Synthesis of immobilized heteropolyanion-based ionic liquids on mesoporous silica SBA-15 as a heterogeneous catalyst for alkylation
Xiaoli Sheng,
Yuming Zhou*,
Yongle Yang,
Yiwei Zhang,
Zewu Zhang,
Shijian Zhou,
Xiaoqin Fu and
Shuo Zhao
School of Chemistry and Chemical Engineering, Southeast University, Nanjing 211189, P.R.China. E-mail: ymzhou@seu.edu.cn; Fax: +86-25-52090617; Tel: +86-25-52090617
First published on 20th June 2014
Abstract
Phosphotungstic acid (H3PW12O40) has been successfully loaded onto sulfonate-functionalized ionic liquid-modified mesoporous silica SBA-15 by total anion-exchange. The immobilized catalysts were characterized by XRD, N2 adsorption, TEM, and FTIR spectroscopy. Characterization results show that the mesopore structure of SBA-15 was maintained well even after surface modification and the subsequent anion-exchange step of [PW12O40]3− (PW). In comparison with the task-specific basic ionic liquid (1-(propyl-3-sulfonate) 3-methyl-imidazolium phosphotungstate), the obtained catalyst showed much higher efficiency in alkylation of o-xylene with styrene. More importantly, such an immobilized task-specific basic ionic liquid could be reused without significant loss of catalytic activity even after recycling six times.
1. Introduction
The alkylation of o-xylene with styrene to afford 1-phenyl-l-arylethane is an industrially important reaction.1 Phenylxylylethane (PXE) is a colorless synthetic liquid with many excellent properties suitable for various applications, e.g., as a solvent for pressure-sensitive record materials, a plasticizer, a heating medium, an electric-insulating oil and a high-boiling solvent.1 Nevertheless, one of the main problems in this reaction is the formation of styrene oligomers generated as byproducts, especially when homogeneous Brønsted or Lewis acid catalysts (such as H2SO4, BF3, HF, AlCl3 or FeCl3) are used.1 The aforementioned traditional acids are generally related to problems of high corrosiveness, poor shape-selectivity, low hydrothermal stability and difficulties in waste acid treatment. The urgent demand for environmentally friendly catalytic technology urges development of heterogeneous catalysts because of their advantages such as high activity and selectivity, ease of separation and reusability. For PXE synthesis, a monumental series of solid acid catalysts such as silica-alumina,2 cation-exchange resins,3 sulfated zirconia/titania,4,5 heteropolyacid salts6 and modified mesoporous silicas7,8 are adopted as catalysts, all of which have promoted the catalytic activity. However, these catalysts have disadvantages as well, for example, the compatibility of reusability, operation loss, and high mass transfer resistance, which limit their practical application in alkylation.Among the recent developments in catalysis, ionic liquids (ILs) have attracted much attention as being environmentally friendly reaction media for their unique properties of high thermal stability, negligible vapor pressure, tunable acidity and selective dissolvability.9,10 Though ionic liquids possessed such promising advantages, their widespread practical application was still hampered by several drawbacks such as unendurable viscosity, high cost, a tedious purification procedure of the product and requirements of long reaction times.11 Therefore, in order to overcome the above shortcomings, an immobilized ionic liquid catalyst combining the advantageous characteristics of ionic liquids, inorganic acids and solid acids had been proposed.12,13
Indeed, rapidly increased reports have become available describing the use of the ILs immobilized on various supports in the Friedel–Crafts reactions.14 Holderich et al.15 developed a process for the alkylation of benzene, toluene, naphthalene, and phenol with dodecene in the presence of immobilized 1-butyl-3-methyl-imidazolium chloride with AlCl3 (Al-IIL). The immobilized IL played an important role in significant reduction of the overall cost of the process. A comparison of different supports showed that carriers based on SiO2 and Al2O3 retained higher amounts of chloroaluminate IL than ZrO2 and TiO2 carriers after the extraction, and only the silica-based supports were active in the alkylation reactions. In 2009, Yin and co-workers16 reported the preparation and catalytic activity of novel periodic mesoporous organosilica (PMO) materials incorporating Lewis acidic chloroindate (III) ionic liquid moieties. It was observed that the immobilization of the Lewis acidic ionic liquids with increasing amount of InCl3 led to a drastic increase in the catalytic activity ranging from 14 to 100 conversions with a constant selectivity of 100%.
It is well known that Lewis acidic chloroindate (III) ionic liquid moieties are limited to anions with a high affinity to oxygen. Therefore, Brønsted acidic ILs used in some acid-catalyzed processes have aroused considerable interest since they show higher stability towards air and water.17 With these points in mind, we designed new heteropolyanion-based ILs containing the sulfonic acid functional group as “task-specific” catalysts immobilized on mesoporous silica SBA-15. The immobilized catalysts were fully characterized by XRD, N2 adsorption, TEM, and FTIR spectroscopy. Using the Friedel–Crafts alkylation as a test reaction, the immobilized TSILs showed high efficiency and stability.
2. Experimental
2.1 Catalyst preparation
Fig. 1 shows the schematic procedures for the synthesis of immobilized HPW-PMIMPS-SBA-15 materials. N-Triethoxysilylpropylimidazole (compound 1) was synthesized by refluxing 53.00 mmol of imidazole with 50.00 mmol of 3-triethoxysilylpropylchloride in toluene (50 mL) at 70 °C for 24 h under a nitrogen atmosphere. 1,3-Propane sulfone (10.50 mmol) was added to 2.72 g of the compound 1, followed by stirring at 50 °C for 8 h under a nitrogen atmosphere. A white precipitate was filtered, washed with diethyl ether three times, and then dried under vacuum. The obtained sample was denoted as 1-(propyl-3-sulfonate)-3-triethoxysilylpropyl-imidazolium (compound 2). 1.00 g of dry SBA-15 prepared according to a method in the literature,18 and 0.40 g of the compound 2 were dispersed in 50 mL of anhydrous toluene, and then refluxed for 24 h under nitrogen protection. After being filtrated and washed with anhydrous ethanol, the obtained precipitate was Soxhlet extracted over ethanol for 48 h. The product was filtrated, washed with anhydrous ethanol, and dried at room temperature under vacuum overnight. The immobilized basic sulfur-containing TSIL is designated as PMIMPS-SBA-15 (compound 3). 1.00 g of the obtained compound 3 was dispersed in 100 mL of deionized water, followed by addition of various amount of 12-tungstophosphoric acid (HPW) according to different weight ratios of HPW to the compound 3 (n, n = 0.10, 0.20, 0.30, or 0.40), and then the resultant suspension was stirred at room temperature for 12 h. The resultant precipitate was filtered, washed with deionized water several times, dried overnight under vacuum, and Soxhlet extracted over ethanol for 12 h. The obtained sample was denoted as xHPW-PMIMPS-SBA-15 (compound 4), where x stands for the weight ratio of HPW to the compound 3.For comparison, the task-specific basic ionic liquid 1-(propyl-3-sulfonate) 3-methyl-imidazolium phosphotungstate ([MIMPS]3PW12O40) was synthesized by the following procedure.19 Methylimidazole (0.11 mol) and 1,3-propane sulfone (0.10 mol) were dissolved in toluene (20 mL) and stirred for 24 h at 50 °C under a nitrogen atmosphere. A white precipitate (MIMPS) was filtered, washed with diethyl ether three times, and then dried under vacuum. MIMPS (0.06 mol) was added to an aqueous solution of H3PW12O40 (0.02 mol), and then the mixture was stirred at room temperature for 24 h. Water was removed under vacuum to give the product as a solid.
2.2 Catalyst characterization
Powder X-ray diffraction (XRD) patterns were obtained on a Rigaku D/max-rC Siemens diffractometer using nickel-filtered Cu Kα as monochromatic X-ray radiation. The scattering intensities were measured over an angle range of 0.58° < 2θ < 40° with a step size D(2θ) = 0.028° and a step time of 8 s. The nitrogen adsorption and desorption isotherms were measured at −196 °C on an ASAP 2020 system. Prior to the measurements, all samples were degassed under vacuum at 80 °C for 4 h. The specific surface area, ABET, was determined from the linear part of the BET equation (P/P0 = 0.05–0.30). The pore size distribution was derived from the adsorption branch of the N2 isotherm using the Barrett–Joyner–Halenda (BJH) method. The total pore volume was estimated from the amount of nitrogen adsorbed at a relative pressure (P/P0) of ca. 0.995. Pore structures of the samples were examined by TEM (Jeol, JEM-2000EXII). Infrared spectra were recorded on a Bruker Tensor-27 FTIR spectrometer. The spectra were acquired by accumulating 32 scans at 4 cm−1 resolution in the range of 400–4000 cm−1. The sample was ground with KBr and pressed into a thin wafer.2.3 Catalytic tests
The alkylation reactions were carried out in a continuously stirred batch reactor under reflux conditions using a three-necked 100 mL round-bottomed flask equipped with a condenser. Preliminary runs were conducted with 7.50 g (0.0721 mol) of styrene, 57.35 g (0.5402 mol) of o-xylene (mole ratio of o-xylene to styrene, 7.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and 1.50 g of catalyst (20% w/w of styrene) at 120 °C for 180 minutes. The required amount of o-xylene was initially added to the reactor at the reaction temperature, followed by the desired amount of the catalyst, and a known amount of styrene was then added to the reaction mixture at the same temperature. After the reaction, unreacted o-xylene was distilled out under atmospheric pressure, and then the collected part was called a crude product. The crude product was analyzed using a GC-9890A gas chromatograph equipped with a OV-1 capillary column and a flame ionization detector (FID). The yield of PXE was defined as follows:
1) and 1.50 g of catalyst (20% w/w of styrene) at 120 °C for 180 minutes. The required amount of o-xylene was initially added to the reactor at the reaction temperature, followed by the desired amount of the catalyst, and a known amount of styrene was then added to the reaction mixture at the same temperature. After the reaction, unreacted o-xylene was distilled out under atmospheric pressure, and then the collected part was called a crude product. The crude product was analyzed using a GC-9890A gas chromatograph equipped with a OV-1 capillary column and a flame ionization detector (FID). The yield of PXE was defined as follows:3. Results and discussion
The low-angle XRD patterns of the siliceous SBA-15, PMIMPS-SBA-15 and 30% HPW-PMIMPS-SBA-15, as well as the high-angle XRD patterns of the Keggin HPW, 30% HPW-PMIMPS-SBA-15 and 40% HPW-PMIMPS-SBA-15, are shown in Fig. 2. The siliceous SBA-15 showed a pattern with a very strong reflection at 2θ = 0.86° for d100 and two other weaker reflections at 2θ = 1.46° and 2θ = 1.68° for d110 and d200, respectively, associated with the quasi-regular arrangement of mesopores with a hexagonal symmetry [17]. In contrast, three reflections of d100, d110, and d200 could clearly be observed for PMIMPS-SBA-15 and 30% HPW-PMIMPS-SBA-15, although the intensities of d100, d110, and d200 reflections decreased slightly and the three diffraction peaks shifted to a higher angle in comparison with the siliceous SBA-15. This can be due to the decreased local order, and/or the reduction of scattering contrast induced by the incorporated organic moieties and PW anions.20 Moreover, the high-angle XRD pattern of the 30% HPW-PMIMPS-SBA-15 does not show the distinct diffraction peaks corresponding to the crystalline HPW phase (Fig. 2B-e versus B-d), which suggests the high dispersion of PW in the channels of the 30% HPW-PMIMPS-SBA-15.21 However, at higher HPW loading (40% HPW-PMIMPS-SBA-15), the high-angle XRD pattern showed some distinct diffraction peaks corresponding to the crystalline HPW phase (Fig. 2B-f), demonstrating the agglomeration of the excessive HPW in the mesopore channels of PMIMPS-SBA-15.N2 adsorption–desorption isotherms record the textural properties of PMIMPS-SBA-15 and xHPW-PMIMPS-SBA-15, as shown in Fig. 3. As can be seen, all samples show typical type-IV isotherms with clear adsorption–desorption hysteresis loops at a relative pressure of 0.65 < P/P0 < 0.85 (Fig. 3 left), characteristic of a mesoporous material with uniform cylindrical channel and narrow pore size distribution [17]. The shape of the isotherms varies with the ionic liquid moieties and PW anions. On the other hand, the BJH pore size distribution (calculated from the analysis of the desorption branch of the isotherms) is presented in Fig. 3 (right). It can be seen that all samples exhibit a fairly uniform pore size distribution (5–7 nm). Effects of the ionic liquid moieties and PW anion loadings on the corresponding surface area, pore diameter and pore volume of HPW-PMIMPS-SBA-15 catalysts are presented in Table 1. As expected, HPW-PMIMPS-SBA-15 present decreased pore volume compared to the parent PMIMPS-SBA-15. Increased PW anion loading leads to a gradual decrease in the pore volume. Especially, some pore blockage occurred when the PW anion content increased over 40%, which suggests that the PW anions are located inside the pore indeed. These details are also reflected in the surface area and pore size of HPW-PMIMPS-SBA-15 catalysts. However, the surface area of the samples up to 30% HPW loaded was more than 300 m2 g−1, and the pore volume was in the range of 0.61–0.70 cm3 g−1 (Table 1), which is sufficient for catalytic performance.
 | ||
| Fig. 3 Nitrogen adsorption–desorption isotherms (left) and pore size distribution (right) of the samples PMIMPS-SBA-15 and xHPW-PMIMPS-SBA-15 (x = 10%, 20%, 30% and 40%). | ||
| Sample | HPW (wt%) | Vtotal (cm3 g−1) | SBETa (m2 g−1) | Pore sizeb (nm) |
|---|---|---|---|---|
| a BET method.b BJH model applied to the desorption branch of the isotherms. | ||||
| PMIMPS-SBA-15 | — | 0.85 | 618 | 5.90 |
| 10% HPW-PMIMPS-SBA-15 | 10 | 0.70 | 446 | 5.60 |
| 20% HPW-PMIMPS-SBA-15 | 20 | 0.66 | 376 | 5.50 |
| 30% HPW-PMIMPS-SBA-15 | 30 | 0.61 | 363 | 5.48 |
| 40% HPW-PMIMPS-SBA-15 | 40 | 0.58 | 330 | 5.42 |
TEM micrographs of the representative sample PMIMPS-SBA-15 and 30% HPW-PMIMPS-SBA-15 (Fig. 4) provide a direct visualization of well-ordered hexagonal arrays of 2D mesoporous channels, particularly in the direction perpendicular to the pore axis (Fig. 4a and c) or along the direction of the pore axis (Fig. 4b and d). The TEM images confirm the intact mesostructures of all the samples, although the PW and ionic liquid moieties were loaded in the channel of SBA-15.
 | ||
| Fig. 4 Typical TEM images of PMIMPS-SBA-15 (a and b) and 30% HPW-PMIMPS-SBA-15 (c and d). The bars in the photos are 20 nm. | ||
Fig. 5 compares the FT-IR spectrum of 30% HPW-PMIMPS-SBA-15 with those of PMIMPS-SBA-15 and neat Keggin-type HPW. In the cases of the samples incorporated with the terminal HPW-based ionic complexes, 30% HPW-PMIMPS-SBA-15 represented an almost identical FT-IR spectrum to that of PMIMPS-SBA-15 (Fig. 5b versus 5c). More specifically, both the samples showed vibration bands at 3160, 3118 cm−1 (+), 1556, 1451, 1170 cm−1 (#), and 1242, 738 cm−1 (*), which can be assigned to the aromatic C–H stretching in imidazolium cations, the feature vibrations of imidazolium moieties, and the C–Si stretching vibrations, respectively.22 Such observations suggest that the grafting process presented here does not change the structure of the ionic liquid. As shown in Fig. 5, four characteristic bands at around 1080, 980, 890 and 800 cm−1 (•) appear distinctively in the FT-IR spectrum of the Keggin-type HPW (Fig. 5a), which are assigned to asymmetric stretching vibration of P–Oa, W = Od, W–Ob–W in corner-shared octahedral and W–Oc–W in edge-shared octahedral, respectively.23 The four bands also appear in the FT-IR spectrum of the supported 30% HPW-PMIMPS-SBA-15, which indicates that the Keggin structure of PW anions is well reserved (Fig. 5c vs. Fig. 5a). Therefore, when combined, Fig. 4 and 5 can be indicative of covalent attachment of HPW and the linker with the SBA-15 surface.
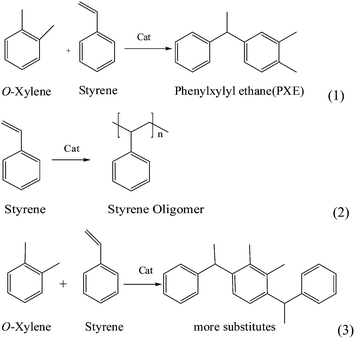
The catalytic activity of different catalysts is showed in Table 2. The detailed reaction scheme is shown in Scheme 1; reaction (1) is the PXE formation reaction, whereas reaction (2) and (3) represent the formation of styrene oligomers and more substitutes, respectively.
| Catalyst | Styrene conversion (%) | PXE yieldc (%) | PXE selectivityd (%) |
|---|---|---|---|
a Reaction conditions: o-xylene:styrene = 7.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, reaction temperature = 120 °C, reaction time = 3.0 h, catalyst loading = 20% (w/w of styrene).b Homogeneous catalyst, 0.30 g.c Isolated yield based on the amount of styrene.d Ratio of the para-to-ortho product. 1, reaction temperature = 120 °C, reaction time = 3.0 h, catalyst loading = 20% (w/w of styrene).b Homogeneous catalyst, 0.30 g.c Isolated yield based on the amount of styrene.d Ratio of the para-to-ortho product. |
|||
| SBA-15 | — | — | — |
| HPWb | 100 | 97.9 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 10% HPW-PMIMPS-SBA-15 | 74.8 | 71.8 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 20% HPW-PMIMPS-SBA-15 | 83.9 | 80.5 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 30% HPW-PMIMPS-SBA-15 | 97.8 | 93.9 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 40% HPW-PMIMPS-SBA-15 | 95.0 | 91.2 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| [MIMPS]3PW12O4 | 94.5 | 90.8 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
As shown in Table 2, the homogeneous HPW shows very high catalytic performances for the reaction, however, it is difficult to separate the HPW from the product mixture. Moreover, the SBA-15 support itself shows no activity performances. Furthermore, 30% HPW-PMIMPS-SBA-15 sample exhibits the highest product yield and good selectivity, indicating that an appropriate concentration of PW anions is advantageous to the reaction. In this case, a significant difference is found among these catalysts as a function of loading the PW anions. The PXE yield decreases in the following order: 30% HPW-PMIMPS-SBA-15 (93.9%) > 40% HPW-PMIMPS-SBA-15 (91.2%) > 20% HPW-PMIMPS-SBA-15 (80.5%) > 10% HPW-PMIMPS-SBA-15 (71.8%). In the case of 40% HPW-PMIMPS-SBA-15, the product yield is found to be less compared to that of the 30% HPW-PMIMPS-SBA-15 catalyst, which is probably due to the blocking of active sites by the dense Keggin structure, which thereby changes the morphology of the support (mesopore diameter and volume) and leads to diffusional constraints. In addition, it is worth noting that the [MIMPS]3PW12O4 catalyst shows good catalytic performances, because it has sufficient PW anion content.
Stability and reusability of the catalyst are very important for any catalytic system. The catalytic reusability of the 30% HPW-PMIMPS-SBA-15 and [MIMPS]3PW12O4 catalysts was evaluated by carrying out the reaction with a used catalyst under the optimized conditions. After completion of the reaction, the catalyst was easily recovered from the reaction mixture by centrifugation, and then used in the subsequent catalytic runs without further activation. The data obtained are shown in Fig. 6. Interestingly, the activity decreased slightly after reuse of the 30% HPW-PMIMPS-SBA-15 catalyst six times. Indeed, the yield of the PXE product was as high as 89.5% even after the 30% HPW-PMIMPS-SBA-15 catalyst was recycled six times. In contrast, the yield of the PXE product significantly decreased from 90.8% to 30.3% as a pure ionic liquid of [MIMPS]3PW12O4 was recycled six times under the same reaction conditions. These results indicate that the immobilized heteropolyanion-based ionic liquid presented here possesses much higher catalytic efficiency in comparison with the corresponding pure ionic liquid. Therefore, an immobilized ionic liquid catalyst on porous carriers used as heterogeneous catalysts can overcome the mass transfer problems in practical application. Indeed, through immobilized ionic liquid catalyst on various supports, we can obtain larger specific surface area and more stability. Moreover, after reaction, it also allows easier separation and better reusability.
4. Conclusions
In summary, the work herein presented fabrication of HPW-based ionic liquid immobilized on mesoporous silica SBA-15 by complete anion exchange. The resultant catalysts showed better catalytic efficiency and reusability than the corresponding task-specific basic ionic liquid in the alkylation of o-xylene with styrene. It was found that the addition of HPW during the synthesis of catalysts played a crucial role in PW anion loading and successive catalytic performance. Moreover, the use of such a heterogeneous catalyst can decrease the consumption of ionic liquid remarkably. As such, the synthesis procedure can be simplified significantly, which may open an opportunity for wide applications of immobilized heteropolyanion-based ionic liquid in catalysis.Acknowledgements
The authors are grateful for the financial supports from National Natural Science Foundation of China (Grant no. 21306023, 21376051, 21106017), Natural Science Foundation of Jiangsu Province of China (Grant no. BK20131288), Fund Project for Transformation of Scientific and Technological Achievements of Jiangsu Province of China (Grant no. BA2011086), Key Program for the Scientific Research Guiding Found of Basic Scientific Research Operation Expenditure of Southeast University (Grant no. 3207043101) and Instrumental Analysis Fund of Southeast University.References
- A. B. Dixit and G. D. Yadav, React. Funct. Polym., 1996, 31, 251–263 CrossRef CAS.
- B. S. Kwak and T. J. Kim, Catal. Lett., 1999, 59, 55–60 CrossRef CAS.
- A. Sato, I. Shimizu and G. Mesalito, US Pat., 4289918A[P], 1981.
- J. H. Lunsford, H. Sang, S. M. Campbell, C. H. Liang and R. G. Anthony, Catal. Lett., 1994, 27, 305–314 CrossRef CAS.
- Y. P. Wang, R. J. Qu and C. H. Wang, Appl. Chem. Ind., 2009, 36, 822–826 Search PubMed.
- F. E. Celik, H. Lawrence and A. T. Bell, J. Mol. Catal. A: Chem., 2008, 288, 87–96 CrossRef CAS PubMed.
- X. L. Sheng, Y. M. Zhou, Y. W. Zhang, M. W. Xue and Y. Z. Duan, Chem. Eng. J., 2012, 179, 295–301 CrossRef CAS PubMed.
- X. L. Sheng, Y. M. Zhou, Y. W. Zhang, Y. Z. Duan and M. W. Xue, Catal. Lett., 2012, 142, 360–367 CrossRef CAS.
- D. R. MacFarlane, J. M. Pringle, K. M. Johansson, S. A. Forsyth and M. Forsyth, Chem. Commun., 2006, 1905–1917 RSC.
- H. Li, P. S. Bhadury, B. A. Song and S. Yang, RSC Adv., 2012, 2, 12525–12551 RSC.
- J. M. Miao, H. Wan and G. F. Guan, Catal. Commun., 2011, 12, 353–356 CrossRef CAS PubMed.
- F. J. Liu, S. F. Zuo, W. P. Kong and C. Z. Qi, Green Chem., 2012, 14, 1342–1349 RSC.
- H. H. Zhao, H. L. Song and L. J. Chou, Microporous Mesoporous Mater., 2013, 181, 182–191 CrossRef CAS PubMed.
- H. H. Zhao, N. Y. Yu, J. Q. Wang, D. Y. Zhuang, Y. Ding, R. Tan and D. H. Yin, Microporous Mesoporous Mater., 2009, 122, 240–246 CrossRef CAS PubMed.
- C. DeCastro, E. Sauvage, M. H. Valkenberg and W. F. Holderich, J. Catal., 2000, 196, 86–94 CrossRef CAS.
- G. J. Wang, N. Y. Yu, L. Peng, R. Tan, H. H. Zhao, D. H. Yin, H. Y. Qiu, Z. H. Fu and D. L. Yin, Catal. Lett., 2008, 123, 252–258 CrossRef CAS.
- J. M. Miao, H. Wan, Y. B. Shao, G. F. Guan and B. Xu, J. Mol. Catal. A: Chem., 2011, 348, 77–82 CrossRef CAS PubMed.
- D. Y. Zhao, J. L. Feng, Q. S. Huo, N. Melosh, G. H. Fredrickson, B. F. Chmelka and G. D. Stucky, Science, 1998, 279, 548–552 CrossRef CAS.
- Y. Leng, J. Wang, D. R. Zhu, X. Q. Ren, H. Q. Ge and L. Shen, Angew. Chem., Int. Ed., 2009, 48, 168–171 CrossRef CAS PubMed.
- S. Jana, B. Dutta, R. Bera and S. Koner, Langmuir, 2007, 23, 2492–2496 CrossRef CAS PubMed.
- G. Karthikeyan and A. Pandurangan, J. Mol. Catal. A: Chem., 2009, 311, 36–45 CrossRef CAS PubMed.
- A. Bordoloi, S. Sahoo, F. Lefebvre and S. B. Halligudi, J. Catal., 2008, 259, 232–239 CrossRef CAS PubMed.
- T. Nakashima and N. Kimizuka, J. Am. Chem. Soc., 2003, 125, 6386–6387 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |