Post-isomorphic substitution of trivalent metal cations for Ca2+ in portlandite crystals†
Jun Hyung Kima,
Man Park*a,
Ahmad Imrana,
Min-Cheol Choia,
Kwang Seop Kima and
Sridhar Komarnenib
aSoil Science Laboratory, School of Applied biosciences, Kyungpook National University, 702-701, Deagu, South Korea. E-mail: manpark@knu.ac.kr; Fax: +82 53953 7233
bMaterials Research Laboratory, Pennsylvania State University, State College, PA 16802, USA. E-mail: komarneni@psu.edu
First published on 20th June 2014
Abstract
Isomorphic substitution, position replacement of one cation by another of similar size, leads to incorporation of a variety of cations into solid crystals without any significant changes to the primary crystal structures. To date, isomorphic substitution has been known to take place almost exclusively via co-crystallization of the cations during formation of the crystals. We report here the discovery of isomorphic substitution of trivalent metal cations for Ca2+ ions in portlandite crystals at room temperature as evidenced by the transient appearance of metastable phase, the formation of Ca-based layered double hydroxides at high pH, the distinct shift of suspension pH after phase transition, and the in situ topochemical reaction. This post-crystallization isomorphic substitution provides an innovative pathway for the synthesis of materials through chemical manipulation of crystals as well as a new insight into interpretation on their weathering and transformation processes.
Introduction
Isomorphic substitution leads to incorporation of a variety of cations into solid crystals without any significant changes to the primary crystal structures, developing diverse chemical functions such as ion exchange, catalysis, electrochemical potential, photosensitivity etc.1−4 To date, isomorphic substitution has been known to take place almost exclusively via co-crystallization of the cations during formation of the crystalline structures.4−8 Once crystallized, structural cations are believed to be very difficult to replace mainly due to strong electrostatic repulsion and high lattice energy as long as the primary structures remain intact. Nevertheless, there have been incessant attempts to elucidate the possibility of post-crystallization isomorphic substitution (hereinafter referred to as ‘post-isomorphic substitution’)8–10 because it can open a new pathway for materials synthesis as well as for understanding the intriguing chemical processes involved in the formation and transformation of inorganic minerals.Substantial degree of isomorphic substitution is observed especially in porous minerals such as zeolites,4,9 swelling phyllosilicates,1,9 layered double hydroxides (LDHs)4,6,7 etc. In general, both layered and network structures of silicates are mainly co-crystallized with tetrahedra of various cations through isomorphic substitution. Similarly, octahedral sheets of phyllosilicates are composed exclusively of octahedra of diverse cations. On the other hand, octahedral sheets of LDHs exhibit much more variations in isomorphic substitution of cations than those of phyllosilicates. Heterogeneous polyhedra like tetrahedron11 and decahedron12 are often co-crystallized in octahedral sheets of LDHs. Consequently, more flexible post-synthesis routes to the incorporation of heterogeneous cations could be applied to LDHs than zeolites and phyllosilicates, including the chemical transformation processes like reconstruction of oxides into the corresponding LDHs13,14 and selective reoxidation of specific framework cations7 as well as the recrystallization processes like localized dissolution-recrystallization and curing of structural defects.5,9,10
Our special attention has been given to phase transition of portlandite, calcium hydroxide, not only because coordination number of Ca2+ increases from six in portlandite to seven in Ca-based LDHs but also because they occur during hydration of calcium aluminate cement, one of the poorly understood chemical processes.12,15,16 Furthermore, Ca-based LDHs are distinguished from other LDHs by unique combination of a capped trigonal antiprismatic decahedral Ca2+ with octahedral trivalent cation (M3+) at a fixed molar ratio of 2Ca2+/M3+.12,17,18 Typically, a water molecule can occupy the seventh apex of the Ca2+-polyhedron to project into interlayer space. Therefore, it can be thermodynamically favorable in portlandite that octahedral M3+ ions substitute for one third of octahedral Ca2+ ions while adding a water molecule to each of the rest, although the formation mechanism of LDHs had been interpreted solely by co-precipitation.4,6 In this study, we demonstrate the post-isomorphic substitution of trivalent metal cations for Ca2+ ions in portlandite crystals at room temperature as evidenced by the transient appearance of metastable phase, the formation of Ca-based layered double hydroxides at high pH, the distinct shift of suspension pH after phase transition, and the in situ topochemical reaction.
Experimental
All the chemicals had a high purity of more than analytical grade. Deionized water was decarbonated by boiling before use. Two different morphologies of portlandite crystals were used in this study; the polyhedral hexagonal tablet crystals were synthesized by hydrothermally treating a mixture of equi-volumes of 1.0 M CaCl2 and 2.0 M NaOH solutions at 353 K for 2 h whereas the hexagonal platelet ones by mixing equi-volumes of 0.2 M CaCl2 with 0.4 M NaOH in the presence of 0.1 M of Na2SO4 followed by 1 h stirring.19 The crystals were separated by filtration, washed with deionized water twice, and dried at 333 K.Plausibility for phase transition of portlandite into Ca-LDHs via the post-isomorphic substitution was evaluated here by simply reacting portlandite with trivalent cations in aqueous solution at room temperature. An aqueous solution of 0.1 M MCl3 or M(NO3)3 (M = trivalent metal cation), typically FeCl3, was titrated to an aqueous suspension of portlandite (1.48 g/100 mL; equal to 0.2 mol L−1) at a flow rate of 0.8–2.0 mL min−1 over stirring. The solid phases reacted at various molar ratios of Ca2+/M3+ were quickly separated by centrifugation for 3 min at 3000 rpm, smeared over slide glass or wide dish, rapidly dried by blowing air, and kept in a tightly capped vessel.
Water solubility of CaFe-LDH was evaluated by measuring the weight of the water-washed CaFe-LDH after equilibrating its suspension (0.2 g/100 mL) for 4 h. Powder XRD patterns were recorded at a scanning speed of 3° min−1 by Ni-filtered Cu Kα radiation at 40 kV and 40 mA (Rigaku JP/D/MAX-2200H). Morphology changes were examined with a scanning electron microscope (Hitachi S-570 equipped with EDS) after Pt-coating.
Results and discussion
Reaction with Fe3+
Portlandite was reacted with Fe3+ by the addition of 0.1 M FeCl3 solution at a flow rate of 0.8 mL min−1 while stirring. On addition of Fe3+, the color of the suspension turned brown to indicate the presence of ferric hydroxide species. Changes in the XRD patterns of the solid phases (Fig. 1) and the suspension pH (Fig. 2) were monitored immediately after the addition of Fe3+.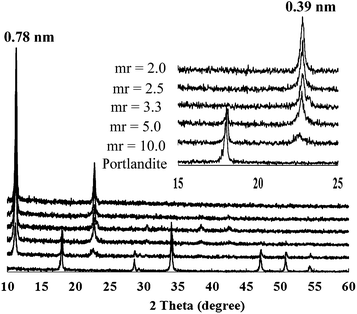 | ||
| Fig. 1 XRD patterns of the solid phases monitored immediately after the addition of Fe3+ at a flow rate of 0.8 mL min−1 (mr = molar ratio of Ca2+/Fe3+). | ||
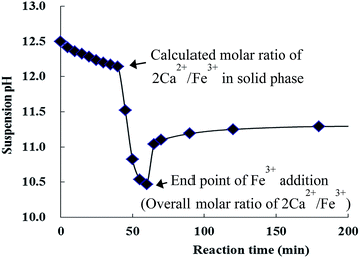 | ||
| Fig. 2 Change in the suspension pH monitored immediately after the addition of Fe3+ at a flow rate of 0.8 mL min−1. | ||
The XRD patterns (Fig. 1) clearly indicated in situ phase transition of portlandite to CaFe-LDH. All the XRD patterns did not show any peak broadening and base-line drifting which were typically found in reconstruction and recrystallization processes.13,14 Relative overall intensity of the peaks assigned to CaFe-LDH increased with the addition of Fe3+ while that of those to portlandite decreased correspondingly. It was remarkable that the phase transition occurred immediately and almost stoichiometrically on adding Fe3+ regardless of the molar ratio of Ca2+/Fe3+. As soon as portlandite reacted with Fe3+, a proportion of portlandite corresponding to a ratio of Ca2+/Fe3+ was almost promptly converted into CaFe-LDH. When Fe3+ solution was rapidly added into the portlandite suspension (above a flow rate of ca. 2 mL min−1), a highly metastable CaFe-LDH phase appeared transiently with a basal spacing of 1.028 nm (Fig. S1†). This phase was quickly converted into the typical CaFe-LDH phase (chloride form with a basal peak at 0.78 nm or nitrate form at 0.86 nm) even in air-dried state. This phase seemed to be highly hydrated18 or intercalated with [Fe(OH)4]− like the [Al(OH)4]−-intercalated AFm phases.15 These results strongly suggested in situ transformation of portlandite into CaFe-LDHs via the post-isomorphic substitution of Fe3+ for Ca2+ rather than localized dissolution-recrystallization route.
A supporting evidence for the post-isomorphic substitution was further provided by the change of the suspension pH during and after the titration of Fe3+ (Fig. 2). The pH curve during the titration revealed two distinct regions, an initial slow decrease followed by a steep drop, which were then followed by a plateau region after the end of titration. The first slow decrease region could be explained by the reactions (I) and (I′), formation of metal hydroxide species,6,20 and the reactions (II) and (II′), incorporation of [M(OH)4]− into portlandite via the post-isomorphic substitution for Ca2+.
| M3+ + 4(OH−) → [M(OH)4]− when the pH ≥ 12 for Fe3+ | (I) |
| M3+ + 3(OH−) → s[M(OH)3] | (I′) |
| [M(OH)4]− + s[Ca3(OH)6] + nH2O → s{[Ca2M(OH)6(H2O)2]+(OH−)·(n − 2)H2O} + Ca2+ + 3(OH−) | (II) |
| 2[M(OH)4]− + s[Ca3(OH)6] + nH2O → s{[Ca2M(OH)6(H2O)2]+·[M(OH)4]−·(n − 2)H2O} + Ca2+ + 4(OH−) | (II′) |
| s{[Ca2M(OH)6(H2O)2]+(OH−)·(n)H2O} + xA− → s{[Ca2M(OH)6(H2O)2]+(OH−)1−x(A−)x·(nH2O)} + x(OH−), | (III) |
Decrease in pH by the reactions (I) and possibly (I′) could be buffered by the corresponding release of OH− by the reactions (II) and (II′) and eventually the reaction (III), hydroxide exchange by counter anion. The reaction (II′) was suggested from the transient appearance of the metastable phase. Interestingly, both the in situ phase transition and the transient appearance could be reasonably explained by rapid intercalation of [Fe(OH)4]− and its prolific substitution for crystallized Ca(OH)2 moiety of adjacent portlandite layers. As the phase transition progresses, the first slow pH decrease region could be expected from delayed equilibrium of the reaction (III) and from lower water solubility of CaFe-LDH than that of portlandite.16 Here, it was worthy to note that CaFe-LDH was formed at the pH of aqueous Ca(OH)2-saturated solution, much higher than that of CaFe-LDH suspension (Fig. S2†). This higher formation pH is a clear indication of the post-isomorphic substitution because crystallization of M2+, M3+-LDH frameworks via coprecipitation route takes place below the pH at which a M2+ hydroxide precipitates alone.6,21 The second steep pH drop region could result mainly from the over-stoichiometric addition of Fe3+ that induces most likely the reaction (I′) consuming free OH−. As the pH decreases below the point of equilibrium water solubility of CaFe-LDH, its dissolution takes place leading to an increase in pH. Even though there is no excess addition of Fe3+, a pH shift would result from the difference in water solubility between portlandite and CaFe-LDH as soon as portlandite disappears. In addition, over-stoichiometric addition of M3+ leads to crystallization of oversaturated Ca2+ into CaFe-LDH via the reaction (IV), typical coprecipitation route.
| [M(OH)3] + 2Ca2+ + (3 − x)(OH−) + xA− + nH2O → s{[Ca2M(OH)6(A−)x(H2O)2−x]+(OH−)1−x·(mH2O)}, where m = n − (2 − x). | (IV) |
Eventually, reactions such as dissolution, coprecipitation and ion exchange reach an equilibrium leading to a pH plateau, the third region. The sharp pH shift between the regions one and three is also indicative of the post-isomorphic substitution because typical coprecipitation route, the reaction (IV), could not induce any pH shift after the formation of LDHs.6,21
The in situ M3+-dependent phase transition excludes both nucleation and crystal growth stages, resulting in the coexistence of portlandite and CaFe-LDH even within one crystal. The substitution of [Fe(OH)4]− for crystallized Ca(OH)2 moiety leads to development of positive layer charge that is compensated by either (OH)− or [Fe(OH)4]− intercalated through simultaneous expansion of interlayer space. Consequently, the phase transition would take place without any significant changes in crystal morphology during the titration. Fig. 3 shows the changes of crystal morphology during the addition of Fe3+.
 | ||
| Fig. 3 Scanning electron micrographs of the solid phases reacted with Fe3+ at various molar ratios of Ca2+/Fe3+. | ||
Two distinct shapes of portlandite crystals, one as polyhedral hexagonal tablet and the other as hexagonal platelet19 were used in this experiment. The pristine tablets were 1–5 μm in diameter and 0.5–1 μm in thickness while the pristine platelets 2–5 μm in diameter and about 1 μm in thickness. After reaction with Fe3+, the tablet crystals were only sliced into thin flakes without any significant changes in diameter. Similarly, the platelet crystals indicated detachment of thickness-sized strips from the rim surface, and steadily decreased in diameter. A proportion of flakes or strips increased with the molar ratio of Fe3+. At higher molar ratio of Ca2+/Fe3+, it was frequently observed that the flakes or the strips were peeling from the surfaces of the crystals (the inserted images of in Fig. 3b). Addition of over-stoichiometric Fe3+ resulted in blunting of the crystal outlines surely due to dissolution of CaFe-LDH. At the molar ratio of 2.0 where over-stoichiometric Fe3+ was added due to water solubility of portlandite, tiny particulates were frequently observed to indicate coprecipitation (Fig. 3d). These results confirm that the phase transition of portlandite into CaFe-LDHs undergoes topochemically as well as stoichiometrically via post-isomorphic substitution.
Reactions with other M3+ ions
The reactions with other trivalent cations such as Al3+, Ga3+, Cr3+ and In3+ also successfully led to the in situ topochemical phase transition of portlandite to CaM-LDHs, as shown in Fig. 4 and 5. Al3+ and Ga3+ exhibited the reaction behaviors very similar to that of Fe3+ although each region in their pH curves exhibited the differences in the molar Ca2+/M3+ ratio and the pH value (Fig. S3†).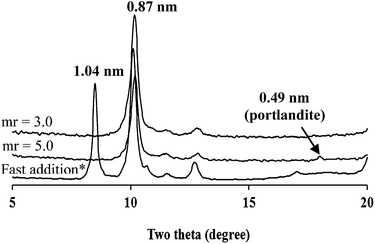 | ||
| Fig. 4 XRD patterns of portlandite reacted with Al3+ (mr = molar ratio of Ca2+/Al3+). *At an addition rate of 2.0 mL min−1 at mr = 5.0. | ||
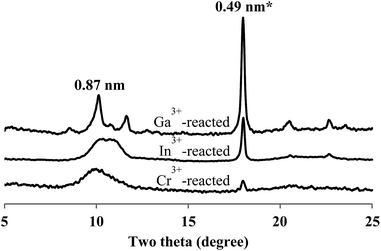
These differences mainly resulted from aqueous acidity of each cation. The highly metastable phase was also observed in the XRD patterns of the reaction products obtained by the fast addition of Al3+ and Ga3+ at the molar Ca2+/M3+ ratio of 5.0. Meanwhile, the XRD patterns derived from Cr3+ and In3+ exhibited much more peak broadening than those from Al3+ and Ga3+. Such low crystallinity of CaIn-LDH seemed to be due to large size of In3+ whereas that of CaCr-LDH due to a coordination environment of Cr3+, CrO4− rather than CrO2−, in spite of its ion size19,20 similar to Fe3+ (Fig. 6). The reactions with both Al3+ and Ga3+ led to the crystal morphologies almost same to those with Fe3+. On the other hand, the crystals from the reactions with In3+ (Fig. 6) and Cr3+ resulted in quite different morphologies; CaIn-LDH as small flakes and CaCr-LDH as tiny particulates (Fig. S4†). This is mainly due to the above mentioned chemical properties of these two cations.
Conclusions
Occurrence of post-isomorphic substitution of heterogeneous cations in already crystallized phases has been elucidated in portlandite crystals as evidenced by the transient appearance of metastable phase, the formation of CaM-LDHs at high pH, the sharp shift of suspension pH after phase transition, and the in situ topochemical reaction. Specifically, it offers an innovative route to simplify synthesis processes and to control crystal morphology for CaM-LDHs. Furthermore, it leads to new understanding on the hydration process of calcium aluminate cement, in particular, the formation and transformation of calcium aluminate hydrate phases that had been explained only by the dissolution and re-precipitation reactions.15,16 Therefore, the post-isomorphic substitution revealed in this study could make a substantial contribution to many scientific and industrial fields related to clay and cement minerals.Acknowledgements
This study was supported by the Rural Development Administration of South Korea.References
- The chemistry of clay minerals, ed. E. Weaver and L. D. Pollard, Elsevier, Amsterdam, 1973, pp. 1–4 Search PubMed.
- (a) S. R. J. Oliver, Chem. Soc. Rev., 2009, 38, 1868 RSC; (b) K. Teramura, S. Iguchi, Y. Mizuno, T. Shishido and T. Tanaka, Angew. Chem., Int. Ed., 2012, 51, 8008 CrossRef CAS PubMed.
- J. M. Zen and A. S. Kumar, Anal. Chem., 2004, 76, 205A CAS.
- A. Vaccari, Catal. Today, 1998, 41, 53–71 CrossRef CAS.
- X. Chen, J. V. Wright, J. M. Conca and L. M. Peurrung, Environ. Sci. Technol., 1997, 31, 624 CrossRef CAS.
- Z. P. Xu and G. Q. Lu, Chem. Mater., 2005, 17, 1055 CrossRef CAS.
- (a) R. Ma, K. Takada, K. Fukuda, N. Iyi, Y. Bando and T. Sasaki, Angew. Chem., Int. Ed., 2008, 47, 86 CrossRef CAS PubMed; (b) R. Ma, J. Liang, X. Liu and T. Sasaki, J. Am. Chem. Soc., 2012, 134, 19915 CrossRef CAS PubMed.
- L. T. Rakov, Geochem. Int., 2006, 44, 1004 CrossRef.
- P. S. Neuhoff and L. S. Ruhl, Chem. Geol., 2006, 225, 373 CrossRef CAS PubMed.
- (a) S. Komarneni, N. Kozai and R. Roy, J. Mater. Chem., 1998, 8, 1329 RSC; (b) M. C. Richardson and P. S. Braterman, J. Mater. Chem., 2009, 19, 7965 RSC.
- L. Poul, N. Jouini and F. Fievet, Chem. Mater., 2000, 12, 3123 CrossRef CAS.
- G. Renaudin and M. Francois, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1999, 55, 835 Search PubMed.
- L. Vieille, I. Rousselot, F. Leroux, J. P. Besse and C. Taviot-Gueho, Chem. Mater., 2003, 15, 4361 CrossRef CAS.
- J. S. Valente, E. Lima, J. A. Toledo-Antonio, M. A. Cortes-Jacome, L. Lartundo-Rojas, R. Montiel and J. Prince, J. Phys. Chem. C, 2010, 114, 2089–2099 CAS.
- N. Ukrainczyk, T. Matusinovic, S. Kurajica, B. Zimmermann and J. Sipusic, Thermochim. Acta, 2007, 464, 7 CrossRef CAS PubMed.
- F. P. Glasser, A. Kindness and S. A. Stronach, Cem. Concr. Res., 1999, 29, 861 CrossRef CAS.
- I. Rousselot, C. Taviot-Gueho, F. Leroux, P. Leone, P. Palvadeau and J. P. Besse, J. Solid State Chem., 2002, 167, 137 CrossRef CAS.
- T. Runcevski, R. E. Dinnebier, O. V. Magdysyuk and H. Pollmann, Acta Crystallogr., Sect. B: Struct. Sci., 2012, 68, 493 CAS.
- S. Galmarini, A. Aimable, N. Ruffray and P. Bowen, Cem. Concr. Res., 2011, 41, 1330 CrossRef CAS PubMed.
- Atlas of Eh-pH diagrams, Takeno, N. Geological Survey of Japan Open File Report No.419, National Institute of Advanced Industrial Science and Technology, Japan, 2005.
- (a) J. W. Boclair and P. S. Braterman, Chem. Mater., 1999, 11, 298 CrossRef CAS; (b) J. W. Boclair, P. S. Braterman, J. Jiang, S. Lou and F. Yarberry, Chem. Mater., 1999, 11, 303 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See E-mail: DOI: 10.1039/c4ra03083d |
| This journal is © The Royal Society of Chemistry 2014 |