One pot synthesis of coordination polymer 2,5-dimercapto-1,3,4-thiadiazole–gold and its application in voltammetric sensing of resorcinol†
Abstract
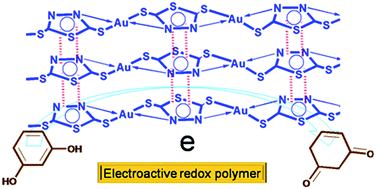
Herein, we report a one pot synthesis of a coordination polymer using 2,5-dimercapto-1,3,4-thiadiazole (DMTD) with gold (Au) and the characterization of the polymer by FTIR, Raman spectroscopy, UV-visible spectroscopy, XPS, XRD, 13CNMR, HRTEM, gel permeation chromatography and TGA. Results show that Au ions get coordinated by the diazole and sulfydryl groups of DMTD throughout the polymeric chain. The polymeric layers are stabilized through π–π stacking and hydrophobic interaction. XRD data reveals the crystalline nature of DMTD–Au coordination polymer. The kinetic parameters, such as the energy of activation and the regression value of the DMTD–Au decomposition, have been determined by exploiting thermogravimetric data. HRTEM analysis establishes the nanostructure of the polymeric material DMTD–Au. Its voltammetric study reveals that the synthesized coordination polymer DMTD–Au exhibits excellent electroactivity towards resorcinol and facilitates fast electron transfer kinetics, which is employed for electro-sensing applications. To the best of our knowledge we are the first to report this coordination polymer DMTD–Au modified carbon paste electrode (DMTD–Au/CPE) for the electrochemical detection of resorcinol (RS). The DMTD–Au modified carbon paste electrode has been found to be highly sensitive towards resorcinol due to its strong interaction through intermolecular hydrogen bonding. Under the optimized conditions, the modified electrode exhibit resorcinol oxidation at low over potential (0.55 V) and the anodic peak current shows a linear response with sensitivity and limit of detection of 0.019 μA nM−1 and 29.77 nM, respectively, at a S/N (signal-to-noise ratio) of 3.

 Please wait while we load your content...
Please wait while we load your content...