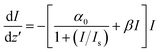DOI:
10.1039/C4RA05649C
(Paper)
RSC Adv., 2014,
4, 39541-39546
Sol–gel prepared Cu2O microspheres: linear and nonlinear optical properties
Received
12th June 2014
, Accepted 13th August 2014
First published on 13th August 2014
Abstract
We report the simple sol–gel based synthesis of size-tunable monodispersed crystalline Cu2O microspheres (CMS) and the measurement of their linear and nonlinear optical properties. Optical absorption spectra show a broad plasmonic absorption band extending from 350 to 1100 nm in general agreement with calculations based on Mie theory, despite the relatively large micron size of the particles. Z-scan measurements (532 nm, 5 ns pulses) reveal that Cu2O microspheres are efficient optical limiters, comparable in strength to nanocarbons and metal/semiconductor nanoparticles. The effective two-photon absorption coefficients are numerically calculated from measured data. These can be tuned systematically by varying the particle size.
1. Introduction
CuO based materials are best known for their high Tc superconducting properties which arise from their electronic structure, where the open d orbital (3d9) of Cu gets bonded with the 2p orbital of O. This particular Cu–O linkage results in d–d Coulomb interactions and charge transfer phenomena. CuO sustains plasmons1 as well as phonons.2 Reports indicate that the high Tc superconductivity of CuO probably originates from its strong electron–phonon coupling,3 and plasmons are known to enhance this superconductivity.4,5 The basic structure of CuO results from oxygen–oxygen and copper–oxygen hybridizations. CuO has the tenorite structure for its unit cell, with the space group of C2h (ref. 5) (monoclinic unit cell). Cluster model calculations predict the existence of two basic structures, viz. (CuO4)5− and (CuO4).5,6 In addition, other one-dimensional and two-dimensional structures also are possible.
One of the major pursuits of modern photonic technologies is the control of phase, amplitude, and direction of light in all-optical and opto-electronic devices.7–11 Photonic materials are promising candidates for ultrafast processes and quantum optical applications such as data encryption and transport. Nonlinear optical materials with high nonlinear susceptibilities are essential for these purposes.12,13 In addition to being good superconductors, CuO based materials are found to perform as nonlinear optical media as well. For instance, the CuO based chain compounds Sr2CuO3, Ca2CuO3 and Nd2CuO4 show an enhanced two-photon absorption efficiency which is attributed to phonon-mediated charge transfer.14–16 Similarly, excimer laser deposited CuO and Cu2O films exhibit a large third order nonlinear susceptibility (χ(3)) under ultrafast laser irradiation, which is comparable to those observed in semiconductor films of ZnO and GaN.17 CuO nanocrystalline films have been found to be good optical limiters with a limiting threshold of 0.3 GW cm−2 and a damage threshold of 1.65 GW cm−2, for 800 nm excitation using mode-locked 50 fs laser pulses at a repetition rate of 80 MHz.18 Ando et al. have reported that the large third order optical nonlinearities found in 3d transition-metal oxide films including CuO can be fast-acting (order of picoseconds) due to band-filling effects.19 Moreover, Cu, CuO and CuO based chain compounds are known to exhibit plasmon oscillations at frequencies which are dependent on the size, shape and environment.20,21 These materials show relatively high optical nonlinearities and a fast decay of electron excitation.22,23
Nano/micro materials exhibit size and shape dependent physical and chemical properties which make them suitable for device applications. For example, nonlinear light transmission of several nanosized metals24 and semiconductors25 have been investigated in the past. D. N. Rao et al.26 have prepared Cu2O micro and nanostructures using the co-precipitation method and investigated morphological changes in these structures, and found that their nonlinear absorption is a combination of saturable absorption and reverse saturable absorption.26 Kuo et al.27 has prepared Cu2O nanocubes using a seed-mediated synthesis approach in aqueous solution, with average size ranging from 40 to 420 nm.28 In the larger nanocubes they observed multiple absorption tails or bands in the visible and near-infrared (NIR) regions. Misra et al.28 investigations provide insights about tuning the SPR, absorption and scattering with varying size and symmetry of Cu2O nano-structures. In view of these studies which illustrate the importance of Cu2O, in the present work we have synthesized Cu2O microspheres (CMS) which are relatively larger in size through a simple sol–gel route, and have studied their linear and nonlinear optical properties. The advantage of the sol–gel technique is that unlike the other methods it is very simple, and can produce nano and micro materials at room temperature without complicated chemical procedures. We found that the linear absorption of the Cu2O microspheres can be modeled with reasonable success using the Mie theory, and that their nonlinear absorption makes them good optical limiters, with an efficiency comparable to that of C60 and carbon nanotubes.
2. Experimental
Cu2O was prepared by adding 200 ml of 0.1 M glucose dissolved in double distilled water to 200 ml of 0.0035 M copper acetate dissolved in double distilled water. When glucose solution is added to copper acetate solution the color changes from dark green to blue. 0.5 M of NaOH dissolved in 100 ml of distilled water is added drop wise to this solution, by which the color changes to dark violet. When kept under stirring for half an hour, precipitates of orange color start depositing. The precipitates are collected after 12 hours, washed with distilled water and ethanol, and left to dry at room temperature. A similar procedure is followed for 0.2 M and 0.1 M concentrations of NaOH as well. The samples are code named CuO5, CuO2 and CuO1 respectively, based on NaOH concentrations of 0.5 M, 0.2 M and 0.1 M used for synthesis. X-Ray powder diffraction (XRD) (Rigaku X-ray spectrometer) measurements have been done using Cu Kα radiation (λ = 1.54 Å) to identify the crystal structure (Fig. 1). The morphology and size of the prepared particles were studied using scanning electron microscopy (Fig. 2). The particles are found to be spherical in shape. Optical absorption spectra (UV-vis) were measured on a Perkin-Elmer spectrophotometer (Fig. 3).
 |
| | Fig. 1 X-ray diffraction patterns of the prepared Cu2O nanopowders. Peaks are indexed against cubic phase Cu2O using JCPDS files #782076 and #351091 data (for diffraction peak marked with *). XRD patterns are moved vertically for clarity. | |
 |
| | Fig. 2 SEM images of CuO2 (first row) and CuO5 (second row) showing spherical morphology. Particle diameter is approximately 1 μm for CuO2 and 2 μm for CuO5 samples. Particle size increases with increasing NaOH concentration used for synthesis. | |
 |
| | Fig. 3 Measured optical absorption spectra of the prepared samples. Spectra are found to change with particle size. | |
3. Results and discussion
Optical absorption studies
Optical absorption spectra of the prepared samples is shown in Fig. 3. From this studies it is obvious that particle size determines the optical absorption spectrum. Mie theory with a quasi-static approximation can be used to calculate the extinction spectrum of metal nanoparticles like Ag, Cu, and Au, and this procedure can be extended to CuO and Cu2O also. According to Mie theory, the scattering (Csca), absorption (Cabs) and extinction (Cext = Csca + Cabs) cross sections (which are sensitive to particle size and surrounding medium) are given by the relations29–31| |
 | (1) |
| |
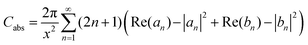 | (2) |
where the Mie coefficients an and bn are functions of the Riccati–Bessel functions (ψ, ζ), refractive index of the material (nmat), refractive index of the surrounding medium (nmed), size of the particle (r), size parameter 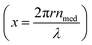 and incident light wavelength (λ). For smaller spherical particles only the dipole term needs to be retained in the above equations, which leads to an extinction cross section given by (quasi-static dipole approximation)
and incident light wavelength (λ). For smaller spherical particles only the dipole term needs to be retained in the above equations, which leads to an extinction cross section given by (quasi-static dipole approximation)| |
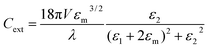 | (3) |
where V is the volume-filling fraction; εm is dielectric constant of the medium; ε1 and ε2 are the real and imaginary part of the complex dielectric constant of the particle. The extinction efficiency or efficiency factor (Qext) is related to the extinction cross section (Cext) by the relation 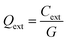 , where G is the cross-sectional area of the particle. Similarly, the absorption and scattering efficiencies are also related to their respective cross sections. Simulations to calculate these efficiencies were performed with the tool “Mieplot v4.3” built by an algorithm based on Mie theory for spheres.32 Frequency dependent refractive index values of bulk Cu2O available from literature were used in these simulations33 with water as the surrounding medium. Absorption, scattering, and extinction efficiencies of Cu2O microspheres of different sizes, calculated from the Mie theory were shown in Fig. 4. A plot of experimentally measured optical absorption spectra of CuO5 and CuO2 samples along with Qabs values calculated for Cu2O particles of various sizes from the Mie theory, is shown in Fig. 5. It is obvious from the figure that the measured optical absorption spectra follow a trend which is similar to that of calculated Qabs. This simulation and the measured optical absorption spectra strongly support the existence of Mie related light scattering in the Cu2O microparticles. The disparities observed between calculated and experimental spectra probably arise from the polydispersity in size and departure from spherical shape of the Cu2O particles, and the use of optical constants of bulk Cu2O in the calculation.
, where G is the cross-sectional area of the particle. Similarly, the absorption and scattering efficiencies are also related to their respective cross sections. Simulations to calculate these efficiencies were performed with the tool “Mieplot v4.3” built by an algorithm based on Mie theory for spheres.32 Frequency dependent refractive index values of bulk Cu2O available from literature were used in these simulations33 with water as the surrounding medium. Absorption, scattering, and extinction efficiencies of Cu2O microspheres of different sizes, calculated from the Mie theory were shown in Fig. 4. A plot of experimentally measured optical absorption spectra of CuO5 and CuO2 samples along with Qabs values calculated for Cu2O particles of various sizes from the Mie theory, is shown in Fig. 5. It is obvious from the figure that the measured optical absorption spectra follow a trend which is similar to that of calculated Qabs. This simulation and the measured optical absorption spectra strongly support the existence of Mie related light scattering in the Cu2O microparticles. The disparities observed between calculated and experimental spectra probably arise from the polydispersity in size and departure from spherical shape of the Cu2O particles, and the use of optical constants of bulk Cu2O in the calculation.
 |
| | Fig. 4 Absorption, scattering, and extinction efficiencies of Cu2O microspheres of different sizes, calculated from the Mie theory. | |
 |
| | Fig. 5 Measured optical absorption spectra of CuO5 samples plotted against calculated Qabs for Cu2O particles of various sizes. Measured spectra follow a trend similar to the calculated spectra. | |
Non-linear optical studies
The open-aperture Z-scan technique34 was employed to investigate the optical nonlinearity of the samples at the excitation wavelength of 532 nm. In the Z-scan technique the sample is scanned along the propagation direction (the z axis) near the focal region of a focused laser beam, while the energy transmitted is recorded as a function of sample position. The normalized transmittance Tnorm., which is the measured transmission normalized to the linear transmission of the sample, is then plotted against the sample position z, to get the Z-scan curve. Considering a Gaussian laser pulse of energy Ein focused using a converging lens, the position-dependent optical fluence F (z) is given by35| |
F(z) = 4(ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)1/2Ein/π3/2ω(z)2 2)1/2Ein/π3/2ω(z)2
| (4) |
where ω(z) is the beam radius, given by| | |
ω(z) = ω(0)[1 + (z/z0)2]1/2
| (5) |
with ω(0) being the focal spot radius, and z0 = πω(0)2/λ is the Rayleigh range (λ is the laser wavelength). The fluence will be a maximum at the focal point (z = 0). In our experiment the samples were prepared by dispersing 3 mg each of the micropowders in doubly distilled water. The concentration is adjusted such that the linear transmission of the dispersion at 532 nm is 80% when taken in a 1 mm path length cuvette. Measurements were done at 532 nm using 5 nanosecond laser pulses obtained from a frequency-doubled Nd:YAG laser (Continuum, MiniLite). The cuvette containing the sample was mounted on a motorized linear translation stage. Laser pulses were fired at a low repetition rate of 0.2 Hz to avoid cumulative sample heating that might otherwise interfere with the measurements.36 The energy reaching the sample per pulse was approximately 50 micro joules. The transmitted energy was measured using a pyroelectric energy probe (laser probe, RjP-735). A fraction of the incident pulse energy was measured by an identical probe for monitoring pulse-to-pulse energy variations. Z-scan curves were then drawn by plotting the normalized transmission Tnorm. against the sample position z. Tnorm. was plotted against the F(z) values as well. These plots can be seen in Fig. 6.
 |
| | Fig. 6 Z-scan curves measured for the prepared samples (insets) along with the corresponding optical limiting curves. Circles are experimental data while solid curves are numerical fits to eqn (6). | |
Nonlinear optical transmission can have contributions from phenomena such as two- or three-photon absorption, excited state absorption (ESA), saturable absorption and nonlinear scattering. We tried fitting the experimental data to different nonlinear transmission equations in order to understand the exact nature of the nonlinearity. The best fit was obtained for a two-photon type absorption concomitant with saturable absorption. Here the intensity dependent nonlinear absorption coefficient α(I) is given by
| |
 | (6) |
where
α0 is the linear absorption coefficient at the wavelength of excitation,
I is the input laser intensity,
Is is the saturation intensity, and
β is the effective two-photon absorption coefficient. The corresponding differential equation describing the nonlinear optical loss, given by
35| |
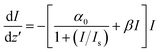 | (7) |
is then numerically solved to obtain
Is and
β from the transmission data. z' is the propagation distance within the sample. The effective two-photon absorption coefficients (
β) and saturation intensities calculated for the present samples are shown in
Table 1. It is seen that
β increases and
Is decreases as the particle size is increased from CuO1 to CuO5.
Table 1 Effective two-photon absorption coefficients and saturation intensities calculated from the Z-scans
| Sample code |
Copper nitrate concentration |
NaOH concentration |
Nature of absorption |
Effective two-photon absorption coefficient, β (m W−1) |
Saturation intensity, Is (W m−2) |
| CuO1 |
0.0035 M |
0.1 M |
Single band |
4.0 × 10−11 |
3.0 × 1012 |
| CuO2 |
0.0035 M |
0.2 M |
Multiple bands |
4.8 × 10−11 |
2.4 × 1012 |
| CuO5 |
0.0035 M |
0.5 M |
Multiple bands |
6.4 × 10−11 |
1.6 × 1012 |
From Table 2 it can be seen that these values are mostly of the same order of magnitude as measured earlier in nanocarbons, quantum dots etc. under similar excitation conditions.
Table 2 Comparison of the effective two-photon absorption coefficients obtained for the Cu2O microspheres with that of nanocarbons and metal/semiconductor nanoparticles (excitation wavelength of 532 nm, pulse width of 5–10 ns)
| Sample |
βeff (m W−1) |
| CuO1[Present work] |
4.0 × 10−11 |
| CuO2[Present work] |
4.8 × 10−11 |
| CuO5[Present work] |
6.4 × 10−11 |
| SWNT (ref. 37) |
2.5 × 10−11 |
| C60 (ref. 37) |
3.3 × 10−11 |
| Bi nanorods38 |
5.3 × 10−11 |
| Graphene oxide39 |
2.8 × 10−11 |
| Graphene sheets39 |
7.1 × 10−11 |
| MWCNT (ref. 40) |
1.7 × 10−10 |
| CdTe quantum dots41 |
5.5 × 10−12 |
| SiO2 (ref. 42) |
1.7 × 10−11 |
| TiO2 (ref. 42) |
2.8 × 10−10 |
In general, the nonlinear optical response of mesoscopic materials depend on a number of parameters such as the size, shape, surface morphology, distribution and host lattice. It may be noted that Cu2O microspheres exhibit a broad absorption spectrum, extending over a large wavelength region. Due to the plasmonic nature of absorption a local field enhancement given by
| |
 | (8) |
is possible, where
El is the local field and
E0 is the applied field,
43 f is the local field factor. Even though this enhancement in the field amplitude will be advantageous to the nonlinear absorption process, the plasmon band being broad, substantial field enhancement is unlikely. The actual physical process behind the observed optical limiting is therefore reverse saturable absorption involving real excited states (which is a fluence dependent process), with a smaller contribution from genuine two-photon absorption (which is an intensity dependent process). The closely lying energy states causing the broad absorption are more conducive for excited state absorption. Being a transmission measurement, the Z-scan cannot distinguish between genuine two-photon absorption and excited state absorption. Therefore, the
β values calculated in the present case are considered as “effective” two-photon absorption coefficients.
44
4. Conclusion
In summary, we have prepared Cu2O microspheres through a simple sol–gel method, and studied their linear and nonlinear optical properties. X-ray diffraction analysis shows that the particles are monoclinic in phase. Theoretical calculations using Mie theory yield a broad absorption band, and measurements give a broad plasmonic absorption band in general agreement with theory. Nonlinear optical measurements reveal an optical limiting behavior arising from an effective two-photon absorption process, with an efficiency similar to that of nanocarbons and metal/semiconductor nanoparticles. The nonlinearity coefficient can be tuned systematically by varying the particle size.
References
- R. Côté and A. Griffin, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 48, 10404 CrossRef.
- P. Calvani, M. Capizzi, S. Lupi, P. Maselli, A. Paolone and P. Roy, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 53, 2756 CrossRef CAS.
- V. Z. Kresin and H. Morawitz, J. Supercond., 1988, 1, 89 CrossRef.
- R. Côté and A. Griffin, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 48, 10404 CrossRef.
- D. Van der Marel and A. Tsvetkov, Czech J. Phys., 1996, 46, 3165 CrossRef CAS.
- J. Ghijsen, L. H. Tjeng, J. Van Elp, H. Eskes, J. Westerink, G. A. Sawatzky and M. T. Czyzyk, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 38, 11322 CrossRef CAS.
- Y. H. Lee, Y. Yan, L. Polavarapu and Q.-H. Xua, Appl. Phys. Lett., 2009, 95, 023105 CrossRef PubMed.
- D. P. Liu, G.-D. Li, Y. Su and J.-S. Chen, Angew. Chem., Int. Ed., 2006, 45, 7370 CrossRef CAS PubMed.
- V. N. Pustovit and T. V. Shahbazyan, J. Chem. Phys., 2012, 136, 204701 CrossRef CAS PubMed.
- T. Omata, Y. Tani, S. Kobayashi, K. Takahashi, A. Miyanaga, Y. Maeda and S. O.-Y. Matsuo, Appl. Phys. Lett., 2012, 100, 061104 CrossRef PubMed.
- B. Anand, R. Podila, K. Lingam, S. Krishnan, S. S. S. Sai, R. Philip and A. M. Rao, Nano Lett., 2013, 13, 5771 CrossRef CAS PubMed.
- J. Titus and M. Thakur, Appl. Phys. Lett., 2007, 90, 121111 CrossRef PubMed.
- F. Z. Henari and A. A. Dakhel, J. Appl. Phys., 2008, 104, 033110 CrossRef PubMed.
- A. Maeda, M. Ono, H. Kishida, T. Manako, A. Sawa, M. Kawasaki, Y. Tokura and H. Okamoto, Phys. Rev. B: Condens. Matter Mater. Phys., 2004, 70, 125117 CrossRef.
- M. Ashida, T. Ogasawara, Y. Tokura, S. Uchida, S. Mazumdar and M. K. Gonokami, Appl. Phys. Lett., 2001, 78, 2831 CrossRef CAS PubMed.
- M. Ono, K. Miura, A. Maeda, H. Matsuzaki, H. Kishida, Y. Taguchi, Y. Tokura, M. Yamashita and H. Okamoto, Phys. Rev. B: Condens. Matter Mater. Phys., 2004, 70, 085101 CrossRef.
- G. Yang, A. Chen, M. Fu, H. Long and P. Lu, Appl. Phys. A: Mater. Sci. Process., 2011, 104, 171 CrossRef CAS.
- A. Chen, G. Yang, H. Long, P. Lu, W. Zhang and H. Wang, Mat. Lett., 2013, 91, 319 CrossRef CAS PubMed.
- M. Ando, K. Kadono, M. Haruta, T. Sakaguchi and M. Miya, Nature, 1995, 374, 625 CrossRef CAS.
- I. Bozovic, Phys. Rev. B: Condens. Matter Mater. Phys., 1990, 42, 1969 CrossRef CAS.
- A. C. Sharma, Solid State Commun., 1989, 70, 1171 CrossRef CAS.
- B. Karthikeyan, J. Thomas and R. Philip, Chem. Phys. Lett., 2005, 414, 346 CrossRef CAS PubMed.
- S. Link and M. A. El-Sayed, J. Phys. Chem. B, 1999, 103, 8410 CrossRef CAS.
- L. Polavarapu, Q.-H. Xu, M. S. Dhoni and W. Ji, Appl. Phys. Lett., 2008, 92, 263110 CrossRef PubMed.
- X. N. Peng, J. B. Li, Y. Z. Yang, H. M. Gong, F. Nan, L. Zhou, X. F. Yu, Z. H. Hao and Q. Wang, J. Appl. Phys., 2012, 112, 074305 CrossRef PubMed.
- H. Sekhar and D. Narayana Rao, J. Nanopart. Res., 2012, 14, 976 CrossRef.
- By: C.-H. Kuo, C.-H. Chen and M. H. Huang, Adv. Funct. Mater., 2007, 17, 3773 CrossRef CAS PubMed.
- B. Sinha, T. Goswami, S. Paul and A. Misra, RSC Adv., 2014, 4, 5092 RSC.
- C. F. Bohren and D. R. Huffman, Absorption and scattering of light by small particles, Wiley, New York, 1983 Search PubMed.
- J. S. Sekhon and S. S. Verma, Plasmonics, 2011, 6, 311 CrossRef CAS.
- J. S. Sekhon and S. S. Verma, Renewable Energy and the Environment, OSA Technical Digest (CD), Optical Society of America, Paper JWE22, 2011 Search PubMed.
- MiePlot v4.1, http://www.philiplaven.com/mieplot.html.
- E. D. Palik, Handbook of optical constants of solids, Academic Press, 1991 Search PubMed.
- S. Bahae, A. Ali Said, T.-H. Wei, J. David Hagan and E. W. Van Stryland, IEEE J. Quantum Electron., 1990, 26, 760 CrossRef.
- R. L. Sutherland, Handbook of Nonlinear Optics, Marcel Dekker, New York, 1996 Search PubMed.
- P. B. Chapple, J. Staromlynska, J. A. Hermann, T. J. Mckay and R. G. Mcduff, J. Nonlinear Opt. Phys. Mater., 1997, 6, 251 CrossRef.
- J. Wang and W. J. Blau, J. Phys. Chem. C, 2008, 112, 2298 CAS.
- S. Sivaramakrishnan, V. S. Muthukumar, S. Sivasankara Sai, K. Venkataramaniah, J. Reppert, A. M. Rao, M. Anija, R. Philip and N. Kuthirummal, Appl. Phys. Lett., 2007, 91, 093104 CrossRef PubMed.
- B. Zhao, B. Cao, W. Zhou, D. Li and W. Zhao, J. Phys. Chem. C, 2010, 114, 12517 CAS.
- T. N. Narayanan, C. S. Suchand Sandeep, M. M. Shaijumon, P. M. Ajayan, R. Philip and M. R. Anantharaman, Nanotechnology, 2009, 20, 285702 CrossRef CAS PubMed.
- J. Khatei, C. S. Suchand Sandeep, R. Philip and K. S. R. Koteswara Rao, Appl. Phys. Lett., 2012, 100, 081901 CrossRef PubMed.
- L. Irimpan, B. Krishnan, V. P. N. Nampoori and P. Radhakrishnan, Opt. Mater., 2008, 31, 361 CrossRef CAS PubMed.
- D. D. Smith, Y. Yoon, R. W. Boyd, J. K. Campbell, L. A. Baker, R. M. Crooks and M. George, J. Appl. Phys., 1999, 86, 6200 CrossRef CAS PubMed.
- M. Rumi and J. W. Perry, Adv. Opt. Photonics, 2010, 2, 451 CrossRef CAS.
|
| This journal is © The Royal Society of Chemistry 2014 |
Click here to see how this site uses Cookies. View our privacy policy here. 

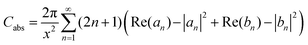
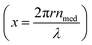 and incident light wavelength (λ). For smaller spherical particles only the dipole term needs to be retained in the above equations, which leads to an extinction cross section given by (quasi-static dipole approximation)
and incident light wavelength (λ). For smaller spherical particles only the dipole term needs to be retained in the above equations, which leads to an extinction cross section given by (quasi-static dipole approximation)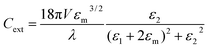
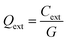 , where G is the cross-sectional area of the particle. Similarly, the absorption and scattering efficiencies are also related to their respective cross sections. Simulations to calculate these efficiencies were performed with the tool “Mieplot v4.3” built by an algorithm based on Mie theory for spheres.32 Frequency dependent refractive index values of bulk Cu2O available from literature were used in these simulations33 with water as the surrounding medium. Absorption, scattering, and extinction efficiencies of Cu2O microspheres of different sizes, calculated from the Mie theory were shown in Fig. 4. A plot of experimentally measured optical absorption spectra of CuO5 and CuO2 samples along with Qabs values calculated for Cu2O particles of various sizes from the Mie theory, is shown in Fig. 5. It is obvious from the figure that the measured optical absorption spectra follow a trend which is similar to that of calculated Qabs. This simulation and the measured optical absorption spectra strongly support the existence of Mie related light scattering in the Cu2O microparticles. The disparities observed between calculated and experimental spectra probably arise from the polydispersity in size and departure from spherical shape of the Cu2O particles, and the use of optical constants of bulk Cu2O in the calculation.
, where G is the cross-sectional area of the particle. Similarly, the absorption and scattering efficiencies are also related to their respective cross sections. Simulations to calculate these efficiencies were performed with the tool “Mieplot v4.3” built by an algorithm based on Mie theory for spheres.32 Frequency dependent refractive index values of bulk Cu2O available from literature were used in these simulations33 with water as the surrounding medium. Absorption, scattering, and extinction efficiencies of Cu2O microspheres of different sizes, calculated from the Mie theory were shown in Fig. 4. A plot of experimentally measured optical absorption spectra of CuO5 and CuO2 samples along with Qabs values calculated for Cu2O particles of various sizes from the Mie theory, is shown in Fig. 5. It is obvious from the figure that the measured optical absorption spectra follow a trend which is similar to that of calculated Qabs. This simulation and the measured optical absorption spectra strongly support the existence of Mie related light scattering in the Cu2O microparticles. The disparities observed between calculated and experimental spectra probably arise from the polydispersity in size and departure from spherical shape of the Cu2O particles, and the use of optical constants of bulk Cu2O in the calculation.


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)1/2Ein/π3/2ω(z)2
2)1/2Ein/π3/2ω(z)2