Paper microfluidics for red wine tasting
Abstract
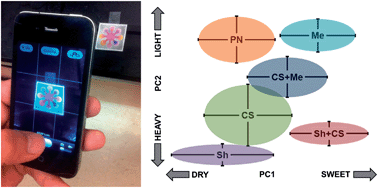
A paper microfluidic chip was designed and fabricated to evaluate the taste of 10 different red wines using a set of chemical dyes. The digital camera of a smartphone captured the images, and its red-green-blue (RGB) pixel intensities were analyzed by principal component analysis (PCA). Using 8 dyes and 2 principal components (PCs), we were able to distinguish each wine by the grape variety and the oxidation status. Through comparing with the flavor map by human evaluation, PC1 seemed to represent the sweetness and PC2 the bodyness of red wine. This superior performance is attributed to: (1) careful selection of commercially available dyes through a series of linear correlation studies with the taste chemicals in red wines, (2) minimization of sample-to-sample variation by splitting a single sample into multiple wells on the paper microfluidics, and (3) filtration of particulate matter through paper fibers. The image processing and PCA procedure can eventually be implemented as a stand-alone smartphone application and can be adopted as an extremely low-cost, disposable, fully handheld, easy-to-use, yet sensitive and specific quality control method for appraising red wine or similar beverage products in resource-limited environments.

 Please wait while we load your content...
Please wait while we load your content...