs has irregular striations along the fiber axis as shown in Fig. 1(a). As such, cross-sections of the boron-undoped and boron-doped RCFs were showed in Fig. 1(e)–(h). Boron-undoped RCFs had a porous texture at the cross-section, with no indication of a sheet-like or fibrillar structures. However, boron-doped RCFs had a sheet-like cross-section with the ordered graphite observable along the fiber axis, which is similar to the boron-doped polyacrylonitrile-based carbon fibers.13 Moreover, as the total of boron-doping increased, the cross-sections of the RCFs were less curved and more well-defined, but had gaps between these sheets. The mechanical properties of boron-doped RCFs treated to 2200 °C were shown in Fig. S2.† It can be seen that doping boron into RCFs at 2200 °C improved greatly their tensile strength from 0.92 to 1.34 GPa and tensile modulus from 49.5 to 80.3 GPa. | ||
| Fig. 1 Evolution of surface and cross-section morphologies by FE-SEM examination of (a and e) B0-2200, (b and f) B10-2200, (c and g) B20-2200 and (d and h) B30-2200. | ||
 | ||
| Fig. 2 Surface and cross-section morphologies by FE-SEM examination of the B10-2400 (a and b) and B100-2600 (c and d). | ||
HTT is another key factor for structural evolution of boron-doped RCFs. The HTT dependence was achieved in the B10-2200 sample. For comparison, SEM images of boron-undoped RCFs treated to 2400 and 2600 °C were also shown in Fig. S3.† Increasing HTT from 2200 to 2400 and 2600 °C leaded to more obvious grooves and folds at the surface of boron-doped RCFs, and cavity features become more prominent along the fiber axis as shown in Fig. 2(a) and (b). The original appearance for RCFs no longer exists (Fig. S3†). Moreover, the orientation of graphite sheets shown at the cross-section was seen more clearly, and the curved feature of graphite sheets was invisible, but graphite sheets do not arrange along the fiber axis as shown in Fig. 2(c) and (d). In addition, the basal planes of graphitic layer must be parallel to the fiber axis in order to produce high modulus and high strength CFs.23 It thus can be seen that at the higher temperatures, the strong erosion of boron vapor, not only changed the original structure of RCFs, but also produced more flaws. Meanwhile, the mechanical properties fail to measure because of great brittleness. For comparison, the mechanical properties of boron-undoped RCFs treated to different temperatures were measured and shown in Fig. S4.†
As well-known, the mechanical properties of RCFs mainly depend on its microstructure. The failure of RCFs was initiated by surface defect. The presence of surface defects obviously affects their tensile strength and modulus (Fig. S2 and S4†). The gaps between graphite sheets at the cross-section of RCFs were identified as the primary internal defect. Their strength and modulus can be improved by reducing/eliminating these flaws.13
3.2 Analysis of XRD patterns
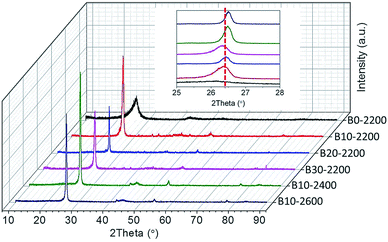
In order to study the effect of boron on the structural properties, XRD patterns for various boron-doped RCFs were shown in Fig. 3. Further, being calculated from the XRD results, the crystal parameters are listed in Table 1. Overall, none of the XRD patterns show the existence of boron. For boron-undoped RCFs, diffraction peaks of (004) were invisible and a broad (002) diffraction peak of hexagonal graphite was observed at 25.34°. The site and width of (002) peaks are significantly linked with the variation in the interlayer spacing.24 For boron-doped RCFs treated to 2200 °C, the (002) peak of the graphite was observed at 26.06°, 26.19°, 25.97° with increasing the B4C concentrations from 10 to 20 and 30%, respectively. The (002) peak position of B10-2200 shifted to a higher diffraction angle side from 26.06 to 26.19°, corresponding to the decrease of interlayer space (d002) and the enlargement of the crystallite size (Lc). This is also consistent with the well-known results that boron can accelerate graphitization of the materials.25 As such, boron can reduce the reaction activation energy of carbon fibers during the graphitization and further promote the transformation of the amorphous carbon to the ideal graphite. The decrease of d002 may be due to the crystalline improvement resulting from the addition of boron, and to the lowering in electron density due to the B substitution.26 For the B4C concentrations higher than 20%, the (002) peak position shifted to a lower diffraction angle side from 26.19 to 25.97°, corresponding to the increase of d002 and the enlargement of Lc. Moreover, an obvious change was observed in the boron-doped RCFs at higher than 2200 °C, as shown in Fig. 3 and S4.† For the boron-doped RCFs, as the HTT increased from 2200 to 2600 °C, the width of the (002) diffraction peak decreased gradually, and the (002) peak position shifted to a lower diffraction angle side from 26.06 to 26.51°, respectively, corresponding to the decrease of d002. On the other hand, the (004) diffraction peak was also observed and make sharper, this may be due to the presence of localised BC3 domains slightly influencing the periodic atomic arrangement of the hexagonal carbon network,15 and the sharper (002) diffraction indicated that an improvement of the crystalline order and a homogenous distribution of boron in the graphitic layer planes.27 Therefore, under this condition (2.16 at.% boron doped, 2400 °C), limited by the structure of rayon,6 it is easy to transform amorphous carbon in RCFs to graphite crystallite, however, more flaws exists in its carbon structure and the basal planes of graphitic layer are not parallel to the fiber axis as shown in Fig. 2(a) and (c).| No. | HTT (°C) | B concentration (at.%) | Crystallite parameters (nm) | |
|---|---|---|---|---|
| Surface analysisa | d002-spacing | Lc | ||
| a Surface analysis was undertaken by XPS. | ||||
| B0-2200 | 2200 | 0 | 0.3513 | 39 |
| B10-2200 | 2200 | 1.40 | 0.3418 | 52 |
| B20-2200 | 2200 | 1.87 | 0.3402 | 31 |
| B30-2200 | 2200 | 3.75 | 0.3430 | 78 |
| B0-2400 | 2400 | 0 | 0.3501 | 41 |
| B10-2400 | 2400 | 2.16 | 0.3370 | 22 |
| B0-2600 | 2600 | 0 | 0.3475 | 45 |
| B10-2600 | 2600 | 4.79 | 0.3361 | 8 |
The surface analysis results by XPS are also listed in Table 1. In spite of relatively high B4C concentrations, the solubility of boron in the graphite is limited to 0.4 at.% under room temperature.14 Therefore, redundant boron atoms may migrate to other sites, such as the interstitial locations. These atoms thus may cause the structural disorder.
3.3 Analysis of Raman spectra
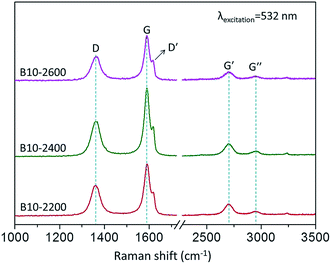
Raman spectra can complement the previous XRD analysis and contribute to a better understanding of the effects of boron on the carbon crystallite in boron-doped RCFs. Fig. 4 and 5 show the first- and second-order Raman spectra of boron-doped RCFs. The results are shown in Table 2. For comparison, Raman spectra of boron-undoped RCFs treated to 2400 and 2600 °C were presented in Fig. S5.† The bandwidth variations and peak shift are two indicators of the disorder or defects of carbon crystalline lattice.18 It can be seen from the Fig. 4 that the width of D-band of RCFs near 1358 cm−1 in the first-order Raman spectra corresponding to the defect lattice vibration mode increases,28 and its peak position shifts toward higher frequency after doping boron. Moreover, Fig. 4 shows that the intensities of D-band have the systematic increase after doping boron. It deduced that the increasing the B concentration leads to an obvious variation in the disordering degree of carbon hexagonal lattice. The second-order Raman spectra show a large number of important information about electronic and vibrational properties.29 One peak (G-band) is at 1580 cm−1 corresponding to an ideal graphitic lattice vibration mode (E2g mode). Based on D-band and G-band, the integrated intensity ratio of ID/IG is used to estimate the amount of defects in carbon materials.30–34 The data of ID/IG for various B-doped RCFs were shown in Table 2. It is noticed that the ID/IG values for boron-doped RCFs were slightly larger than the one for boron-undoped RCFs, indicating that boron-doped RCFs possesses more defects than boron-undoped RCFs due to the boron doping.35 Another peak at 2689 cm−1 (G′-band) is the overtone of the D-band.34 The intensity of the G'-peak is also sensitive to the presence of structural disorder.30 After doping boron, the G′-band peaks of RCFs slightly downshifted and the intensity decreased as the increase of the B4C concentration, indicating the increase of amount of defects. Moreover, the disorder-induced G′′-band of the boron-doped RCFs also appears at 2957 cm−1.18 These results indicate that the presence of boron atoms in the carbon network have disrupted the formation of graphene sheets.25 Other weak peaks in the spectra also show certain carbon structures. Most noticeable is that the shoulder peak (D′-band) at about 1620 cm−1 in the spectra of boron-doped RCFs is strengthened. Usually, the shoulder peak reflects two cases, including the vibration of carbon atoms in the surface layer of the graphite lattice and the vibration of displacement lattice in the carbon network. Therefore, it well indicates the displacement and solid solution of boron in the carbon lattice.22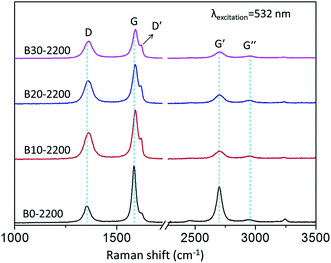 | ||
| Fig. 4 Room temperature Raman spectra of boron-doped RCFs treated to 2200 °C with different boron concentrations. | ||
| No. | HTT (°C) | Raman shift/cm−1 | FWHM/cm−1 | ID/IG | ||||
|---|---|---|---|---|---|---|---|---|
| D-band | G-band | D′-band | G′′-band | D-band | G-band | |||
| B0-2200 | 2200 | 1355 | 1585 | — | — | 39.6 | 23.1 | 0.35 |
| B10-2200 | 2200 | 1360 | 1591 | 1618 | 2950 | 50.1 | 31.0 | 0.57 |
| B20-2200 | 2200 | 1361 | 1590 | 1618 | 2948 | 50.5 | 33.1 | 0.61 |
| B30-2200 | 2200 | 1362 | 1592 | 1622 | 2958 | 49.6 | 29.8 | 0.60 |
| B10-2400 | 2400 | 1368 | 1589 | 1616 | 2951 | 20.1 | 29.0 | 0.53 |
| B10-2600 | 2600 | 1362 | 1588 | 1617 | 2952 | 28.3 | 28.8 | 0.58 |
Raman spectra of the boron-doped RCFs prepared at different HTT were also presented in Fig. 5. For comparison, Raman spectra of boron-undoped RCFs treated to 2400 and 2600 °C were also shown in Fig. S5.† It can be found that in Fig. 5 no obvious change were observed with a gradual increase of HTT, as Hagio et al.27 reported, indicating that the carbon crystalline structure in boron-doped RCFs do change less. The result was consistent with a relatively narrow distribution of the d002 spacing of 0.3361–0.3418 nm from the XRD analysis (Table 1).
3.4 Analysis of 11B-NMR
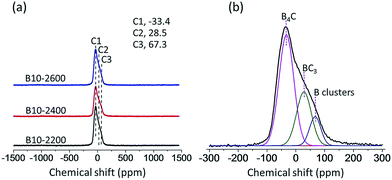
In order to learn more about the environment of boron species in the boron-doped RCFs, we perform the MAS 11B-NMR measurements as shown in Fig. 6. From the curves in Fig. 6(a), we can see that three 11B-NMR spectra of boron-doped RCFs at different temperatures disclose the similar line shape and show an asymmetric shape, which has been deconvoluted and shown in Fig. 6(b). It is clear that the boron atoms may exist in three different chemical environments. The chemical shift and line shape in MAS 11B-NMR spectra may be directly correlated with the coordination environment of doping boron.36After Gaussian fitting, we can see that three peaks may be assigned to B4C, boron clusters and ideal BC3 as shown in Fig. 6(b). The low-chemical shift component C1 at −33.4 ppm assigned to B4C.37 The observed chemical shift C2 at 28.5 ppm was assigned to BC3 induced by the substitution in the graphitic structure, and the chemical shift C3 at 67.3 ppm was assigned to boron clusters.38 Combined with SEM and XRD results, at high temperatures gaseous boron atoms can be doped into carbon structure of RCFs, and the doped boron atoms exist in three chemical environments, including B4C, boron clusters and BC3. It may be due to the gather of boron atoms to form boron clusters and B4C and when the substitution limit is reached. Moreover, at the temperature of higher than 2200 °C gaseous boron atoms invaded into the interior of RCFs, which directly resulted in the gaps between the graphite sheets as shown in Fig. 2.
3.5 Structural analysis by HR-TEM
A local characterization by HR-TEM was performed to portray more explicitly the microstructure of the boron-doped RCFs. Fig. 7 presents HR-TEM images of boron-doped RCFs with different boron concentrations. Four HR-TEM images in Fig. 7 disclosed the presence of the crystalline border except for the boron-undoped RCFs in Fig. 7(a). As Serin et al.39 reported, the presence of boron atoms in graphite lattice is consistent with the disorders. Moreover, the HR-TEM lattice images can disclose the orientation of the graphitic basal plane.40 As the total of boron doping, the graphitic basal planes twisted increases, which provides an assertive evidence for substituting carbon atoms in the graphite sheet by boron atoms. Thus, the substitution reaction slightly influenced the periodic atomic arrangement of the hexagonal carbon lattice.25 As a result, as shown in Fig. 7(b)–(d), the products of B10-2200, B20-2200 and B30-2200 mainly consisted of the graphite layer planes doped with boron. The magnified images gave the spacing of the (002) lattice fringes of 0.351 nm in Fig. 7(a), 0.342 nm in Fig. 7(b), 0.340 nm in Fig. 7(c), and 0.343 nm in Fig. 7(d). These results are in agreement with the XRD results as shown in Table 1. Further, electron diffraction verifies the previous results calculated from XRD and measured the spacing of the (002) lattice fringes. SAED patterns of four products in Fig. 7 show a (002) rings and weaker (004) rings visible in the XRD patterns. It indicates that the d002 spacing also varies in a small scale, and the crystallite size (Lc) is small. As such, these results from the HR-TEM analysis indicate that boron atoms have entered the graphite lattice by substituting for carbon at the trigonal sites and by intruding into graphite interlayer as interstitial atoms.3.6 Oxidation resistance measured by TGA
In this work, boron atoms were firstly doped into RCFs. Oxidation resistance of boron-undoped and boron-doped RCFs was determined using TGA. The results were shown in Fig. 8. Fig. 8(a) shows the TGA curves of boron-doped RCFs treated to 2200 °C with different boron concentrations. It can be seen that the oxidation onset temperature is close to 600, 700, 710, and 750 °C for the samples of B0-2200 (0 at.% B doped), B10-2200 (1.40 at.% B doped), B20-2200 (1.87 at.% B doped) and B30-2200 (3.75 at.% B doped), respectively. The result indicates the improvement of oxidation resistance of RCFs induced by doping boron, and their oxidation resistance increases with the increase of the boron concentration. On the other hand, increasing the HTT also improves the oxidation resistance of boron-doped RCFs. The result was shown in Fig. 8(b). The oxidation onset temperature is close to 700, 750, and 790 °C for the samples of B10-2200, B10-2400, and B10-2600, respectively. These results were consistent with our previous work.17 Thus, boron-doped RCFs also have higher oxidation inhibition, similar to boron-doped polyacrylonitrile-based CFs. For comparison, boron-undoped RCFs treated to different temperatures were also presented in Fig. 8(c). The more detailed comparisons of oxidation resistance were shown in Fig. S7.† It was found that for boron-doped RCFs treated to 2400 and 2600 °C, the improvement of oxidation resistance induced by doping boron is not remarkable. It may be due to the high degree of graphitization obtained at high temperature. Moreover, the mechanisms on the improvement of oxidation resistance have been explained in many references,17,19,41 and thus we would not discuss here. The systematic studies on the oxidation resistance of boron-doped carbon materials are underway and will be reported in future contributions.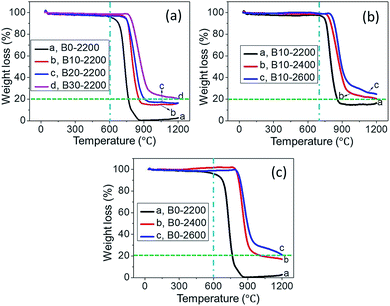 | ||
| Fig. 8 TGA curves of boron-doped RCFs treated to 2200 °C with different boron concentrations (a), with different HTTs (b), and boron-undoped RCFs treated to different temperatures (c). | ||
4. Conclusions
For the first time, boron-doped RCFs with tunable amount of boron have been prepared by exposing the RCFs in a vapor of boron at high temperatures. The fabrication of boron-doped RCFs was very sensitive to the temperature and the concentration of B4C in the graphite crucible. As increasing from 2200 to 2600 °C in the graphite crucible containing the 10% B4C, the amount of boron in RCFs increased from 1.40 to 4.79 at.%. Further, a systematic analysis of the structural evolution of RCFs induced by doping boron was performed by means of SEM, TEM, XRD and Raman spectroscopy. Boron promotes the transformation of the amorphous carbon to the ideal graphite, and produces more flaws. After doping boron at high temperatures, the original structure of RCFs no longer exists because of the stronger erosion of boron vapor. The evidence of BC3 in the signal components of 11B-NMR spectra indicated that highly coordinated boron atoms substituted the carbon atoms at the trigonal sites within the graphene sheet. Other than the substitutional boron, the presence of the small B4C and boron clusters generates the gaps between graphite sheets in RCFs. Moreover, the TGA curves indicated that doping boron greatly improved the oxidation inhibition of RCFs up to 800 °C.Acknowledgements
This work was supported by the National Basic Research Program of China (no. 2011CB605802), the National Defense Science and Technology Innovation Program of China Academy of Sciences, National Key Technology Research and Development Program of the Ministry of Science and Technology of China (2013BAC14B00), Shanxi province science and technology tacking fund (no. 201203013009-3) and School science foundation of North University of China. The authors are very grateful to Prof. Yongnan Zhao at Tianjin Polytechnic University for providing help and advice in English language.References
- X. Zhang, Y. Lu, H. Xiao and H. Peterlik, J. Mater. Sci., 2014, 49, 673–684 CrossRef CAS.
- H. Rong, Z. Ryu, J. Zheng and Y. Zhang, Carbon, 2002, 40, 2291–2300 CrossRef CAS.
- C. Sauder, J. Lamon and R. Pailler, Carbon, 2004, 42, 715–725 CrossRef CAS PubMed.
- A. Ramos, I. Camean and A. B. Garcia, Carbon, 2013, 59, 2–32 CrossRef CAS PubMed.
- X. Qin, Y. Lu, H. Xiao, Y. Wen and T. Yu, Carbon, 2012, 50, 4459–4469 CrossRef CAS PubMed.
- C. Pradere, J. C. Batsale, J. M. Goyhénèche, R. Pailler and S. Dilhaire, Carbon, 2009, 47, 737–743 CrossRef CAS PubMed.
- S. Wang, Z. H. Chen, W. J. Ma and Q. S. Ma, Ceram. Int., 2006, 32, 291–295 CrossRef CAS PubMed.
- Z. Wangxi, L. Jie and W. Gang, Carbon, 2003, 41, 2805–2812 CrossRef.
- I. M. K. Ismail and W. P. Hoffman, Carbon, 1991, 29, 587–594 CrossRef CAS.
- D. Li, H. Wang and X. Wang, J. Mater. Sci., 2007, 42, 4642–4649 CrossRef CAS.
- H. Xiao, Y. Lu, W. Zhao and X. Qin, J. Mater. Sci., 2014, 49, 794–804 CrossRef CAS.
- T. Liu, R. Y. Luo, S. H. Yoon and I. Mochida, J. Power Sources, 2010, 195, 1714–1719 CrossRef CAS PubMed.
- H. Q. Wang, Q. G. Guo, J. H. Yang, Z. J. Liu, Y. Zhao, J. Li, Z. H. Feng and L. Liu, Carbon, 2013, 56, 296–308 CrossRef CAS PubMed.
- C. E. Lowell, J. Am. Ceram. Soc., 1967, 50, 142–144 CrossRef CAS PubMed.
- T. Shirasaki, A. Derre, M. Menetrier, A. Tressaud and S. Flandrois, Carbon, 2000, 38, 1461–1467 CrossRef CAS.
- J. Y. Howe and L. E. Jones, Carbon, 2004, 42, 461–467 CrossRef CAS PubMed.
- H. Wang, Q. Guo, J. Yang, Z. Liu, Y. Zhao, J. Li, Z. Feng and L. Liu, Carbon, 2013, 56, 296–308 CrossRef CAS PubMed.
- M. Endo, T. Hayashi, S. H. Hong, T. Enoki and M. S. Dresselhaus, J. Appl. Phys., 2001, 90, 5670–5674 CrossRef CAS PubMed.
- L. E. Jones and P. A. Thrower, Carbon, 1991, 29, 251–269 CrossRef CAS.
- L. E. Jones and P. A. Thrower, J. Chim. Phys. Phys.-Chim. Biol., 1987, 84, 1431–1438 CAS.
- Y. Zhao, Z. Liu, H. Wang, J. Shi, J. Zhang, Z. Tao, Q. Guo and L. Liu, Carbon, 2013, 53, 313–320 CrossRef CAS PubMed.
- J. E. Chen, X. Xiong, P. Xiao and H. B. Zhang, Carbon, 2010, 48, 2341–2346 CrossRef CAS PubMed.
- E. Fitzer, Carbon, 1989, 27, 621–645 CrossRef.
- N. Iwashita, C. R. Park, H. Fujimoto, M. Shiraishi and M. Inagaki, Carbon, 2004, 42, 701–714 CrossRef CAS PubMed.
- S. C. Lyu, J. H. Han, K. W. Shin and J. H. Sok, Carbon, 2011, 49, 1532–1541 CrossRef CAS PubMed.
- M. Endo, C. Kim, T. Karaki, T. Tamaki, Y. Nishimura, M. J. Matthews, S. D. M. Brown and M. S. Dresselhaus, Phys. Rev. B: Condens. Matter Mater. Phys., 1998, 58, 8991–8996 CrossRef CAS.
- T. Hagio, M. Nakamizo and K. Kobayashi, Carbon, 1989, 27, 259–263 CrossRef CAS.
- L. Nikiel and P. W. Jagodzinski, Carbon, 1993, 31, 1313–1317 CrossRef CAS.
- M. S. Dresselhaus, G. Dresselhaus, R. Saito and A. Jorio, Phys. Rep., 2005, 409, 47–99 CrossRef PubMed.
- R. Rozada, J. Paredes, S. Villar-Rodil, A. Martínez-Alonso and J. D. Tascón, Nano Res., 2013, 6, 216–233 CrossRef CAS PubMed.
- M. B. Vázquez-Santos, E. Geissler, K. László, J.-N. Rouzaud, A. Martínez-Alonso and J. M. D. Tascón, J. Phys. Chem. C, 2011, 116, 257–268 Search PubMed.
- L. M. Malard, M. A. Pimenta, G. Dresselhaus and M. S. Dresselhaus, Phys. Rep., 2009, 473, 51–87 CrossRef CAS PubMed.
- Z. Ni, Y. Wang, T. Yu and Z. Shen, Nano Res., 2008, 1, 273–291 CrossRef CAS PubMed.
- M. A. Pimenta, G. Dresselhaus, M. S. Dresselhaus, L. G. Cancado, A. Jorio and R. Saito, Phys. Chem. Chem. Phys., 2007, 9, 1276–1290 RSC.
- Z. H. Sheng, H. L. Gao, W. J. Bao, F. B. Wang and X. H. Xia, J. Mater. Chem., 2012, 22, 390–395 RSC.
- Y. Lee, D. Y. Han, D. Lee, A. J. Woo, S. H. Lee and Y. K. Kim, Carbon, 2002, 40, 403–408 CrossRef CAS.
- M. Murakami, T. Shimizu, M. Tansho, Y. Takano, S. Ishii, E. A. Ekimov, V. A. Sidorov and K. Takegoshi, Diamond Relat. Mater., 2009, 18, 1267–1273 CrossRef CAS PubMed.
- M. Murakami, T. Shimizu, M. Tansho and Y. Takano, Sci. Technol. Adv. Mater., 2008, 9, 044103 CrossRef.
- V. Serin, R. Brydson, A. Scott, Y. Kihn, O. Abidate, B. Maquin and A. Derré, Carbon, 2000, 38, 547–554 CrossRef CAS.
- G. S. Zhou, Y. Q. Liu, L. L. He, Q. G. Guo and H. Q. Ye, Carbon, 2011, 49, 2883–2892 CrossRef CAS PubMed.
- L. R. Radovic, M. Karra, K. Skokova and P. A. Thrower, Carbon, 1998, 36, 1841–1854 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra07892f |
| This journal is © The Royal Society of Chemistry 2014 |