A general approach towards efficient catalysis in Pickering emulsions stabilized by amphiphilic RGO–Silica hybrid materials†
Abstract
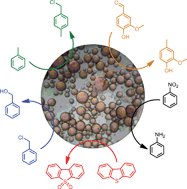
A general approach towards efficient emulsion catalysis has been achieved using amphiphilic RGO–silica hybrid materials with suitable surface wettability and mesoporous structures. On the basis of the promising hybrids, a Pickering emulsion with droplets from 20–100 μm was formed and a broad range of reactions was facilitated.

 Please wait while we load your content...
Please wait while we load your content...