Direct comparison on the structural and optical properties of metal-catalytic and self-catalytic assisted gallium nitride (GaN) nanowires by chemical vapor deposition†
V. Purushothamana,
P. Sundara Venkatesha,
R. Navamathavanb and
K. Jeganathan*a
aCentre for Nanoscience and Nanotechnology, School of Physics, Bharathidasan University, Tiruchirappalli, 620024, India. E-mail: kjeganathan@yahoo.com
bSemiconductor Materials Process Laboratory, School of Advanced Materials Engineering, Engineering College, Research Center for Advanced Materials Development (RCAMD), Chonbuk National University, Deokjin-dong 664-14, Chonju 561-756, Korea
First published on 29th August 2014
Abstract
The structural and optical properties of GaN nanowires (NWs) grown by catalytic and self-catalytic-assisted vapor liquid solid approach using chemical vapor deposition (CVD) are reported. Liquid gallium droplets were used as the nucleation centre for the growth of self-catalyst assisted NWs, whereas pre-deposited Ni and Au thin films were employed as the seed layer for the catalyst-assisted growth on Si (111) substrates. Electron microscopy analyses revealed that the growth rates and densities of the structural defects of the NWs strongly vary with the nature of the catalyst. The high resolution electron microscopy and selected area electron diffraction studies exhibit a high crystalline quality of Ni-catalyst-assisted GaN NWs, whereas self-catalytic NWs contain defects such as stacking faults and cubic inclusion. Temperature-dependent photoluminescence on the ensembles of NWs illustrates the absence of the characteristic yellow luminescence band of GaN for the Ni-assisted vapor–liquid–solid approach, implying the high optical quality of GaN NWs by CVD. The results show that the quality of the self-catalytic GaN NWs grown by CVD is yet to be improved for device applications.
Introduction
III-nitrides are direct band-gap compound semiconductors with a high breakdown field that plays a vital role in current optoelectronics and high temperature, high power electronics.1–3 Semiconductor NWs hold significant promise as building blocks for novel devices in various areas such as electronics, optoelectronics, photovoltaics, piezotronics, and thermoelectrics.4–7 The most celebrated approach for the formation of NWs is the vapor–liquid–solid (VLS) process,8 in which the external metal catalyst acts as the liquid forming seeds to promote the anisotropic NWs. Briefly, a thin metal layer containing a substrate is preheated to form a eutectic alloy and the precursors are allowed into the growth environment. The precursors dissociate at the growth temperature and incorporate into the catalyst droplet; subsequently, the droplet is supersaturated with elements and precipitate in the liquid–solid interface (substrate) to elongate as NWs.8 A decade ago, a major modification in VLS growth was formulated in which the catalyst remained solid throughout the growth; this mechanism is termed the vapor–solid–solid (VSS) process.9 Despite important advances and impressive results on NW-based applications, the reliance on gold and other metals in the fabrication process is a significant barrier to advances in the integration of NWs. In particular, gold is a forbidden element in complementary metal-oxide semiconductor (CMOS) processing, because it forms mid-gap electronic states in Si that degrade device performance.10 Any fabrication process that relies on gold will be very difficult to integrate with CMOS beyond the laboratory level.11 In addition, the widespread use of a catalyst raises the question of whether some of the seed material is incorporated into the lattice of NWs and adversely affects their properties, thus preventing high-performance applications.12 Hence, there are increasing efforts to grow NWs without any metal catalyst. In this self-catalytic approach,13–16 the excess of one of the materials forms liquid droplets that may act as the catalyst for the growth of NWs.17–20 Depending on the growth parameters, the self-catalytic growth may be VLS or VSS.21 Significant efforts have been dedicated to the self-catalytic growth of III–V NWs such as GaAs and InP.22–25 Nevertheless, only a few reports are available in the literature for the self-catalytic growth of III-nitride NWs by CVD.26,27Despite significant effort for detecting any contamination by the catalyst material, understanding the differences in the quality of the NWs grown by catalytic and self-catalytic approaches is less explored. Chèze et al.28 compared the structural and optical properties of catalyst-assisted and catalyst-free GaN NWs grown by molecular beam epitaxy (MBE), in which they showed that the catalyst-free GaN NWs possessed high structural and optical quality, whereas the Ni-seeded GaN NWs contained a high density of stacking faults and comparatively poor luminescence properties. From their results, it is evident that the catalyst-assisted GaN NWs grown at a lower temperature (750 °C) tend to have higher stacking-fault-related defects. In addition, they also suggested that Ni incorporation in GaN NWs could be the possible reason for the poor optical quality of NWs. On the other hand, high-temperature growth of catalyst-assisted GaN NWs by other techniques, such as MOCVD, results in very high structural quality as compared with the catalyst-assisted MBE NWs. CVD is an inexpensive and simple method to grow NWs by both the catalytic and self-catalytic approach as compared with MBE and MOCVD. Hence, we attempted to compare the structural and optical properties of GaN NWs grown by both the pathways under identical growth conditions to deduce the effect of the catalyst. Ni-assisted GaN NWs demonstrate excellent optical properties compared with the Au-catalyst and self-catalytic assisted GaN NWs.
Experimental section
GaN NWs were fabricated by the direct reaction of metal Ga (ALFA ACEAR – 99.999%) and ammonia (NH3-99.999%) in a horizontal CVD reactor. For both the catalyst and self-catalyst assisted growth types, Si(111) was used as the substrate. Prior to loading, the substrates were cleaned by a standard RCA procedure and 5 nm thin metal layers (Au or Ni) were deposited by e-beam evaporation with a base vacuum of 1 × 10−6 mbar for catalyst-assisted growth. Metal Ga (0.1 g) was put in a quartz crucible and placed in the alumina boat. Si substrates were kept downstream to the source Ga at a distance of 10 mm. Then, the alumina boat was inserted into the growth reactor. The reactor was evacuated to 10−3 mbar and filled with nitrogen (N2-99.999%). N2 gas was allowed to flow for 2 h before the growth to overcome residual oxygen. The temperature of the furnace was increased to the growth temperature of 900 °C at a rate of at 15 °C min−1. N2 and NH3 flow of 500 and 200 SCCM (standard cubic centimeter), respectively, were used as carrier and reactant gases. A detailed growth process for both the catalytic and self-catalytic approach has already been published.26,27,29 In short, for self-catalytic GaN NWs, initially N2 was flown for 2 min to transport Ga vapor to deposit the Ga droplet layer on the Si substrate, which in turn acts as the catalyst for the subsequent growth of NWs. Then, NH3 was introduced into the reactor without interrupting N2 flow to avoid the surface nitridation of Ga. The continuous supply of precursors led to the nucleation and subsequent growth of GaN NWs. In the case of catalyst-assisted GaN NWs (Ni and Au), both the precursors were introduced simultaneously because the initial Ga deposition step is not essential because the metal catalyst droplets activate the growth of NWs. The growth duration was fixed at 1 h for all the samples. At completion, the reactor was turned off to naturally cool to room temperature. All the growth parameters including flow rate, source to substrate distance, temperature and growth duration were kept constant for all the experiments.The morphology and compositional variation of GaN NWs were studied using field emission scanning electron microscopy (FESEM) (Carl Zeiss-Σigma) equipped with energy dispersive X-ray analysis (EDX) (Oxford instruments). EDX was recorded at 10 keV with a point resolution of around 10 nm. High resolution transmission electron microscope (HRTEM) (JEM 2010, JEOL, Japan) was used at an operating voltage of 200 kV to examine the crystallinity of a single GaN nanowire. The GaN nanowires were carefully removed from the Si substrate by the sonication process and then collected in the transmission electron microscopy (TEM) grid. Low temperature photoluminescence (PL) spectra for the ensemble of NWs were recorded using the He–Cd laser of 325 nm (30 mW) focused through a objective lens with a spot size of ∼10 μm, and the resulting luminescence signal was collected through a monochromator (Horiba Jobin Yvon – 0.55 M) and a charge-coupled device. The samples were placed in a closed cycle He cryostat with the temperature varied from 10 to 300 K.
Results and discussion
Fig. 1(a–f) shows the electron microscopic images of self-catalytic assisted GaN NWs grown by chemical vapor deposition. Fig. 1(a) shows the self-catalytic GaN NWs, with an average diameter and length of 150 ± 20 nm and 1.8 μm, respectively. The average growth rate of NWs was calculated to be 30 nm min−1 (for more electron microscopy images please refer to ESI S1†). In the case of self-catalytic GaN NWs, the Ga metal droplet is normally not observed on the apex because the growth proceeding at higher temperature results in the consumption or desorption of Ga.30 Fig. 1(b) shows the TEM image recorded on the top part of a single GaN NW, which reveals the absence of any such catalytic particle. Fig. 1(c) shows the HRTEM image recorded on the top of the single GaN NW, which contains defects such as stacking faults.31 Fig. 1(d) shows the HRTEM image recorded on the body of the single NW, which shows some extended defects.In addition to the wurtzite structure, the presence of cubic inclusions and stacking faults is also evidenced by the corresponding selected area electron diffraction pattern (SAED) pattern (Fig. 1(e)) taken from (0001) zone axis. HRTEM spectra was recorded for several NWs, and all the NWs show very similar defects for self-catalytic GaN NWs. The stacking faults and other defects in self-catalytic GaN NWs were attributed to the change in supersaturation during the growth, despite the low growth rate.32 HRTEM results reveal that the self-catalytic GaN nanowires contain defects and are polycrystalline in nature, which in turn affects the optical properties of the nanowires. However, deeper investigations of the HRTEM analysis are required to map the defect formation and propagation, which is beyond the scope of this manuscript. EDX recorded on the NWs shows slightly N-rich conditions, with the atomic ratio of 48.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 51.6% for Ga and N. Here, excess N can be attributed to the N-rich growth conditions that favor the growth of anisotropic NWs under the self-catalytic approach.
51.6% for Ga and N. Here, excess N can be attributed to the N-rich growth conditions that favor the growth of anisotropic NWs under the self-catalytic approach.
Fig. 2(a) shows the tilt-view FESEM image of the high yield Ni-catalyst assisted GaN NWs. The average diameter and length of the NWs are 120 ± 10 nm and 10 μm, respectively. Fig. 2(b) shows the magnified view of a single NW with the Ni-catalyst on the top of the NW. The calculated average axial growth rate is 165 nm min−1. Fig. 2(c) shows the low-magnification HRTEM image recorded on a single NW, which reveals that the NW is long and straight with a flat surface. To probe the crystalline nature of the catalytic alloy and GaN NWs, HRTEM analysis was carried out. Fig. 2(c) presents a detailed HRTEM examination on the interface between the NW and the catalyst droplet of a single GaN NW, which indicates that the interface is abrupt and smooth. Fig. 2(d), the high-magnification HRTEM image of a single NW shows it to be free of domain boundaries and cubic inclusion having a single-crystalline nature. The crystalline nature of GaN NWs was further confirmed by SAED. Fig. 2(e) shows the SAED pattern, recorded perpendicular to the NW long axis.
Detailed analysis of diffraction pattern shows that the NW assumed the wurtzite structure and grew along the c-direction. HRTEM and SAED observations for many NWs and at different locations of each NW showed similar diffraction pattern and lattice fringes (d = 0.52 nm) oriented along the (0001) crystallographic direction. A detailed analysis of the chemical composition of the catalytic particle and the novel VLS approach, in which the incorporation of Ga and N adatoms follows catalytic and triple phase boundary can be found elsewhere.19 Unlike in the MBE-grown GaN NWs by Chèze et al.,33 here the Ni assisted GaN NWs have high structural quality. In general, MBE-grown NWs are often reported34 to be superior to CVD-grown. However, the poor quality of MBE-grown Ni-catalyst assisted GaN NWs can be attributed to the low-temperature growth at 730 °C, whereas CVD GaN NWs were grown at 950 °C. Further, the EDX recorded on the ensembles of GaN NWs shows an almost equimolar Ga to N ratio, in addition to the presence of other elements such as Si, O and Ni, as shown in Fig. 2(f). The presence of O from the elemental analysis has been attributed to the strong native oxide of the Si substrate or oxygen onto the ensembles of NWs during the atmospheric transfer of samples for SEM imaging. Elemental analysis has also been carried out during the TEM analysis on a single GaN NW that reveals the absence of O and Si impurities at the detection limit.
Fig. 3(a) shows the FESEM image of Au-catalyst assisted GaN NWs. The average diameter and length of the NWs were 150 ± 20 nm and 6 μm, respectively. The calculated average axial growth rate was 100 nm min−1. No specific relationship can be established between the diameter and length (d vs. L) of the GaN nanowires for both the catalyst- and self-catalyst assisted growth of NWs. Fig. 3(b–d) show the HRTEM images recorded on the body of the NW, which reveals that the NWs are free from defects, such as cubic inclusion, and have a single crystalline nature. The NW diameter is apparently very homogeneous from the top to bottom without any tapering effect; however, branching and overlapping of NWs can be seen. Fig. 3(d) shows the high-magnification HR-TEM lattice image of the NW taken normal to the growth axis. This image shows the (0001) growth direction of the NW. The lattice images of the NW are very clear without any vacancy defects and stacking faults. From the HR-TEM data, the interplanar spacing was observed to be ∼0.51 nm. The crystalline nature of GaN NWs was further confirmed by SAED. Fig. 3(e) shows the SAED pattern recorded perpendicular to the NW long axis. Elemental analyses carried out on the ensembles of NWs show an equimolar ratio of Ga to N in addition to the presence of Si, O and Au. The structural quality of Au-assisted GaN NWs and Ni-assisted GaN NWs are very similar, which is in contradiction to the earlier report by Zhou et al.,35 in which they reported that the Au-assisted MOCVD grown GaN NWs contain basal stacking faults, whereas the Ni assisted GaN NWs were free from such defects. Nevertheless, we expect that such quality difference may be due to the change in growth temperature along with the substrate effects because Zhou et al.35 have used an r-plane sapphire substrate. It is worth noting that we have employed Si(111)-substrates for the fabrication of GaN NWs regardless of using different catalysts. Seed particles, defects and compositional variations can strongly affect the NWs.36,37
Fig. 4 shows the XRD pattern recorded for catalyst and self-catalyst assisted GaN NWs. All the diffraction peaks are well indexed to the wurtzite GaN (JCPDS Card no: 898624). The peak positions and their corresponding full width half maximum (FWHM) of the dominant (100), (002) and (101) planes are shown in the ESI S1.† Ni-catalyst GaN NWs shows sharp diffraction peaks, and evidently their FWHM values for (100), (002) and (101) planes are narrow as compared with Au-catalyst and self-catalyst GaN NWs. The abovementioned result is clearly evident of the highly crystalline nature of the Ni-catalyst GaN NWs. Despite the poor crystalline nature of self-catalyst GaN NWs, the peak positions of diffraction planes match well with the bulk GaN, evidencing lattice relaxation. Because the XRD pattern was recorded for the ensembles of GaN NWs, the diffraction from various hexagonal planes was also observed. The cubic and oxide phases were absent in all GaN NWs irrespective of the growth process. However, the presence of a weak SiN peak can be attributed to the nitridation of the Si surface at the growth temperature during the initial stage of nucleation. To further investigate whether the optical properties of the GaN nanowires differ in growth using Au and Ni seed particles or by self-catalysis, PL studies were performed on the as-grown ensemble nanowires. Low temperature photoluminescence recorded at 10 K for self-catalyst and catalyst-assisted GaN NWs are shown in Fig. 5. At first sight, two important observations can be made from the luminescence studies. The first is that self-catalytic GaN NWs contain defect-related emissions, as compared with the catalyst-assisted GaN NWs. The second observation is that Ni-assisted GaN NWs (Fig. 5) show dominant emission around the GaN band edge without yellow luminescence that is frequently observed in GaN thin films and nanowires. All the data for the comparison of luminescence properties of catalyst and self-catalyst NWs are given in Table 1.
 | ||
| Fig. 5 (a–c) Photoluminescence spectra of GaN NWs recorded at 10 K (a) self-catalyst GaN NWs (b) Ni-assisted GaN NWs and (c) Au-assisted GaN NWs. | ||
| Luminescence properties of NWs | D0X | NBE | Defect levels | ||||
|---|---|---|---|---|---|---|---|
| E (eV) | FWHM (meV) | E (eV) | FWHM (meV) | DAP at 300 K (eV) | BL at 300 K (eV) | YL at 300 K (eV) | |
| Self catalytic NWs | 3.468 | 127.4 | 3.407 | 174 | 3.20 | 3.07 | 2.22 |
| Ni-GaN NWs | 3.485 | 63.9 | 3.441 | 146 | 3.25 | 3.00 | Nil |
| Au-GaN NWs | 3.484 | 74.7 | 3.440 | 178 | 3.29 | 2.96 | 2.01 |
Lorentzian peak fitted spectra of Ni, Au and self-catalytic GaN NWs recorded at 10 K show free donor-bound exciton (D0X) transitions at 3.485, 3.484 and 3.468 eV, respectively, with the full width at half maximum (FWHM) of 63.9, 74.7 and 122 meV, respectively. As temperature increases, D0X peak positions of Ni, Au and self-catalytic GaN NWs shift to lower energy, and the peaks are centered at 3.441, 3.440 and 3.407 eV with the FWHM of 146, 148 and 178 meV, respectively, for 300 K. The FWHM of the D0X peaks are higher as compared with the MOCVD-38 and MBE-39 grown NWs, likely because the PL spectra were obtained on an ensemble of GaN nanowires. PL spectra were recorded for a number of samples grown by both catalytic and self-catalytic assisted GaN NWs. None of the Ni-catalyst assisted GaN NWs exhibited the yellow band (YL@10 K), which indicative of very low defect densities. However, the YL band is clearly visible for Au–GaN NWs but is dominantly present for self-catalytic GaN NWs.
As shown in Fig. 5, in a typical 10 K PL spectra of Ni, Au and self-catalyzed GaN nanowires, all NWs exhibited a strong ultraviolet emission in addition to the deep level emission at 3.25–29 eV pertaining to shallow donor acceptor recombination (DAP).40 The DAP band of all the NW samples shifts to higher energies (from 3.31 eV to 3.34 eV) with an increase in temperature due to the thermal escape of electrons from long-lived distant pairs contributing at the low-energy side of a PL band due in turn to weaker Coulomb interaction.41 The shallow DAP has been attributed42 to defects such as SiGa and ON because both the defects are quite possible in our growth approach. Si has high solubility in both Ni and Au, and hence NWs can have Si interstitials and/or SiGa-type defects. Further, it has been widely reported that the catalytic-grown GaN NWs on Si substrates can be unintentionally doped with Si.43 In addition to Si, oxygen could also be incorporated into the growing NW from the native oxide layer of Si. Another other source for oxygen could be from the atmospheric transfer of samples for analysis. The EDX recorded for all the ensemble of NWs shows small traces of oxygen with less than 1 at.% in all the cases; however, elemental analysis carried out through TEM analysis does not show any traces of O in a single GaN NW up to the detection limit.
At 10 K, GaN NWs samples exhibit blue band emission at 2.9–3.1 eV because of the transitions from the conduction band or a shallow donor to a relatively deep acceptor.44 In particular, self-catalytic GaN NWs show a dominant blue luminescence (BL) band at low temperature as compared with the Ni- and Au-assisted GaN NWs. The intensity of the BL band decreases on increasing temperature from 10 to 300 K; the quenching of the BL band at high temperature can be attributed to the escape of holes from the acceptor to the valence band, and/or the bound holes may non-radiatively recombine with free electrons in the conduction band. However, in the case of Ni- and Au-catalyst assisted GaN NWs, the BL band increases with increasing temperature and shows the tendency opposite to that of self-catalytic NWs. In general, the BL band is related to a shallow acceptor (SiN or CN); the origin of this BL band is still a topic of considerable debate in literature.38
In most of the unintentionally and intentionally doped n-typeGaN samples grown by the various techniques available, the room-temperature PL spectrum contains a near band-edge emission at about 3.42 eV and the YL band centering at 2.20–2.25 eV (Fig. 6(a), 7(a) and 8(a)). Here, at 10 K, the YL band was present in both the Au- and self-catalytic NWs but the peak was too weak to be observed for Ni-GaN NWs. In the case of catalytic NWs, the YL band is suppressed at low temperature and at 10 K; Ni-assisted GaN NWs do not possess any visible YL band. For self-catalytic GaN NWs, the YL band broadens (450 meV) and increases with temperature. The increase in the YL band with temperature is typical for defects with strong electron–phonon coupling. There are several contradictory results in the literature concerning the origin of the YL band and the effects of doping on its intensity. First principle calculations predict45 that several VGa-related defects may be responsible for YL in undoped GaN, which confirms that the Ni-GaN NWs are free from defects such as VGa, VN and complex defects related that gives rise to emission at 2.2 eV.28,29 This can be related to the high solubility of Ga in Ni-catalyst particles, which continuously supply Ga-adatoms to the growth interface for the crystallization of GaN, thereby reducing Ga-vacancy related defects.
 | ||
| Fig. 6 (a) Temperature-dependent photoluminescence spectra recorded for self-catalytic GaN NWs for various temperature ranges; (b) its corresponding Varshni fitting. | ||
 | ||
| Fig. 7 (a) Temperature-dependent photoluminescence spectra recorded for Ni-catalyst-assisted GaN NWs for various temperature ranges; and (b) its corresponding Varshni fitting. | ||
 | ||
| Fig. 8 (a) Temperature-dependent photoluminescence recorded spectrum for Au-catalyst assisted GaN NWs for various temperature ranges; and (b) its corresponding Varshni fitting. | ||
Further, the effective ionic radius of Ni (60 pm) is smaller than that of Ga (62 pm); thus, the Ga vacancies can be easily replaced by the Ni ions. In general, the Ga vacancies (negatively charged) and the Ni ions (3+) are in the opposite charge states. Hence, the suppression of the YL band in Ni-GaN NWs can be attributed to the compensation of deep-level defects such as Ga vacancies.46 However, deep investigations are warranted to confirm the presence of Ni in the lattice of GaN. Using the Varshni formula47 and band-tail model, the temperature-dependent emission energy can be fitted by.
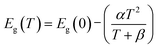 | (1) |
Summary
We have compared the structural and luminescence properties of GaN nanowires grown with different self-catalyst and catalytic approaches. The self-catalytic GaN NWs contain a high density of structural defects such as stacking faults and cubic inclusions, whereas Ni- and Au-catalyst GaN NWs possess high structural quality along with the absence of such defects. The PL spectra of the self-catalytic GaN NWs exhibit defect-related BL and YL bands in addition to the broad band-edge emission. For Ni- and Au-assisted GaN NWs, the PL emission is significantly stronger with donor bound exciton at ∼3.485 eV. Further, Ni-GaN NWs do not have any visible YL band, demonstrating superior structural and optical quality as compared with Au- and self-catalytic GaN NWs. The poor optical quality of CVD-grown self-catalytic GaN NWs has been attributed to the presence of defects and structural disorder, as evidenced by electron microscopy.Acknowledgements
One of the authors, K.J., thanks the Department of Science and Technology (DST), Govt. of India for financial assistance under the Nanomission project no. SR/NM/NS-77/2008. V.P. acknowledges CSIR, Govt. of India for the award of a senior research fellowship (SRF).Notes and references
- Y. Li, J. Xiang, F. Qian, S. Gradecak, Y. Wu, H. Yan, D. A. Blom and C. M. Lieber, Nano Lett., 2006, 6, 1468–1473 CrossRef CAS PubMed.
- F. A. Ponce and D. P. Bour, Nature, 1997, 386, 351–359 CrossRef CAS.
- J. W. Orton and C. Foxon, Rep. Prog. Phys., 1998, 61, 1–75 CrossRef CAS.
- H. M. Kim, Y. H. Cho, H. Lee, S. I. Kim, S. R. Ryu, D. Y. Kim, T. W. Kang and K. S. Chung, Nano Lett., 2004, 4, 1059–1064 CrossRef CAS.
- X. Duan and C. M. Lieber, J. Am. Chem. Soc., 2000, 122, 188–189 CrossRef CAS.
- S. Y. Bae, H. W. Seo, J. Park, H. Yang, J. C. Park and S. Y. Lee, Appl. Phys. Lett., 2002, 81, 126 CrossRef CAS PubMed.
- Z. J. Li, X. L. Chen, H. J. Li, Q. Y. Tu1, Z. Yang, Y. P. Xu and B. Q. Hu, Appl. Phys. A: Mater. Sci. Process., 2001, 72, 629–632 CrossRef CAS.
- R. S. Wagner and W. C. Ellis, Appl. Phys. Lett., 1964, 4, 89 CrossRef CAS PubMed.
- A. I. Persson, M. W. Larsson, S. Stenstrom, B. J. Ohlsson, L. Samuelson and L. R. Wallenberg, Nat. Mater., 2004, 3, 677–681 CrossRef CAS PubMed.
- A. J. Tavendale and S. J. Pearton, J. Phys. C: Solid State Phys., 1983, 16, 1665 CrossRef CAS.
- K. A. Dick and P. Caroff, Nanoscale, 2014, 6, 3006–3021 RSC.
- J. E. Allen, E. R. Hemesath, D. E. Perea, J. L. Lensch-Falk, S. Y. Li, F. Yin, M. H. Gass, P. Wang, A. L. Bleloch and R. E. Palmer, Nat. Nanotechnol., 2008, 3, 168–173 CrossRef CAS PubMed.
- R. Q. Zhang, Y. Lifshitz and S. T. Lee, Adv. Mater., 2006, 15, 635–640 CrossRef.
- J. Noborisaka, J. Motohisa and T. Fukui, Appl. Phys. Lett., 2005, 86, 213102 CrossRef PubMed.
- M. He, I. Minus, P. Zhou, S. N. Mohammed, J. B. Halpern, R. Jacobs, W. L. Sarney, L. S. Riba and R. D. Vispute, Appl. Phys. Lett., 2000, 77, 3731 CrossRef CAS PubMed.
- B. Mandl, J. Stangl, T. Mårtensson, A. Mikkelsen, J. Eriksson, L. S. Karlsson, G. Bauer, L. Samuelson and W. Seifert, Nano Lett., 2006, 6, 1817–1821 CrossRef CAS PubMed.
- P. J. Poole, J. Lefebvre and J. Fraser, Appl. Phys. Lett., 2003, 83, 2055 CrossRef CAS PubMed.
- E. Stach, P. Pauzauskie, T. Kuykendall, J. Goldberger, R. He and P. Yang, Nano Lett., 2003, 3, 865 CrossRef.
- Y. Zhang, H. Jia and D. Yu, J. Phys. D: Appl. Phys., 2004, 37, 413 CrossRef CAS.
- C. J. Novotny and P. K. L. Yu, Appl. Phys. Lett., 2005, 87, 203111 CrossRef PubMed.
- V. Purushothaman, V. Ramakrishnan and K. Jeganathan, RSC Adv., 2012, 2, 4802–4806 RSC.
- C. Colombo, D. Spirkoska, M. Frimmer, G. Abstreiter and A. Fontcuberta i Morral, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 77, 155326 CrossRef.
- A. Fontcuberta i Morral, C. Colombo, G. Abstreiter, J. Arbiol and J. R. Morante, Appl. Phys. Lett., 2008, 92, 063112 CrossRef PubMed.
- F. Jabeen, V. Grillo, S. Rubini and F. Martelli, Nanotechnology, 2008, 19, 275711 CrossRef PubMed.
- J. Noborisaka, J. Motohisa and T. Fukui, Appl. Phys. Lett., 2005, 86, 213102 CrossRef PubMed.
- V. Purushothaman, V. Ramakrishnan and K. Jeganathan, CrystEngComm, 2012, 14, 8390–8395 RSC.
- V. Purushothaman and K. Jeganathan, J. Phys. Chem. C, 2013, 117, 7348–7357 CAS.
- C. Chèze, L. Geelhaar, O. Brandt, W. M. Weber, H. Riechert, S. Münch, R. Rothemund, S. Reitzenstein, A. Forchel, T. Kehagias, P. Komninou, G. P. Dimitrakopulos and T. Karakostas, Nano Res., 2010, 3, 528–536 CrossRef PubMed.
- V. Purushothaman and K. Jeganathan, J. Nanopart. Res., 2013, 15, 1789 CrossRef.
- J. Noborisaka, J. Motohisa and T. Fukui, Appl. Phys. Lett., 2005, 86, 213102 CrossRef PubMed.
- D. Tham, C. Y. Nam and J. E. Fischer, Adv. Funct. Mater., 2006, 16, 1197 CrossRef CAS.
- N. A. Jeon, S. A. Dayeh and L. J. Lauhon, Nano Lett., 2013, 13(8), 3947–3952 CrossRef CAS PubMed.
- C. Chèze, L. Geelhaar, O. Brandt, W. M. Weber, H. Riechert, S. Münch, R. Rothemund, S. Reitzenstein, A. Forchel, T. Kehagias, P. Komninou, G. P. Dimitrakopulos and T. Karakostas, Nano Res., 2010, 3, 528–536 CrossRef PubMed.
- C. Chèze, L. Geelhaar, B. Jenichen and H. Riechert, Appl. Phys. Lett., 2010, 97, 153105 CrossRef PubMed.
- X. Zhou, J. Chesin, S. Crawford and S. Gradecak, Nanotechnology, 2012, 23, 285603 CrossRef PubMed.
- M. M. Brewster, X. A. Zhou, S. K. Lim and S. Gradecak, J. Phys. Chem. Lett., 2011, 2, 586 CrossRef CAS.
- S. K. Lim, S. Crawford, G. Haberfehlner and S. Gradecak, Nano Lett., 2013, 13(2), 331–333 CrossRef CAS PubMed.
- A. A. Talin, G. T. Wang, E. Lai and R. J. Anderson, Appl. Phys. Lett., 2008, 92, 093105 CrossRef PubMed.
- N. Thillosen, K. Sebald, H. Hardtdegen, R. Meijers, R. Calarco, S. Montanari, N. Kaluza, J. Gutowski and H. Lüth, Nano Lett., 2006, 6(4), 704–708 CrossRef CAS PubMed.
- M. C. Reshchikov and H. Morkoc, J. Appl. Phys., 2005, 97, 061301 CrossRef PubMed.
- D. G. Thomas, J. J. Hopfield and W. M. Augustyniak, Phys. Rev., 1965, 140, 202 CrossRef CAS.
- S. M. Lee, M. A. Belkhir, X. Y. Zhu, Y. H. Lee, Y. G. Hwang and T. Frauenheim, Phys. Rev. B: Condens. Matter Mater. Phys., 2000, 61, 16033 CrossRef CAS.
- J. Liu, X. M. Meng, Y. Jiang, C. S. Lee, I. Bello and S. T. Lee, Appl. Phys. Lett., 2003, 83, 4241 CrossRef CAS PubMed.
- A. Y. Polyakov, M. Shin, J. A. Freitas Jr, M. Skowronski, D. W. Greve and R. G. Wilson, J. Appl. Phys., 1996, 80, 6349 CrossRef CAS PubMed.
- C. G. Van de Walle and J. Neugebauer, J. Appl. Phys., 2004, 95, 3851 CrossRef CAS PubMed.
- G. Devaraju, A. P. Pathak, N. Srinivasa Rao, V. Saikiran, F. Enrichi and E. Trave, Nucl. Instrum. Methods Phys. Res., Sect. B, 2011, 269, 1925–1928 CrossRef CAS PubMed.
- V. P. Varshni, Physica, 1967, 149–154 CrossRef.
- M. O. Manasreh, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 53, 16425–16428 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra05388e |
| This journal is © The Royal Society of Chemistry 2014 |




