Enhanced visible photocatalytic activity of Cu2O nanocrystal/titanate nanobelt heterojunctions by a self-assembly process
Abstract
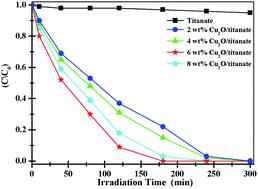
In the present work, we introduce a facile and widely used route to fabricate Cu2O nanocrystal/titanate nanobelt heterojunctions with high yield, good dispersion and tight interaction, and which are self-assembled via the linker molecule 3-mercaptopropionic acid. Their photocatalytic activity for the degradation of methyl orange solution is enhanced by decorating with Cu2O nanocrystals, especially in the visible light region. The degradation rates of 6 wt% Cu2O/titanate heterojunctions are both the highest at 100% after 80 and 180 minutes irradiation under UV and visible light irradiation, while those of the pure titanate nanobelts are only 20% and 3%, respectively. These lower values for the pure titanate nanobelts are due to the p–n heterojunctions suppressing the recombination of electron–hole pairs in the titanate nanobelts, where the Cu2O nanocrystals act as electron traps aiding electron–hole separation. Additionally, a synergistic effect from the tight contact between Cu2O nanocrystals and titanate nanobelts also efficiently enhances the photodegradation of the Cu2O/titanate heterojunctions.

 Please wait while we load your content...
Please wait while we load your content...