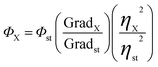Nitrogen-doped carbon dots with heterogeneous multi-layered structures
Fengyi Du†
*a,
Jing Yuan†a,
Miaomiao Zhang†a,
Jianan Li†b,
Zhou zhoua,
Zhang Lia,
Mulan Caoc,
Junhui Chena,
Lirong Zhanga,
Xia Liua,
Aihua Gonga,
Wenrong Xua and
Qixiang Shao*a
aSchool of Medical Science and Laboratory Medicine, Jiang Su University, ZhenJiang 212013, PR China. E-mail: biodfy@ujs.edu.cn; Fax: +86 05115038449; Tel: +86 05115038449
bDepartment of Otolaryngology Head and Neck Surgery, Chinese PLA General Hospital, Beijing 100853, PR China
cCollege of Biological and Chemical Engineering, Panzhihua University, Panzhihua 617000, PR China
First published on 12th August 2014
Abstract
Photoluminescent carbon dots (CDs) with graphite-like phase structure have been widely reported. Herein, we first report nitrogen-doped carbon dots (NCDs) with heterogeneous multi-layered structures by bottom-up hydrothermal carbonization process. The formation and surface passivation of NCDs was accomplished simultaneously. The as-prepared NCDs possess stable photoluminescence, solubility in water and various functional groups on their surface. XRD results clearly showed that there were two kinds of interlayer lattice spaces in the phase structure of prepared NCDs, which corresponded to the {002} plane of graphite and the {001} plane of graphite oxide. We further demonstrated that NCDs with good biocompatibility were easily and quickly internalized by cancer cells and are particularly suitable for multicolour real-time cellular imaging.
1 Introduction
Over the last decade, fluorescent carbon dots (CDs)1,2 have attracted tremendous attention due to their remarkable optical properties, water dispensability, chemical inertness, good photostability, low cytotoxicity, and excellent biocompatibility, compared with organic dyes and other semiconductor nanodots with heavy metal cores. Taking advantage of these properties, the CDs have a wide range of technological applications in bio-imaging,3 drug delivery,4 photocatalysis,5 bio-sensing,6 optoelectronic devices,7 and energy conversion/storage devices.8 Fluorescent carbon nanoparticles were firstly produced by Sun and his co-workers.9 Due to their non-toxicity, biocompatibility, and harmless-to-environment, CDs were considered as a brand-new class of fluorescent materials.10Up to now, the strategies for synthesizing CDs are mainly classified into two groups; namely the top-down and bottom-up methods.1 Top-down methods generally needs to break down the large carbonaceous materials and polymerize into the nanoparticles by using physical or chemical methods, including arc discharge,11 plasma treatment,12 laser ablation,13–15 and electrochemical oxidation.16 In contrast, the bottom-up methods involves the carbonization of small molecular precursors into the CDs, consisting of hydrothermal carbonization,17,18 micro-wave hydrolysis19 and combustion methods.20 Compared to the top-down approaches, it is more conducive to control the chemical compositions and physical characters by bottom-up approaches. Consequently, bottom-up methods are mostly adopted for the synthesis of various CDs with excellent optical properties.
Despite the controversial feature of photoluminescent (PL) origins, the involvement of surface states in the radiative recombination of CDs has been widely accepted.21–24 These surface defects create emission energy traps for the nanoparticles through introducing traps states with different energy levels, leading to excitation-wavelength dependent emissive properties. Furthermore, Wang25 and Cushing26 demonstrated that a common origin of PL luminescence in CDs and grapheme quantum dots (GQDs) was assigned to special edge states. It is well known that band gap of grapheme is need to open by reducing its size to the nanometer scale and introducing defects or by modifying its two-dimensional (2D) carbon framework with functional groups to enable fluoresecence. According to the above evidences, we speculate that CDs and GQDs have similar phase structure and surface functional groups, as shown in Scheme 1.
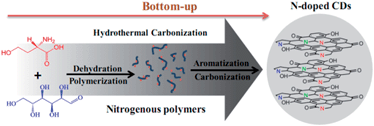 | ||
| Scheme 1 Schematic synthesis of multi-layered NCDs from hydrothermal carbonization of glucose and serine. | ||
Herein, the N-doped CDs were synthesized by hydrothermal carbonization with serine and glucose as precursors. Based on XRD results, we explore that the N-doped CDs have common phase structure of both graphite and graphite oxide, and further identify that the functional groups bear responsibility for dependent emissive fluorescence. Finally, the excellent multicolour N-doped CDs are applied in the bioimaging due to their good PL properties and biocompability.
2 Experimental
2.1 Materials
Glucose, serine and quinine sulfate (98%, suitable for fluorescence), 3-(4,5-dimethyl-2-thiazoyl)-2,5-diphenyltetrazolium bromide (MTT, 98%) were obtained from Aladdin. NaH2PO4, Na2HPO4, and H2SO4 were obtained from Guangfu Fine Chemical (Tianjin, China). Fetal bovine serum (FBS) and Dulbecco Minimum Essential Medium (DMEM) medium were purchased from Invitrogen China limited (China, Shanghai). And all of the chemicals were analytical reagent grade and were used without further purification. All the aqueous solution was prepared using double distilled water.2.2 Synthesis of the fluorescent carbon dots
The experimental procedure is described as follows: 1 g of glucose and 1.13 g of serine (the molar ratio is 1/2) were dissolved in 20 mL of deionized water. The mixture was placed into a stainless steel autoclave with a Teflon liner of 50 mL capacity. Subsequently, the autoclave was placed in an oven of 180 °C for 4 hours. After the reaction was finished, the solution was filtrated by filter membrane to remove black precipitates. The solution was dialyzed using dialysis membrane with molecular weight cut-off of 1000 for four days. Then, the dialyzed fluid was collected and frozen dried. Finally, the NCDs powers were obtained and saved for further characterization and application.2.3 Instrumentation and characterizations
The chemical structure of NCDs was identified by Fourier Transform Infrared (FT-IR) spectra (Nicolet Nexus 470, America). The morphologies of the CDs were examined by high resolution transmission electron microscopy (HRTEM) on a JEM-2100 microscope (Jeol, Japan) with an accelerating voltage of 200 kV. UV-vis absorption was characterized by UV-2450 UV-vis Spectrophotometer (Shimadzu, Japan). PL emission measurements were performed using Cary Eclipse Fluorometer (Varian, America). The carbon structure of the NCDs was recorded by Raman spectra on a Renishaw1000 microspectrometer with excitation wavelength of 514.5 nm. The crystal structure of the NCDs was characterized via X-ray diffraction (XRD) patterns on a Rigaku-D/MAX2500 diffractometer (Rigaku, Japan) equipped with Cu Kα (λ = 0.15405 nm) radiation at a scanning speed of 4° min−1 in the range from 5° to 80°. The composition of the NCDs was inferred from X-ray photoelectron spectroscopy (XPS) by a Kratos AXISUl Ultraspectrometer with a Al Kα X-ray as excitation source (1486.71 eV).2.4 Measurement of fluorescence quantum yields
The quantum yield (Φ) of the as-synthesized carbon nanodots was determined by a comparative method.27 As the standard measurement, the quinine sulfate (literature quantum yield: 54%) was dissolved in 0.1 M H2SO4 (refractive index (η) = 1.33) and the CDs were dissolved in distilled water (η = 1.33) at different concentrations. All the absorbant values of the solutions at the excitation wavelength were measured with UV-vis spectrophotometer. Photoluminescence (PL) emission spectra of all the sample solutions were recorded by FLS920 fluorometer at an excitation wavelength of 360 nm. The samples were then measured by PL spectrometer in order to get the PL emission intensity at the excitation wavelength with which the C-dots and the reference have the same UV absorbance. Then the quantum yield was calculated by the following equation:The st and X denote standard group and test group respectively. Φ is the fluorescence quantum yield, Grad is the gradient from the plot of integrated fluorescence intensity vs. absorbance, and η is the refractive index of the solvent. In order to minimize the re-absorption effects, absorbance in the 10 mm fluorescence cuvette should never exceed 0.1 at the excitation wavelength.
2.5 Cell viability assay
The cytotoxicity of NCDs was evaluated on A549 cells (adenocarcinomic human alveolar basal epithelial cells) using a modified MTT assay. Cells were seeded in a 96-well plate at a density of 4 × 104 cells per well and incubated for 24 h at 37 °C and 5% CO2, and then the growth medium was replaced with DMEM medium containing different concentrations of CDs. Each sample was prepared in triplicates. After incubation for 24 h, 20 μL of 2.5 mg mL−1 of MTT solution (in 0.01 M sterilized PBS, pH = 7.4) was added to each well and incubated for another 4 h at 37 °C under 5% CO2. After all medium removed, each well was added with DMSO (150 μL) and shaken for 15 min. The absorbance of each well was measured at 490 nm using Synergy HT Multi-Mode Microplate Reader (BioTek, USA) with pure DMSO as blank. Non-treated cells (in DMEM) were used as control and the relative cell viability (mean ± SD, n = 3) was expressed as Abssample/Abscontrol × 100%.2.6 Cell imaging
A549 cells were chosen to demonstrate the feasibility of NCDs for the cell imaging. For fluorescent images, three different kinds of cells were seeded in a 24-well plate containing 15 mm diameter glass cover-slips, in culture medium containing 10% FBS incubated for 24 h at 37 °C and 5% CO2. The NCDs probes containing medium were then added to the cells at a concentration of 100 μg mL−1. Cells were incubated at 37 °C under 5% CO2 for 6 h. After washing, confocal lasers scanning fluorescence microscopy (Zeiss LSM-710) was used to observe the fluorescence in the cells with different wavelengths laser to excite the NCDs.3 Results and discussion
3.1 The preparation and morphology of NCDs
The successful preparation of NCDs was carried out by using glucose acid as carbon source and serine as surface passivation agent according to the Scheme 1. To prepare fluorescent NCDs effectively, the influence of reaction time and surface passivation agent on the PL of NCDs was investigated. On the one hand, the PL intensity of the reaction solution increased with the increase of reaction time at early stage (as shown in Fig. 1a). With the reaction time, the PL intensity of the reaction solution reached maximum value at 4 hours, which was similar to previous researches. When time exceeded 5 hours, the PL intensity became relatively weak, which was supposed to form larger particles. On the other hand, the appropriate amount of surface passivation was essential for the NCDs to get optimum PL. Fig. 1b exhibited the influence of serine dosage on the PL intensity of NCDs. First, the PL intensity enhanced with an increase in serine dosage, and then the maximum PL intensity was achieved when the ratio of glucose to serine is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. In our previous works, we have demonstrated that single glucose or amino could barely form the carbon quantum dots under this condition of hydrothermal reactionx. The amino groups accelerated the dehydration and carbonization of glucose and serine to form the carbon quantum dots. Consequently, excessive amounts of amino acid or glucose could not achieve higher PL intensities. So, a proper amount of serine could accelerate dehydration process and the formation of NCDs. In the following sections, all samples used for characterization were synthesized under the conditions of 1 g glucose, 1.13 g serine and 4 hours hydrothermal treatment, unless specifically mentioned.
2. In our previous works, we have demonstrated that single glucose or amino could barely form the carbon quantum dots under this condition of hydrothermal reactionx. The amino groups accelerated the dehydration and carbonization of glucose and serine to form the carbon quantum dots. Consequently, excessive amounts of amino acid or glucose could not achieve higher PL intensities. So, a proper amount of serine could accelerate dehydration process and the formation of NCDs. In the following sections, all samples used for characterization were synthesized under the conditions of 1 g glucose, 1.13 g serine and 4 hours hydrothermal treatment, unless specifically mentioned.
The formation mechanism of NCDs via hydrothermal reaction may undergo dehydration, polymerization, aromatization and carbonization. In this experiment, a certain amount of serine in the raw solution was necessary to enhance the NCDs formation which was critical for carbonization degree. The morphology of NCDs was characterized by TEM. As shown in Fig. 1c, the NCDs exhibited uniform dispersion and discrete quasi spherical shape without apparent aggregation. Fig. 1d displayed that the diameter distribution of NCDs matched well with Gaussian fitting curve. The average size of NCDs was about 2.3 nm, which was determined by statistical analysis of more than one hundred particles by using the Image J software.
3.2 The characterization of the NCDs
The optical properties of NCDs were characterized by UV-vis spectrum and photoluminescence (PL) spectrum. The NCDs aqueous solution exhibited a long-term homogeneous phase without any noticeable precipitation under ambient conditions at room temperature. The absorption spectrum (Fig. 2a) showed that no obvious absorption peak was observed. The NCDs aqueous solution was pale yellow and transparent in daylight but changed to bright blue under UV excitation, which could be easily observed with naked eyes (Fig. 2a, inset). The quantum yield measured by using quinine bisulfate as standard sample was 13.4%. As shown in Fig. 2b, the NCDs displayed broad range of emission wavelengths (preeminent multicolor fluorescent emission) and excitation-dependent emission. The strongest emission peak shifts from 420 nm to 480 nm and the intensity decrease gradually as the excitation from 340 nm to 400 nm. The strongest fluorescence emission was observed centered at approximately 450 nm under 350 nm excitation. Thus, the NCDs exhibited different emission colors including blue, green and red when they were excited by different excitation wavelengths. This phenomenon has been widely observed in the luminescent carbon nanomaterials,28,29 which may be due to the optical selection of different surface defect states near the Fermi level of CDs.30 Meanwhile, almost no bleaching was observed after continuous irradiation with UV light for 8 h (Fig. 2c), suggesting the emission was quite stable. When the excitation wavelength changes from 330 nm to 400 nm, the photoluminescence (PL) peak correspondingly shifts from 425 nm to 460 nm.The carbon structures of the NCDs were confirmed by Raman spectra. In order to properly explain the Raman spectra, fitting process was conducted on the original Raman data by PeakFit (v4.12) software. A good fit (R2 = 0.998) was found when four Lorenztians located at 1179 cm−1, 1323 cm−1 (D), 1449 cm−1, 1566 (G) cm−1 were used to deconvolute the Raman spectrum in Fig. 3a. The bands at 1179 cm−1 and 1449 cm−1 might be related to the phonon frequencies at K and M point of graphite equivalent Brillouin zone. The vibration peak at 1318 cm−1 in Raman spectrum (Fig. 2d) was ascribed to the D band, which arised from sp3 dangling bonds of the disorder carbon structure. The G bands at 1563 cm−1 corresponded to the mode of graphite, which attributed to the vibration of sp2 band carbon atoms. The relatively intensity of the disorder D band to the crystalline G band (ID/IG) was about 0.75, which confirmed the partial amorphous nature of the NCDs.
3.3 The chemical structure and composition of the NCDs
The chemical structure and composition of the NCDs was investigated using FITR spectrum and XPS pattern. As shown in Fig. 3a, the FTIR exhibited characteristic absorption bands of O–H and N–H stretching vibrations of amine groups at 3384 cm−1, C–H stretching vibrations at 2935 cm−1, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations at 1619 cm−1, C
O stretching vibrations at 1619 cm−1, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations at 1456 cm−1, C–N stretching vibrations at 1382 cm−1 and C–H stretching vibrations at 1074 cm−1. The XPS survey spectrum of the as-produced NCDs (Fig. 3b) showed three typical peaks at 284.0, 400.0 and 530.6 eV, which indicated that the NCDs were mainly composed of carbon, nitrogen and oxygen atoms. The high-resolution spectrum of N1S revealed the presence of pyridinic type (399.5 eV), pyrrolic type (400.6 eV) and graphitic type (401.1 eV) N atoms. The O/C atomic ratio for NCDs (about 0.34) was much lower than that of the precursor glucose (1
C stretching vibrations at 1456 cm−1, C–N stretching vibrations at 1382 cm−1 and C–H stretching vibrations at 1074 cm−1. The XPS survey spectrum of the as-produced NCDs (Fig. 3b) showed three typical peaks at 284.0, 400.0 and 530.6 eV, which indicated that the NCDs were mainly composed of carbon, nitrogen and oxygen atoms. The high-resolution spectrum of N1S revealed the presence of pyridinic type (399.5 eV), pyrrolic type (400.6 eV) and graphitic type (401.1 eV) N atoms. The O/C atomic ratio for NCDs (about 0.34) was much lower than that of the precursor glucose (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) due to the chemical transformation during the synthesis processes. The N/C atomic ratio was calculated to be 0.09, confirming the successful incorporation of N atoms into the NCDs by hydrothermal process. The elemental analysis experiment demonstrated that the amount of nitrogen was about 7.34%, carbon is 55.32, hydrogen was 3.62%, oxygen was 66.72% (calculated). These results revealed that the molecular structures of NCDs were mainly contained amino groups, plentiful carbonyl, carboxylate and hydroxyl groups. Obviously, the functional groups could be attributed to degradation of carbohydrates via hydrothermal reaction. As a result, the presence of functional groups located at their surface endowed the NCDs with high solubility and enhanced the optical properties.
1) due to the chemical transformation during the synthesis processes. The N/C atomic ratio was calculated to be 0.09, confirming the successful incorporation of N atoms into the NCDs by hydrothermal process. The elemental analysis experiment demonstrated that the amount of nitrogen was about 7.34%, carbon is 55.32, hydrogen was 3.62%, oxygen was 66.72% (calculated). These results revealed that the molecular structures of NCDs were mainly contained amino groups, plentiful carbonyl, carboxylate and hydroxyl groups. Obviously, the functional groups could be attributed to degradation of carbohydrates via hydrothermal reaction. As a result, the presence of functional groups located at their surface endowed the NCDs with high solubility and enhanced the optical properties.
3.4 The phase structure of the NCDs
The phase structural properties of the NCDs were investigated by XRD, where pristine graphite power and graphite oxide power as comparison, as shown in Fig. 4. Two diffraction peaks of NCDs around 29.6° and 42.1° can be observed in the Fig. 4, corresponding to the {002} and {100} planes of graphitic framework. Compared to the ordered crystal structure of graphite (002 planes, 2θ = 26.5°), the diffraction peak of NCDs around 29.6° (d3 = 0.3 nm) with a upward shift become weak and broad, which is attributed to highly disorder carbon and decrease in the sp2 (C–C) layers spacing in the carbonization process. The main peak of NCDs around 12.08 (d4 = 0.735 nm) is similar to the typical diffraction peak (2θ = 10.6°, 001 plane) of graphite oxide (GO), which is attributed to an increase in sp3 layers spacing. During this process, many functional groups, such as hydroxyl, carbonyl, epoxy and amino groups were bonded to the edges of basal planes of the crystal structure, inducing an increase in the interlayer spacing. The decrease in the three peaks intensity and the increase of the full width half maximum (FWHM) are due to the amorphous carbon structure of the prepared NCDs. The result further indicated these NCDs with poor crystalline nature possessed heterogeneous multi-layered structure, which was consistent with many other reports about CDs.31–333.5 Application in biomedical imaging
As a new kind of fluorescent probes, the potential application of NCDs was evaluated for bioimaging in biomedicine. Firstly, the inherent cytotoxicity of NCDs was assessed in human lung adenocarcinoma of A549 cells through MTT assay. Fig. 5a showed that NCDs exhibited extremely low cytotoxicity with cells viability about 90% even at a concentration of 300 μg mL−1 and in 48 hours exposure time. In addition, the bright-field optical images also validated no morphological change after NCDs treatment, suggesting the as-synthesized bagasse-derived CDs had good biocompatibility.The potential application of the as-prepared NCDs was evaluated by laser scanning confocal microscopy. After incubation with NCDs for 6 h, the A549 cells were washed to remove extracellular NCDs. We found that the labelled cells were brightly illuminated with multicolor images due to strong fluorescence emitting from NCDs which distributed in cytosol (Fig. 5b). Consistent with optical results (Fig. 2), the NCDs exhibited blue, green and red color upon excitation at 405 nm, 488 nm and 543 nm. Furthermore, the combination of different fluorescent images demonstrated that emissive site displayed highly co-localization in the cytoplasm (Fig. 5c). These emissive traps, such as C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and O
O and O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–OH, could introduce surface defects with different energy levels, endowing CDs with excitation-dependent properties under various excitation energies.25,34–36 These results suggested the excitation-dependent emission was ascribed to emissive traps in the radiative transition of NCDs instead of size-dependent effect. It was noted that the NCDs were clearly observed in cell membrane and cytoplasm regions, especially around cell nucleus, while the photoluminescence of NCDs was very weak in the cell nucleus. In view of this result, we suppose that large amount of NCDs can be readily internalized into the cells across the cell membrane. It might be explained by the fact that uptaked NCDs by cells could be spread to cytoplasm after escaping from endosome in a short time. These results gave us the conclusion that CDs were of low cytotoxicity with cells, which agreed well with the results of previous investigation on the interaction of living cells with other CDs. Thus, it can be seen that NCDs can be effectively applied in cell imaging and biological labelling.
C–OH, could introduce surface defects with different energy levels, endowing CDs with excitation-dependent properties under various excitation energies.25,34–36 These results suggested the excitation-dependent emission was ascribed to emissive traps in the radiative transition of NCDs instead of size-dependent effect. It was noted that the NCDs were clearly observed in cell membrane and cytoplasm regions, especially around cell nucleus, while the photoluminescence of NCDs was very weak in the cell nucleus. In view of this result, we suppose that large amount of NCDs can be readily internalized into the cells across the cell membrane. It might be explained by the fact that uptaked NCDs by cells could be spread to cytoplasm after escaping from endosome in a short time. These results gave us the conclusion that CDs were of low cytotoxicity with cells, which agreed well with the results of previous investigation on the interaction of living cells with other CDs. Thus, it can be seen that NCDs can be effectively applied in cell imaging and biological labelling.
4 Conclusions
In summary, photoluminescent NCDs with heterogeneous multi-layered structure have been first observed by bottom-up hydrothermal carbonization process. The as-prepared NCDs possess stable photoluminescence, solubility in water and various functional groups on their surface. The Raman and XRD illustrated the poor crystalline nature and heterogeneous multi-layer phase structure of NCDs. The two main kinds of interlayer lattice spaces in NCDs were similar with the {002} plane of graphite and {001} plane of graphite oxide. We further demonstrated that NCDs with good biocompatibility were easily and fastly internalized by cancer cells and particularly suitable for multicolour real-time cellular imaging. Investigations on the clear formation and phase structure of the NCDs are in progress.Acknowledgements
The research was supported by the National Natural Science Foundation of China (no. 81301316, 31200676, 81273202, 81372718), Doctoral Fund of Ministry of Education of China (no. 20123227120008), China Postdoctoral Science Foundation (2013M540425, 2014T70486, 2013M542520), Senior Talents Scientific Research Foundation of Jiangsu University (no. 13JDG022, 11JDG113). Zhenjiang City Social Development Fund (no. SH2013026).Notes and references
- S. N. Baker and G. A. Baker, Angew. Chem., Int. Ed., 2013, 49, 6726–6744 CrossRef PubMed.
- H. T. Li, Z. H. Kang, Y. Liu and S. T. Lee, J. Mater. Chem., 2013, 22, 24230–24253 RSC.
- A. Zhu, Q. Qu, X. Shao, B. Kong and Y. Tian, Angew. Chem., Int. Ed. Engl., 2012, 51, 7185–7189 CrossRef CAS PubMed.
- Q. L. Wang, X. X. Huang, Y. J. Long, X. L. Wang, H. J. Zhang and R. Zhu, Carbon, 2013, 59, 192–199 CrossRef CAS PubMed.
- H. T. Li, X. D. He, Z. H. Kang, H. Hui, Y. Liu and J. L. Liu, Angew. Chem., Int. Ed., 2010, 49, 4430–4434 CrossRef CAS PubMed.
- M. Liong, A. N. Hoang, J. Chung, N. Gural, C. B. Ford and C. Min, Nat. Commun., 2013, 1752 CrossRef PubMed.
- X. Guo, C. F. Wang, Z. Y. Yu, L. Chen and S. Chen, Chem. Commun., 2013, 48, 2692–2694 RSC.
- J. J. Huang, Z. F. Zhong, M. Z. Rong, X. Zhou, X. D. Chen and M. Q. Zhang, Carbon, 2014, 70, 190–198 CrossRef CAS PubMed.
- Y. P. Sun, B. Zhou, Y. Lin, W. Wang, K. A. Fernando and P. L. Pathak, J. Am. Chem. Soc., 2006, 128, 7756–7757 CrossRef CAS PubMed.
- S. Sahu, B. Behera, T. K. Maiti and S. Mohapatra, Chem. Commun., 2013, 48, 8835–8837 RSC.
- X. Y. Xu, R. Ray, Y. L. Gu, H. J. Ploehn, L. Gearheart, K. Raker and W. A. Scrivens, J. Am. Chem. Soc., 2004, 126, 12736–12737 CrossRef CAS PubMed.
- J. Wang, C. F. Wang and S. Chen, Angew. Chem., Int. Ed. Engl., 2012, 51, 9297–9301 CrossRef CAS PubMed.
- L. Cao, X. Wang, M. J. Meziani, F. S. Lu, H. F. Wang, P. J. G. Luo, Y. Lin, B. A. Harruff, L. M. Veca, D. Murray, S. Y. Xie and Y. P. Sun, J. Am. Chem. Soc., 2007, 129, 11318–11319 CrossRef CAS PubMed.
- S. L. Hu, P. K. Bai, S. R. Cao and J. Sun, Chem. Res. Chin. Univ., 2009, 30, 1497–1500 CAS.
- S. Hu, K. Niu, J. Sun, J. Yang, N. Zhao and X. Du, J. Mater. Chem., 2009, 19, 484–488 RSC.
- J. Lu, J. Yang, J. Wang, A. Lim, S. Wang and K. Loh, ACS Nano, 2009, 3, 2367–2375 CrossRef CAS PubMed.
- Q. Liang, W. Ma, Y. Shi, Z. Li and X. Yang, Carbon, 2013, 60, 421–428 CrossRef CAS PubMed.
- C. Zhu, J. Zhai and S. Dong, Chem. Commun., 2012, 48, 9367–9369 RSC.
- S. Qu, X. Wang, Q. Lu, X. Liu and L. Wang, Angew. Chem., Int. Ed. Engl., 2012, 51, 12215–12218 CrossRef CAS PubMed.
- R. Zhang, Y. Liu, L. Yu, Z. Li and S. Sun, Nanotechnology, 2013, 24, 225601 CrossRef PubMed.
- X. Li, S. Zhang, S. Kulinich, Y. Liu and H. Zeng, Sci. Rep., 2014, 4, 4976 Search PubMed.
- Y. Yang, J. Cui, M. Zheng, C. Hu, S. Tan, Y. Xiao, Q. Yang and Y. Liu, Chem. Commun., 2012, 48, 380–382 RSC.
- Z. Wu, P. Zhang, M. Gao, C. Liu, W. Wang, F. Leng and C. Huang, J. Mater. Chem. B, 2013, 1, 2868–2873 RSC.
- X. Zhai, P. Zhang, C. Liu, T. Bai, W. Li, L. Dai and W. Liu, Chem. Commun., 2012, 48, 7955–7957 RSC.
- L. Wang, S. Zhu, H. Wang, S. Qu, Y. Zhang, J. Zhang, Q. Chen, H. Xu, W. Han, B. Yang and H. Sun, ACS Nano, 2014, 8, 2541–2547 CrossRef CAS PubMed.
- S. Cushing, M. Li, F. Huang and N. Wu, ACS Nano, 2014, 8, 1002–1013 CrossRef CAS PubMed.
- C. Zhu, J. Zhai and S. Dong, Chem. Commun., 2012, 48, 9367–9369 RSC.
- B. De and N. Karak, RSC Adv., 2013, 3, 8286–8290 RSC.
- J. Zhou, X. Shan, J. Ma, J. Gu, Y. Qian, Z. Chen and J. Feng, RSC Adv., 2014, 4, 5465–5468 RSC.
- Y. Xu, M. Wu, Y. Liu, X. Z. Feng, X. B. Yin and X. W. He, Chemistry, 2013, 19, 2276–2283 CrossRef CAS PubMed.
- L. Tang, R. Ji, X. Li, G. Bai, C. Liu, J. Hao, J. Lin, H. Jiang, K. Teng, Z. Yang and S. Lau, ACS Nano, 2014, 8, 6312–6320 CrossRef CAS PubMed.
- H. Ding, P. Zhang, T. Wang, J. Kong and H. Xiong, Nanotechnology, 2014, 25, 205604 CrossRef PubMed.
- Z. Zhang, J. Hao, J. Zhang, B. Zhang and J. Tang, RSC Adv., 2012, 2, 8599–8601 RSC.
- H. Nie, M. Li, Q. Li, S. Liang, Y. Tan, L. Sheng, W. Shi and S. X. Zhang, Chem. Mater., 2014, 26, 3104–3112 CrossRef CAS.
- S. Zhu, J. Zhang, S. Tang, C. Qiao, L. Wang, H. Wang, X. Liu, B. Li, Y. Li and W. Yu, Adv. Funct. Mater., 2012, 22, 4732 CrossRef CAS PubMed.
- L. Bao, Z. Zhang, Z. Tian, L. Zhang, C. Liu, Y. Lin, B. Qi and D. Pang, Adv. Mater., 2011, 23, 5801–5806 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2014 |