Implications of surface passivation on physicochemical and bioimaging properties of carbon dots†
Abstract
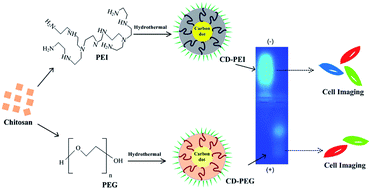
The prevalence of surface functionalized carbon dots (CDs) with intriguing fluorescence properties has given a new dimension to the field of bioimaging and is perceived as a promising alternative to quantum dots (QDs). In the present work, polyethylene glycol (PEG) and polyethyleneimine (PEI) passivated CDs have been synthesized by one-step hydrothermal carbonization of chitosan. We have made a comparative analysis of the physicochemical and bioimaging properties of PEI based carbon dots (CD-PEI) and PEG based carbon dots (CD-PEG). This article further provides an insight into the role of surface functionality in controlling the bioimaging efficiencies of CDs. The concentration dependent cytotoxic effects of CD-PEI and CD-PEG were studied on normal (BHK-21) and cancer (A549) cell lines and we explored the competitive performance of CD-PEI compared to CD-PEG for bio-applications.

 Please wait while we load your content...
Please wait while we load your content...