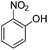
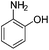
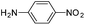
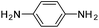
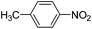
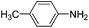
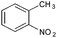
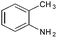
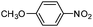
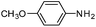
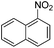
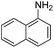
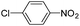
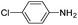
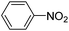
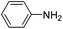
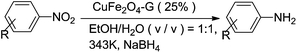Copper ferrite–graphene hybrid: a highly efficient magnetic catalyst for chemoselective reduction of nitroarenes†
Haiyan Zhang,
Shutao Gao,
Ningzhao Shang,
Chun Wang* and
Zhi Wang
College of Science, Agricultural University of Hebei, Baoding 071001, Hebei, China. E-mail: chunwang69@126.com; Fax: +86-312-7528292; Tel: +86-312-7528291
First published on 4th July 2014
Abstract
In this paper, a superparamagnetic copper ferrite–graphene hybrid nanocomposite (CuFe2O4–G) was synthesized by a hydrothermal method and used as an efficient catalyst for the reduction of nitroarenes for the first time. The method has been applied to a broad range of compounds with different properties. The CuFe2O4–G catalyst can be readily recovered and reused for at least five consecutive cycles without significant loss of its catalytic activity.
Introduction
Development of new catalytic transformations with easy separation and recyclability of the catalyst is an essential task in chemical synthesis. Aromatic amines are important starting materials and intermediates for the manufacture of a great variety of chemicals such as dyes, pesticides, herbicides, and agrochemicals.1–3 They are generally synthesized by the chemical reduction of nitroarenes. Iron-compound,4 mixed metal compounds,5 transition metals (Cu, Ni)6 and noble metals (Pt, Pd, Au)7–9 are commonly employed as the catalyst for the reduction reaction. However, some of the methods employed have drawbacks, for example using expensive reagents, prolonged reaction time, using toxic and volatile solvent, unsatisfactory yield, etc. Hence, the development of an inexpensive, facile and effective method for the synthesis of aromatic amines is of high demand.The copper ferrite, CuFe2O4 with a spinel structure, is one of the most important ferrites in powder form, has been widely applied in sensors, electronics and catalysts owing to its advantages of environmentally compatible, moisture insensitive, high dispersion, high reactivity and easy separation at an external magnet in recent years. Amini et al.10 investigated the low temperature CO oxidation over mesoporous CuFe2O4 nanopowders synthesized by a novel sol–gel method. Parella et al.11 explored the catalytic application of CuFe2O4 nanoparticles for the Friedel–Crafts acylation. Feng et al.6 investigated the catalytic activity of CuFe2O4 nanoparticles for the reduction of 4-nitrophenol to 4-aminophenol with excess amount of NaBH4.
Graphene(G), discovered in 2004 by Geim and co-workers,12 has enjoyed tremendous research interest in a wide range of fields over the past decades. Graphene-related nanomaterials exhibit a great application potential in catalysis13 thanks to their large specific surface area, easy modification, remarkable electrical conductivity, excellent adsorptivity, ultrathin thickness, superior structural flexibility, and high chemical and thermal stability. To date, various kinds of catalysts, including TiO2,14 SnO2,15 ZnO,16 Pt,17,18 Pt–Ru,19 Pt–Pd,20 Pd21 and so on, have been supported on graphene-based templates for catalyzing different chemical transformations, energy conversion and photocatalytic reactions. Recently, superparamagnetic graphene–Fe3O4 nanocomposite (G–Fe3O4) was fabricated and used as an efficient catalyst for the reduction of nitroarenes with hydrazine hydrate as reductant, which is a toxic reagent.22 Copper ferrite–graphene hybrid was developed as an effective photocatalyst for the degradation of methylene blue under visible light irradiation, which also showed excellent electrochemical behaviors for use as the anode in lithium-ion batteries.23 However, to our best knowledge, there has been no report yet about the use of copper ferrite–graphene (CuFe2O4–G) magnetic nanocomposite as catalyst for organic synthesis.
In the present work, the catalytic activity of magnetic CuFe2O4–G nanocomposite for the chemoselective reduction of nitroarenes was investigated using sodium borohydride as a hydrogen donor. The results demonstrate that the combination of CuFe2O4 with graphene results in a dramatic enhancement of the catalytic activity of CuFe2O4.
Results and discussions
Material characterizations
X-ray powder diffraction analysis was used to identify the crystal structure of the CuFe2O4–G (0.25) and CuFe2O4. As shown in Fig. 1, except some Cu impurity peaks at 43.0° and 50.6°, all peaks were indexed to be CuFe2O4 (JCPDS 77-0010), in detail, the peaks at 30.5°, 35.2°, 57.0°, 62.8° and 74.1° are attributed to (220), (311), (511), (440) and (533) crystal planes of CuFe2O4. While no typical diffraction peak of reduced graphene oxide was observed. It is speculated that the GO in the CuFe2O4–G(0.25) heteroarchitecture was fully exfoliated due to the crystal growth of CuFe2O4 nanoparticles between the interlayer of GO sheets, which result in the low diffraction intensity of GO. The formation of copper was identified based on the reported data (JCPDS 85-1326) at 43.0° and 50.6°. The reason of existence of metallic copper was that some of Cu2+ was reduced to copper metal in the formation process of CuFe2O4 due to the strong reducing capability of ethylene glycol.6Fig. 2 shows the SEM image of CuFe2O4–G (0.25). It can be seen that CuFe2O4–G(0.25) were composed of quasi-sphere particle with particle sizes of about 100 nm.
The FTIR spectra were recorded to further testify the hybrid material. As shown in Fig. 3, the spectrum of GO is in good agreement with previous work.24 The broad, intense band at 3250 cm−1 is assigned to the stretching of O–H. The peak at 1728 cm−1 corresponds to the stretching of the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and –COOH groups on GO sheets. The peak at 1616 cm−1 (aromatic C
O and –COOH groups on GO sheets. The peak at 1616 cm−1 (aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) can be ascribed to the skeletal vibrations of unoxidized graphene domains. The C–O bond is associated with the band at 1047 cm−1. From the FTIR spectrum of GO and CuFe2O4–G (0.25) samples, it is can be clearly seen that the graphene oxide exhibits an obvious characteristic absorption peak at about 1728 cm−1 corresponds to the stretching of the –C
C) can be ascribed to the skeletal vibrations of unoxidized graphene domains. The C–O bond is associated with the band at 1047 cm−1. From the FTIR spectrum of GO and CuFe2O4–G (0.25) samples, it is can be clearly seen that the graphene oxide exhibits an obvious characteristic absorption peak at about 1728 cm−1 corresponds to the stretching of the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and –COOH groups. However, it is can not be seen from the FTIR spectrum of CuFe2O4–G (0.25). It turned out that graphene oxide was reduced to graphene due to the strong reducing capability of ethylene glycol. From the FTIR spectrum of CuFe2O4 and CuFe2O4–G (0.25) samples, the spectra show two main absorption corresponding to the stretching vibration of the tetrahedral and octahedral sites around 586 and 400 cm−1, respectively. The observed values illustrate that the frequency bands appearing at 586 and 400 cm−1 are responsible for the formation of CuFe2O4. The absorption band at 1596 cm−1 on spectrum referred to the vibration of remainder H2O in the sample.6
O and –COOH groups. However, it is can not be seen from the FTIR spectrum of CuFe2O4–G (0.25). It turned out that graphene oxide was reduced to graphene due to the strong reducing capability of ethylene glycol. From the FTIR spectrum of CuFe2O4 and CuFe2O4–G (0.25) samples, the spectra show two main absorption corresponding to the stretching vibration of the tetrahedral and octahedral sites around 586 and 400 cm−1, respectively. The observed values illustrate that the frequency bands appearing at 586 and 400 cm−1 are responsible for the formation of CuFe2O4. The absorption band at 1596 cm−1 on spectrum referred to the vibration of remainder H2O in the sample.6
Catalytic activity CuFe2O4–G
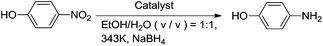
To obtain the optimal reaction conditions, the effect of type of catalyst and catalyst dosage on the reduction of p-nitrophenol were initially investigated (Table 1). As shown in Table 1, no product was obtained in the absence of the catalyst (Table 1, entry 1), indicating that the catalyst was necessary for the reaction. Only low yields of the product were obtained with either graphene, Fe3O4 or Fe3O4–graphene (with 25 wt% graphene) as catalyst (Table 1, entries 2–4). While CuFe2O4 could catalyze the reduction of p-nitrophenol efficiently, 99% yield was obtained when the reaction was performed at 343 K for 16 min (Table 1, entry 5). The combination of CuFe2O4 with appropriate amount of graphene results in a dramatic enhancement of the catalytic activity of CuFe2O4. The reaction time required to complete the reaction decreased with increasing the loading amount of graphene in CuFe2O4–G until 25 wt% (Table 1, entries 7–9). Compare with CuFe2O4 catalyst, the reaction time could lower down to 9 min from 16 min with CuFe2O4–G (0.25) as the catalyst, which can be attributed to its unique heteroarchitechture, which provides the remarkable synergistic effect between the CuFe2O4 and the graphene sheets. The presence of graphene could enhance the adsorption of reactant molecules onto the catalytic sites of the CuFe2O4–G through the π–π stacking and/or electrostatic interaction. However, the catalytic activity of CuFe2O4–G decreased when the content of graphene was increased to 35% (Table 1, entry 6). The large amount of graphene in CuFe2O4–G might decrease the number of the catalytic sites, which resulted in the decreasing of the catalytic activity of CuFe2O4–G (0.35). The above results also indicating that there is the most synergistic effect between CuFe2O4 and the graphene sheets. The reaction time could be further reduced with increasing the dosage of CuFe2O4–G (0.25) (Table 1, entries 11 and 12). But a longer reaction time was required to complete the reaction with decreasing the reaction temperature from 343 K to 333 K (Table 1, entry 13).| Entry | Catalyst | Catalyst (mg) | Time (min) | Yield (%) |
|---|---|---|---|---|
a Reaction condition: p-nitrophenol (1 mmol), solvent (10 mL, EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water = 1 water = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), NaBH4 (5 mmol), temperature (343 K).b Temperature (333 K). 1), NaBH4 (5 mmol), temperature (343 K).b Temperature (333 K). |
||||
| 1 | — | 20 | 500 | — |
| 2 | G | 20 | 500 | Trace |
| 3 | Fe3O4 | 20 | 60 | 26 |
| 4 | Fe3O4–G(0.25) | 20 | 60 | 39 |
| 5 | CuFe2O4 | 20 | 16 | 99 |
| 6 | CuFe2O4–G(0.35) | 20 | 35 | 99 |
| 7 | CuFe2O4–G(0.25) | 20 | 9 | 99 |
| 8 | CuFe2O4–G(0.15) | 20 | 12 | 99 |
| 9 | CuFe2O4–G(0.1) | 20 | 14 | 99 |
| 10 | CuFe2O4–G(0.25) | 10 | 12 | 99 |
| 11 | CuFe2O4–G(0.25) | 30 | 4 | 99 |
| 12 | CuFe2O4–G(0.25) | 50 | 3 | 99 |
| 13 | CuFe2O4–G(0.25) | 20 | 40b | 99 |
Fig. 4 showed the UV-vis absorption spectra of the reduction of p-nitroaniline by NaBH4 at various reaction times in the presence of CuFe2O4–G (0.25). The observed peak at 385 nm for the p-nitroaniline shows a gradual decrease in intensity with time and a new peak appeared at 295 nm indicating the formation of p-phenylene diamine. The results indicated that CuFe2O4–G (0.25) exhibited considerably high activity for the reduction of nitroarenes with sodium borohydride as the hydrogen donor.
 | ||
| Fig. 4 Successive UV-vis absorption spectra of the reduction of p-nitroaniline with NaBH4 in the presence of CuFe2O4–G (0.25). | ||
To show the generality of this model reaction, the reduction of a series of nitro aromatics was studied under the optimized reaction conditions. As shown in Table 2, aromatic nitro compounds containing various electrons donating (Table 2, entries 1–6) or electron-withdrawing groups (Table 2, entries 8) were converted to the corresponding amino aromatics in good yields and all the reactions could proceed smoothly. Moreover, the reduction was also successfully carried out on bulkier molecule such as 1-nitronaphthalene with high yield (Table 2, entry 7).
The reusability and recycling of CuFe2O4–G(0.25) was also investigated. The catalyst was separated from the reaction mixture using an external magnet, washed with ethanol for three times, dried at 60 °C in a vacuum oven for 2 h and reused in another reaction. The catalytic activity of CuFe2O4–G(0.25) did not show any significant decrease even after five runs.
Experimental
Materials and instrumentation
Ferric trichloride hexahydrate (FeCl3·6H2O), copper sulfate pentahydrate (CuSO4·5H2O), sodium borohydride (NaBH4), glycol, sodium hydroxide (NaOH), ethanol were all obtained from Chengxin Chemical Reagents Company (Baoding, China). Nitroaromatics were purchased from Aladdin Reagent Limited Company. The water used throughout the work was double-distilled on a SZ-93 automatic double-distiller from Shanghai Yarong Biochemistry Instrumental Factory (Shanghai, China).IR spectra (cm−1) were measured with a WQF-510 spectrometer. UV-vis absorption spectra were measured with a UV-vis spectrometer (UV-3600, Shimadzu). The size and morphology of the catalyst were observed by scanning electron microscopy (SEM) using a Hitachi S4800 field emission electron microscope operated at 30 kV. The XRD patterns of the samples were recorded with a Rigaku D/max 2500 X-ray diffractometer using Cu Kα radiation (40 kV, 150 mA) in the range 2θ = 10°–80°. 1H NMR (400 MHz) spectra were obtained with a Brucker AVANCE 400 spectrometer (DMSO-d6) using TMS as an internal standard.
Synthesis of CuFe2O4–graphene heteroarchitecture
Graphite oxide (GO) was prepared according to the procedure reported by us.25,26 CuFe2O4–graphene heteroarchitectures with differing graphene content (10, 15, 25, 35 wt%) were synthesized by a modified hydrothermal method reported in the literature.A typical experiment procedure for the synthesis of CuFe2O4–graphene heteroarchitecture with 25 wt% graphene content is as follows: 40 mg of GO was dispersed into 30 mL of ethylene glycol with sonication for 12 h. 0.1675 g (0.67 mmol) of CuSO4·5H2O and 0.3618 g (1.34 mmol) of FeCl3 were added to 10 mL of ethylene glycol and sonicated for 1 h. The above two solutions were then mixed together and stirred for 30 min. After that, the mixture was adjusted to pH of 8 with 6 mol L−1 NaOH aqueous solution and stirred for 30 min, yielding a stable bottlegreen homogeneous emulsion. The resulting mixture was transferred into a 70 mL Teflon-lined stainless steel autoclave and heated to 180 °C for 24 h under autogenous pressure. After the reaction mixture was cooled down to room temperature, the precipitate was filtered, washed with distilled water and ethanol, and dried in a vacuum oven at 30 °C for 12 h. The product was labeled as CuFe2O4–G (0.25). For comparison, the same method was used to synthesize CuFe2O4 without adding GO.
Reduction reactions of nitroarenes
In a 25 mL round bottom flask, nitroarenes (1.0 mmol, 1 eq.) was dissolved in a mixture of 10 mL H2O–EtOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). Then, sodium borohydride (5.0 mmol, 5 eq.) and CuFe2O4–G (0.25) (20 mg) were added. The mixture was stirred at 70 °C for an appropriate time depending upon the nature of the substrate. Upon completion of the reaction (monitored by TLC), the mixture was cooled to room temperature and the catalyst was separated by a magnet for recycling tests. The reaction mixture was extracted with ether (3 × 10 mL). The organic phase were combined together and dried over anhydrous MgSO4. The solvent was evaporated under vacuum. The pure products were obtained by silica-gel column chromatography using petroleum ether
1, v/v). Then, sodium borohydride (5.0 mmol, 5 eq.) and CuFe2O4–G (0.25) (20 mg) were added. The mixture was stirred at 70 °C for an appropriate time depending upon the nature of the substrate. Upon completion of the reaction (monitored by TLC), the mixture was cooled to room temperature and the catalyst was separated by a magnet for recycling tests. The reaction mixture was extracted with ether (3 × 10 mL). The organic phase were combined together and dried over anhydrous MgSO4. The solvent was evaporated under vacuum. The pure products were obtained by silica-gel column chromatography using petroleum ether![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethylacetate (4
ethylacetate (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as the eluent. The products were identified by IR and 1H NMR.
1) as the eluent. The products were identified by IR and 1H NMR.
Conclusions
In conclusion, an inexpensive and magnetically recyclable catalyst, CuFe2O4–G (0.25), was synthesized by a hydrothermal method. CuFe2O4–G (0.25) was used as an efficient catalyst for the reduction of nitroarenes. The results demonstrate that the combination of CuFe2O4 with graphene results in a dramatic enhancement of the catalytic activity of CuFe2O4, which can be attributed to the remarkable synergistic effect between the CuFe2O4 and the graphene sheets. The CuFe2O4–G (0.25) catalyst can be readily isolated from the reaction mixture using an external magnet due to its superparamagnetism and it can be reused at least five consecutive cycles without significant loss of its catalytic activity. We believe that this method is important addition to known procedures for reduction of aromatic nitro compounds either on a lab as well as on larger scale.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (no. 31171698), the Innovation Research Program of Department of Education of Hebei for Hebei Provincial Universities (LJRC009) and Natural Science Foundation of Hebei Province (B2011204051).Notes and references
- P. L. Gkizis, M. Stratakis and I. N. Lykakis, Catal. Commun., 2013, 36, 48 CrossRef CAS PubMed.
- M. M. Dell'Anna, P. Mastrorilli, A. Rizzuti and C. Leonelli, Appl. Catal., A, 2011, 401, 134 CrossRef PubMed.
- A. Tanaka, K. Fuku, T. Nishi, K. Hashimoto and H. Kominami, J. Phys. Chem. C, 2013, 117, 16983 CAS.
- L. Pehlivan, E. Métay, S. Laval, W. Dayoub, P. Demonchaux, G. Mignani and M. Lemaire, Tetrahedron Lett., 2010, 51, 1939 CrossRef CAS PubMed.
- S. Ghosh, Appl. Catal., A, 2004, 268, 61 CrossRef CAS PubMed.
- J. Feng, L. Su, Y. Ma, C. Ren, Q. Guo and X. Chen, Chem. Eng. J., 2013, 221, 16 CrossRef CAS PubMed.
- R. Nie, J. Wang, L. Wang, Y. Qin, P. Chen and Z. Hou, Carbon, 2012, 50, 586 CrossRef CAS PubMed.
- A. K. Shil, D. Sharma, N. R. Guha and P. Das, Tetrahedron Lett., 2012, 53, 4858 CrossRef CAS PubMed.
- J. Li, C.-y. Liu and Y. Liu, J. Mater. Chem., 2012, 22, 8426 RSC.
- E. Amini, M. Rezaei and M. Sadeghinia, Chin. J. Catal., 2013, 34, 1762 CrossRef CAS.
- R. Parella, A. Naveen Kumar and S. A. Babu, Tetrahedron Lett., 2013, 54, 1738 CrossRef CAS PubMed.
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, Y. Zhang, S. V. Dubonos, I. V. Grigorieva and A. A. Firsov, Science, 2004, 306, 666 CrossRef CAS PubMed.
- B. F. Machado and P. Serp, Catal. Sci. Technol., 2012, 2, 54 CAS.
- Y. Liang, H. Wang, H. Sanchez Casalongue, Z. Chen and H. Dai, Nano Res., 2010, 3, 701 CrossRef CAS PubMed.
- J. Zhang, Z. Xiong and X. S. Zhao, J. Mater. Chem., 2011, 21, 3634 RSC.
- B. Li and H. Cao, J. Mater. Chem., 2011, 21, 3346 RSC.
- E. Yoo, T. Okata, T. Akita, M. Kohyama, J. Nakamura and I. Honma, Nano Lett., 2009, 9, 2255 CrossRef CAS PubMed.
- Y. Li, L. Tang and J. Li, Electrochem. Commun., 2009, 11, 846 CrossRef CAS PubMed.
- L. Dong, R. R. S. Gari, Z. Li, M. M. Craig and S. Hou, Carbon, 2010, 48, 781 CrossRef CAS PubMed.
- S. Guo, S. Dong and E. Wang, ACS Nano, 2009, 4, 547 CrossRef PubMed.
- A. R. Siamaki, A. E. R. S. Khder, V. Abdelsayed, M. S. El-Shall and B. F. Gupton, J. Catal., 2011, 279, 1 CrossRef CAS PubMed.
- H.-Y. Zhang, C. Feng, N.-Z. Shang, S.-T. Gao, C. Wang and Z. Wang, Lett. Org. Chem., 2013, 10, 17 CAS.
- Y. Fu, P. Xiong, H. Chen, X. Sun and X. Wang, Ind. Eng. Chem. Res., 2012, 51, 725 CrossRef.
- N. Shang, C. Feng, H. Zhang, S. Gao, R. Tang, C. Wang and Z. Wang, Catal. Commun., 2013, 40, 111 CrossRef CAS PubMed.
- W. Wang, Y. Li, Q. Wu, C. Wang, X. Zang and Z. Wang, Anal. Methods, 2012, 4, 766 RSC.
- C. Wang, C. Feng, Y. Gao, X. Ma, Q. Wu and Z. Wang, Chem. Eng. J., 2011, 173, 92 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra05059b |
| This journal is © The Royal Society of Chemistry 2014 |