A cyano and end-to-end azido bridged 3D copper(ii)–copper(i) mixed-valence coordination polymer and its transformation to copper nitride nanoparticles†
Abstract
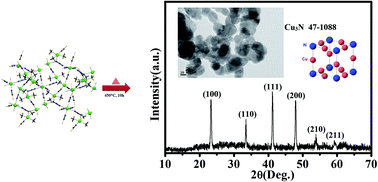
A new cyano and end-to-end azido bridged 3D copper(II)–copper(I) mixed valence polymer, {[Cu5(CN)6(NH3)6(N3)2]}n (1), copper-azide systems without cyanide {[Cu(NH3)2(N3)2]4}n (2), and a cyano-bridged copper analogue without azide {[Cu3(NH3)3(CN)4]}n (3) have been prepared and their structures have been revealed by X-ray crystallography. The cyano groups acting as μ2,η2 and azido anions have a rare asymmetric μ-1,1,3 bridging mode connecting the copper centers in 1 to form an infinite 3-D layered motif, whereas 2 consists of one dimensional polymer chains bridged by two azide moieties in μ-1 and μ-1,1,3 modes and 3 is a two dimensional CuIICuI infinite polymeric network formed by cyanide bridges. The solid-state transformation of all the three compounds has also been investigated. The present investigation suggests application of {[Cu5(CN)6(NH3)6(N3)2]}n (1) as a precursor for copper nitride nanoparticles.

 Please wait while we load your content...
Please wait while we load your content...