Structural properties and electrical characteristics of Ho2O3 and HoTixOy gate dielectrics for a-InGaZnO thin-film transistors
Abstract
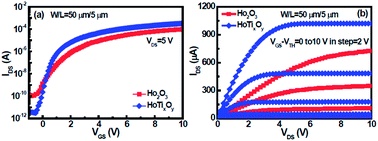
In this study, we developed high-κ Ho2O3 and HoTixOy gate dielectrics for amorphous indium–gallium–zinc oxide (a-IGZO) thin-film transistor (TFT) applications. X-ray diffraction, atomic force microscopy, and X-ray photoelectron spectroscopy were used to study the structural, morphological and chemical features of Ho2O3 and HoTixOy dielectric films. Compared with the Ho2O3 dielectric, the a-IGZO TFT incorporating the high-κ HoTixOy gate dielectric exhibited very good electrical characteristics, such as a high Ion/off ratio of 1.1 × 108, a high field effect mobility of 20.6 cm2 V−1 s−1, a low threshold voltage of 0.23 V, and a low subthreshold swing of 183 mV decade−1. These results are probably due to the incorporation of Ti into the Ho2O3 film, resulting in the formation of a smooth surface and a low density of interface states at the oxide/channel interface. In addition, the stability of high-κ Ho2O3 and HoTixOy a-IGZO TFTs was investigated under positive gate-bias stress (PGBS) and negative gate-bias stress (NGBS). The electron charge trapping at the dielectric–channel interface resulted from the PGBS, whereas the oxygen vacancies occurred in the a-IGZO under the NGBS.

 Please wait while we load your content...
Please wait while we load your content...