Terpenes in honey: occurrence, origin and their role as chemical biomarkers
Igor Jerković*a and
Piotr Marek Kuśb
aDepartment of Organic Chemistry, Faculty of Chemistry & Technology, University of Split, N. Tesle 10/V, HR-21000 Split, Croatia. E-mail: igor@ktf-split.hr; Fax: +385-21-329-461; Tel: +385-21-329-436
bDepartment of Pharmacognosy, Wrocław Medical University, ul. Borowska 211a, 50-556 Wrocław, Poland
First published on 1st July 2014
Abstract
The major developments and concepts in the study of terpenes (mainly monoterpenes) as well as C9-, C10-, C13-, C14- and C15- norisoprenoids in honey are summarized. Their occurrence and biosynthetic correlations (terpene transformations, particularly the generation of linalool derivatives as well as carotenoid degradation and the corresponding variety of norisoprenoids) are discussed considering the plant/nectar/bee-stomach composition and hive conditions (e.g. warm and acidic conditions that can lead to oxidative degradation of compounds). Terpenes up to C15 have been found as major compounds of the essential oils, but honeys from the same plant sources exhibit only partial similarity to the oil composition (e.g. lavender, sage or citrus essential oils/honeys). The formation of heat-derived or prolonged storage artefacts in honey (mainly the products of Maillard reactions and Strecker degradation reactions) influence the honey composition, and hotrienol is particularly labile among the chemical groups discussed. Stimulated by the challenges of fingerprinting methods and structure identification, exploratory studies (including our published results) on terpenes and norisoprenoids are summarized with emphasis on identifying specific or nonspecific chemical markers of the botanical origin of honey. In general, nonspecific biomarkers dominate in different honey types (e.g. monoterpenes: linalool, linalool oxide isomers or lilac alcohol/aldehyde isomers; norisoprenoids: isophorone and vomifoliol derivatives) while only a few specific biomarkers are found (e.g. anhydrolinalool oxide isomers, 3,4-dihydro-3-oxoedulan isomers, 3-oxo-retro-α-ionol isomers, kamahine A-C or meliracemoic acid).
1. Introduction
Volatile organic compounds (VOCs) are present in honey at very low concentrations as more or less complex mixtures of different organic functionalities (e.g. alcohols, carbonyl compounds, carboxylic acids, esters, phenols and others) with relatively low molecular weights.1 It has been pointed out that careful analysis of VOCs could be a useful tool for characterization of the botanical origin of honey accompanying melissoplaynological analysis and other routine physical–chemical parameters.1–3 The precise definition of a specific honey type is very difficult because of the different proportions of possible sources of nectar derived from a great variety of plants.4 In addition, detection of honey adulteration is also a major problem.3 Therefore, searching for new, more advanced methodologies for honey authentication has been encouraged.1–3 Owing to the presence of characteristic compounds, the chemical profile of VOCs represents a “chemical fingerprint” indicating more precisely the honey floral origin and/or its geographic origin.1,5 The “fingerprint” may contain specific (present in one honey type) or nonspecific (present in several honey types) chemical markers of the botanical origin.6,7 Gas chromatography with flame ionization detection (GC/FID) and quadrupole mass spectrometry (GC/MS) combine high separation efficiency and sensitivity and provide qualitative and quantitative data for the identification of the VOCs of honey.2,5 However, these techniques require prior isolation and concentration of VOCs to separate them from carbohydrates and water which are the main constituents of honey.5,7A large biodiversity of identified organic compounds is found in honey: volatiles (e.g. monoterpenes, norisoprenoids or benzene derivatives), phenolics, carbohydrates, nitrogen containing compounds and others.1,8 The contribution of catalase and other natural products to the antibacterial activity of honey was reviewed in 2000.9 Bioorganic studies on the VOCs of honey have been the focus of attention in the last 20 years. Several reviews exist from 1998 covering general isolation techniques for VOCs,2,5,7 the significance of honey VOCs (e.g. determination of floral and geographical origin1) and the biomedical activities of honey and the contribution from its VOCs.6 Various terpenes, including those occurring in honeys, are well known to possess versatile biological activities. Several of them exert, inter alia, antimicrobial, antioxidant and cancer-preventing effects. However, the concentration of these compounds in honey is low, thus they may only contribute to the overall biological activity of honey together with its other components.6 Our recent review focused in detail on low molecular volatile benzene derivatives from honey.10 As a continuation of our work and in contrast to the existing reviews, the present paper is targeted on summarizing the occurrence of terpenes (particularly monoterpenes) and C9-, C10-, C13-, C14- and C15-norisoprenoids in honey. Their extraction/analysis methods are presented in short as well as the chemical structures identified with a short overview of their possible formation pathways and with emphasis toward identifying the chemical markers of the honey’s botanical origin. In addition, only the partial similarity of terpenes from the essential oils and honey from the same plant source is presented. However, norisoprenoids in honeys appear to originate from the floral source.
2. Concepts in the study of honey volatiles
Several techniques have been applied for the isolation and concentration of VOCs from honey, including dynamic headspace purge-and-trap systems (DHS), static headspace extraction (SHE), headspace solid-phase microextraction (HS-SPME), solvent extraction (SE), ultrasound-assisted solvent extraction (USE) and Likens–Nickerson simultaneous distillation-extraction methodology (SDE).2,5,11–18 Selection of the appropriate isolation protocol should take into consideration that heat should not be applied to honey and that oxygenated VOCs can be water-soluble. Therefore, hydrodistillation (HD), the technique most often used for the isolation of essential oils, cannot be applied for the isolation of the VOCs in honey.19 SDE in semi micro apparatus is used only for the concentration of previously obtained solvent extracts and removal of non-volatile compounds.5 Recent studies have shown that the chemical composition of the headspace and solvent extract from the same honey sample has always been very different, representing complementary results for comprehensive characterisation of the sample.17,18,20–22VOCs of honey have been found to be the plant-derived constituents (phytochemicals) in their native form or transformed by the bees, compounds generated by the bees or transferred from the combs, but also as products of thermal treatment and/or prolonged storage as well as microbial or environmental contaminants.1,6 Little work has been done to correlate the chemical constituents of the plant to those of the corresponding honey. It was suggested23 that most of the compounds on which the assessment of the floral origin is based seem to originate from the nectar. Chemical components of several nectars have been investigated.24,25 The immediate contribution of the bees and combs to honey volatiles (proposed blank trial probe for honey chemical profiling) has confirmed that terpenes and norisoprenoids do not originate from the bees and the comb environment. Namely, the “saccharose probe” obtained mainly contained higher fatty alcohols and long-chain linear hydrocarbons.26 When considering the formation pathways of isoprenenoids and C9-, C10-, C13-, C14- and C15-norisoprenoids, targeted fingerprints of honey/nectar and honey/bee-stomach are important. The warm (about 30 °C) and acidic (pH ca. 3.5) conditions in the hive should also be considered27 since they can lead to oxidative degradation (reduction reactions are not favoured). Terpenes are produced by biochemical pathways that are described in further detail later in this section.
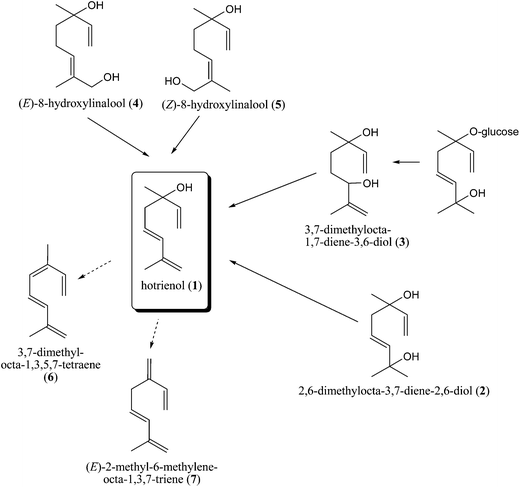
Heat-derived or prolonged storage artefacts in honey are mainly the products of Maillard reactions (among carbohydrates and amino compounds) and Strecker degradation reactions (among amino acids and dicarbonyl compounds). Among the VOCs, these artefacts belong to compounds of furan/pyran type as well as to aliphatic and aromatic aldehydes and others.19,28 Honey bioorganic studies have mainly been directed toward phytochemicals and their transformation by the bees. However, it is very difficult sometimes to distinguish transferred phytochemicals from the products of prolonged storage or heat treatment, especially when the honey origin and production is unknown. Two examples are phenylacetaldehyde and hotrienol. Phenylacetaldehyde is known to be a heat-derived artefact produced by Strecker degradation.10 However, it can be generated in honey from amino acid phenylalanine by enzyme catalysis.10,29 Its content in non-heated honey depends on the phenylalanine content (differs greatly and depends on the botanical origin of honey) and on the storage conditions. Hotrienol (3,7-dimethylocta-1,5,7-trien-3-ol) is a particularly labile compound among terpenes and is known to be a thermally generated product, but there are findings that support its natural occurrence in non-thermally treated honey.27 For example, hotrienol (1) is the principal component detected in leatherwood (Eucryphia lucida Baill.) honey headspace, while 2,6-dimethylocta-3,7-diene-2,6-diol (2) and hotrienol (1) were major compounds in the extracts.30 The diol (2) has also been detected in the nectar of leatherwood flowers, but increased polarity is likely to render it insufficiently volatile to contribute significantly to the headspace.
Much lower proportions of hotrienol (1) are found in unripe than in ripe honey, suggesting that hotrienol (1) is probably formed during honey ripening30 within the hive conditions (temperature, pH, enzymes that can lead to the oxidative degradation or the cleavage of glycosidic bonds). Hotrienol (1) can be derived either from the dehydration of 2,6-dimethylocta-3,7-diene-2,6-diol (2), or from the allylic rearrangement and dehydration of 3,7-dimethylocta-1,7-diene-3,6-diol (3) that can even be liberated from the corresponding glucoside or from dehydration30–32 of 8-hydroxylinalool isomers (4, 5) (Fig. 1). Dehydration of the diol (3) appeared to occur only at the C(2) position. No evidence could be found by GC/MS of honey extract for the presence of C(6) dehydrated compounds 2,6-dimethylocta-3,5,7-triene-2-ol or 2-methy1-6-methyleneocta-3,7-diene-2-ol. Likewise, it might be expected that hotrienol (1) would further dehydrate to 3,7-dimethylocta-1,3,5,7-tetraene (cosmene; 6) or (E)-2-methyl-6-methyleneocta-1,3,7-triene (7). The octatriene (7) is stable at room temperature.33 However, cosmene (6) has been reported to be highly unstable,34 and at present, neither cosmene (6) nor its isomers have been detected in the extracts of leatherwood honey.
3. Honey isoprenes: occurrence and main biosynthetic correlations
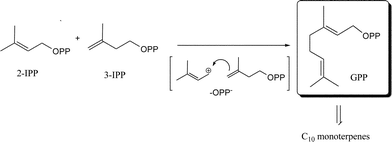
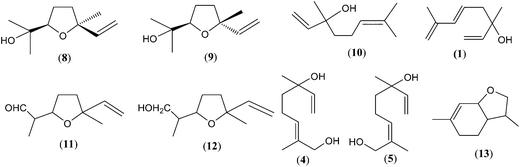
Volatile terpenes are often represented by C5-compounds (hemiterpenes), C10-compounds (monoterpenes) and C15-compounds (sesquiterpenes) that are the major constituents of the plant volatilome and the essential oils.35 The degree of volatility of these terpenes depends on their structures (i.e. oxygenation, architecture of the hydrocarbon backbone, saturation of carbon double bonds, etc.). All terpenes (isoprenoids) are produced from the precursor 2-isopentenyl pyrophosphate (2-IPP), also known as dimethylallyl pyrophosphate (DMAPP) and its isomer 3-isopentenyl pyrophosphate (3-IPP) (Fig. 2). They are synthesized by the mevalonate (MVA) pathway in the cytoplasm or deoxyxylulose-5-phosphate (DXP), also known as the methylerythritol phosphate (MEP) pathway, in the chloroplasts.35,36 Some exchange and/or cooperation occurs between these two pathways.37Regular monoterpenes derived from geranyl pyrophosphate (GPP) are the most common honey terpenes. Among them, linalool derivatives such as cis- and trans-linalool oxides (8, 9), linalool (10), hotrienol (1), lilac aldehyde isomers (11), lilac alcohol isomers (12), (E)- and/or (Z)-8-hydroxylinalool (4, 5), dill ether isomers (13) and others are the most abundant (Fig. 3).
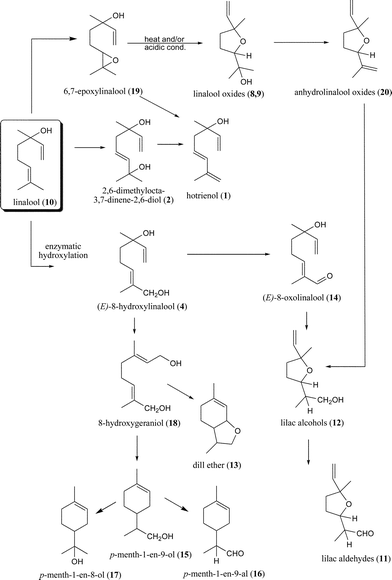
Starting from linalool (10), a variety of compounds can be formed, as shown in Fig. 4. Direct hydroxylation of linalool (10) at the C(8) position forms (E)-8-hydroxylinalool (4) which can give various products. Considering the acidic nature of honey, reduction reactions are not favoured and it is plausible to assume that (E)-8-hydroxylinalool (4) is transformed, via (E)-8-oxolinalool (14), by enzymatic ω-oxidation to lilac alcohols (12) that undergo oxidation to yield lilac aldehydes (11). Lilac alcohols (12) and aldehydes (11) have been found in the headspace of different honey types, e.g. Citrus spp., Paliurus spina-christi Mill. and Coriandrum sativum L.27,38,39 Dill ether (13), p-menth-1-ene-9-ol (15), p-menth-1-ene-9-al (16) and p-menth-1-ene-8-ol (α-terpineol; 17) are also derived from (E)-8-hydroxylinalool (4) and via the allylic rearrangement and cyclisation of 8-hydroxygeraniol (18). However, their presence in honey is limited. Alternatively, epoxidation of linalool (10) gives 6,7-epoxylinalool (19), which undergoes further reactions to form isomeric furanoid linalool oxides (8, 9) and anhydrolinalool oxides (20) that can further yield lilac alcohols (12). Two isomeric anhydrolinalool oxides (20) were identified for the first time in the headspace of citrus honey and later in coriander honey.27,39 Hotrienol (1) can be formed from the corresponding (E)/(Z)-2,6-dimethylocta-2,7-diene-1,6-diol [(E)/(Z)-8-hydroxylinalool] (4, 5). Biogenetic studies have shown that lilac aldehydes (11) are formed from linalool (10) by direct hydroxylation of linalool (10) at C(8) to (E)-8-hydroxylinalool (4) and further to (E)-8-oxolinalool (14) which is converted to lilac alcohols (12) that undergo oxidation to lilac aldehydes (11).40
The bioconversion of linalool (10) by honeybees under closed beehive conditions was investigated by Alissandrakis et al.41 The bees were fed with sugar syrup containing linalool, while the control bees were fed with linalool-free syrup. The research showed that the formation of furan/pyran linalool oxides (8, 9) as well as 2,6-dimethylocta-3,7-diene-2,6-diol (2) was favoured, probably catalysed by the enzymes secreted by the bees. Linalool oxides (8, 9) are possibly formed from linalool (10) and via the 6,7-epoxide intermediary (19),42 most likely involving P-450 hydroxylation.43 (E)-8-Hydroxylinalool (4) is formed from linalool after ω-hydroxylation, in the presence of P-450 hydroxylase.43 On the other hand, important linalool derivatives in honeys such as lilac aldehydes (11), cis- and trans-anhydrolinalool oxides (20) and (Z)-8-hydroxylinalool (5) were absent in the obtained probe, suggesting the requirement of a plant-derived enzyme for their bioconversion. The same stands for (E)-8-hydroxylinalool (4), a major volatile compound in citrus honey, although small amounts were present in the probe. To evaluate the effect of hive acidic conditions on the conversion of linalool (10), a methanol solution of linalool (1%) was mixed with acidified water (pH 3.4) and maintained at 30 °C for 10 days under constant stirring.41 Linalool derivatives were not generated and almost three-quarters of the linalool remained intact, while one-quarter was converted to myrcene, α-phellandrene, α-terpinene, limonene, (Z)-β-ocimene, (E)-β-ocimene, γ-terpinene and terpinolene. Therefore, acidic hive conditions do not lead to the formation of typical linalool derivatives found in honeys.
3.1. Comparison of bee-stomach/honey
The gathered nectar is stocked in the bee-stomach, which can contain up to 60 μL of the liquid. The enzymes in the saliva degrade nectar sucrose into glucose and fructose and cleave the glycosides. Upon returning to the hive, the content of the stomach is regurgitated into the waxy honeycomb and is ripened into honey. There are only a few studies in general comparing the bee-stomach/VOCs of honey. However, these studies are important for understanding the possible transformations of the nectar VOCs in the bee-stomach environment and their transfer to honey.In the honeybee-stomach containing linden (Tilia cordata Mill.) nectar, the aliphatic compounds, isoprenoids and the alkaloids remained unchanged.24 However, the active glycosidases are present in honeybee saliva and the liquid isolated from the bee stomach after gathering linden nectar24 contained new monoterpenic alcohols (three linalool derivatives: 3,7-dimethylocta-1,5-diene-3,7-diol (2), 3,7-dimethylocta-1,6-diene-3,5-diol (21) and 2,6-dimethylocta-2,7-dienal (22)) (Fig. 5) probably originating from glycosidase activity (cleavage of the nectar glycosides). These alcohols were also identified in Citrus spp. and Tilia cordata Mill. honeys.18,24 Aliphatic compounds that resemble the “Queen's substance” (9-oxodec-2-enoic acid) were also found.24
The contents of the stomach of 70 bees caught at the entrance of the hive on their way back from Mentha spp. nectar gathering were isolated by USE and analysed by GC/MS.44 The bee-stomach organic extract was mainly composed of aliphatic fatty acids and higher hydrocarbons with structures related to the composition of cuticular waxes and less to pheromones. The major identified terpene in the bee-stomach was 3,7-dimethylocta-1,5-dien-3,7-diol (terpendiol I; 2) probably originating from glycosidase activity (the cleavage of the nectar glycosides) in the saliva. From other identified groups of natural compounds, methyl syringate and vomifoliol can be considered to be plant-derived phytochemicals in the bee-stomach. Comparison of Mentha spp. honey organic extractives with the corresponding bee-stomach extractive44 indicated that methyl syringate and vomifoliol were transferred to honey while terpendiol I (2) was partially transformed to hotrienol (1) in ripened honey.
3.2. Comparison of nectar/honey/flower extracts
There are only a few studies in which the chemical constituents of the flower extracts have been correlated with those of the corresponding honey or nectar. The nectar VOCs could be composed in general of all the essential groups of natural products: compounds of fatty acid degradation, phenylpropanoids, norisporenoids, monoterpenes and others.Hotrienol (1) was the principal compound detected by headspace analysis in leatherwood (Eucryphia lucida Baill.) honey.30 2,6-Dimethylocta-3,7-diene-2,6-diol (2; precursor of hotrienol) was the principal compound in methylated and unmethylated ether extract of leatherwood honey and was found in the plant extracts. Methylated ether extracts of honey also contained different aromatic substances.30
A comparison of the extracts of Citrus spp. flowers and Citrus spp. honey showed the presence of similar monoterpenes. Extracts from the flowers of four Citrus species (lemon, orange, sour orange and tangerine) contained linalool (10) as the predominant compound.18 The extracts from citrus honey were predominated by an array of linalool derivatives (more than 80% of the total extract). (E)-2,6-Dimethylocta-2,7-diene-1,6-diol (4) was the predominant compound, while significant proportions of 2,6-dimethylocta-3,7-diene-2,6-diol (2) and (Z)-2,6-dimethylocta-2,7-diene-1,6-diol (5) were also present.18
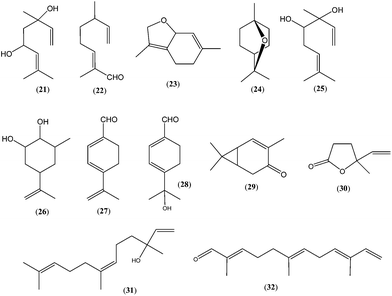
Analysis of linden honey and linden flowers24 led to the identification of a unique monoterpenoid ether called “linden ether” (2,4,5,7a-tetrahydro-3,6-dimethylbenzofuran; 23). The linden nectar, among others, contained norisoprenoids (vomifolione, vomifoliol and 3,5,5-trimethyl-4-(3-oxobutyl)cyclohex-2-ene-1-one) and a complex mixture of monoterpenes such as linden ether (23), 1,8-cineol (24), diols (3,7-dimethylocta-1,6-diene-3,4-diol (25) and 3-methyl-5-(1-methylethenyl)cyclohexan-1,2-diol (26)), p-metha-1,3,8-triene-7-al (27) as well as 8-hydroxy-p-mentha-1,3-diene-7-al (28)24 (Fig. 5).
The volatile profiles of apple honey and apple blossoms showed some similarities; several compounds found in honey were previously reported to be constituents of apple flowers.45,46 Besides linalool (10), terpenes in apple honey (mainly hotrienol (1), terpendiol I (2), (E)-8-hydroxylinalool (4), furan-type linalool oxides (8, 9) and car-2-en-4-one (29)) were different from those in apple flowers (limonene, car-3-ene, α-pinene, α-humulene, terpinolene, geraniol, (Z)- and (E)-citral, and other compounds).
In summary, only partial similarities among the VOCs from nectar/honey/flower extracts were found, but in all the investigated extracts, the array of linalool derivatives was most common. Differences among honey and flower volatiles were expected, since floral scent compounds are often constituents of several parts of the flower and may not be collected with nectar.
3.3. Comparison of honey/essential oils from the same plant
It is well known that terpenes are the major essential oil constituents in a wide range of aromatic plants. The bees often collect nectar from aromatic plants, and terpenes are found in corresponding honeys. Therefore, the aim of this paragraph is to compare the qualitative similarity among the essential oil composition and the corresponding honey terpenes, since significant quantitative differences exist. However, it should be pointed out that only partial qualitative similarity is expected since nectar composition differs from the essential oils. On the other hand, the comparison should take into consideration that the different isolation methods used for the VOCs in honey (e.g. USE, HS-SPME) and essential oil (e.g. HD) could also cause different chemical compositions of the isolates obtained. Lavandula spp., Coriandrum sativum L., Citrus spp., Salvia officinalis L., Mentha spp. and Rosmarinus officinalis L. honeys/essential oils were selected for the comparison (Table 1 and Fig. 6).| Plant source | Major honey terpenes | Method of isolation | Major essential oil terpenes | Method of isolation |
|---|---|---|---|---|
| a CH2Cl2 – extraction with CH2Cl2; SDE – simultaneous distillation extraction; SPE – solid phase extraction; HS-SPME – headspace solid phase microextraction; USE – ultrasonic solvent extraction; SE – solvent extraction; HD – hydrodistillation; SD – steam distillation; “–” – terpenes not found. | ||||
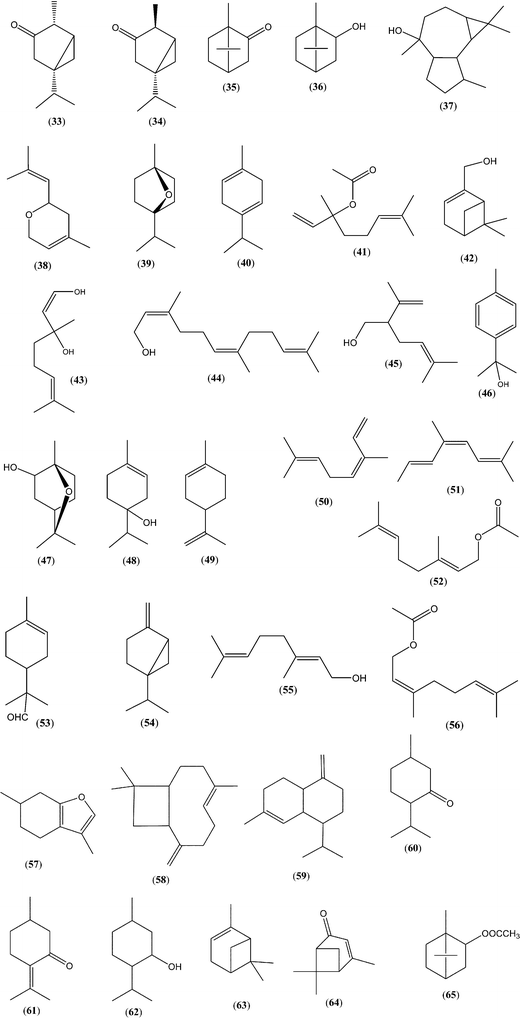
| Lavandula stoechas L. (French lavender) | — | CH2Cl2/SDE47 | Flowers: camphor (35), 1,4-cineol (39), γ-terpinene (40), 1,8-cineol (24), linalool (10), linalyl acetate (41) | HD48 |
| Lavandula angustifolia x latifolia L. (lavandin) | cis-/trans-Linalool oxide (8, 9), linalool (10), hotrienol (1), epoxylinalool isomers (19), myrtenol (42), 2,6-dimethylocta-3,7-diene-2,6-diol (2), 1,8-cineol isomers (24), 1-hydroxylinalool (43), farnesol (44) | SPE49 | Flowers: linalool (10), linalyl acetate (41), α-terpineol (17), lavandulol (45), 1,8-cineol (24), cis-/trans-linalool oxide (8, 9) | HD50 |
| — | CH2Cl2/SDE47 | SD50 | ||
| Lavandula latifolia L. (spike lavender) | cis-/trans-Linalool oxide (8, 9), linalool (10), hotrienol (1), α-terpineol (17), epoxylinalool isomers (19), myrtenol (42), p-cymen-8-ol (46), 2-hydroxycineol (47), 2,6-dimethylocta-3,7-diene-2,6-diol (2), 1,8-cineol isomers (24), 1-hydroxylinalool (43) | SPE49 | Flowers: linalool (10), camphor (35), 1,8-cineol (24), borneol (36), α-terpineol (17), terpinen-4-ol (48), farnesol (44) | HD51 |
| Lavandula angustifolia L. (English lavender) | — | CH2Cl2/SDE47 | Flowers: 1,8-cineol (24), borneol (36), camphor (35) | HD52 |
| Lavender (unidentified) specie) | cis-/trans-Linalool oxide (8, 9), linalool (10), hotrienol (1), α-terpineol (17), lilac alcohol isomers (12), epoxylinalool isomers (19), p-cymen-8-ol isomers (46) | CH2Cl2/SDE53 | See previous Lavandula spp. | HD50–52 |
| Lavandula hybrida Reverchon II (lavandin) | Hotrienol (1) | HS-SPME54 | Seeds: linalool (10), 1,8-cineol (24), limonene (49), cis-β-ocimene (50), camphor (35), linalyl acetate (41), terpinen-4-ol (48), alloocimene (51), α-terpineol (17) | HD55 |
| Hotrienol (1), 3,7-dimethylocta-1,5-diene-3,7-diol (2) | USE54 | |||
| Coriandrum sativum L. (coriander) | cis-/trans-Linalool oxide (8, 9), cis-/trans-anhydrolinalool oxide (20), lilac aldehyde/alcohol isomers (11, 12), p-menth-1-en-9-al (16) | HS-SPME39 | Flowers: linalool (10); seeds: linalool (10), geranyl acetate (52), γ-terpinene (40), linalyl acetate (41), others | HD56,57 |
| (E)-/(Z)-2,6-Dimethylocta-2,7-diene-1,6-diol (4, 5) | USE39 | |||
| Citrus spp. (citrus) | cis-/trans-Anhydrolinalool oxides (20), lavender lactone (30), dill ether (13), p-menth-1-en-9-al isomers (16), nerolidol (31) | HS-SPME27 | Flowers: sabinene (54), linalool (10), limonene (49), geraniol (55), linalyl acetate (41), geranyl acetate (52), neryl acetate (56), (E)-farnesol (44), (E)-nerolidol (31) | HD59,60 |
| Linalool (10), cis-/trans-linalool oxide (8, 9), α-terpineol (17), terpineal (53), lilac aldehyde/alcohol isomers (11, 12), nerolidol (31), sinensal (32) | CH2Cl2/SDE58 | |||
| (E)-/(Z)-2,6-Dimethylocta-2,7-diene-1,6-diol (4, 5), 2,6-dimethylocta-3,7-diene-2,6-diol (2), hotrienol (1) | USE17 | |||
| Salvia officinalis L. (Dalmatian sage) | — | USE61 | Flowers: α-/β-thujones (33, 34), 1,8-cineol (24), camphor (35), borneol (36), viridiflorol (37) | HD62 |
| Mentha spp. (Mentha aquatica L. and Mentha pulegium L.) (mint) | Hotrienol (1), cis-/trans-linalool oxides (8, 9), linalool (10), nerol oxide (38) | HS-SPME44 | Mentha aquatica L. (leaves and flowers): menthofuran (57), 1,8-cineol (24), trans-caryophyllene | HD63 |
| 3,7-Dimethylocta-1,5-diene-3,7-diol (2) | USE44 | |||
| (58), γ-cadinene (59), viridiflorol (37), linalool (10), others | HD64 | |||
| Mentha pulegium L. (leaves and flowers): menthone (60), pulegone (61), neo-menthol (62) | ||||
| Rosmarinus officinalis L. (rosemary) | Lilac aldehyde isomers (11) | CH2Cl2/SDE53 | Leaves and flowers: α-pinene (63), 1,8-cineol (24), verbenone (64), camphor (35), borneol (36), bornyl acetate (65), trans-caryophyllene (58), linalool (10) | HD65 |
| cis-/trans-Linalool oxides (8, 9), hotrienol (1), car-2-en-4-one (29), nerol, geraniol (55) | SDE14 | |||
| Hotrienol (1), car-2-en-4-one (29), 2,6-dimethylocta-3,7-diene-2,6-diol (2) | SE14 | |||
| Car-2-en-4-one (29), 2,6-dimethylocta-3,7-diene-2,6-diol (2), hotrienol (1), cis-/trans-linalool oxides (8, 9) | SPE14 | |||
 | ||
| Fig. 6 Typical structures of isoprenoids from honey and/or essential oil from Table 1. | ||
Lavender is a popular aromatic plant. The genus Lavandula contains several species. Honey from Spike lavender (Lavadula latifolia L.) is among the high-quality honeys. In recent years, cultivation of hybrid lavender species known as lavandin (L. agnustifolia x latifolia L.) has proliferated widely. Table 1 summarizes the terpenes found in different lavender/lavandin honeys and the corresponding essential oils. It can be seen that the method of isolation applied strongly influenced the isolation of terpenes within the same honey type: lavandin honey processed with CH2Cl2/SDE did not contain terpenes, while when processed with SPE, an array of terepenes was found. Linalool (10), cis-/trans-linalool oxides (8, 9) and 1,8-cineol (24) were found as common compounds in honey and the oil of L. agnustifolia x latifolia and L. latifolia. However, both honeys contained other linalool derivatives not found in the corresponding oils. Significant qualitative differences were found in L. hybrida Reverchon II honey and the oil.
Linalool (10) was found as a common constituent of Coriandrum sativum L. honey and in the essential oils in flowers/seeds. Linalool derivatives were more widespread in the honey and significant qualitative differences were found between its extract and headspace.
Citrus spp. honey headspace contained cis-/trans-anhydrolinalool oxides (20), lavender lactone (30), dill ether (13), p-menth-1-en-9-al isomers (16) and nerolidol (31) while terpenic diols and hotrienol (1) were present in the extracts. Linalool (10), cis-/trans-linalool oxide (8, 9), α-terpineol (17), lilac aldehyde/alcohol isomers (11, 12), nerolidol (31) and sinensal (32) were found in the honey by SDE. Common compounds in the essential oil from Citrus spp. flower were linalool (10) and nerolidol (31).
Salvia officinalis L. is a well known aromatic plant with essential oil composed of α-/β-thujones (33, 34), 1,8-cineol (24), camphor (35), borneol (36), and viridiflorol (37), whereas the corresponding honey extracts did not contain any terpenes (Fig. 6).
Mentha spp. honey contained hotrienol (1), cis-/trans-linalool oxides (8, 9), linalool (10), nerol oxide (38) and 3,7-dimethylocta-1,5-diene-3,7-diol (2) and the corresponding oils differed significantly in terpene qualitative composition.
Major differences were also found in Rosmarinus officinalis L. honey and the essential oil (Table 1).
The variety of linalool derivatives in different honeys could be related to warm (about 30 °C) and acidic honey conditions in the hive that could lead to the oxidative degradation of linalool (usually present in the plant). Moreover, bee and nectar enzymes lead to various reactions during ripening and storage. Therefore, oxidation reactions have been favoured as linalool (10) was found in honey in very low proportions compared to the amount in nectar.
3.4. Chiral isoprenoid distribution in honey
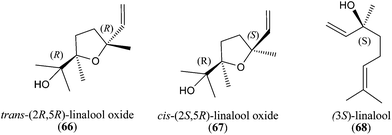
Natural chiral terpenes generally have a characteristic distribution of enantiomers that is related to the stereoselectively controlled biogenetic formation mechanisms. It is well known that the key building blocks of terpenes, dimethylallyl pyrophosphate (DMAPP) and isopentenyl pyrophosphate (IPP), can be produced via the mevalonate pathway from (R)-mevalonate and its subsequent phosphorylated metabolites (R)-mevalonate-5-phosphate and (R)-mevalonate-pyrophosphate. Combination of DMAPP and IPP via the enzyme prenyl transferase yields geranyl diphosphate (GPP). Linalyl PP and neryl PP are isomers of geranyl PP, and are likely to be formed from geranyl PP.36 These three compounds, by relatively modest changes, can lead to a range of linear chiral or achiral monoterpenes. The range of monoterpenes encountered is extended considerably by cyclization reactions, and monocyclic or bicyclic systems can be created. Generally, in the available literature, data on the enantiomeric composition/distribution of honey chiral terpenes is lacking. There are two ways to perform chiral separations of VOCs: (1) one-dimensional gas chromatography with chiral stationary phases, (2) multidimensional gas chromatography.66The enantiomeric ratio of major headspace chiral VOCs in Slovakian monofloral honeys (45 unifloral samples) was determined67 using GC with a chiral stationary phase (DB-FFAP column coated with permethyl-β-cyclodextrin). The isomeric ratios of linalool (10), cis- and trans-furanoid linalool oxides (8, 9), hotrienol (1) and four isomers of lilac aldehydes (11) were determined, but without identification of the corresponding enantiomers. It was found that the distribution of enantiomers in honey samples was partially dependent on the botanical origin.68 The first eluting enantiomer of trans-linalool oxide (8) could be found in a wide range in acacia, rapeseed, sunflower and linden honeys. The first eluting enantiomer of cis-linalool oxide (9) was found in the highest percentage in sunflower honey, compared to other samples. In acacia honey, the second eluting enantiomer of lilac aldehyde B (11) was present in higher purity than in other samples studied. This could be used to distinguish between acacia and rapeseed honeys, which is interesting as these plants blossom in the same period. The first eluting enantiomer of hotrienol (1) dominated in acacia honey. Linden honey could be distinguished from other monofloral honeys based on the content of the first eluting enantiomer of lilac aldehyde isomer D (11).
trans-(2R,5R)-Linalool oxide (66), cis-(2S,5R)-linalool oxide (67) and (3S)-linalool (68) prevailed (Fig. 7) in the headspace of orange honey,67 determined by chiral GC (MEGA-DEX DMP Beta column coated with dimethylpentyl-β-cyclodextrine). Linalool oxides can not only be generated from linalyl 6,7-epoxide, but also from 3,7-dimethyloct-1-ene-3,6,7-triol whose cyclization includes partial racemization at C(3).69 The VOCs in orange honey are directly correlated with those present in the flowers (linalool derivatives). (3S)-Linalool (68) dominated in the headspace of orange flowers and the same enantiomeric ratio was found in orange honey.
4. Honey norisoprenoids: occurrence and main biosynthetic correlations
Norisoprenoids are carotenoid derived compounds with 3,5,5-trimethylcyclohex-2-enic structures (degraded-carotenoid-like structures) (Fig. 8).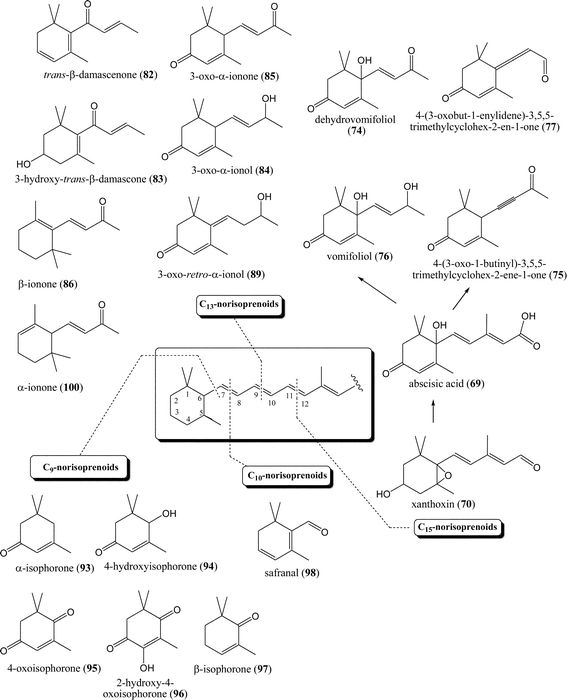 | ||
| Fig. 8 Relationships between the classes of degraded carotenoids (C9-, C10-, C13- and C15-norisoprenoids) with typical compounds identified in honeys. | ||
Carotenoids are tetraterpenoids, i.e. C40-compounds, which are composed of 8 isoprenic C5-units and differentiate between carotenes (pure hydrocarbons) and oxygen-bearing xanthophylls, such as lutein, zeaxanthin, violaxanthin and neoxanthin. The biosynthesis of carotenoids proceeds via 1-deoxy-D-xylulose-5-phosphate or mevalonate pathways of IPP biosynthesis.70,71
Carotenoids undergo chemical and enzymatic reactions generating norisoprenoids. These mechanisms involve two types of reaction:72 (1) enzymatic, catalysed by dioxygenases, as one-step direct carotenoid degradation or via glycosylation and breakdown of stored glycosides by glycosidase enzymes and (2) non-enzymatic reactions involving one or several steps of carotenoid degradation, stimulated by light, oxygen, temperature and acid hydrolysis. The breakdown products of carotenoids are carbonyl compounds with 13, 11, 10 or 9 carbon atoms retaining the terminal group of their carotenoid parent as illustrated in Fig. 8.
C13-compounds are the most abundant norisoprenoids in nature. They can be divided into: (1) compounds with megastigmane structure, including the family of ionones and damascones with oxygen at different positions, e.g. with a keto group at C(9) or at C(7) and (2) compounds with megastigmane structure but without oxygen in the lateral chain. In addition to the most widespread C13-norisoprenoids, C9-, C10- and C15-norisoprenoids have also frequently been detected in nature.36 Cleavage of the carotenoid chain is generally considered to proceed at different double bonds (Fig. 8). Cleavage of the C(11)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(12) bond forms C15-norisoprenoids via abscisic acid (ABA; 69), a well-known growth hormone formed after the cleavage of C40-carotenoids. Zeaxanthin is the first committed abscisic acid precursor.36 A series of enzyme-catalyzed epoxidations and isomerizations, and final cleavage of C40-carotenoid by dioxygenation reaction yield the precursor xanthoxin (70), which is then further oxidized to abscisic acid (69).36
C(12) bond forms C15-norisoprenoids via abscisic acid (ABA; 69), a well-known growth hormone formed after the cleavage of C40-carotenoids. Zeaxanthin is the first committed abscisic acid precursor.36 A series of enzyme-catalyzed epoxidations and isomerizations, and final cleavage of C40-carotenoid by dioxygenation reaction yield the precursor xanthoxin (70), which is then further oxidized to abscisic acid (69).36
4.1. C14/C15-Norisoprenoids and degradation products
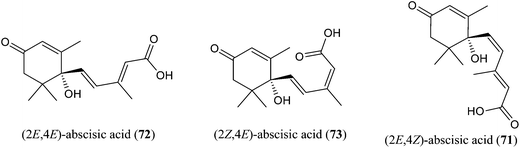
trans,cis-Abscisic acid [(E,Z)-ABA; 71] along with trans,trans-abscisic acid [(E,E)-ABA; 72] were found (Fig. 9) in New Zealand Salix nectar honey.73 Willow nectar honey from Croatia is characterized by the presence of two ABA isomers,74 with a concentration of (Z,E)-ABA (73) three times higher than (E,E)-ABA (98.2 and 31.7 mg kg−1; 72). Abscisic acid (ABA) was found in Polish Salix spp. honey (up to 53.7 mg kg−1) with the isomeric ratio (Z,E)-ABA (73)–(E,E)-ABA (72) = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.22 The presence of ABA isomers is correlated with the occurrence of other norisprenoid compounds in the volatile fraction of willow honey20,75 and confirms the common origin through a degraded carotenoid pathway. (Z,E)-ABA (73) and (E,E)-ABA (72) cannot be considered specific markers for willow honey, because they were found in several honeys such as crow ash (Guioa semiglauca F. Muell.), eucalyptus (Eucalyptus spp.), heath banksia (Banksia ericifolia L.f.), tea tree (Melaleuca quinquenervia (Cav.) S.T.Blake), jelly bush (Leptospermum polygalifolium Salisb.), heather (Erica spp.), sunflower (Helianthus annuus L.), manuka (Leptospermum scoparium J.R.Forst. & G.Forst.) and strawberry tree (Arbutus unedo L.) honeys.76–79 Nevertheless, ABAs can be used to discriminate Salix spp. nectar honey from honeydew honey, because the latter completely lacks these C15-norisoprenoid compounds.
2.22 The presence of ABA isomers is correlated with the occurrence of other norisprenoid compounds in the volatile fraction of willow honey20,75 and confirms the common origin through a degraded carotenoid pathway. (Z,E)-ABA (73) and (E,E)-ABA (72) cannot be considered specific markers for willow honey, because they were found in several honeys such as crow ash (Guioa semiglauca F. Muell.), eucalyptus (Eucalyptus spp.), heath banksia (Banksia ericifolia L.f.), tea tree (Melaleuca quinquenervia (Cav.) S.T.Blake), jelly bush (Leptospermum polygalifolium Salisb.), heather (Erica spp.), sunflower (Helianthus annuus L.), manuka (Leptospermum scoparium J.R.Forst. & G.Forst.) and strawberry tree (Arbutus unedo L.) honeys.76–79 Nevertheless, ABAs can be used to discriminate Salix spp. nectar honey from honeydew honey, because the latter completely lacks these C15-norisoprenoid compounds.
C13-norisoprenoid dehydrovomifoliol (4-hydroxy-4-(3-oxo-1-butenyl)-3,5,5-trimethylcyclohex-2-ene-1-one; 74) probably arises through degradation of abscisic acid (69).80 It was found in Calluna vulgaris L. honey along with structurally similar 4-(3-oxo-1-butinyl)-3,5,5-trimethylcyclohex-2-ene-1-one (75).81 Research into the absolute configuration by HPLC isolation followed by NMR, MS, IR, UV and polarimetry determined (S)-dehydrovomifoliol (74) in Calluna vulagaris L. honey82 in the range 186–264 mg kg−1, in Erica arborea L. honey (56 mg kg−1) and with minor quantities (1–6 mg kg−1) in eucalyptus, chestnut, linden, rape and buckwheat honeys. Structurally related vomifoliol (76) was identified, e.g. in Mentha spp. and Prunus mahaleb L. honeys.44,83 The allenic diketone 4-(3-oxobut-1-enylidene)-3,5,5-trimethylcyclohex-2-ene-1-one (77), appeared predominant in Calluna vulgaris L. honey with significantly higher concentrations than in Erica arborea L. honey. Dehydrovomifoliol (74) was also found in the honey of Salvia officinalis L.61 and leatherwood (Eucryphia lucida Baill.).30 It is direct a precursor of aromatic compounds such as vitispirans and theaspirans.84
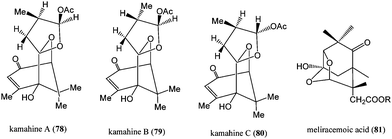
The isolation and characterisation of unusual nor-sesquiterpenoids C14-spiroketals kamahines A, B and C (Fig. 8; 78–80) was performed from the methylated diethyl ether extracts of kamahi (Weinmannia racemosa L. f.) honey samples.85 Kamahines A–C (78–80) were dominant constituents, the levels of which ranged from 43 to 144 mg kg−1. Interconversion of kamahines A–C (78–80) via reversible ring opening and closure of the hemiacetal entity present in these compounds leads to an equilibrated mixture of kamahines A (78), B (79), and C (80) in ca. 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio in solvents such as chloroform.86 The presence of a broad, late eluting peak in the GC/MS profiles of the methylated kamahi honey extracts85 is inconsistent with the proposal that interconversion between isomeric forms of kamahines A–C (78–80) also occurs in the vapour phase.
1 ratio in solvents such as chloroform.86 The presence of a broad, late eluting peak in the GC/MS profiles of the methylated kamahi honey extracts85 is inconsistent with the proposal that interconversion between isomeric forms of kamahines A–C (78–80) also occurs in the vapour phase.
In addition, the isolation and structural elucidation of meliracemoic acid (2,5-methano-hexahydro-1,3-benzodioxolane analogue; 81) was performed from New Zealand honeys.87 Meliracemoic acid (Fig. 10) is believed to be the rearrangement product of abscisic acid (69). Structural similarities between kamahines A–C (78–80) and meliracemoic acid (81) exist (both are 14-carbon degraded carotenoid-like compounds) and suggest that they may arise via rearrangement of a common precursor and that this may be a consequence of biosynthetic processes in the plant, rather than oxidative processes within honey.85 Relatively high levels of meliracemoic acid methyl ester, ranging from 9.3 to 21 mg kg−1, were detected in all of methylated diethyl ether extracts of the kamahi honey samples.85
4.2. C13-norisoprenoids
Besides degradation of abscisic acid (which produces e.g. dehydrovomifoliol (74), vomifoliol (76), the allenic and acetylenic diketones), cleavage of the C(9)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(10) bond of the carotenoid precursor directly generates C13-norisoprenoids such as trans-β-damascenone (82), 3-hydroxy-trans-β-damascone (83), 3-oxo-α-ionol (84) or 3-oxo-α-ionone (85) (Fig. 8). Biosynthetic interconversion among the C13-norisoprenoids formed is also possible. β-Damascenone (82) and β-ionone (86), smelling like honey, exhibit the lowest odour threshold values of all C13-norisoprenoids.88
C(10) bond of the carotenoid precursor directly generates C13-norisoprenoids such as trans-β-damascenone (82), 3-hydroxy-trans-β-damascone (83), 3-oxo-α-ionol (84) or 3-oxo-α-ionone (85) (Fig. 8). Biosynthetic interconversion among the C13-norisoprenoids formed is also possible. β-Damascenone (82) and β-ionone (86), smelling like honey, exhibit the lowest odour threshold values of all C13-norisoprenoids.88
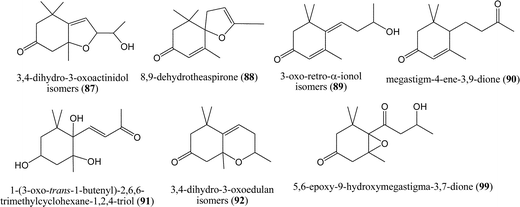
The dione 3-oxo-α-ionone (85) was the most abundant compound of eucalyptus honey ultrasonic solvent extract.89 C13-Norisoprenoids were dominant in number and concentration (up to 14.7 mg kg−1) in Australian blue gum (Eucalyptus leucoxylon F. Muell) and yellow box (Eucalyptus melliodora A. Cunn. Ex Schauer) honeys.90 Eight of these compounds were identified in honey for the first time (Fig. 9), four bicyclic diastereoisomers of 3,4-dihydro-3-oxoactinidols (87; only found in Australian honeys), bicyclic 8,9-dehydrotheaspirone (88; higher abundance in blue gum honey), 3-oxo-retro-α-ionols (89; 9-hydroxymegastigm-4,6-diene-3-ones) and megastigm-4-ene-3,9-dione (90) (Fig. 11).
Examination of the extractives of more than 200 New Zealand honey samples revealed the occurrence of 1-(3-oxo-trans-l-butenyl)-2,6,6-trimethylcyclohexan-1,2,4-triol isomers (91) to be confined to the samples that include thyme component, while the occurrence of abscisic acid isomers (69) was indicative of a willow component.73 The study indicates that in unifloral thyme honey, 1-(3-oxo-trans-1-butenyl)-2,6,6-trimethylcyclohexane-1,2,4-triol (91) was present at a level greater than 40 μg g−1.
The dominant C13-norisoprenoid in Centaurea cyanus L. honey headspace was 3,4-dihydro-3-oxoedulan (92; 2,3,5,6,8,8a-hexahydro-2,5,5,8a-tetramethyl-7H-1-benzopyran-7-one, also known as 2,5,5,8a-tetramethyl-2,3,5,6,8,8a-hexahydro-7H-chromen-7-one).91 However, this is the first report of 3,4-dihydro-3-oxoedulan (92) with high abundance in honey headspace (characteristic for C. cyanus L. honey). The formation of oxoedulan by intramolecular acid-catalyzed conjugate addition may well take place in acidic medium (e.g., hive conditions). Polyhydroxylated C13-norisoprenoids are natural precursors of edulans.92 The oxoedulan structure contains epoxide with the oxo group in position 3, so it is more volatile in comparison with its hydroxylated precursors, which can exhibit hydrogen donor and acceptor properties in the formation of hydrogen bonds in the honey-water solution. Therefore, 3,4-dihydro-3-oxoedulan (92) was abundant in the headspace.
4.3. C9-/C10-norisoprenoids
Degradation of carotenoid precursors producing different C9-norisoprenoids can yield most commonly α-isophorone (93), 4-hydroxy-α-isophorone (94), 4-oxoisophorone (95), 2-hydroxy-4-oxoisophorone (96), and others (Fig. 6). α-Isophorone (93), β-isophorone (97) and 4-oxoisophorone (95) were recognized by dynamic headspace extraction (DHE) as floral origin markers of strawberry-tree (Arbutus unedo L.) honey.93 The α-isophorone (93)/β-isophorone (97) ratio varied from 4 to 8, whereas the α-isophorone (93)/4-oxoisophorone (95) ratio was found to range from 11 to 20. α-Isophorone (93) is another 3,5,5-trimethylcyclohex-2-ene derivative isolated by CH2Cl2 dissolution followed by SDE and is liable to contribute to authenticating Calluna vulgaris L. honey81 where it can reach a concentration of 1453 μg g−1. High α-isophorone (93) contents, however, were observed in sunflower and eucalyptus honey samples, thus restricting the use of this compound as the specific floral marker.81 Smaller percentages of a variety of C9-norisoprenoids can be found in different honeys.1,91Only a few representatives of C10-norisoprenoids, which arise from cleavage of the C(7)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(8) bond in the carotenoid precursor (Fig. 8), were found in different honey types. Most typical is safranal (98) found in smaller percentages in Salix spp. honey20 and Prunus mahaleb L. honey.83 It was a useful marker of “corontillo” (Escallonia pulverulenta Ruiz & Pav.) honey among three unifloral Chilean honeys,94 although was present among the minor honey VOCs.
C(8) bond in the carotenoid precursor (Fig. 8), were found in different honey types. Most typical is safranal (98) found in smaller percentages in Salix spp. honey20 and Prunus mahaleb L. honey.83 It was a useful marker of “corontillo” (Escallonia pulverulenta Ruiz & Pav.) honey among three unifloral Chilean honeys,94 although was present among the minor honey VOCs.
4.4. Comparison of honey/plant norisoprenoids
Norisoprenoids constitute one of the most important classes of flavour compounds identified in nature. Derived from carotenoids, they occur in many higher plants, especially tobacco, tea, flower scents, fruits, spices, grapes and wine.88 A high norisoprenoid content has been found in heather and strawberry tree honey.81,93 Although they both come from plants belonging to the Ericaceae family, they appeared distinguishable on the basis of a few specific compounds, i.e. C10-norisoprenoids and 5,6-epoxy-9-hydroxymegastigma-3,7-dione (99) in strawberry tree honey. Heather honey (Calluna vulgaris L.) samples can be distinguished from tree heath (Erica arborea L.) honeys on the basis of their 4-(3-oxo1-butinyl)-3,5,5-trimethylcyclohex-2-ene-1-one (78) and dehydrovomifoliol (74) contents.81 Honeys derived from plants from the same family show several similarities that may be related to metabolism specific to the botanical family, but also show differences that are related to the specific plant.There have only been a few attempts to correlate floral source with the presence of norisoprenoids originating either in the nectar or in biochemical modifications carried out by the bees. In general, norisoprenoids in honeys appear to originate from the floral source. A recent papers89 reports a more detailed comparison of eucalyptus honey and flower extracts. More than 50% of the flower extract was comprised of 3-oxo-α-ionone (85), but its concentration in eucalyptus honey was not higher than in other unifloral Greek honeys. 3-Oxo-α-ionol (84) was found in the floral and honey extracts. The ratio of 3-oxo-α-ionone (85)/dehydrovomifoliol (74) in the flower extract was 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and less than 2
1 and less than 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in the honey extract. Other compounds found both in honey and flower extract include 4-hydroxyisophorone (94), 8,9-dehydrotheaspirone (88), 7,8-dihydro-3-oxo-α-ionone (89) and 6,7-dehydro-7,8-dihydro-3-oxo-α-ionol (89). The flowers should be extracted immediately after harvest. Analysing the extract obtained a week after the flowers had been kept at −18 °C gave a completely different composition, with 3-oxo-α-ionone (85) being a trace component and terpenic compounds predominating.89
1 in the honey extract. Other compounds found both in honey and flower extract include 4-hydroxyisophorone (94), 8,9-dehydrotheaspirone (88), 7,8-dihydro-3-oxo-α-ionone (89) and 6,7-dehydro-7,8-dihydro-3-oxo-α-ionol (89). The flowers should be extracted immediately after harvest. Analysing the extract obtained a week after the flowers had been kept at −18 °C gave a completely different composition, with 3-oxo-α-ionone (85) being a trace component and terpenic compounds predominating.89
In addition, norisoprenoids were isolated by hydrodistillation with minor percentages in the flowers, leaves and bark of Prunus mahaleb L., particularly C13-norisoprenoids (E)-β-damascenone (82), (E)-α-ionone (100) and (E)-β-ionone (86), as well as the C9-norisoprenoid α-isophorone (93).95 Partial similarity was noticed in comparison with Prunus mahaleb L. honey extracts.83
5. Isoprenoids and norisoprenoids as chemical biomarkers of the botanical origin of honey
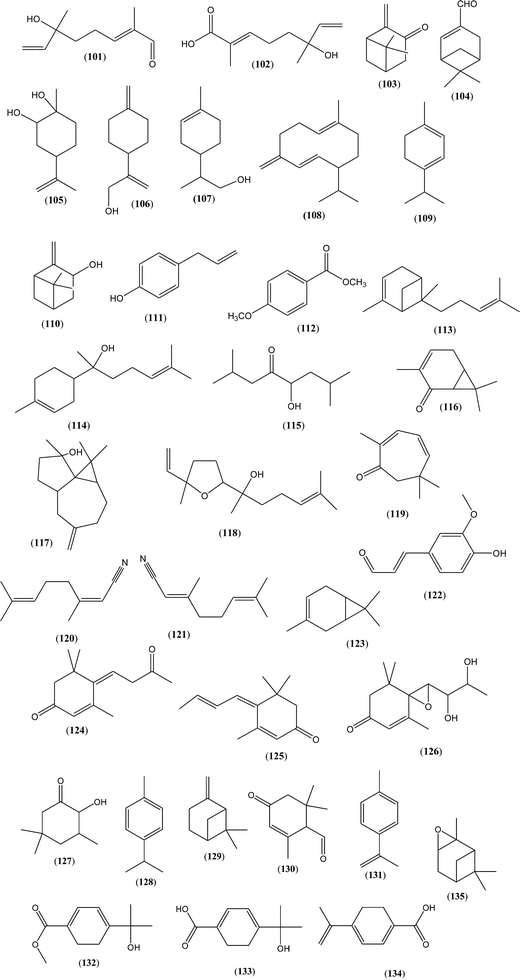
A large variety of terpenes frequently occur in many honey types. Often, molecules from this group may serve as specific (available only in one honey type) or non-specific (available in more honey types but in different quantities) markers of botanical origin (Table 2 and Fig. 12). Most of those compounds have a great impact on honey olfactory characteristics. However, the contribution of a specific compound to overall honey aroma depends on the extent to which the concentration exceeds its odour threshold (the possibility of synergistic and/or antagonistic interactions exists). Thus, even compounds present in low concentrations may contribute strongly to the honey aroma, if odour activity values (OAVs) are greater than 1.1,5,6 These proprieties make such compounds even more attractive as markers, since they have a significant impact on the specific aroma of the honey type.| Plant source | Several major or significant terpenes and norisoprenoids found in different honey types | Method of isolation |
|---|---|---|
| a SDE – simultaneous distillation-extraction; USE – ultrasonic solvent extraction; DHS – dynamic headspace extraction; SPE – solid phase microextraction; HS-SPME – headspace solid-phase microextraction; HPLC-DAD – high-performance liquid chromatography with diode-array detection; SE – solvent extraction; SOL – analysis of aqueous solution; * – percentage isomer distribution; deriv. – analysis of the sample was performed after derivatization. | ||
| Amorpha fruticosa L. (false indigo) | trans- and cis-linalool oxides (8, 9) | HS-SPME, USE103 |
| Arbutus unedo L. (strawberry tree) | α-Isophorone (93); β-isophorone (97); 4-oxoisophorone (95) | DHS93 |
| trans,trans-(72; 48%) and cis,trans-abscisic acid (73; 52%)*; unedone (126) | SOL117 | |
| Calluna vulgaris L. (heather) | Dehydrovomifoliol (74); isophorone (93); 4-(3-oxo-1-butinyl)-3,5,5-trimethylcyclohexe-2-ene-1-one (75); 4-(3-oxobut-1-enylidene)-3,5,5-trimethylcyclohex-2-ene-1-one (77) | SDE81 |
| cis,trans-Abscisic acid (73) | SPE116 | |
| α-Isophorone (93), 2-hydroxy-3,5,5-trimethyl-cyclohexanone (127) | HS-SPME114 | |
| Carduus nutans L. (nodding thistle) | (E)-2,6-Dimethyl-6-hydroxyocta-2,7-dienoic acid (102); (E)-2,6-dimethylocta-3,7-diene-2,6-diol (2); (Z)-2,6-dimethylocta-2,7-diene-1,6-diol (5); (E)-2,6-dimethyl-6-hydroxyocta-2,7-dienal (101); lilac aldehyde (11) and lilac alcohol (12) isomers | SE, deriv.102 |
| Centaurea cyanus L. (cornflower) | (Z/E)-3-Oxo-retro-α-ionol (89); 3-oxo-α-ionol (84); 3-oxo-α-ionone (85) | SPE118 |
| 3,4-Dihydro-3-oxoedulan (92); 4-oxoisophorone (95) | HS-SPME91 | |
| (Z/E)-3-Oxo-retro-α-ionol (89) | USE115 | |
| Citrus sinensis (L.) osbeck (orange) | (E)-2,6-Dimethylocta-2,7-diene-1,6-diol (4) | SE105 |
| Citrus spp. (citrus) | Linalool (10); trans- and cis-linalool oxides (8, 9); α-terpineol (17); terpineal (53); lilac aldehyde (11); lilac alcohol (12); sinensal (32) isomers | SDE58 |
| Limonene-1,2-diol (105; 8-p-menthene-1,2-diol) | HS-SPME11 | |
| (E)-/(Z)-2,6-Dimethylocta-2,7-diene-1,6-diol (4, 5), 2,6-dimethylocta-3,7-diene-2,6-diol (2), hotrienol (1) | USE17 | |
| Lilac aldehyde (11), lilac alcohol (12) isomers; cis-linalool oxide (9); p-menth-1-en-9-al (16); p-menth-1-en-9-ol (15); hotrienol (1) | USE18 | |
| Lilac aldehyde (11); anhydrolinaloxide (20); 1-p-menthen-9-al (16) isomers; dill ether (13); nerolidol (31) | HS-SPME27 | |
| p-Menth-1(7),8(10)-dien-9-ol (106) | HS-SPME119 | |
| cis-Linalool oxide (9); lilac aldehyde (11), lilac alcohol (12) and sinensal (32) isomers; limonyl alcohol (107); p-menth-1-en-9-al (16) | SDE53 | |
| Lilac aldehydes (11) | HS-SPME114 | |
| Coriandrum sativum L. (coriander) | cis/trans-Anhydrolinalool oxides (20); p-menth-1-en- 9-al (16); trans-linalool oxide (8); (E/Z)-2,6-dimethylocta-2,7-diene-1,6-diol (4, 5) | HS-SPME,39 USE39 |
| Erica arborea L. (tree heath) | Isophorone (93); 4-(3-oxobut-1-enylidene)-3,5,5-trimethylcyclohex-2-en-1-one (77) | SDE81 |
| α-Isophorone (93); 2-hydroxy-3,5,5-trimethyl-cyclohexanone (127) | HS-SPME114 | |
| Escallonia pulverulenta Ruiz & Pav. (corontillo) | Safranal (98) | SPME94 |
| Eucalyptus globulus Labill. (blue gum) | exo-2-Hydroxycineol (47); dehydrovomifoliol (74); 3-oxo-α-ionone (85); 3-oxo-α-ionol (84); 7,8-dihydro-3-oxo-α-ionone (89); 6,7-dehydro-7,8-dihydro-3-oxo-α-ionone (124); 6,7-dehydro-7,8-dihydro-3-oxo-α-ionol (89); 8,9-dehydrotheaspirone (88) | USE89 |
| 5-Hydroxy-2,7-dimethyloctan-4-one (115); spathulenol (117); car-3-en-2-ol (116); p-cymene (128); p-cymen-8-ol (46) isomers; 2-hydroxycineol (47); 4-oxoisophorone (95); 3-oxo-α-ionone (85) | SDE53 | |
| Dehydrovomifoliol (74); 3-oxo-α-ionone (85) | SE105 | |
| Nerolidol (31) | SDE28 | |
| Eucalyptus leucoxylon F. Muell (blue gum – South Australian) | 3,4-Dihydro-3-oxoactinidol (87) isomers; 3-oxo-α-ionol (84); 3-oxo-α-ionone (85); 8,9-dehydrotheaspirone (88); dehydrovomifoliol (74) | SE90 |
| Eucalyptus melliodora A. Cunn. ex Schauer (yellow box) | 3,4-Dihydro-3-oxoactinidol (91) isomers; 3-oxo-α-ionol (84); dehydrovomifoliol (74); 3-oxo-α-ionone (85) | SE90 |
| Eucryphia cordifolia Cav. (ulmo) | Isophorone (93); 4-oxoisophorone (95) | HS-SPME94 |
| Eucryphia lucida Baill (leatherwood) | 3,7-Dimethylocta-1,5,7-trien-3-ol (1; hotrienol); 2,6-dimethylocta-3,7-diene-2,6-diol (2) | HS-SPME30 |
| Fagopyrum esculentum Moench. (buckwheat) | β-Damascenone (82) | SE120 |
| Galactites tomentosa moench. (thistle) | α-Linalool (10); lilac aldehyde (11; isomer IV); hotrienol (1), 4-oxoisophorone (95); cis-linalool oxide (9) | HS-SPME, DHS98 |
| Gossypium L. (cotton) | Neryl (120) and geranyl (121) nitrile; cis-/trans-furanoid linalooxide (8, 9) | USE, HS-SPME112 |
| Hedysarum coronarium L. (sulla) | Vomifoliol (76) | USE121 |
| Helianthus annuus L. (sunflower) | α-Pinene (63) | DHS108 |
| Lavandula angustifolia x latifolia (lavandin) | Hotrienol (1) | HS-SPME54 |
| Farnesol (44); dehydrovomifoliol (74) | SPE49 | |
| Lavandula spp. (lavender) | Nerolidol oxide (118), hotrienol (1) | SDE53 |
| Leptospermum polygalifolium salisb. (jelly bush) | trans,trans-(72; 68%) and cis,trans-abscisic acid (73; 32%)* | SPE77 |
| Leptospermum scoparium J. R. Forst. & G. Forst. (manuka) | trans,trans-(72; 95%) and cis,trans-abscisic acid (73; 5%)* | SPE77 |
| Malus domestica Borkh. (apple) | Hotrienol (1); lilac aldehyde isomers (11); terpendiol I (2); linalool (10); vomifoliol (76) | HS-SPME,45 USE45 |
| Mentha spp. (mint) | Hotrienol (1); vomifoliol (76) | HS-SPME, USE44 |
| Paliurus spina-christi Mill. (Christ's Thorn) | Lilac aldehyde isomers (11) | HS-SPME38 |
| Pinus brutia Ten. (turkish pine) honeydew | α-Pinene (63); β-damascenone (82) | HS-SPME111 |
| Car-3-ene (123) | DHS113 | |
| Prunus cerasus L. (sour cherry) | Lilac aldehydes (11); vomifoliol (76) | HS-SPME, USE,96 |
| Prunus dulcis L. (Mill.) D. A. Webb (almond tree) | Eucarvone (119) | HS-SPME110 |
| Prunus mahaleb L. (mahaleb cherry) | Vomifoliol (76) | USE83 |
| Quercus frainetto Ten. (oak) honeydew | cis- and trans-linalool oxides (8, 9) | HS-SPME104 |
| Quillaja saponaria Mol. (soapbark) | Megastigmatrienone (125); β-pinene (129) | HS-SPME94 |
| Robinia pseudoacacia L. (acacia) | Hotrienol (1); cis-linalool oxide (9); linalool (10) | HS-SPME119 |
| cis,trans-Abscisic acid (73) | SE105 | |
| cis-Linalool oxide (9) | DHS108 | |
| Rosmarinus officinalis L. (rosemary) | cis-Linalool oxide (9); hotrienol (1); lilac aldehyde (11) and lilac alcohol (12) isomers; eucarvone (119); isophorone (93), 4-oxoisophorone (95) | HS-SPME119 |
| Lilac aldehydes (11) | SDE53 | |
| Rhododendron L. (rhododendron) | Lilac aldehyde (11); β-damascenone (82) | HS-SPME99 |
| Rhus succedanea L. (haze) | Lilac aldehydes (11) | DHS100 |
| Salix spp. (willow) | Linalool derivatives, borneol (36); pinocarvone (103); myrtenal (104), verbenone (64); trans-β-damascenone (82); vomifoliol (76); trans,trans-(72; 34%), cis,trans-abscisic acid (73; 66%)* | HS-SPME, USE, SOL22 |
| trans,trans-(72; 24%), cis,trans-abscisic acid (73; 76%)* | HPLC-DAD74 | |
| 3-Hydroxy-trans-β-damascone (83); safranal (98); lilac alcohols (12) | HS-SPME, USE20 | |
| trans,trans-(72; 28.4%), trans,cis-abscisic acid (71; 71.6%) | SE, deriv.73 | |
| Salvia officinalis L. (Dalmatian sage) | 2,6,6-Trimethyl-4-oxocyclohex-2-ene-1-carbaldehyde (130) | USE61 |
| Solidago canadensis L. (Canada goldenrod) | Germacrene-D (108); nerolidol (31) | SDE106 |
| Thymus spp. (thyme) | Hotrienol (1), cis-linalool oxide (9); p-menth-1-en-9-al (16); lilac aldehydes (11) | HS-SPME119 |
| Tilia spp. (linden) | Menthol (62); 8-p-menthene-1,2-diol (105); p-cymenene (131) | SDE107 |
| 8-p-Menthen-1,2-diol (105) | HS-SPME11 | |
| 4-(1-Hydroxy-1-methylethyl)cyclohexa-1,3-dienecarboxylic acid methyl ester (132); 4-(1-hydroxy-1-methylethyl)-cyclohexa-1,3-dienecarboxylic acid (133); 4-(1-methylethenyl)-cyclohexa-1,3-dienecarboxylic acid (134) | SE105 | |
| α-Terpinene (109); α-pinene oxide (135); p-cymene (128) | DHS108 | |
| Turbina corymbosa L. (christmas vine) | trans-Pinocarveol (110); chavicol (111); trans-α-bergamotene (113); α-bisabolol (114) | HS-SPME109 |
| Weinmannia racemosa L.f. (kamahi) | Kamahine isomers (78–80); meliracemoic acid (81) | SE, deriv.86 |
 | ||
| Fig. 12 Selected structures of compounds in Table 2. | ||
Many honey types are particularly rich in monoterpenes. Owing to low molecular weight and low boiling point, they are often the main components of the honey headspace and dominate in the volatile profiles obtained by HS-SPME and dynamic headspace purge-and-trap techniques (DHS). Norisoprenoids, compounds that derive from carotenoid degradation, can be divided into two groups: more volatile C9- and C10-compounds and usually less volatile C13 or semi-volatile/non-volatile C15-molecules. Similar to monoterpenes, C9-norisoprenoids are present particularly in the extracts obtained by HS-SPME and DHS techniques. C13-norisoprenoids with some exceptions (e.g. 3,4-dihydro-3-oxoedulan (92), trans-β-damascenone (82)) are generally not sufficiently volatile to be present in extracts obtained with the above mentioned techniques but can be extracted, for example, by solvent extraction (SE), solid phase microextraction (SPE) or ultrasonic solvent extraction (USE) and analysed by GC-FID/MS and/or HPLC, UPLC combined with diode array detection (DAD) and/or MS detectors.
One of the most common terpenes occurring in honey is linalool (10) derived from flower nectar. In the beehive, it is transformed to various derivatives such as furan and pyran linalool oxides (8, 9) as well as terpendiol I (2) that is attributed to the action of the bee enzymes, while plant-derived enzyme is needed for bioconversion to lilac aldehydes (11), cis- and trans-anhydrolinalool oxides (20), (E)-2,6-dimethyl-6-hydroxyocta-2,7-dienal (101) (Fig. 12) and 8-hydroxylinalool (4).41 Other linalool derivatives, such as lilac aldehyde isomers (11) were found to be abundant in the honey types from numerous plants: sour cherry (Prunus cerasus L.), citrus trees (Citrus spp.), thyme (Thymus spp.), Christ's Thorn (Paliurus spina-christ Mill.), apple tree (Malus domestica Borkh.), purple milk thistle (Galactites tomentosa Moench.), rhododendron (Rhododendron L.), rosemary (Rosmarinus officinalis L.), wax tree (Rhus succedanea L.), cashew (Anacardium occidentale L.), and nodding thistle (Carduus nutans L.).18,38,45,53,96–102 In willow (Salix spp.), citrus, rosemary, and nodding thistle honeys, a significant percentage of closely related lilac alcohol (12) was also reported.22,58,97,102 Linalool oxides (8, 9) were mentioned as significant volatile compounds and potential markers in coriander (Coriandrum sativum L.), citrus, thyme, false indigo (Amorpha fruticosa L.), oak honeydew (Quercus frainetto Ten.), purple milk thistle, acacia (Robinia pseudoacacia L.), and rosemary, and present in minor quantities in soapbark (Quillaja saponaria Mol.) honeys.39,58,94,97,98,103,104. (E)-8-Hydroxylinalool (4) was found to be a marker in orange honey105 and (Z)-8-hydroxylinalool (5) in Greek citrus honey.27 The same isomer was also found in New Zealand's thistle honey.102 Other derivatives, such as 2,6-dimethylocta-3,7-diene-2,6-diol (2), were reported in leatherwood (Eucryphia lucida Baill.) and nodding thistle honeys.30,102 In the latter, (E)-2,6-dimethyl-6-hydroxyocta-2,7-dienoic acid (102) and (E)-2,6-dimethyl-6-hydroxyocta-2,7-dienal (101) were also reported as principal compounds102 (Fig. 12).
Terpendiol I (2) and hotrienol (1; which is formed from the former during the ripening of honey)27 were also reported to be significant VOCs in citrus, mint (Mentha spp.), thyme, apple, lavender (Lavandula spp.), leatherwood, purple milk thistle, acacia and rosemary honey.18,30,45,53,97,98 Since most of the linalool derivatives were abundant in several honey types, they can be mostly useful only as non-specific markers of botanical origin. However, some compounds from this group are less common, e.g. cis/trans-anhydrolinalool oxides (20), and they were found in significant percentages in coriander honey and could be used as specific markers of this honey type.39
Less commonly occurring terpenes, such as borneol (36), pinocarvone (103), myrtenal (104) and verbenone (64) in willow honey,22 α-terpineol (17), terpineal (53), sinensal (32), limonene-1,2-diol (105; 8-p-menthene-1,2-diol), p-menth-1-ene-9-al (16), p-menth-1-ene-9-ol (15), p-menth-1(7),8(10)-dien-9-ol (106) and limonyl alcohol (107) in citrus honey,53,58,97 and p-menth-1-ene-9-al (16) in thyme honey97 may support the characterization of honey types. Similarly, germacrene-D (108) and nerolidol (31) were found in Canada goldenrod (Solidago canadensis L.),106 menthol (62), 8-p-menthene-1,2-diol (105), α-terpinene (109), and others in linden (Tilia spp.),11,107,108 trans-pinocarveol (110), chavicol (111), methyl anisate (112), trans-α-bergamotene (113) and α-bisabolol (114) in Christmas vine (Turbina corymbosa L (Raf.)),109 eucarvone in almond tree (Prunus dulcis L. (Mill.) D. A. Webb),110 safranal (98) in corontillo (Escallonia pulverulenta Ruiz & Pav) honey,109 α-pinene (63) in Turkish pine (Pinus brutia Ten.) honeydew,111 exo-2-hydroxycineol (47), 5-hydroxy-2,7-dimethyloctan-4-one (115), car-3-en-2-ol (116), spathulenol (117), and nerolidol (31) in blue gum (Eucalyptus globulus Labill.),89 nerolidol oxide (118) in lavender,53 eucarvone (119) in rosemary,97 neryl and geranyl nitrile (120, 121), and coniferaldehyde (122) in cotton,112 α-pinene (63) in sunflower108 and car-3-ene (123) in Turkish pine honey113 as well as other compounds. Very specific are kamahine A–C (78–80) and meliracemoic acid (81) found only in Kamahi (Weinmannia racemosa L.f.) honey.86
C9-Norisprenoids are also quite common but usually their percentage in headspace is not high. Some authors attributed a high concentration of 4-oxoisophorone (95; 2,6,6-trimethylcyclohex-2-ene-1,4-dione) to eucalyptus honey,53 isophorone (93) and its oxidized derivative 4-oxoisophorone (95) to ulmo (Eucryphia cordifolia Cav.) honey,94 α-isophorone (93) as a non-specific marker of heather and tree heath (Calluna vulgaris L., Erica arborea L.),114 and α-isophorone (93), β-isophorone (97) and 4-oxoisophorone (95) to strawberry tree (Arbutus unedo L.) honey.93 The latter was also found in purple milk thistle.98 However, those compounds are also quite common in other honey types.
Among the C13 compounds, vomifoliol (76; present in sulla (Hedysarum coronarium L.), mint, willow, sour cherry (Prunus cerasus L.), and yellow box (Eucalyptus mellidora A.Cunn. ex Schauer) honeys), dehyderovomifoliol (74; present, for example, in blue gum, yellow box, and heather honeys), trans-β-damascenone (82; present in Rhododendron, Turkish pine honeydew, willow, and buckwheat (Fagopyrum esculentum Moench.) honeys), and 3-oxo-α-ionol (84) and its oxidized derivative 3-oxo-α-ionone (85; present in blue gum, yellow box, and cornflower (Centaurea cyanus L.) honeys) were found most frequently in various honeys.89,90,115 Similarly, the isomers of abscisic acid (69) were reported in several honey types, such as acacia, manuka (Leptospermum scoparium J. R. Forst. & G. Forst.), jelly bush (Leptospermum polygalifolium Salisb.), heather, willow as well as strawberry tree honey, but with various occurrences of the isomers and their ratios.74,77,116,117 Other less frequent molecules were 7,8-dihydro-3-oxo-α-ionone (89) with its derivative 6,7-dehydro-7,8-dihydro-3-oxo-α-ionone (124) with corresponding alcohol 6,7-dehydro-7,8-dihydro-3-oxo-α-ionol (89) or 8,9-dehydrotheaspirone (88) occurring in blue gum honey.89 Similarly, 4-(3-oxobut-1-enylidene)-3,5,5-trimethylcyclohex-2-ene-1-one (77) present in tree heath and heather honey,81 and 4-(3-oxo-1-butynyl)-3,5,5-trimethylcyclohex-2-en-1-one (78) that is present only in the latter,81 megastigmatrienone (125) occurring in soapbark,94 megastigm-4-ene-3,9-dione (89) in yellow gum (Eucalyptus leucoxylon F.Muell.) together with a small quantity of isomeric 3-oxo-retro-α-ionols (89)90 that were not as abundant as (Z/E)-3-oxo-retro-α-ionols (89) in cornflower honey accompanied additionally by a significant percentage of 3-oxo-α-ionol (84) and 3,4-dihydro-3-oxoedulan (92).91,115,118
Although a large group of terpenes and norisoprenoids occur in several different honey types, they can be useful as non-specific markers of botanical origin or in combination with other characteristic compounds. Additionally, the relative percentage among the isomers may also be important.117 A high abundance of several compounds may be used as specific markers, e.g. a significant quantity of 3,4-dihydro-3-oxoedulan (92), which dominated the headspace of cornflower honey, till now was found abundant only in this honey type.91 A further example is another C13 compound – unedone (126), which can be used as a specific marker for Arbutus unedo L. honey.117
Acknowledgements
This work has been fully supported by the Croatian Science Foundation under the project (8547) “Research of natural products and flavours: chemical fingerprinting and unlocking the potential”.References
- V. Kaškonienė and P. R. Venskutonis, Compr. Rev. Food Sci. Food Saf., 2010, 9, 620–634 CrossRef PubMed.
- E. Anklam, Food Chem., 1998, 63, 549–562 CrossRef CAS.
- S. Bogdanov, K. Ruoff and L. Persano Oddo, Apidologie, 2004, 35, 4–17 CrossRef.
- L. Persano Oddo and S. Bogdanov, Apidologie, 2004, 35, 2–3 CrossRef.
- L. F. Cuevas-Glory, J. A. Pino, L. S. Santiago and E. Sauri-Duch, Food Chem., 2007, 103, 1032–1043 CrossRef CAS PubMed.
- C. E. Manyi-Loh, R. N. Ndip and A. M. Clarke, Int. J. Mol. Sci., 2011, 12, 9514–9532 CrossRef CAS PubMed.
- I. Jerković and Z. Marijanović, Croat. J. Food Sci. Technol., 2009, 1, 28–34 Search PubMed.
- J. M. Alvarez-Suarez, S. Tulipani, S. Romandini, A. Vidal and M. Battino, Curr. Anal. Chem., 2009, 5, 293–302 CrossRef CAS.
- R. J. Weston, Food Chem., 2000, 71, 235–239 CrossRef CAS.
- I. Jerković, Synlett, 2013, 24, 2331–2334 CrossRef PubMed.
- L. Piasenzotto, L. Gracco and L. Conte, J. Sci. Food Agric., 2003, 83, 1037–1044 CrossRef CAS.
- A. C. Soria, I. Martínez-Castro and J. Sanz, J. Sep. Sci., 2003, 26, 793–801 CrossRef CAS.
- E. Alissandrakis, P. A. Tarantilis, P. C. Harizanis and M. Polissiou, J. Sci. Food Agric., 2005, 85, 91–97 CrossRef CAS.
- L. Castro-Vázquez, M. S. Pérez-Coello and M. D. Cabezudo, Chromatographia, 2003, 57, 227–233 Search PubMed.
- L. C. Vázquez, M. C. Díaz-Maroto, E. Guchu and M. S. Pérez-Coello, Eur. Food Res. Technol., 2006, 224, 27–31 CrossRef PubMed.
- R. A. Pérez, C. Sánchez-Brunete, R. M. Calvo and J. L. Tadeo, J. Agric. Food Chem., 2002, 50, 2633–2637 CrossRef PubMed.
- E. Alissandrakis, P. A. Tarantilis, C. Pappas, P. C. Harizanis and M. Polissiou, Eur. Food Res. Technol., 2009, 229, 365–373 CrossRef CAS PubMed.
- E. Alissandrakis, D. Daferera, P. A. Tarantilis, M. Polissiou and P. C. Harizanis, Food Chem., 2003, 82, 575–582 CrossRef CAS.
- I. Jerković, J. Mastelić, Ž. Marijanović, Z. Klein and M. Jelić, Ultrason. Sonochem., 2007, 14, 750–756 CrossRef PubMed.
- I. Jerković and Z. Marijanović, Chem. Biodiversity, 2010, 7, 2309–2325 Search PubMed.
- I. Jerković, Z. Marijanović, C. I. G. Tuberoso, D. Bubalo and N. Kezić, Mol. Diversity, 2010, 14, 237–248 CrossRef PubMed.
- I. Jerković, P. M. Kuś, C. I. G. Tuberoso and M. Šarolić, Food Chem., 2014, 145, 8–14 CrossRef PubMed.
- S. T. Tan, A. L. Wilkins, P. C. Molan, P. T. Holland and M. A. Reid, J. Apic. Res., 1989, 28, 212–222 Search PubMed.
- R. Naef, A. Jaquier, A. Velluz and B. Bachofen, Chem. Biodiversity, 2004, 1, 1870–1879 CAS.
- C. I. G. Tuberoso, E. Bifulco, I. Jerković, P. Caboni, P. Cabras and I. Floris, J. Agric. Food Chem., 2009, 57, 3895–3900 CrossRef CAS PubMed.
- I. Jerković, Z. Marijanović, I. Ljubičić and M. Gugić, Chem. Biodiversity, 2010, 7, 1217–1230 Search PubMed.
- E. Alissandrakis, P. A. Tarantilis, P. C. Harizanis and M. Polissiou, Food Chem., 2007, 100, 396–404 CrossRef CAS PubMed.
- J. Bonvehí Serra and F. Ventura Coll, J. Sci. Food Agric., 2003, 83, 275–282 CrossRef.
- K. Beckmann, G. Beckh, C. Lüllmann and K. Speer, Dtsch. Lebensm.-Rundsch., 2007, 103, 154–158 CAS.
- C. Y. Rowland, A. J. Blackman, B. R. D’Arcy and G. B. Rintoul, J. Agric. Food Chem., 1995, 43, 753–763 CrossRef CAS.
- P. J. Williams, C. R. Strauss and B. Wilson, J. Agric. Food Chem., 1980, 28, 766–771 CrossRef CAS.
- H. Wintoch, A. Morales, C. Duque and P. Schreier, J. Agric. Food Chem., 1993, 41, 1311–1314 CrossRef CAS.
- G. Mignani, D. Morel, Y. Colleuille and C. Mercier, Tetrahedron Lett., 1986, 27, 2591–2594 CrossRef CAS.
- L. P. Christensen, J. Lam and T. Thomasen, Phytochemistry, 1990, 29, 3155–3156 CrossRef CAS.
- M. E. Maffei, J. Gertsch and G. Appendino, Nat. Prod. Rep., 2011, 28, 1359–1380 RSC.
- P. M. Dewick, Medicinal Natural Products: A Biosynthetic Approach, John Wiley & Sons, Ltd., Chichester, UK, 3rd edn., 2009 Search PubMed.
- T. W. Hallahan and R. Croteau, Arch. Biochem. Biophys., 1989, 269, 313–326 CrossRef CAS.
- I. Jerković, C. I. G. Tuberoso, Z. Marijanović, M. Jelić and A. Kasum, Food Chem., 2009, 112, 239–245 CrossRef PubMed.
- I. Jerković, M. Obradović, P. M. Kuś and M. Šarolić, Chem. Biodiversity, 2013, 10, 1549–1558 Search PubMed.
- M. Kreck, S. Püschel, M. Wüst and A. Mosandl, J. Agric. Food Chem., 2003, 51, 463–469 CrossRef CAS PubMed.
- E. Alissandrakis, E. Mantziaras, P. A. Tarantilis, P. C. Harizanis and M. Polissiou, Eur. Food Res. Technol., 2010, 231, 21–25 CrossRef CAS.
- P. Winterhalter, D. Katzenberger and P. Schreier, Phytochemistry, 1986, 25, 1347–1350 CrossRef CAS.
- R. A. Raguso and E. Pichersky, Plant Species Biology, 1999, 14, 95–120 CrossRef.
- I. Jerković, G. Hegić, Z. Marijanović and D. Bubalo, Molecules, 2010, 15, 2911–2924 CrossRef PubMed.
- P. M. Kuś, I. Jerković, C. I. G. Tuberoso and M. Šarolić, Chem. Biodiversity, 2013, 10, 1638–1652 Search PubMed.
- G. Buchbauer, L. Jirovetz, M. Wasicky and A. Nikiforov, J. Agric. Food Chem., 1993, 41, 116–118 CrossRef CAS.
- C. Guyot-Declerck, S. Renson, A. Bouseta and S. Collin, Food Chem., 2002, 79, 453–459 CrossRef CAS.
- B. Malika, J. Pharmacogn. Phytother., 2012, 4, 96–101 CrossRef.
- L. Castro-Vázquez, V. Leon-Ruiz, M. E. Alañon, M. S. Pérez-Coello and A. V. González-Porto, Food Control, 2014, 37, 362–370 CrossRef PubMed.
- S. Périno-Issartier, C. Ginies, G. Cravotto and F. Chemat, J. Chromatogr. A, 2013, 1305, 41–47 CrossRef PubMed.
- D. Herraiz-Peñalver, M. Á. Cases, F. Varela, P. Navarrete, R. Sánchez-Vioque and J. Usano-Alemany, Biochem. Syst. Ecol., 2013, 46, 59–68 CrossRef PubMed.
- V. Hajhashemi, A. Ghannadi and B. Sharif, J. Ethnopharmacol., 2003, 89, 67–71 CrossRef.
- L. Castro-Vázquez, M. C. Díaz-Maroto, M. A. González-Viñas and M. S. Pérez-Coello, Food Chem., 2009, 112, 1022–1030 CrossRef PubMed.
- I. Jerković and Z. Marijanović, Chem. Biodiversity, 2009, 6, 421–430 Search PubMed.
- J. Mastelić and D. Kuštrak, Acta Pharm., 1997, 47, 133–138 Search PubMed.
- R. Nurzyńska-Wierdak, Acta Agrobot., 2013, 66, 53–60 CrossRef.
- K. Msaada, M. Ben Taarit, K. Hosni, M. Hammami and B. Marzouk, Sci. Hortic., 2009, 122, 116–124 CrossRef CAS PubMed.
- L. Castro-Vázquez, M. C. Díaz-Maroto and M. S. Pérez-Coello, Food Chem., 2007, 103, 601–606 CrossRef PubMed.
- M. G. Miguel, S. Dandlen, A. C. Figueredo, J. G. Barroso, L. G. Pedro, A. Duarte and J. Faisca, Acta Hortic., 2008, 773, 89–94 CAS.
- E. Sarrou, P. Chatzopoulou, K. Dimassi-Theriou and I. Therios, Molecules, 2013, 18, 10639–10647 CrossRef CAS PubMed.
- I. Jerković, J. Mastelić and Z. Marijanović, Chem. Biodiversity, 2006, 3, 1307–1316 Search PubMed.
- M. Maksimović, D. Vidić, M. Miloš, M. Edita Šolić, S. Abadžić and S. Siljak-Yakovlev, Biochem. Syst. Ecol., 2007, 35, 473–478 CrossRef PubMed.
- I. Jerković and J. Mastelić, Croat. Chem. Acta, 2001, 74, 431–439 Search PubMed.
- B. Teixeira, A. Marques, C. Ramos, I. Batista, C. Serrano, O. Matos, N. R. Neng, J. M. F. Nogueira, J. A. Saraiva and M. L. Nunes, Ind. Crops Prod., 2012, 36, 81–87 CrossRef CAS PubMed.
- G. De Mastro, C. Ruta, A. Mincione and M. Poiana, Acta Hortic., 2004, 629, 471–482 CAS.
- K. A. Krock and C. L. Wilkins, Trends Anal. Chem., 1994, 13, 13–17 CrossRef CAS.
- A. Pažitná, A. Janáčová and I. Špánik, J. Food Nutr. Res., 2012, 51, 236–241 Search PubMed.
- A. Verzera, G. Tripodi, C. Condurso, G. Dima and A. Marra, Food Control, 2014, 39, 237–243 CrossRef CAS PubMed.
- F. Luan, A. Mosandl, M. Gubesch and M. Wüst, J. Chromatogr. A, 2006, 1112, 369–374 CrossRef CAS PubMed.
- H. K. Lichtenthaler, Annu. Rev. Plant Physiol. Plant Mol. Biol., 1999, 50, 47–65 CrossRef CAS PubMed.
- H. K. Lichtenthaler, J. Schwender, A. Disch and M. Rohmer, FEBS Lett., 1997, 400, 271–274 CrossRef CAS.
- P. Winterhalter and R. Rouseff, in Carotenoid-Derived Aroma Compounds, eds. P. Winterhalter and R. Rouseff, ACS Symposium Series, American Chemical Society, Washington, DC, 2001, pp. 1–17 Search PubMed.
- S. T. Tan, A. L. Wilkins, J. P. T. Holland and T. K. McGhie, J. Agric. Food Chem., 1990, 38, 1833–1838 CrossRef CAS.
- C. I. G. Tuberoso, I. Jerković, E. Bifulco and Z. Marijanović, Chem. Biodiversity, 2011, 8, 872–879 CAS.
- I. Jerković, Z. Marijanović and C. Tuberoso, Planta Med., 2009, 75, PB32 CrossRef PubMed.
- F. Ferreres, P. Andrade and F. A. Tomás-Barberán, J. Agric. Food Chem., 1996, 44, 2053–2056 CrossRef CAS.
- L. Yao, N. Datta, F. A. Tomás-Barberán, F. Ferreres, I. Martos and R. Singanusong, Food Chem., 2003, 81, 159–168 CrossRef CAS.
- L. Yao, Y. Jiang, R. Singanusong, N. Datta and K. Raymont, Food Chem., 2004, 86, 169–177 CrossRef CAS PubMed.
- L. Yao, Y. Jiang, R. Singanusong, N. Datta and K. Raymont, Food Res. Int., 2005, 38, 651–658 CrossRef PubMed.
- S. T. Tan, A. L. Wilkins, P. T. Holland and T. K. McGhie, J. Agric. Food Chem., 1989, 37, 1217–1221 CrossRef CAS.
- C. Guyot, V. Scheirman and S. Collin, Food Chem., 1999, 64, 3–11 CrossRef CAS.
- M. Häusler and A. Montag, Z. Lebensm.-Unters. Forsch., 1989, 189, 113–115 CrossRef.
- I. Jerković, Z. Marijanović and M. Malenica-Staver, Molecules, 2011, 16, 2507–2518 CrossRef PubMed.
- M. Häusler and A. Montag, Dtsch. Lebensm.-Rundsch., 1991, 87, 35–36 Search PubMed.
- S. J. Broom, Structure and stereochemistry of some degraded carotenoids from honey, Ph.D. Thesis, The University of Waikato, Hamilton, New Zealand, 1998 Search PubMed.
- S. J. Broom, A. L. Wilkins, Y. Lu and R. M. Ede, J. Org. Chem., 1994, 59, 6425–6430 CAS.
- R. M. Ede, A. L. Wilkins, Y. Lu and S. T. Tan, Tetrahedron Lett., 1993, 34, 6795–6798 CrossRef CAS.
- H.-D. Belitz, W. Grosch and P. Schieberle, Food Chemistry, Springer-Verlag, Berlin, 4th edn, 2009 Search PubMed.
- E. Alissandrakis, P. A. Tarantilis, C. Pappas, P. C. Harizanis and M. Polissiou, LWT–Food Sci. Technol., 2011, 44, 1042–1051 CrossRef CAS PubMed.
- B. R. D'Arcy, G. B. Rintoul, C. Y. Rowland and A. J. Blackman, J. Agric. Food Chem., 1997, 45, 1834–1843 CrossRef.
- P. M. Kuś, Z. Marijanović and I. Jerković, Chem. Nat. Compd., 2013, 49, 961–964 CrossRef.
- M. Herderich and P. Winterhalter, J. Agric. Food Chem., 1991, 39, 1270–1274 CrossRef CAS.
- F. Bianchi, M. Careri and M. Musci, Food Chem., 2005, 89, 527–532 CrossRef CAS PubMed.
- G. Montenegro, M. Gómez, G. Casaubon, A. Belancic, A. M. Mujica and R. C. Peña, Φyton, 2009, 78, 61–65 Search PubMed.
- J. Mastelić, I. Jerković and M. Mesić, Flavour Fragrance J., 2006, 21, 306–313 CrossRef.
- P. M. Kuś, I. Jerković, C. I. G. Tuberoso, Z. Marijanović and M. Šarolić, Nat. Prod. Commun., 2013, 8, 651–654 Search PubMed.
- A. C. Soria, I. Martínez-Castro and J. Sanz, Food Res. Int., 2008, 41, 838–848 CrossRef CAS PubMed.
- F. Bianchi, A. Mangia, M. Mattarozzi and M. Musci, Food Chem., 2011, 129, 1030–1036 CrossRef CAS PubMed.
- H. Z. Senyuva, J. Gilbert, S. Silici, A. Charlton, C. Dal, N. Gürel and D. Cimen, J. Agric. Food Chem., 2009, 57, 3911–3919 CrossRef CAS PubMed.
- M. Shimoda, Y. Wu and Y. Osajima, J. Agric. Food Chem., 1996, 44, 3913–3918 CrossRef CAS.
- R. F. A. Moreira, L. C. Trugo, M. Pietroluongo and C. A. B. De Maria, J. Agric. Food Chem., 2002, 50, 7616–7621 CrossRef CAS PubMed.
- A. L. Wilkins, Y. Lu and S. T. Tan, J. Agric. Food Chem., 1993, 41, 873–878 CrossRef CAS.
- I. Jerković, Z. Marijanović, J. Kezić and M. Gugić, Molecules, 2009, 14, 2717–2728 CrossRef PubMed.
- I. Jerković and Z. Marijanović, Molecules, 2010, 15, 3744–3756 CrossRef PubMed.
- E. Schievano, E. Morelato, C. Facchin and S. Mammi, J. Agric. Food Chem., 2013, 61, 1747–1755 CrossRef CAS PubMed.
- M. Amtmann, J. Food Compos. Anal., 2010, 23, 122–129 CrossRef CAS PubMed.
- C. Guyot, A. Bouseta, V. Scheirman and S. Collin, J. Agric. Food Chem., 1998, 46, 625–633 CrossRef CAS PubMed.
- B. S. Radovic, M. Careri, A. Mangia, M. Musci, M. Gerboles and E. Anklam, Food Chem., 2001, 72, 511–520 CrossRef CAS.
- L. Ceballos, J. A. Pino, C. E. Quijano-Celis and A. Dago, J. Food Qual., 2010, 33, 507–528 CrossRef CAS PubMed.
- E. de la Fuente, M. L. Sanz, I. Martinez Castro, J. Sanz and A. I. Ruiz Matute, Food Chem., 2007, 105, 84–93 CrossRef CAS PubMed.
- S. Silici, Turk. J. Biol., 2011, 35, 641–645 CAS.
- E. Alissandrakis, A. C. Kibaris, P. A. Tarantilis, P. C. Harizanis and M. Polissiou, J. Sci. Food Agric., 2005, 85, 1444–1452 CrossRef CAS.
- C. Tananaki, A. Thrasyvoulou, J. Giraudel and M. Montury, Food Chem., 2007, 101, 1687–1693 CrossRef CAS PubMed.
- E. de la Fuente, I. Martínez-Castro and J. Sanz, J. Sep. Sci., 2005, 28, 1093–1100 CrossRef CAS.
- P. M. Kuś, I. Jerković, C. I. G. Tuberoso, Z. Marijanović and F. Congiu, Food Chem., 2014, 142, 12–18 CrossRef PubMed.
- I. Jasicka-Misiak, A. Poliwoda, M. Dereń and P. Kafarski, Food Chem., 2012, 131, 1149–1156 CrossRef CAS PubMed.
- C. I. G. Tuberoso, E. Bifulco, P. Caboni, F. Cottiglia, P. Cabras and I. Floris, J. Agric. Food Chem., 2010, 58, 384–389 CrossRef CAS PubMed.
- S. Oelschlaegel, L. Pieper, R. Staufenbiel, M. Gruner, L. Zeippert, B. Pieper, I. Koelling-Speer and K. Speer, J. Agric. Food Chem., 2012, 60, 11811–11820 CrossRef CAS PubMed.
- A. C. Soria, J. Sanz and I. Martínez-Castro, Eur. Food Res. Technol., 2009, 228, 579–590 CrossRef CAS.
- Q. Zhou, C. L. Wintersteen and K. R. Cadwallader, J. Agric. Food Chem., 2002, 50, 2016–2021 CrossRef CAS PubMed.
- I. Jerković, C. I. G. Tuberoso, M. Gugić and D. Bubalo, Molecules, 2010, 15, 6375–6385 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2014 |