NaY(MoO4)2 microcrystals with controlled faceting and their tunable photoluminescence properties after doping with Eu3+†
Lin Xua,
Xiaoyan Yanga,
Haimeng Lub,
Chenhui Hua and
Wenhua Hou*ab
aKey Laboratory of Mesoscopic Chemistry of MOE, School of Chemistry and Chemical Engineering, Nanjing University, Nanjing, 210093, P. R. China. E-mail: whou@nju.edu.cn; Fax: +86-25-83317761; Tel: +86-25-83686001
bNanjing University, Yangzhou Institute of Chemistry and Chemical Engineering, Yangzhou, 211400, P. R. China
First published on 4th March 2014
Abstract
We present a facile synthesis of NaY(MoO4)2 microcrystals with controlled morphologies and tunable faceting via an ethylenediaminetetraacetic acid (EDTA)-mediated hydrothermal method. Three different types of NaY(MoO4)2 microcrystals, namely, sharp bipyramids, truncated bipyramids, and quasi-cubes, can be selectively produced, and the percentage of {001} and {101} facets in the tetragonal bipyramidal NaY(MoO4)2 microcrystals can be engineered through the judicious choice of an appropriate amount of EDTA. The preferential adsorption of EDTA on {001} facets of the NaY(MoO4)2 crystal is responsible for the morphology evolution of the NaY(MoO4)2 microcrystals from sharp bipyramids to quasi-cubes. The present synthetic approach is also extendable to achieve NaHo(MoO4)2 and NaTb(MoO4)2 microcrystals with different shapes and tunable exposed facets. It is found that the luminescence properties of the NaY(MoO4)2:Eu3+ microcrystals are highly dependent on their shape and size, and the sharp NaY(MoO4)2:Eu3+ bipyramids exhibit the strongest emission intensity due to their larger size, fewer defects and less exposed facets. As such, the sharp NaY(MoO4)2:Eu3+ bipyramids may have potential applications in light display systems as well as optoelectronic devices, while the present EDTA-mediated hydrothermal synthetic strategy represents a potentially general route to the generation of nano/microcrystals with shape- and exposed facet-control.
1. Introduction
Over the past few years, elaborate architectural manipulation of inorganic micro/nanocrystals with uniform controlled morphologies and tunable size has attracted enormous scientific and technological interest in multidisciplinary research fields owing to the close correlation between morphologies and the corresponding physicochemical properties.1–4 Particularly, different exposed facets with distinct surface atomic arrangements and coordination intrinsically determine the surface atom densities, bonding, electronic structures, and chemical reactivities.5–8 Therefore, the ability to understand, predict and control faceting is of great importance to elucidate the shape–property relationship and enable us to manipulate their properties as desired. Hitherto, tremendous efforts have been progressively devoted to explore and fabricate materials with specific exposed facets, which exhibit remarkable facet-dependent properties and have widespread potential applications in various fields, such as heterogeneous catalysis,9–11 photocatalysis,12–14 electrocatalysis,15,16 gas sensor,17 molecular adsorption,18,19 battery20 and so forth.Generally speaking, the equilibrium form of a crystal tends to possess a minimum surface energy since high-energy facets have fast growth rates and ultimately diminish during crystal growth, whereas the facets with the lowest surface energy will dominate the surfaces of a crystal. Therefore, tuning the product's shape could be readily achieved by altering the relative order of the surface energy of different crystallographic facets. Thus, introducing appropriate additives with specific functional groups offers a great opportunity to tune and control the final morphology since the additive can selectively adhere to some specific crystallographic facet and thus in turn modify the crystal growth dynamically.21–24 Some small chelating agents, such as citric acid (CA), and sodium tartrate (Na2tar) are commonly employed to control the nucleation, growth and alignment of crystals.25,26 Recently, we have developed a versatile EDTA-mediated hydrothermal approach through which several geometrical parameters of the final structures, such as the building blocks, the size distribution, and consequently the overall morphology, can be tailored in a highly precise and controllable manner.27–31 Collectively, these results unambiguously highlight that the EDTA-mediated hydrothermal technique has the capacity to address some of the challenges in crystal-facet engineering.
Double alkaline rare-earth molybdates ARE(MoO4)2 (A = Na or K, RE = trivalent rare earth cation), which share the sheelite-like (CaWO4) isostructure, exhibit excellent thermal and hydrolytic stability and can be widely used in quantum electronics,32 white light-emitting diodes (W-LED),33 and efficient phosphors.34 In these compounds, RE3+ ions occupy the lattice sites without centro-symmetry, and the concentration quenching effect hardly occurs in ARE(MoO4)2 doped with RE ions.35 Recently, stimulated by both intriguing properties and promising applications, much attention has been dedicated to the controlled synthesis of NaY(MoO4)2 with different shapes and the investigation of their size/shape-dependent properties.36–41 Despite these demonstrations, however, the precise architectural manipulation of NaY(MoO4)2 crystals with well-defined morphologies and accurately tunable exposed facets still remains a research focus and a challenging issue, so the exploration of an efficient and more controlled method for creating such novel architectures will be of general interest.
Here we report a facile route to the synthesis of NaY(MoO4)2 microcrystals with controlled morphology and tunable exposed facets via an EDTA-mediated hydrothermal method that involves EDTA as the capping and structure-directing agent. In this work, we demonstrate that three different types of NaY(MoO4)2 microcrystals, namely, sharp bipyramids, truncated bipyramids, and quasi-cubes, can be selectively produced, and the percentage of {001} and {101} facets in the tetragonal bipyramidal NaY(MoO4)2 microcrystals can be engineered by the amount of EDTA. It is found that the luminescence properties of NaY(MoO4)2:Eu microcrystals are highly dependent on their shape and size, and the sharp NaY(MoO4)2:Eu3+ bipyramids exhibit the strongest emission intensity due to their larger size, less defects and less exposed facets. The present synthetic route will greatly contribute to the further design of nano/microcrystals with well-defined morphology and tunable exposed facets.
2. Experimental section
2.1 Preparation of samples
All reagents were analytical grade and used as purchased without further purification. In a typical procedure, 1.00 mmol (0.226 g) of Y2O3 was dissolved in 8 mL of 10% HNO3. Subsequently, a predetermined amount of EDTA (ranging from 0.2 to 0.45 g) was added to form an Y3+–EDTA complex. After vigorous stirring for 20 min, 15 mL aqueous solution containing 0.57 mmol (0.704 g) of (NH4)6Mo7O24 was added dropwise into the above solution under continuous stirring. Then, the pH value of the resulting white suspension was adjusted to 5.0 and the final suspension volume was kept at 35 mL through the addition of 2.0 M NaOH solution. After an additional agitation for 30 min, the as-obtained white colloidal precipitate was transferred into a 40 mL Teflon-lined autoclave (filled up to 80% of its total volume), and the autoclave was sealed and maintained at 180 °C for 24 h. After the autoclave was allowed to air-cool to room temperature, the final product was collected by filtration, washed with distilled water and absolute alcohol several times, and subsequently dried at 60 °C and kept for further characterization.2.2 Characterization
The phase purity and crystallinity of the products were identified by X-ray diffraction (XRD) on a Philip-X'Pert X-ray diffractometer equipped with a Cu Kα radiation (λ = 1.5418 Å) at a scanning rate of 0.02° s−1 in a 2θ range of 10–80°. The product morphology was investigated by scanning electron microscopy (SEM JEOL JEM-6300F) and transmission electron microscopy (TEM JEOL JEM-2100, operated at an accelerating voltage of 200 kV). For TEM observation, the sample was dispersed in ethanol by ultrasonic treatment and then dropped on carbon–copper grids. Photoluminescence excitation and emission spectra were recorded on a 48000DSCF luminescence spectrometer at room temperature.3. Results and discussion
3.1 Structural study
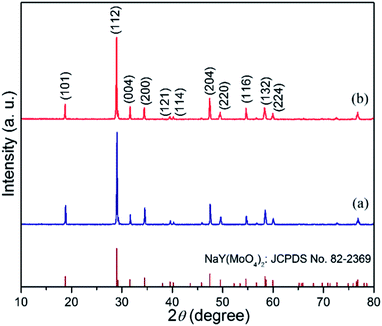
The composition and phase purity of the products were characterized by XRD. The XRD patterns of the products prepared in the presence of different amounts of EDTA at pH 5.0 are displayed in Fig. 1. Compared with the standard card of tetragonal NaY(MoO4)2 (JCPDS #82-2369), all diffraction peaks of the different samples can be readily indexed to pure tetragonal phase of NaY(MoO4)2 with the space group of I41/a. The lattice constants calculated from XRD data are a = b = 5.196 Å, and c = 11.30 Å, respectively. The strong and sharp diffraction peaks indicate the high crystallinity of the as-synthesized products. No evident peaks of other impurity phases were detected, revealing a high phase purity of the as-obtained products. By further observation, it can be seen that the relative diffraction intensity ratio of (004)/(101) exhibits a slight enhancement as the amount of EDTA increases, indicating that the samples obtained in the EDTA-rich system are preferentially oriented along the (001) plane, which is further confirmed by HRTEM and SAED.3.2 Morphology
SEM images (Fig. 2) illustrate the range of morphologies achievable through the present approach, depending on the amount of EDTA under otherwise identical reaction conditions. We observed the gradual morphological evolution of the obtained NaY(MoO4)2 microstructures as the amount of EDTA was increased. As illustrated in Fig. 2a–c, the product obtained in the presence of 0.20 g EDTA is almost entirely composed of well-defined uniform tetragonal micro-bipyramids with a longitudinal length of around 15.0 μm and four hemline lengths of about 7.2 μm. No other morphologies are detected, indicating a high yield of these microstructures. Interestingly, geometrical parameters of the micro-bipyramids, such as interplanar angles and edge-length ratios, are essentially identical regardless of the actual sizes. This observation indicates that an overall crystallographic relationship exists among the interplanars that constitute the bipyramid. Magnified images shown in Fig. 2b and c indicate that the bipyramid possesses a very smooth surface and is bound by eight isosceles triangular crystal planes. The angle α of (65.35°) measured between the pyramidal facets and the waist basal plane (Fig. 2b) matches closely with the theoretical value (65.13°) between the 〈101〉 and 〈001〉 directions in a tetragonal phase of NaY(MoO4)2. From the above investigation, we can propose that the as-obtained bipyramids might be exclusively enclosed by eight {101} facets and the longitudinal direction of the bipyramid is 〈001〉. This interpretation will be unambiguously supported by HRTEM below. | ||
| Fig. 2 SEM images of the NaY(MoO4)2 microstructures obtained at pH = 5 with the assistance of different amounts of EDTA. (a–c) 0.20 g EDTA. (d–f) 0.35 g EDTA. and (g–i) 0.45 g EDTA. | ||
As the amount of EDTA is increased to 0.35 g, the product turns out to be truncated bipyramids which are significantly truncated perpendicular to the 〈001〉 direction. Meanwhile, the average longitudinal length of the NaY(MoO4)2 truncated bipyramids is observed to decrease to 12.1 μm, as displayed in Fig. 2d–f. A further increased addition of EDTA (0.45 g) results in the formation of quasi-cubes with a high percentage of {001} facets. Moreover, the average thickness of the quasi-cubes is observed to reduce significantly to 3.9 μm (Fig. 2g–i). Thus, the present synthetic technique provides an effective means to synthesize truncated NaY(MoO4)2 bipyramids with tunable fraction of {001} facets with respect to the {101} facets by varying the amount of EDTA, and truncation and shortening of bipyramids become evident with the addition of EDTA.
Detailed morphological and surface structural information of the as-obtained NaY(MoO4)2 microstructures was obtained by TEM measurements. Fig. 3a displays the TEM image of an individual NaY(MoO4)2 bipyramid obtained in the presence of 0.20 g EDTA. Consistent with the SEM images shown in Fig. 2a–c, the as-obtained product exhibits a rhombus shape with a smooth surface. HRTEM image (Fig. 3b) recorded at the edge of the bipyramid (the red frame area) demonstrates the single-crystalline nature of the bipyramid. The lattice interplanar spacing in the HRTEM image is determined to be 0.47 nm, corresponding to the (101) planes of tetragonal NaY(MoO4)2. Fig. 3c depicts the TEM image of an individual quasi-cube synthesized with the assistance of 0.45 g EDTA. The product appears in the form of cuboidal shape, which is in good accordance with the SEM images shown in Fig. 2g–i. From the HRTEM image (Fig. 3d) taken from the fringe of the quasi-cube (the blue frame area), the lattice interplanar spacing was determined to be 0.26 nm, corresponding to the (020) planes of tetragonal NaY(MoO4)2. The corresponding SAED pattern (inset of Fig. 3d) further highlights the single-crystalline feature of the quasi-cube and can be indexed to the tetragonal NaY(MoO4)2. These results also demonstrate that the basal plane of a quasi-cube is a (001) plane, consistent with the hypothesis during SEM observation.
Generally, rare earth compounds have similar crystal structures. As a result, they may have similar physical and/or chemical properties. In order to demonstrate the power and versatility of the present EDTA-induced facet-controlled approach, microstructures with tunable exposed facets of the other two rare earth molybdates, namely, NaHo(MoO4)2 and NaTb(MoO4)2, have also been obtained through a similar protocol by using Ho2O3 and Tb2O5 as parent oxides. Fig. 4 illustrates the representative SEM images of the resultant NaHo(MoO4)2 and NaTb(MoO4)2 samples obtained in the presence of different amount of EDTA. As expected, at low EDTA content (0.20 g EDTA), both samples exhibit a bipyramidal morphology (Fig. 4a and c). While at higher EDTA content (0.35 g EDTA), the two products appear as quasi-cubic morphology (Fig. 4b and d). These results further confirm that the use of more EDTA results in the preferential exposed of the {001} facet of tetragonal NaRE(MoO4)2 (RE = rare earth). The above results also indicate that the present facile EDTA-induced facet-controlled approach can be extended to fabricate other rare earth molybdates with controlled exposed facets.
3.3 Formation mechanism of the tetragonal NaY(MoO4)2 bipyramids and quasi-cubes
Given the ability to engineer the exposed facets of the as-obtained microstructures by varying the amount of EDTA, a better understanding of the mechanism underlying product formation is desirable and would allow the approach to be applied generally. In the present study, when EDTA is absent from the reaction system while the other reaction parameters remain unchanged, the resulting product is only composed of irregular aggregated nanosheets (Fig. S1†). However, when an appropriate amount of EDTA is added into the reaction system, new physicochemical processes can occur and bring out new morphologies (Fig. 2). This result indicates that the addition of EDTA is integral to the generation of single-crystalline, facet-controlled particles.It is commonly accepted that the evolution of crystal shape during growth is primarily driven by continuously decreasing the total surface energy of the crystal, and finally terminates at the minimum surface energy point in a given growth environment. The final shape of a crystal is determined by the relative specific energy of each crystal facet, according to Gibbs–Curie–Wulff theorem.42 Therefore, manipulation of particle's shape through the use of capping agent is typically considered as a thermodynamic means of structure direction, since the interactions between selective adsorbate and facet with specific atomic configurations can alter the relative energies of different facets. The morphology of a crystal can also be considered in terms of growth kinetics: the fastest growing planes should disappear to leave behind the slowest growing planes as facets of the product.43 As we know, EDTA possesses four carboxyl groups (–COOH) and two lone pairs of electrons on two nitrogen atoms which can act as binding sites. These characters endow EDTA with a strong chelating ability and an excellent capping ability, which can effectively modulate the growth rates of different facets. In this work, when EDTA is added into Y(NO3)3 solution, it can coordinate with Y3+ ions to form the intermediate complex of Y3+–EDTA, which greatly decreases the concentration of free Y3+ in solution and helps to control the nucleation and growth of high-quality crystals in view of the dynamic process. Then, the introduction of molybdate into Y3+–EDTA leads to the competition of MoO42− for Y3+ with EDTA and the formation of NaY(MoO4)2 nanoparticles. The released EDTA may preferentially and selectively bind to {101} and {001} facets of NaY(MoO4)2 nuclei through bidentate coordination to the metal ions due to the higher Na+/Y3+ packing density on {101} and {001} facets than on other facets, based on the structure model of surface cleavage of NaY(MoO4)2 crystal illustrated in Fig. 5. Such a preferential adsorption of EDTA can effectively restrict the crystal growth along 〈101〉 and 〈001〉 directions. According to Donnay–Harker rules,44 the surface energy of {001} facet is higher than that of {101} facet in NaY(MoO4)2 tetragonal structure and the surface energy of {001} facet might be still higher than that of {101} facet even after EDTA adsorption at a low EDTA content. Thus, the faster growth rate along 〈100〉, 〈010〉, and 〈001〉 directions than that of along 〈101〉 direction leads to the generation of bipyramidal morphology with exposed {101} facets. With increasing the amount of EDTA, more EDTA will preferentially adsorb on {001} facets of NaY(MoO4)2 crystal nuclei and thus inhabit their growth. As a result, the high-energy {001} facets become more and more stable and the percentage of {001} facets becomes larger at higher EDTA content.
 | ||
| Fig. 5 Surface cleavage of NaY(MoO4)2 crystal: (a) (100) plane, (b) (101) plane, and (c) (001) plane. The red, cyan and green spheres represent O, Mo and Y or Na, respectively. | ||
From the above analyses, it is apparent that EDTA plays dual important roles in the manipulation of the exposed facets of NaY(MoO4)2 microstructures. First, it serves as a chelating agent to form a stable complex with Y3+ and further kinetically controls the reaction rate. Second, as a capping agent, it can change the free energies of different crystallographic surfaces and thus alter their growth rates, and eventually determine the product morphology. Significantly, studying the roles of EDTA may open up new opportunities to fabricate nano/micro-materials with tunable size and exposed facets. The morphological evolution process as a function of EDTA amount is schematically illustrated in Fig. 6.
 | ||
| Fig. 6 Illustration of the morphological evolution process from sharp bipyramid to quasi-cube as a function of EDTA amount. | ||
3.4 Photoluminescence properties
NaY(MoO4)2 has been demonstrated to be an efficient host for other lanthanide ions, bringing out various luminescence properties.36–41 Herein, Eu3+ was selected as the doping ion to investigate the luminescence properties of NaY(MoO4)2 bipyramids with different truncation degrees obtained with the assistance of different amounts of EDTA. It is noteworthy that doping with Eu3+ (5 mol%) alters neither the crystal structure nor the morphology of the host material due to the similarity of Eu3+ to Y3+ (Fig. S2†). Room temperature excitation and emission spectra of different NaY(MoO4)2:Eu3+ samples are presented in Fig. 7. | ||
| Fig. 7 Room-temperature (a) excitation (λem = 615 nm) and (b) emission (λex = 396 nm) spectra of NaY(MoO4)2:Eu3+ microcrystals with different sizes and truncation degrees. | ||
As shown in Fig. 7a, the excitation spectrum of NaY(MoO4)2:Eu3+ sharp bipyramids consists of the characteristic f–f transition lines within the Eu3+ 4f6 configuration between 350 and 450 nm. These transition lines can be assigned as the transitions from the 7F0 ground state to the different excited states of Eu3+, i.e., 5D4 (362 nm), 5G2 (383 nm), 5L6 (396 nm), and 5D3 (417 nm), respectively. The PL emission spectra of the three samples upon excitation into the strongest 7F0–5L6 transition of Eu3+ at 396 nm are displayed in Fig. 7b. As labeled in Fig. 7b, the obtained emission spectra consist of a series of 5D0–7FJ (J = 1, 2, 3, 4) transition lines of Eu3+, with the 5D0–7F2 transition centered at 615 nm being the most prominent group. It is well-established that the intensities of the transitions between different J-number levels are sensitively dependent on the symmetry of the local environment of Eu3+ activators in terms of Judd–Ofelt theory.45,46 If Eu3+ is located in a site with an inversion center, the 5D0–7F1 magnetic dipole transition should be dominant, while in a C1 site without an inversion center, the 5D0–7F2 electric dipole transition will be preponderant. In the present case, the most prominent red emission centered at 615 nm ascribed to 5D0–7F2 transition is evidently far stronger than the orange emission at 592 nm corresponding to the 5D0–7F1 transition in three samples, indicating that Eu3+ ions occupy the sites without inversion center and have a low crystal field symmetry. Meanwhile, the dominant red emission centered at 615 nm means high a red color purity, which is quite crucial to practical applications in the future. This exciting result fulfils the current urgent demand of exploring efficient phosphors with high a red-color purity and implies that the synthesized NaY(MoO4)2:Eu3+ microstructures with efficient luminescence properties may have practical applications in fluorescence lamps, field emission displays (FED) and plasma display panels (PDP).
From Fig. 7b, it can be clearly seen that the emission spectra of the three samples are similar in shape, but obviously different in the intensity, indicating that the luminescence properties are closely correlated with the morphology and size of the products. Among three NaY(MoO4)2:Eu3+ microcrystals, the NaY(MoO4)2:Eu3+ bipyramids with sharp tips outperform the truncated counterparts in terms of the emission intensity. Moreover, the emission intensity decreases as the truncation degree increases. The possible reasons for this luminescence feature can be elucidated as follows. Firstly, as illustrated in Fig. S2,† the sharp NaY(MoO4)2:Eu3+ bipyramids are much bigger than the truncated counterparts and the average size of the resultant NaY(MoO4)2:Eu3+ microcrystals decreases with the increment of EDTA amount, thus the surface area of NaY(MoO4)2:Eu3+ microcrystals increases along with a decrease in size. The large surface area may result in a large number of defects into the phosphor crystal, which have a serious impairment in the photoluminescence intensity for phosphors because they provide nonradiative recombination routes for electrons and holes. If the surface area is significantly reduced, which results from the increased crystallite size, the sharp NaY(MoO4)2:Eu3+ bipyramids with fewer defects would show great a improvement in photoluminescence intensity.47 Secondly, in the case of truncated NaY(MoO4)2:Eu3+ bipyramids, the more exposed facets with a higher surface and more defects will increase light scattering and thus decrease their emission intensity.48 Finally, since EDTA is employed as a structure-directing agent during the synthesis, it may remain on the surface of the final NaY(MoO4)2:Eu3+ microcrystals, and it is reasonable to assume that the more EDTA added, the more it remains. The organic groups in EDTA are generally considered as fluorescence quenchers due to high energy vibrations of the groups.49 Accordingly, the quasi-cubic NaY(MoO4)2:Eu3+ microcrystals exhibit the weakest photoluminescence emission under identical measurement conditions. Therefore, on the basis of the aforementioned explanations, the different luminescence behaviors can be reasonably attributed to the synergistic effects of the morphology and size, exposed facets and surface chemistry of the NaY(MoO4)2:Eu3+ microcrystals.
4. Conclusion
In summary, a facile and effective EDTA-mediated hydrothermal approach was employed to synthesize tetragonal NaY(MoO4)2 microcrystals with shape and facet control. The judicious addition of an appropriate amount of EDTA allows for the morphology of the final NaY(MoO4)2 to be precisely tuned from sharp bipyramids to truncated bipyramids and further to quasi-cubes. The preferential adsorption of EDTA on {001} facets of NaY(MoO4)2 crystal is responsible for the morphology evolution of NaY(MoO4)2 microcrystals with different aspect ratios and tunable exposed facets. Specifically, the present synthetic approach is also extendable to achieve NaHo(MoO4)2 and NaTb(MoO4)2 microcrystals with different shapes and tunable faceting. The photoluminescence measurement indicates that the luminescence properties of NaY(MoO4)2:Eu3+ microcrystals are highly dependent on their shape, size and exposed facets, and the sharp NaY(MoO4)2:Eu3+ bipyramids exhibit the strongest emission intensity, due to their larger size, less defects and less exposed facets. As such, the sharp NaY(MoO4)2:Eu3+ bipyramids may have potential applications in light display systems as well as optoelectronic devices, while the present EDTA-mediated hydrothermal synthetic strategy represents a potentially versatile and reliable means of achieving nano/microcrystals with well-defined morphology and tunable exposed facets.Acknowledgements
This project was supported by the National Natural Science Foundation of China (Grant no. 21073084 and 20773065), Specialized Research Fund for the Doctoral Program of Higher Education (SRFDP, Grant no. 20130091110010), Natural Science Foundation of Jiangsu Province (Grant no. BK2011438), National Basic Research (973) Program of China (Grant no. 2009CB623504), National Science Fund for Talent Training in Basic Science (Grant no. J1103310), and the Modern Analysis Center of Nanjing University.References
- X. Liu, D. Wang and Y. Li, Nano Today, 2012, 7, 448 CrossRef CAS PubMed.
- C. X. Burda, C. R. Narayanan and M. A. El-Sayed, Chem. Rev., 2005, 105, 1025 CrossRef CAS PubMed.
- Y. Xia, Y. J. Xiong, B. Lim and S. E. Skrabalak, Angew. Chem., Int. Ed., 2009, 48, 60 CrossRef CAS PubMed.
- W.-N. Li, J. Yuan, X.-F. Shen, S. Gomez-Mower, L.-P. Xu, S. Sithambaram, M. Aindow and S. L. Suib, Adv. Funct. Mater., 2006, 16, 1247 CrossRef CAS.
- G. Liu, J. C. Yu, G. Lu and H.-M. Cheng, Chem. Commun., 2011, 47, 6763 RSC.
- D.-F. Zhang, H. Zhang, L. Guo, K. Zheng, X.-D. Han and Z. Zhang, J. Mater. Chem., 2009, 19, 5220 RSC.
- B. Lv, Z. Liu, H. Tian, Y. Xu, D. Wu and Y. Sun, Adv. Funct. Mater., 2010, 20, 3987 CrossRef CAS.
- M. H. Huang and P.-H. Lin, Adv. Funct. Mater., 2012, 22, 14 CrossRef CAS.
- X. Xie, Y. Li, Z. Q. Liu, M. Haruta and W. Shen, Nature, 2009, 458, 746 CrossRef CAS PubMed.
- K. M. Bratlie, H. Lee, K. Komvopoulos, P. Yang and G. A. Somorjai, Nano Lett., 2007, 7, 3097 CrossRef CAS PubMed.
- K. Zhou and Y. Li, Angew. Chem., Int. Ed., 2012, 51, 602 CrossRef CAS PubMed.
- T. R. Gordon, M. Cargnello, T. Paik, F. Mangolini, R. T. Weber, P. Fornasiero and C. B. Murray, J. Am. Chem. Soc., 2012, 134, 6751 CrossRef CAS PubMed.
- H. Wang, J. Yang, X. Li, H. Zhang, J. Li and L. Guo, Small, 2012, 8, 2802 CrossRef CAS PubMed.
- Y. Bi, S. Ouyang, N. Umezawa, J. Cao and J. Ye, J. Am. Chem. Soc., 2011, 133, 6490 CrossRef CAS PubMed.
- J. W. Hong, S. W. Kang, B.-S. Choi, D. Kim, S. B. Lee and S. W. Han, ACS Nano, 2012, 6, 2410 CrossRef CAS PubMed.
- M. Jin, H. Zhang, Z. Xie and Y. Xia, Energy Environ. Sci., 2012, 5, 6352 CAS.
- X. Han, M. Jin, S. Xie, Q. Kuang, Z. Jiang, Y. Jiang, Z. Xie and L. Zheng, Angew. Chem., Int. Ed., 2009, 48, 9180 CrossRef CAS PubMed.
- M. J. Siegfried and K.-S. Choi, J. Am. Chem. Soc., 2006, 128, 10356 CrossRef CAS PubMed.
- C. G. Read, E. M. P. Steinmiller and K.-S. Choi, J. Am. Chem. Soc., 2009, 131, 12040 CrossRef CAS PubMed.
- X. Xiao, X. Liu, H. Zhao, D. Chen, F. Liu, J. Xiang, Z. Hu and Y. Li, Adv. Mater., 2012, 24, 5762 CrossRef CAS PubMed.
- Y. W. Jun, M. F. Casula, J. H. Sim, S. Y. Kim, J. Cheon and A. P. Alivisatos, J. Am. Chem. Soc., 2003, 125, 15981 CrossRef CAS PubMed.
- T. Zhang, W. Dong, M. Keeter-Brewer, S. Konar, R. N. Njabon and Z. R. Tian, J. Am. Chem. Soc., 2006, 128, 10960 CrossRef CAS PubMed.
- L. F. Gou and C. J. Murphy, Nano Lett., 2003, 3, 231 CrossRef CAS.
- C. Li, K. L. Shuford, M. Chen, E. J. Lee and S. O. Cho, ACS Nano, 2008, 2, 1760 CrossRef CAS PubMed.
- Z. Xu, X. Kong, C. Li, Z. Hou, C. Zhang, D. Yang, G. Li and J. Lin, Inorg. Chem., 2010, 49, 6706 CrossRef CAS PubMed.
- L. Qian, J. Zhu, Z. Chen, Y. Gui, Q. Gong, Y. Yuan, J. Zai and X. Qian, Chem.–Eur. J., 2009, 15, 1233 CrossRef CAS PubMed.
- L. Xu, J. Shen, C. Lu, Y. Chen and W. Hou, Cryst. Growth Des., 2009, 9, 3129 CAS.
- L. Xu, C. Lu, Z. Zhang, X. Yang and W. Hou, Nanoscale, 2010, 2, 995 RSC.
- L. Xu, X. Yang, Z. Zhai, X. Chao, Z. Zhang and W. Hou, CrystEngComm, 2011, 13, 4921 RSC.
- L. Xu, X. Yang, Z. Zhai and W. Hou, CrystEngComm, 2011, 13, 7267 RSC.
- L. Xu, X. Yang, Z. Zhai, D. Gu, H. Pang and W. Hou, CrystEngComm, 2012, 14, 7330 RSC.
- A. A. Kaminskii, J. J. Eichler, K. Ueda, N. V. Klassen, B. S. Redkin, L. E. Li, J. Findeisen, D. Jaque, J. Garcia-Sole, J. Fernandez and R. Balda, Appl. Opt., 1999, 38, 4533 CrossRef CAS.
- Y. Hu, W. Zhuang, H. Ye, D. Wang, S. Zhang and X. Huang, J. Alloys Compd., 2005, 390, 226 CrossRef CAS PubMed.
- F. Esteban-Betegón, C. Zaldo and C. Cascale, Chem. Mater., 2010, 22, 2315 CrossRef.
- F. Shi, J. Meng and Y. Ren, J. Solid State Chem., 1996, 121, 236 CrossRef CAS.
- W. Bu, Z. Chen, F. Chen and J. Shi, J. Phys. Chem. C, 2009, 113, 12176 CAS.
- Z. Xu, C. Li, G. Li, R. Chai, C. Peng, D. Yang and J. Lin, J. Phys. Chem. C, 2010, 114, 2573 CAS.
- S.-S. Liu, D.-P. Yang, D.-K. Ma, S. Wang, T.-D. Tang and S.-M. Huang, Chem. Commun., 2011, 47, 8013 RSC.
- J. Liu, B. Xu, C. Song, H. Luo, X. Zou, L. Han and X. Yu, CrystEngComm, 2012, 14, 2936 RSC.
- Y. Li, G. Wang, K. Pan, W. Zhou, C. Wang, N. Fan, Y. Chen, Q. Feng and B. Zhao, CrystEngComm, 2012, 14, 5015 RSC.
- Y. Li, G. Wang, K. Pan, Y. Qu, S. Liu and L. Feng, Dalton Trans., 2013, 42, 3366 RSC.
- J. W. Mullin, Crystallization, Butterworth-Heinemann, Oxford, 3rd edn, 1997 Search PubMed.
- Z. L. Wang, J. Phys. Chem. B, 2000, 104, 1153 CrossRef CAS.
- J. D. H. Donnay and D. Harker, Am. Mineral., 1937, 22, 446 CAS.
- B. R. Judd, Phys. Rev., 1962, 127, 750 CrossRef CAS.
- G. S. Ofelt, J. Chem. Phys., 1962, 37, 511 CrossRef CAS PubMed.
- J. Yang, C. Li, Z. Cheng, X. Zhang, Z. Quan, C. Zhang and J. Lin, J. Phys. Chem. C, 2007, 111, 18148 CAS.
- C.-L. Lo, J.-G. Duh, B.-S. Chiou, C.-C. Peng and L. Ozawa, Mater. Chem. Phys., 2001, 71, 179 CrossRef CAS.
- Y. Zeng, Z. Li, L. Wang and Y. Xiong, CrystEngComm, 2012, 14, 7043 RSC.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra00244j |
| This journal is © The Royal Society of Chemistry 2014 |