Reduced graphene oxide as a highly efficient adsorbent for 1-naphthol and the mechanism thereof†
Mohamed Mukthar Ali and
K. Y. Sandhya*
Department of Chemistry, Indian Institute of Space Science and Technology, Thiruvananthapuram, Kerala 695 547, India. E-mail: sandhya@iist.ac.in
First published on 24th September 2014
Abstract
Herein, adsorption and removal of 1-naphthol from water using reduced graphene oxide (rGO) is reported. The adsorption isotherms of different rGOs at different temperatures (303 and 323 K) fit well with Langmuir adsorption isotherms. At 303 K, the maximum adsorption capacities (qmax) of the relatively lower and higher surface area rGOs (l-rGO and h-rGO) are 3.57 and 5.55 mmol g−1 respectively. The qmax for the adsorption of 1-naphthol by h-rGO, at 323 K, is a remarkable 8.6 mmol g−1, which is the highest so far reported in the literature. The qmax value is greater than that of the h-rGO's water dispersible graphene counterpart reported in the literature. Acidic and alkaline pH increases the adsorption. Examination of the adsorption mixture using atomic force microscopy shows that the rGO is dispersed as few layered graphene nanosheets. The remarkably higher qmax is attributed to the higher surface area of rGO in aqueous solution due to its exfoliation by 1-naphthol. In summary, rGO is a promising material for the removal of 1-naphthol from water and our findings help in better understanding the mechanism of the adsorption of 1-naphthol and similar compounds by rGO.
1. Introduction
Dissolved organic matter in wastewater is a major concern for health, the environment and the ecosystem. The textile, leather, paper, printing, plastic industries etc. use various chemicals, most of them, aromatic in nature. Due to their non-degradability, aromatic compounds are persistent pollutants and removal of them from wastewater remains a challenge for industries. Aromatic compounds in wastewater cause damage to many forms of life. Potential carcinogenicity of aromatic hydrocarbons and their derivatives emphasize the importance of their removal from the aqueous system. Different techniques such as photodegradation1 sonochemical degradation,2 coagulation,3 biodegradation4 and adsorption5 etc. are employed to remove the pollutants. From these methods, adsorption is the most popular one because of its cost-effectiveness, simplicity, efficiency, fastness and due to the fact that it does not lead to the formation of harmful secondary products.Activated carbon powder is a well-known adsorbent which has been widely studied and applied due to its high adsorption capabilities.6,7 Recently, carbon nanomaterials such as carbon nanotubes (CNT)8 and graphene9,10 are attracting the attention in the management of aromatic pollutants due to their aromatic benzene rings and higher specific surface areas (SA) which facilitates the adsorption of aromatic compounds efficiently. Graphene due to its exceptionally high theoretical SA (∼2600 m2 g−1) and cost effectiveness is receiving greater attention from research community as an adsorbent. Li et al. used few-layered graphene nanosheets made by ultrasonication of a 1-methyl-2-pyrrolidinone (NMP) suspension, for the adsorption of fluoride from aqueous solutions achieving an adsorption capacity of 35 mg g−1.11 Functionalized/modified graphene is used for removal of heavy metal ions12 such as Pb2+, Cd2+ and Cr6+.13 Adsorption and removal of dyes such as methyl orange, methylene blue, rhodamine B, etc.14,15 from aqueous solutions using chemically exfoliated reduced graphene oxide (rGO)16,17 are investigated. Pei et al.18 reported the adsorption characteristics of naphthalene based aromatic effluents with different functional groups on GO and rGO. Removal of gaseous pollutants such as NOx, SOx and CO etc., inorganic cations, anions and organic dyes from environment are investigated using graphene materials.19 Aggregation of graphene in aqueous solution is a major issue which reduces the adsorption efficiency of rGO. Therefore, for better performance, water dispersible (modified) graphene materials20 are investigated as adsorbents for removal of pollutants. In accordance, an aqueous dispersible sulphonated graphene material which forms few layer graphene nanosheets in water exhibits the highest maximum adsorption capacity (qmax) for aromatic compounds such as naphthalene and 1-naphthol.9,21 1-Naphthol is an aromatic pollutant present in the environmental system which acts as precursor for insecticides and dyes. It causes hemolysis, nephritis, irritation to respiratory organs and skin and is a potential carcinogenic agent. Iron oxide modified graphene material (iron oxide-rGO)10 was reported to have an efficiency of 1.68 mmol g−1. However, none of these reports investigated the adsorption of 1-naphthol using (unmodified) rGO.
In this paper, we show that rGO has the highest adsorption efficiency for 1-naphthol, higher than the water dispersible counterpart reported in the literature. We conducted the adsorption experiment with two different rGOs, a relatively lower SA rGO (l-rGO) and higher SA rGO (h-rGO) and at different temperatures (303 and 323 K). The qmax values at 303 K of l-rGO and h-rGO are 3.57 mmol g−1 (515 mg g−1) and 5.55 mmol g−1 (800 mg g−1), respectively. At 323 K, the qmax value is a remarkable 8.6 mmol g−1 (1241.8 mg g−1) for h-rGO, evidently the highest value so far reported for 1-naphthol. Analysis of the adsorption mixture using atomic force microscopy (AFM) reveals that 1-naphthol disperses rGO as nanosheets of ≤10 nm thickness. The results show that rGO is a super adsorbent material for 1-naphthol and suggest that it may act as a highly efficient adsorbent for similar aromatic compounds, which are capable of dispersing rGO in aqueous solutions. The adsorption efficiency may depend on the structure of the adsorbate, the number of benzene rings and other factors.
2. Experimental
2.1 Raw materials
Graphite (21 μm) was purchased from Sigma Aldrich India Co. Ltd. Intercalated graphite (IC) was purchased from Anthracite industries. Potassium permanganate (99%), hydrogen peroxide (30%), hydrochloric acid (35%), sulfuric acid (98%) and 1-naphthol (99% of reagent grade) were purchased from Merck, India Ltd. Distilled water was used for all the experiments.2.2 Preparation of GO
Two different methods were used to prepare GO, in the attempt to make different SA rGOs. The GOs used for preparing comparatively lower and higher SA rGOs were represented as l-GO and h-GO, respectively. Preparation procedure of l-GO (modified Hummer's method) was described elsewhere.22l-GO was synthesized by reacting commercial graphite powder with H2SO4 and KMnO4 followed by addition of H2O2. The solid brown product was filtered and washed repeatedly with 1 M HCl and then with distilled water and finally dried under vacuum at room temperature.
For h-GO preparation, IC was used in the place of graphite. IC was thermally expanded by heating at 1050 °C for 30 seconds under argon atmosphere23 to produce the thermally expanded graphite (TEG). In a typical reaction, 2 g of TEG was mixed with 240 mL of conc. H2SO4 in a three neck RB flask. The flask was kept in an ice bath and then 16 g of KMnO4 were slowly added under stirring and the mixture was stirred at 90 °C for 24 h. Then the mixture was cooled to room temperature and 400 mL of water were added, followed by the slow addition of 50 mL of H2O2 till the color of the suspension turned to light brown. The product was washed with HCl solution three times and plenty of water to bring the down the pH to 5. The suspension was then centrifuged and the product was dried under vacuum at 70 °C overnight.
2.3 Preparation of rGO
In a typical procedure24 200 mg of GO and 200 mL water were introduced in a three neck flask and sonicated to yield a homogeneous yellow brown dispersion. To the homogenous solution 2 mL of hydrazine hydrate were added and the resultant solution was refluxed at 100 °C under a water cooled condenser for 24 h. The rGO gradually precipitated out as a black solid. The product was filtered off and washed with water and methanol and then dried at 60 °C under vacuum overnight. l-GO and h-GO after reduction yields l-rGO and h-rGO, respectively.2.4 Characterization
Perkin Elmer spectrum100 Fourier transform infra-red (FTIR) spectrophotometer was used for recording IR spectra. X-ray diffraction (XRD) patterns were obtained using Xpert-pro diffractometer with Kα wavelength (1.54 Å) of Cu metal. Thermogravimetric Analysis (TGA) was done using TA Q50. Absorption spectra were recorded using CARY 100 Bio UV-Vis spectrophotometer. Brunauer–Emmett–Teller (BET) SA analyses were done using Tristar II micromeritics SA analyzer. Degasification of the samples for SA analyses were done at 120 °C for 8 h, prior to nitrogen (N2) adsorption isotherm. N2 adsorption isotherms were carried out at 77 K using liquid N2 as a coolant. p/p0 values in the range of 0.05 to 0.25 were used for calculating the BET SA values. Raman spectral analyses were carried out using WiTech alpha 300R with 532 nm excitation laser. AFM analyses were carried out in Agilent 5500 scanning probe microscope.2.5 Adsorption studies
For adsorption studies, 5.26 mmol L−1 of 1-naphthol solution was used as stock solution. For the contact time studies, 0.1 mg mL−1 of rGO in water and 1 mmol L−1 of 1-naphthol solution were used. Effect of pH experiments were carried out using 1 mmol L−1 1-naphthol solutions. For adsorption isotherm studies, 0.1 mg mL−1 of rGO in water was used. All adsorption studies were done using a 20 mL sample tube with a working volume of 8 mL. Agitation was provided with a mechanical hand-shaker in dark conditions. Adsorbent was removed from the mixture by centrifugation and the supernatant was analysed using UV-Vis spectrophotometer. Absorbance value at 290 nm was used for determining the 1-naphthol concentration.3. Results and discussion
Fig. 1 provides the FTIR spectra, thermograms, N2 adsorption–desorption isotherms and Raman spectra of the l-GO and l-rGO. The formation of rGO is explained using the results of l-GO and l-rGO. The IR spectrum of l-GO (Fig. 1A) displays peaks at 3400 and 1640 cm−1 corresponding to the stretching and bending of hydroxyl (–OH) groups, peaks at 1718 and 1623 cm−1 are due to –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and skeletal –C
O and skeletal –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C– stretching vibrations, respectively. Epoxy C–O–C stretching peak is observed at 1223 cm−1. No peaks are observed in the IR spectrum of l-rGO indicating reduction of GO to rGO. The thermogram of l-GO shows two-step weight loss, around 100 and 200 °C and are attributed to the loss of water molecules and the removal of oxygen functional groups from GO respectively. The residual mass for l-GO at 750 °C is 35% and that of the rGO is ∼85%. The weight loss in l-rGO is attributed to the removal of edge functional groups. The results confirm the formation of rGO.
C– stretching vibrations, respectively. Epoxy C–O–C stretching peak is observed at 1223 cm−1. No peaks are observed in the IR spectrum of l-rGO indicating reduction of GO to rGO. The thermogram of l-GO shows two-step weight loss, around 100 and 200 °C and are attributed to the loss of water molecules and the removal of oxygen functional groups from GO respectively. The residual mass for l-GO at 750 °C is 35% and that of the rGO is ∼85%. The weight loss in l-rGO is attributed to the removal of edge functional groups. The results confirm the formation of rGO.
The results of BET SA analyses and N2 adsorption–desorption isotherms of l-GO and l-rGO are shown in Fig. 1C. The comparatively lower SA of GO (24 m2 g−1) is assigned to the interactions of the functional groups which lead to the stacking of layers. In rGO due to the removal of functional groups, the rGO layers assume a random aggregation of graphene layers by the weak π–π interactions and edge functional groups leading to higher SA. The N2 adsorption–desorption isotherms and the SA values of l-rGO and h-rGO are given in Fig. S1 and Table S1,† respectively. The SAs of l-rGO and h-rGO are 287 and 418 m2 g−1, and the pore sizes are 4.8 and 2.7 nm, respectively. The Raman spectra of GO and rGO show the D (due to defect) and G (graphitic) peaks at 1360 and 1580 cm−1 and ID/IG ratio of rGO shows an increase which agrees with the earlier report.24 Thus, the characterization results confirm the formation of different SA rGOs.
 | ||
| Fig. 1 (A): FTIR spectra; (B): TGA spectra; (C): N2 adsorption–desorption isotherms and (D): Raman spectra of (a): l-GO and (b): l-rGO. | ||
3.1 Adsorption of 1-naphthol by the rGOs
It is observed that the adsorption of 1-naphthol by l-rGO reaches equilibrium quicker than that of h-rGO (Fig. S2†). The h-rGO reaches the equilibrium in ∼50 h while l-rGO reaches much earlier. Kinetic parameters are given in Table S2† and shows that the adsorption of l-rGO fits with pseudo first order while that of the h-rGO fits with pseudo second order kinetics (ESI†). This difference is attributed to the difference in their pore sizes (Table S1†). In accordance to an earlier study,9 5 days of time is used in our adsorption experiments. The amount of 1-naphthol adsorption at equilibrium is calculated using the following equation.
 | (1) |
The adsorption isotherms provide the details of interaction between adsorbate and adsorbent at equilibrium. The linear form of Langmuir adsorption isotherm25 can be expressed as:
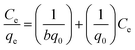 | (2) |
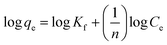 | (3) |
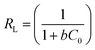 | (4) |
| Adsorbent | Parameters | l-rGO at 303 K | h-rGO at 303 K |
|---|---|---|---|
| Langmuir adsorption model | q0 (mmol g−1) | 3.574 | 5.552 |
| b (L mmol−1) | 3.432 | 16.46 | |
| R2 | 0.9746 | 0.9663 | |
| Freundlich adsorption model | (1/n) | 0.338 | 0.061 |
| Kf (mmol1−n g−1 Ln) | 0.49 | 3.66 | |
| R2 | 0.91484 | 0.601 |
The maximum adsorption capacity (qmax) obtained for the adsorption of 1-naphthol by l-rGO and h-rGO at 303 K, are 3.57 and 5.56 mmol g−1 (515.46 and 800.7 mg g−1), respectively. The values are significantly higher than the qmax of water dispersible sulphonated graphene material (2.3–2.5 mmol g−1) with a similar SA (387 m2 g−1),9 iron oxide modified rGO (rGO-iron oxide) (1.686 mmol g−1),10 carbon nanotube (0.377 mmol g−1)30 and many other adsorbents in the literature. For comparison, the qmax values of various adsorbents reported in the literature and our values are compiled in Table 2. The values clearly indicate that rGO has the highest adsorption efficiency for 1-naphthol compared to that of the other adsorbents. The comparatively lower value of rGO-iron oxide to that of the rGO may be due to the presence of iron oxide on graphene sheets which might limit the SA available for 1-naphthol adsorption by π–π interaction. It must be highlighted that the adsorption capacity exhibited by rGO is higher than its water dispersible sulphonated graphene counterpart. However, the highest value reported for the adsorption of 1-naphthol is 6.4 mmol g−1 at 333 K by the water dispersible sulphonated graphene.21 Also rGO-iron oxide exhibits an increase from 1.686 mmol g−1 at 303 K to a 4.03 mmol g−1 at 323 K. Hence we conducted adsorption experiments at higher temperature.
3.2 Adsorption at higher temperature
Previous reports of 1-naphthol by graphene materials demonstrate that the adsorption is endothermic in nature. We conducted the adsorption experiments at higher temperature (323 K) using h-rGO. The h-rGO was chosen for higher temperature studies because of its higher qmax value compared to that of the l-rGO and in addition its SA is similar to that of the sulphonated graphene. The adsorption isotherms and the parameters obtained at higher temperature experiments are given in Fig. 3 and Table 3, respectively. In accordance with room temperature results, the Langmuir adsorption isotherm fits better than the Freundlich isotherm. The qmax value at 323 K is a remarkable 8.6 mmol g−1 and is the highest value reported for the adsorption of 1-naphthol. The result clearly indicates that rGO is an excellent nanomaterial for the adsorption and removal of 1-naphthol from aqueous solutions. In an attempt to find the rationale for the remarkably higher value of rGO we further conducted experiments and analyses using the adsorption mixture.| Adsorbent | Parameters | h-rGO at 303 K | h-rGO at 323 K |
|---|---|---|---|
| Langmuir adsorption model | q0 (mmol g−1) | 5.55 | 8.61 |
| b (L mmol−1) | 16.46 | 689 | |
| R2 | 0.9663 | 0.9993 | |
| Freundlich adsorption model | (1/n) | 0.061 | 0.160 |
| Kf (mmol1−n g−1 Ln) | 3.66 | 4.15 | |
| R2 | 0.601 | 0.8922 |
3.3 Mechanism of adsorption of 1-naphthol by rGO and the structure of 1-naphthol adsorbed rGO (rGO–naphthol)
It is well known that rGO, due to its hydrophobic nature, is not dispersible in water. We observed that the initially aggregated rGO in the adsorption mixture during adsorption forms a stable dispersion. The digital photo images of the dispersions of h-rGO at 323 K and l-rGO at 303 K are given in Fig. 4A and Fig. S3,† respectively. The images evidently show that 1-naphthol disperses rGO in water and the dispersion of rGO in water increases with the concentration of 1-naphthol. The digital photo images of the adsorption mixture of h-rGO and 1 mmol L−1 of 1-naphthol at 303 and 323 K given in Fig. 4B demonstrate that the dispersion of rGO increases with increasing temperature. The exfoliation of rGO can be initiated by the sliding mechanism of 1-naphthol into the graphene layers due to π–π interactions. The exfoliated graphene layers are well stabilized by the π–π interaction between rGO and 1-naphthol and the interaction of –OH groups of 1-naphthol with water molecules. The dispersion of rGO in water as few layered graphene nanosheets will result in an increase in the SA of rGO, which in turn increases the adsorption capacity of rGO. Dispersion of graphene sheets in aqueous solutions by pyrene derivatives32–34 is been reported. Very recently Kim et al.35 reported the exfoliation of graphite in various organic solvents by ultrasonicating in presence of naphthalene and achieved graphene concentration of 0.15 mg mL−1 in NMP solvent using this procedure. The report mentions that the naphthalene adsorbed over graphene layers acts as a molecular wedge and exfoliates graphite. To the best of our knowledge, this is a first report of a naphthalene moiety dispersing and stabilizing graphene nanosheets in aqueous solution. The sulphonated graphene material dispersed as few layered nanosheets exhibits comparatively lower qmax value. Therefore, we assume that rGO is dispersed as few layered graphene nanosheets comprising of bi, tri-layers etc. in water. The comparatively lower qmax of water dispersible sulphonated graphene may be due to the presence of bulky sulphonated groups which may hinder the adsorption of 1-naphthol.In the attempt to prove our assumptions, we further characterized the dispersions and dry rGO–naphthol samples using XRD, TGA and AFM techniques. The XRD patterns (Fig. 4C) of h-rGO–naphthol show a sharp peak at 20.1° and the broad characteristic peak of rGO at 26.5° while that of the rGO shows only the broad peak around 26.5°. The broad peak centered on 26.5° is due to the typical (002) random ordering of graphene sheets with an interlayer spacing of 0.36 nm. The peak at 20.1° in the rGO–naphthol is sharp and corresponds to an interlayer spacing of ∼0.44 nm. This increased spacing is attributed to the 1-naphthol adsorbed rGO layers. The presence of two peaks (20.1° and 26.5°) in the XRD spectrum indicates that the rGO–naphthol has the typical (002) random ordering of graphene sheets and the higher spacing due to the 1-naphthol adsorbed graphene sheets. The thermograms of l-rGO, 1-naphthol and l-rGO–naphthol with different 1-naphthol content are provided in Fig. S3.† The thermograms confirm the presence of 1-naphthol in the rGO–naphthol and suggest that the 1-naphthol molecules are trapped in between the rGO layers due to which the sublimation of 1-naphthol becomes unfavorable.
Height profile of the AFM images (Fig. 4D) reveal graphene nanosheets of thickness between 1–8 nm, (additional AFM image with height profiles is given in Fig. S4†). The images evidently support the formation of graphene nanosheets in water by 1-naphthol. Our results show that 1-naphthol can disperse and stabilize rGO as graphene nanosheets in water, which in turn enhances the adsorption efficiency. The finding also suggests that similar aromatic compounds with appropriate groups can disperse rGO to graphene nanosheets in water and the adsorption efficiency of rGO for such compounds may possibly be high and depends on the number of benzene rings, its structure and other factors. A schematic of the mechanism of dispersion of rGO by 1-naphthol is depicted in Fig. 5 and illustrates the exfoliation of graphene into few layered nanosheets in water due to the adsorption of 1-naphthol.
 | ||
| Fig. 5 Illustration of the mechanism of dispersion of rGO to few layered graphene nanosheets by 1-naphthol in water. | ||
3.4 Adsorption at different pH values
Investigation of adsorption capacity of adsorbents at different pH is important because of the presence of different ions in the wastewater which make it acidic or alkaline in nature. These conditions will greatly influence the nature of interactions between adsorbate and adsorbent and hence affects the efficiency between the adsorbent and adsorbate. Effect of pH on the adsorption of 1-naphthol on l-rGO is shown in Fig. S5.† At lower and higher pH values, the adsorption capacity increases dramatically compared to that of the neutral condition. We assume the higher adsorption at lower pH values might be due to: (i) lower solubility of 1-naphthol in water at lower pH values (Table S3† shows that the solubility decreases slightly with decreasing pH), and due to (ii) the protonation of –OH groups of 1-naphthol which make the benzene ring an electron-depleted Lewis acid and induce strong interaction with the electron rich rGO surfaces acting as an electron-rich Lewis base, thus increasing the adsorption.9 Similar to that of the acidic conditions, dramatic increase in the 1-naphthol adsorption is observed for alkaline conditions. It is reported that it might be due to the ionisation of the phenolic –OH group in 1-naphthol and thereby induced side reactions.9 We believe that the strong adsorptive interaction between hydroxyl-substituted aromatics and rGO is mainly due to the electron-donating effect of the hydroxyl group due to the lone pair, which caused a strong electron-donor–acceptor (EDA) interaction between the adsorbates and the π-electron-depleted regions on the graphene surfaces of the rGO. Another type of EDA interaction is the π–π EDA interaction between the π-electron-rich aromatic ring(s) of the adsorbates and the π-electron-depleted regions on the rGO due to the defects. It is possible to have in the rGO, both electron-rich and depleted sites, caused primarily by the surface defects.364. Conclusions
In conclusion, rGO exhibits the highest adsorption efficiency for 1-naphthol compared to that of any adsorbents reported in the literature. At 303 K, qmax values of l-rGO and h-rGO are 3.57 and 5.55 mmol g−1, respectively and at 323 K, the qmax of h-rGO for 1-naphthol is 8.6 mmol g−1. This is the highest qmax value reported for 1-naphthol so far. The adsorption is endothermic in nature. Acidic and alkaline pH increases the adsorption. The adsorption isotherms of the rGOs fit well with the Langmuir isotherm model. The higher and remarkable efficiency exhibited by rGO is assigned to the ability of 1-naphthol, to disperse rGO as few layered graphene nanosheets in water. AFM images confirm the exfoliation of rGO to nanosheets of 1–8 nm thickness by 1-naphthol. Our results point to the fact that modification of rGO to water dispersible graphene does not necessarily increase the adsorption efficiency, especially for compounds such as 1-naphthol and similar aromatic compounds, which are capable of dispersing rGO in aqueous solution. The result suggests that rGO can be dispersed in aqueous solution by naphthalene moieties having appropriate side groups. Our findings give a better understanding about the mechanism of adsorption of 1-naphthol by rGO. The results suggest that (unmodified) rGO is a super adsorbent for 1-naphthol and will probably be a very good adsorbent material for other aromatic compounds with structural similarities with 1-naphthol which can equally disperse rGO in water.Acknowledgements
The authors thank the Director, IIST for the research facilities and Dean, R & D and Head, Department of Chemistry for their support and encouragements.Notes and references
- L. Zhang, P. Li, Z. Gong and X. Li, Photocatalytic degradation of polycyclic aromatic hydrocarbons on soil surfaces using TiO2 under UV light, J. Hazard. Mater., 2008, 158, 478–484 CrossRef CAS PubMed.
- E. Taylor, B. Cook and M. Tarr, Dissolved organic matter inhibition of sonochemical degradation of aqueous polycyclic aromatic hydrocarbons, Ultrason. Sonochem., 1999, 6, 175–183 CrossRef CAS.
- A. Raj, M. Sander, V. Janardhanan and M. Kraft, A study on the coagulation of polycyclic aromatic hydrocarbon clusters to determine their collision efficiency, Combust. Flame, 2010, 157, 523–534 CrossRef CAS PubMed.
- T. Barkay, S. Navon-Venezia, E. Ron and E. Rosenberg, Enhancement of Solubilization and Biodegradation of Polyaromatic Hydrocarbons by the Bioemulsifier Alasan, Appl. Environ. Microbiol., 1999, 65, 2697–2702 CAS.
- M. Rafatullah, O. Sulaiman, R. Hashim and A. Ahmad, Adsorption of methylene blue on low-cost adsorbents: a review, J. Hazard. Mater., 2010, 177, 70–80 CrossRef CAS PubMed.
- C. Valderrama, X. Gamisans, X. de las Heras, A. Farran and J. Cortina, Sorption kinetics of polycyclic aromatic hydrocarbons removal using granular activated carbon: intraparticle diffusion coefficients, J. Hazard. Mater., 2008, 157, 386–396 CrossRef CAS PubMed.
- R. Walters and R. Luthy, Equilibrium adsorption of polycyclic aromatic hydrocarbons from water onto activated carbon, Environ. Sci. Technol., 1984, 18, 395–403 CrossRef CAS PubMed.
- K. Yang, L. Zhu and B. Xing, Adsorption of Polycyclic Aromatic Hydrocarbons by Carbon Nanomaterials, Environ. Sci. Technol., 2006, 40, 1855–1861 CrossRef CAS.
- G. Zhao, L. Jiang, Y. He, J. Li, H. Dong, X. Wang and W. Hu, Sulfonated Graphene for Persistent Aromatic Pollutant Management, Adv. Mater., 2011, 23, 3959–3963 CrossRef CAS PubMed.
- X. Yang, C. Chen, J. Li, G. Zhao, X. Ren and X. Wang, Graphene oxide–iron oxide and reduced graphene oxide–iron oxide hybrid materials for the removal of organic and inorganic pollutants, RSC Adv., 2012, 2, 8821–8826 RSC.
- Y. Li, P. Zhang, Q. Du, X. Peng, T. Liu, Z. Wang, Y. Xia, W. Zhang, K. Wang, H. Zhu and D. Wu, Adsorption of fluoride from aqueous solution by graphene, J. Colloid Interface Sci., 2011, 363, 348–354 CrossRef CAS PubMed.
- X. Deng, L. Lu, W. Li and F. Luo, The adsorption properties of Pb(II) and Cd(II) on functionalized graphene prepared by electrolysis method, J. Hazard. Mater., 2010, 183, 923–930 CrossRef CAS PubMed.
- J. Zhu, S. Wei, H. Gu, S. Rapole, Q. Wang, Z. Luo, N. Haldolaarachchige, D. Young and Z. Guo, One-pot synthesis of magnetic graphene nanocomposites decorated with core@double-shell nanoparticles for fast chromium removal, Environ. Sci. Technol., 2012, 46, 977–985 CrossRef CAS PubMed.
- T. Liu, Y. Li, Q. Du, J. Sun, Y. Jiao, G. Yang, Z. Wang, Y. Xia, W. Zhang, K. Wang, H. Zhu and D. Wu, Adsorption of methylene blue from aqueous solution by graphene, Colloids Surf., B, 2012, 90, 197–203 CrossRef CAS PubMed.
- G. Ramesha, A. Kumara, H. Muralidhara and S. Sampath, Graphene and graphene oxide as effective adsorbents toward anionic and cationic dyes, J. Colloid Interface Sci., 2011, 361, 270–277 CrossRef CAS PubMed.
- K. Parvez, Z. S. Wu, R. Li, X. Liu, R. Graf, X. Feng and K. Mullen, Exfoliation of Graphite into Graphene in Aqueous Solutions of Inorganic Salts, J. Am. Chem. Soc., 2014, 136, 6083–6091 CrossRef CAS PubMed.
- M. A. Gross, M. J. Sales, M. A. G. Soler, M. A. Pereira-da-silva, M. F. P. Da silva and L. G. Paterno, Reduced graphene oxide multilayers for gas and liquid phases chemical sensing, RSC Adv., 2014, 4, 17917–17924 RSC.
- Z. Pei, L. Li, L. Sun, S. Zhan, X. Q. Shan, S. Yang and B. Wen, Adsorption characteristics of 1,2,4-trichlorobenzene, 2,4,6-trichlorophenol, 2-naphthol and naphthalene on graphene and graphene oxide, Carbon, 2013, 51, 156–163 CrossRef CAS PubMed.
- S. Wang, H. Sun, H. M. Ang and M. O. Tade, Adsorptive remediation of environmental pollutants using novel graphene based nanomaterials, Chem. Eng. J., 2013, 226, 336–347 CrossRef CAS PubMed.
- P. Montes-Navajas, N. G. Asenjo, R. Santamaria, R. Menedez, A. Corma and H. Garcia, Surface Area Measurement of Graphene Oxide in Aqueous Solutions, Langmuir, 2013, 29, 13443–13448 CrossRef CAS PubMed.
- G. Zhao, J. Li and X. Wang, Kinetic and thermodynamic study of 1-naphthol adsorption from aqueous solution to sulfonated graphene nanosheets, Chem. Eng. J., 2011, 173, 185–190 CrossRef CAS PubMed.
- G. Venugopal, K. Krishnamoorthy, R. Mohan and S. J. Kim, An investigation of the electrical transport properties of graphene-oxide thin films, Mater. Chem. Phys., 2012, 132, 29–33 CrossRef CAS PubMed.
- Q. Zhen, W. Hing Ip, X. Lin, N. Yousefi, K. K. Yeung, Z. Li and J. K. Kim, Transparent Conductive Films Consisting of Ultralarge Graphene Sheets Produced by Langmuir Blodgett Assembly, ACS Nano, 2011, 5, 6039–6051 CrossRef PubMed.
- S. Stankovich, D. Dikin, R. Piner, K. Kohlhaas, A. Kleinhammes, Y. Jia, Y. Wu, S. T. Nguyen and R. S. Ruoff, Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide, Carbon, 2007, 45, 1558–1565 CrossRef CAS PubMed.
- I. Langmuir, The adsorption of gases on plane surfaces of glass, mica and platinum, J. Am. Chem. Soc., 1918, 40, 1361–1403 CrossRef CAS.
- J. Xu, L. Wang and Y. Zhu, Decontamination of Bisphenol A from Aqueous Solution by Graphene Adsorption, Langmuir, 2012, 28, 8418–8425 CrossRef CAS PubMed.
- H. M. F. Freundlich, Uber die adsorption in losungen, Z. Phys. Chem., 1906, 57, 385–470 CAS.
- T. Weber and R. Chakkravorti, Pore and solid diffusion models for fixed bed adsorbers, AIChE J., 1974, 20, 228–238 CrossRef CAS.
- B. H. Hameed, A. T. M. Din and A. L. Ahmad, Adsorption of methylene blue onto bamboo-based activated carbon: kinetics and equilibrium studies, J. Hazard. Mater., 2007, 141, 819–825 CrossRef CAS PubMed.
- G. D. Sheng, D. D. Shao, X. M. Ren, X. Q. Wang, J. X. Li, Y. X. Chen and X. K. Wang, Kinetics and thermodynamics of adsorption of ionizable aromatic compounds from aqueous solutions by as-prepared and oxidized multi-walled carbon nanotubes, J. Hazard. Mater., 2010, 178, 505–516 CrossRef CAS PubMed.
- J. Wei, R. Zhu, J. Zhu, F. Ge, P. Yuan, H. He and C. Ming, Simultaneous sorption of crystal violet and 2-naphthol to bentonite with different CECs, J. Hazard. Mater., 2009, 166, 195–199 CrossRef CAS PubMed.
- A. Schlierf, H. Yang, E. Gebremedhn, E. Treossi, L. Ortolani, L. Chen, A. Minoia, V. Morandi, P. Samorì, C. Casiraghi, D. Beljonne and V. Palermo, Nanoscale insight into the exfoliation mechanism of graphene with organic dyes: effect of charge, dipole and molecular structure, Nanoscale, 2013, 5, 4205–4216 RSC.
- D. Parviz, S. Das, T. Ahmed, F. Irin, S. Bhattacharia and M. J. Green, Dispersions of Non-Covalently Functionalized Graphene with Minimal Stabilizer, ACS Nano, 2012, 6, 8857–8867 CrossRef CAS PubMed.
- J. W. T. Seo, A. A. Green, A. L. Antaris and M. C. Hersam, High-concentration aqueous dispersions of graphene using nonionic, biocompatible block copolymers, J. Phys. Chem. Lett., 2011, 2, 1004–1008 CrossRef CAS.
- E. J. Kim, J. Xu, D. K. Dang, V. T. Tran, X. Liu, J. S. Chung, S. H. Hur, W. M. Choi and P. A. Kohl, Liquid-phase exfoliation of graphene in organic solvents with addition of naphthalene, J. Colloid Interface Sci., 2014, 418, 37–42 CrossRef PubMed.
- W. Chen, L. Duan, L. Wang and D. Zhu, Adsorption of hydroxyl- and amino-substituted aromatics to carbon nanotubes, Environ. Sci. Technol., 2008, 42, 6862–6868 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Details N2 adsorption isotherm, effect of contact time on adsorption, effect of pH, digital photo images & AFM images, kinetic fittings and parameters and solubility at lower pH values are available. See DOI: 10.1039/c4ra05702c |
| This journal is © The Royal Society of Chemistry 2014 |



