Modulation of the organelle specificity in Re(i) tetrazolato complexes leads to labeling of lipid droplets†
Abstract
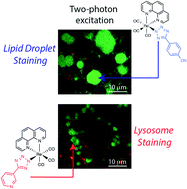
The biological behaviour in terms of cellular incubation and organelle specificity for two complexes of the type fac-[Re(CO)3(phen)L], where phen is 1,10-phenanthroline and L is either 3-pyridyltetrazolate or 4-cyanophenyltetrazolate, are herein investigated. The emission signal detected from the live insect Drosophila and human cell lines, generated by exploiting two-photon excitation at 830 nm to reduce cellular damage and autofluorescence, suggests photophysical properties that are analogous to those measured from dilute solutions, meaning that the complexes remain intact within the cellular environment. Moreover, the rhenium complex linked to 4-cyanophenyltetrazolate shows high specificity for the lipid droplets, whereas the complex bound to 3-pyridyltetrazolate tends to localise within the lysosomes. This differential localisation implies that in these complexes, organelle specificity can be achieved and manipulated by simple functional group transformations thus avoiding more complex bioconjugation strategies. More importantly, these results highlight the first example of phosphorescent labeling of the lipid droplets, whose important cellular functions have been recently highlighted along with the fact that their role in the metabolism of healthy and diseased cells has not been fully elucidated.

 Please wait while we load your content...
Please wait while we load your content...