The self-aggregation of fluorophore-triphenylamine nanostructures with tunable luminescent properties: the effect of acidity and rare earth ions†
Qi-yu Chena,
Lin Kong*a,
Yu-peng Tianab,
Xian-yun Xua,
Long-Mei Yanga,
Gao-bin Zhanga,
Wen-bin Jiaa and
Jia-xiang Yang*ab
aAnhui University, Department of Chemistry, Hefei, Anhui, China. E-mail: kong_lin2009@126.com; jxyang@ahu.edu.cn
bState Key Laboratory Materials, Shandong University, Jinan 502100, P. R. China
First published on 28th February 2014
Abstract
A D–π–A type triphenylamine derivative 3-(4-(diphenylamino)phenyl)acrylic acid (abbreviated as L) is designed, synthesized and characterized by single crystal X-ray diffraction analysis. The self-aggregation of L nanostructures in different acidity aqueous solutions and/or under the guidance of rare earth (RE) ions is studied, along with the corresponding optical properties. In strong acidic conditions, L molecules self-assemble to form two dimensional nanostructures. While in neutral aqueous solutions, L tends to form nanorods. In the strong alkali environment, nanofibers were obtained. Moreover, in acidic conditions, the existence of Tm3+ induces the L molecules to self-aggregate into nanoplates. Flower-like six branched structures are observed when Yb3+ is used. The changed morphology leads to tunable linear optical properties.
1. Introduction
Over the past few decades, increasing interest has been received about functional organic materials composed of optically and electronically active constituents in nanoscience and nano-technology.1–3 The tunable physical properties of organic nanomaterials can be controlled through their size, shape and crystallinity, which are expected to provide an opportunity to control the optoelectronic properties and further device performance.4–6 A multitude of cooperative and simultaneous intermolecular interactions, such as hydrogen bond, van der Waals, π–π stacking and electrostatic attractions, can affect the final structure and further adjust the corresponding properties.7–9 In addition, the concentration, temperature, surfactants and solution acidity can also be used to alter or control the above-mentioned interactions and further cause the change of morphology.10–13 However, to the best of our knowledge, little research has been carried about using RE metal ions to induce and/or control the morphology and/or particle size of an organic nanostructure.Rare earth ions have been extensively studied in the fundamental and technical field over the past decades due to their particular characteristics arising from the 4f and/or 5f electronic shells,14,15 which have a great potential to be used in luminescent devices, magnets, catalysts and other functional materials.16–18 Compounds consisting of RE ions also have significant commercial applications in radiation intensifying screens, X-ray-computed tomography, oxygen storage and medical imaging radiation detectors.19,20 It is well known that there are significant intermolecular interactions between carboxylate compounds and the RE ions.21 According to the hard–soft acid–base principle, RE ions should have a high tendency to combine with the O atoms of these carboxylate compounds. Thus, the interactions between the RE ions and the oxygen atoms of the carboxylate ligands are somewhat strong. Up to now, the use of RE ions as efficient inductive agents to build organic nanostructures has rarely been reported. Therefore, in this study, the interaction between the RE ion and carboxylate derivative is used to regulate the morphology.
In many functional organic materials, triphenylamine and its derivatives are well-known for their 3-D propeller shapes which have been widely investigated and applied in organic field-effect transistors and organic light-emitting diodes, as well as second order nonlinear optical devices.22–24 In many cases, triphenylamine groups can be employed as electron-donors and used to prepare D–π–A type molecules (D = donor, A = acceptor) through linkage with an electron-acceptor group, which are regarded as ideal candidates for red emitting and nonlinear optical materials.25,26 To increase the interactions between the RE ions and the organic matrix, the carboxylic group is introduced into the triphenylamine group, which can also improve the solubility of the as-prepared organic compound in water as well and further present a bright future for the application.
Considering all the aspects above, in this work, an ICT structural triphenylamine derivative, L (Scheme 1), was designed and synthesized, in which the triphenylamine group was employed as an electron-donor unit and the carboxyl group was employed as an electron-acceptor unit. Furthermore, nanostructures of L were prepared under different conditions. The influence of the solution acidity (pH), Tm3+ and Yb3+ ions on the morphology and the corresponding linear optical properties of L were discussed. It is found that pH plays an important role in the formation of the morphology. The morphology of L has a tendency to accumulate and form semi two dimensional nanostructure in the excessive acid aqueous solution and one dimensional nanorods in a neutral aqueous solution. However, in the alkaline solution, the surface of the L nanostructures was etched due to the acid–base reaction and eventually nanofibers were observed. Furthermore, the interactions between L and the RE ions through a simple process at room temperature result in a completely different morphology. In brief, by controlling the acidity of the solution and the RE metal ions, the self-assembly of 1-D or semi 2-D organic materials can be achieved. The existence of Tm3+ induces nanoplates in acidic conditions. A flower-like six branched structure is obtained when Yb3+ is used in acidic conditions.
2. Experimental section
2.1 Materials and instrument
All reagents used in synthesis route were of analytical reagent grade. The pH of the solution was adjusted by adding hydrochloric acid or sodium hydroxide in water and demarcated with the pH meter of PHS-3CT. Metal salts Tm(NO3)3·5H2O, Yb(NO3)3·5H2O were prepared from the corresponding metal oxidate and concentrated nitric acid.The X-ray diffraction measurement of single crystal was performed on a Bruker SMART CCD area detector using graphite-monochromated Mo Ka radiation (λ = 0.71073 Å) at 298(2) K. The intensity data were collected in the variable ω-scan mode. The structures were solved by direct methods and difference Fourier syntheses. All non-hydrogen atoms were refined anisotropically and hydrogen atoms were introduced geometrically. Calculations were performed with the SHELXTL-97 program package.27 The morphologies were obtained by using a field-emission scanning electron microscope (FESEM, Hitachi S-4800). The UV-vis absorption spectra were obtained on a UV-3100 spectrophotometer in the wavelength range 200–700 nm. The fluorescence spectra were measured at room temperature using a Hitachi F-7000 spectrophotometer.
2.2 The synthesis of L
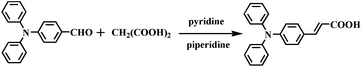
4-Diphenylaminobenzaldehyde (0.5 g, 1.8 mmol) and malonic acid (0.0187 g, 1.8 mmol) were dissolved in pyridine (5 mL) and 3–5 drops of piperidine was added as a catalyzer. The above mixture was refluxed for 4 h and monitored by TLC to ensure a complete reaction. Then, water was added to the mixture and the precipitate was purified through recrystallization in ethanol solution to get a yellow solid. Yield: 95%. 1H NMR (400 MHz, d6-DMSO): δ (ppm) 12.22 (s, 1H); 7.56 (d, 2H, J = 6.8 Hz); 7.50 (d, 1H, J = 16.0 Hz); 7.36 (t, 4H, J = 7.6 Hz); 7.13 (t, 2H, J = 7.2 Hz); 7.09 (d, 4H, J = 8.0 Hz); 6.89 (d, 2H, J = 8.0 Hz); 6.34 (d, 1H, J = 16.0 Hz); 13C NMR (100 MHz): δ (ppm) 167.54, 146.34, 143.49, 129.75, 129.54, 127.21, 125.01, 124.15, 120.93, 116.39. FT-IR (KBr, cm−1): ν = 3029 (m, νCOO), 1681 (s, νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1622 (w, νC
O), 1622 (w, νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1584 (s), 980 (w), 829 (w), 753 (m), 698 (s).
C), 1584 (s), 980 (w), 829 (w), 753 (m), 698 (s).
Yellow crystals of L suitable for X-ray diffraction were obtained by the slow evaporation of a dichloromethane–methanol mixed solution (VCH2Cl2/VMeOH = 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at room temperature for a week.
1) at room temperature for a week.
2.3 The preparation of aggregates
A stock ethanol solution of L with a concentration of 1.0 × 10−3 mol L−1 (abbreviated as M) was prepared. Stock aqueous solutions of different acidity (pH values of which were 1, 3, 4, 5, 7, 9 and 13, respectively) and stock aqueous solutions of RE ions (Tm3+, Yb3+) were also prepared.The organic nanostructures were prepared through a simple reprecipitation process. For studying the effect of the acidity on the aggregation, 200 μL of an L–ethanol solution was injected into 2 mL of high-purity water with different acidity under stirring. For exploring the effect of RE ions on the aggregation, the aqueous solution of Tm3+ and/or Yb3+ was rapidly added into 2 mL of high-purity water and/or aqueous solutions with different acidity with stirring for 3 min. Then, 200 μL of L–ethanol solution was injected into the above solution under stirring. The samples were stirred for 10 min and then left undisturbed for 12 h at room temperature for stabilization.
2.4 TD-DFT calculation
The molecular orbital calculations of time-dependent density functional theory (TD-DFT) at the pbe1pbe/6-31g level basis set (Gaussian 09) were performed to study the electronic structures of L.28 The molecular geometry used for the calculation was obtained from the X-ray diffraction crystallographic data.3. Results and discussion
3.1 Structural characterization
L crystallized in the monoclinic form with the space group P2(1)/n as shown in Fig. 1. The crystallography data are summarized in Table 1. Selected bond lengths and bond angles are listed in ESI Table S1.† In the molecule, the bond lengths were all of aromatic character. The linkage bond length between the benzene ring and the carboxyl group was quite conjugated with C16–C19 being 1.399(12) Å. The C19![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C20 double bond was nearly coplanar to the adjacent benzene and carboxyl group with the torsion angle of C17–C16–C19–C20 being 176.01(16)° and O2–C21–C20–C19 being 179.90(15)°. The structural features suggested that all nonhydrogen atoms were highly conjugated and nearly coplanar, which would favour the electronic delocalization in the whole molecule.
C20 double bond was nearly coplanar to the adjacent benzene and carboxyl group with the torsion angle of C17–C16–C19–C20 being 176.01(16)° and O2–C21–C20–C19 being 179.90(15)°. The structural features suggested that all nonhydrogen atoms were highly conjugated and nearly coplanar, which would favour the electronic delocalization in the whole molecule.
| Empirical formula | C21H17NO2 | a [Å] | 13.716(5) |
| Formula weight | 315.36 | b [Å] | 8.084(5) |
| Crystal system | Monoclinic | c [Å] | 15.868(5) |
| Space group | P2(1)/n | β [deg] | 100.602(5) |
| Temperature (K) | 298(2) | V [Å3] | 1729.4(14) |
| Z | 4 | Dcalcd[g cm−3] | 1.211 |
| F (000) | 664 | Goodness of fit on F2 | 1.032 |
| Crystal size (mm) | 0.30 × 0.20 × 0.20 | Reflections collected/unique | 2366 |
| Final R indices [I > 2sigma(I)] | R1 = 0.0409, wR2 = 0.1329 | R indices (all data) | R1 = 0.0549, wR2 = 0.1497 |
The adjacent molecules are stacked through C–H⋯C weak interactions along the same direction (b axis) at a short intermolecular distance between 2.804 and 2.854 Å to form a 1-D structure as shown in Fig. S1a.† There is also an O2–H2⋯O1 intermolecular hydrogen bond with the H⋯O distance of 1.842 Å (C–H⋯O = 178.59°), which is very important in supramolecular crystal packing to form 2-D sheets in the ab plane (Fig. S1b†). There also exists a C10–H10⋯O1 intermolecular hydrogen bond with the H⋯O distance of 2.349 Å (C–H⋯O = 163.56°), which connects with the C–H⋯C weak interactions mentioned above to form 2-D sheets in the bc plane. Such weak interactions are somewhat strong and supposed to be the main driving forces for the molecules self-assembly along a certain direction to form bulk materials and/or nanomaterials.
3.2 The effect of the solvent on the aggregation of L
In the present work, the L nanostructures are simply prepared in several solvents with no addition of any surfactant, template or catalyst. The SEM images of the L self-assembly aggregations from different solvents are shown in Fig. 2. The solvent-induced morphological change of L is remarkable. Monodisperse spherical nanoparticles are observed in a DMF–H2O mixed solvent with an average diameter of about 80 nm and a good dispersion (Fig. 2a). Nanobelts are observed in acetonitrile–H2O with the length of about 10 μm and the width of about 500 nm (Fig. 2b), which have a tendency to aggregate. The main products in acetone–H2O are nanorods with lengths of about 1.3 μm and an average width of about 140 nm (Fig. 2c), which are directionally arranged one by one. Moreover, nanorods with lengths of about 10 μm and an average width of about 200 nm are obtained in ethanol–H2O (Fig. 2d). This solvent-based morphological variation can be attributed to the different intensity of the noncovalent intermolecular interactions between L–L and L–solvent.29 Owing to the fact that the morphology of L obtained from ethanol was more irregular than that obtained from the other solvents, the following experiments were carried out in an ethanol solution in this work. | ||
| Fig. 2 The morphologies of L from different mixed solvents: (a) DMF–H2O, (b) acetonitrile–H2O, (c) acetone–H2O, (d) ethanol–H2O. | ||
3.3 The effect of the acidity on the aggregation of L
As L is a carboxylic acid derivative, the acidity of the solution may change the weak intermolecular interactions and further influence the morphology. In order to study the influence of the acidity of the solution on the morphology, experiments were conducted by adjusting the acidity of the EtOH–water mixed solution as shown in Fig. 3. When the experiment was carried out in the neutral conditions of pH = 7, nanorods with smooth surfaces were observed with lengths and diameters of ∼10 μm and ∼200 nm, respectively (Fig. 3d), which fit well with the result shown in Fig. 2d. | ||
| Fig. 3 The SEM images of the L nanostructures prepared in an aqueous solution with different acidities (a) pH = 1, (b) pH = 3, (c) pH = 5, (d) pH = 7, (e) pH = 9, (f) pH = 13. | ||
As the acidity of the solution varied, a remarkable change in the L self-aggregation was observed. When the acidity of the solution increased, the morphology gradually changed from nanorods to ordered superimposed flake-like semi 2-D structures. In detail, when the acidity was pH = 5 (Fig. 3c), there is a slight change in the morphology compared to that obtained in the neutral conditions. The nanorods in the decentralized state appeared and the length decreased to ∼1.5 μm. When the acidity increased to pH = 4, two different morphologies of a stacked structure and dispersed nanorods coexisted (Fig. S2†). The stacked structure was made of multiple nanorods arranged orderly with a length of about 3 μm and a thickness of about 1.5–2 μm. The dispersed nanorods had an average length of about 4 μm. The results indicated that the driving force of the formation of different nanostructure was probably a complex balance of intermolecular interactions.
Furthermore, as the acidity increased to pH = 3, the aggregated nanorods were observed to form a semi 2-D structure (Fig. 3b). The length and diameter of a single nanorod were ∼1 μm and 200 nm, respectively. These nanorods gathered to form a semi 2-D structure with ∼20 μm length and ∼15 μm width. The border between the adjacent nanorods was clearly observed, which showed the aggregation and weak intermolecular interactions along two directions. When the acidity of the preparation environment increased to pH = 1, superimposed flake-like structures were obtained (Fig. 3a). A single nanoflake possessed a width of about 300 nm, a thickness of 70–80 nm and an average length of about 1.2–1.6 μm. Several nanoflakes stacked closely and the interface was clearly observed. The nanoflake had a high morphological purity as other morphologies were seldom captured under such conditions.
Another interesting trend of morphological change was observed when the L molecules were put in an alkaline medium. When the acidity of the aqueous solution reached to pH = 9, one-armed nanotubes were obtained (Fig. 3e) with a length of about 3–5 μm and an average bore diameter of 100 nm. The formation of one-armed nanotubes was considered as coming from the erosion effect of the nanorods. The surface of the nanotubes was rough, which may be due to the erosion effect caused by the interactions between the alkali and carboxylic group. These one-armed nanotubes arranged orderly and stacked into a quasi 2-D structure. Furthermore, when the acidity reached to pH = 13, nanofibers arose with the length of ∼30 μm and width of about 200 nm (Fig. 3f), which was a marked distinction compared with the nanorods obtained in pH = 7. Moreover, the growth direction of these nanofibers was basically the same in the visual field, which revealed that the intermolecular interactions along this direction were the main driving forces to form the nanofibers.
When an L–EtOH solution was injected into pure water, the sudden change in the environment of the L molecules induced molecular segregation from ethanol and precipitation in water to form seed crystals. In the subsequent growth process, the strong intermolecular D–A dipole–dipole interactions between adjacent L molecules acted as the driving force for the aggregation of the L molecules. The limited solubility of L in the poor solvent (water), together with the strong intermolecular dipole–dipole interactions, drove L molecules to align orderly and assemble mainly along the direction of the dipole moment, and thus 1-D nanorods were formed.
While in the acid solution, the carboxylic acid unit of L was fully protonated. Thus, the hydrogen bond between the carboxyl groups of L and water would be strong. The process would decrease the probability of the formation of O–H⋯O hydrogen bonds. Thus, the proportion and the corresponding strength of the C–H⋯C weak interactions between the triphenylamine group and the π–π weak interactions between the conjugated groups of adjacent molecules would increase and be comparable to that of O–H⋯O hydrogen bonds. The three types of weak interactions would be the main driving force for the molecular packing which leads to the creation of accumulation from 1-D morphology to 2-D structures.30 However, when the environment changed to alkaline condition, the carboxyl group was expected to be deprotonated, which caused the intermolecular hydrogen bond to weakened and even disappeared. Thus, the C–H⋯C weak interactions were dominant in the molecular self-assembly, resulting in a more obviously 1-D orientational growth.31
3.4 The morphology of the L nanostructure induced by RE ions
Through the above study, it is concluded that the solvent and acidity of the solution have a significant effect on the self-aggregation of the L molecules. Furthermore, the effect of the RE metal ions on the morphology of L was studied. A more interesting change in the morphology was expected based on the strong interactions between the RE metal ions and the O atoms of L. To explore the aim, the aqueous solution of Tm3+ (4 × 10−4 M) was added to the reaction system. A cross-like structure was observed, which had a length of about 6–10 μm and the larger angle made of two nanorods was about 100° (Fig. 4a). As the concentration of Tm3+ increased, the molecular interactions of a certain direction became enhanced and led to the increased length of the nanorods and a small quantity of impurities. The phenomenon was possibly caused by the hydrolysis of Tm3+ (Fig. S3†). In order to eliminate this hydrolysis effect, the solution was adjusted to acidic conditions. When the Tm3+ ions were added into the acidic aqueous solution, the morphology of L further changed. For example, when the sample was prepared in pH = 3 and the concentration of Tm3+ was 2 × 10−6 M, unordered rectangular nanoplates were obtained (Fig. 4b). However, as the concentration of Tm3+ gradually increased to 6 × 10−6 M, the nanoplate structures grew along a particular direction shown in Fig. 4c (the white solid lines are the virtual growth direction of the nanostructures). When the concentration of Tm3+ increased to 2 × 10−5 M, the nanoplate structure finally grew into a large size sheet-like structure with a length of about 30–50 μm and a width of about 4–20 μm (Fig. 4d). To further explore the influence of the different RE ions on the aggregation of the L molecules, another kind of RE ion, Yb3+, was investigated. A completely different morphology compared with that of Tm3+ was observed as shown in Fig. 5. Moreover, the concentration of the Yb3+ ions also influenced the L aggregation mode. When an aqueous solution of Yb3+ (8 × 10−5 M) was used, a flower-like nanorod structure was obtained, among which six nanorods extended in six directions revolved round the centre point (Fig. 5a) to form the six-branched flower. When the concentration of Yb3+ increased to 2 × 10−4 M, the length of the nanorods increased to ∼40 μm and the nanostructure gathered gradually (Fig. 5b). Also, to eliminate the hydrolysis effect, Yb3+ (6 × 10−5 M) was added in a solution of pH = 3, and regular hexagonal (Fig. 5c) and hexagonal prism (Fig. S4†) images can be observed with the average side length being 2 μm. These hexagonal flakes were believed to be the gem of the flower-like six-branched structures.3.5 Linear optical properties and theoretical calculations
Furthermore, the theoretical spectral characteristic showed two main transitions. One was from the configuration HOMO to LUMO with λabs = 367 nm (oscillator strength fHOMO–LUMO being 0.6745). The other optical transition corresponded to the configuration HOMO to LUMO+1 with λabs = 296 nm. The calculated results fit with the experimental data and the corresponding absorption bands were observable on the absorption spectra of the L–EtOH solution.
Furthermore, the UV-vis absorption spectra of the L nanostructures obtained in different acidity aqueous solutions were thoroughly studied. The results revealed that the π–π* transition of the whole conjugated structure changed a little with the change of the acid and alkali environment. The absorption band centred at 362 nm in acidic solution of pH = 1 and/or 3, which was 356 nm in the neutral environment of pH = 7, while it centred at 359 nm in an alkaline solution of pH = 13. At the same time, the absorption band from the triphenylamine fragment varied much more, which centred at 284 nm, 293 nm and 300 nm in acidic, neutral and alkaline solutions, respectively. Moreover, the ratio of the two peak values showed fluctuation. In an acidic solution of pH = 1 and 3, the absorbance ratio of A284/A362 was 0.3768, where the suffix represents the monitor wavelength. In a neutral solution of pH = 7, the absorbance ratio of A293/A365 increased to 0.5687. In an alkaline solution of pH = 13, the absorbance ratio showed a further increase to A300/A359 = 1.0869. The results meant that the electronic structure of the whole conjugated structure changed little with the different acidity of the solution. The phenomenon was reasonable in that the acidity of the solution changed the H atom of the carboxylic group, which was not included in the whole conjugated structure. On the other hand, the different acidity of the solution violently influenced the electronic structure of the triphenylamine fragment. The higher the acidity used, the more the influence occurred.
As for the fluorescence emission, there was a stable blue-shifted wavelength band which varied from acidic conditions to a neutral medium, and then to an alkaline medium. When the pH value of the solution was 1, the emission band centred at 484 nm. When the pH value increased to 7, the emission band blue shifted for about 9 nm to 475 nm, which further blue shifted for about 23 nm to 461 nm as pH = 13. The results meant that there was a great environmental influence on the fluorescence with the increased pH value.
The changes of the absorption and emission spectra in different acidity solutions indicated that, to a certain extent, the intermolecular interactions between the L–L molecules and/or L–solvent were different in the acid–base aqueous solution environment, as mentioned above, which then brought about significant changes in the morphology, size and optical properties.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at this moment), the emission showed a significant red shift to 470 nm with a quenched intensity. In the presence of the Tm3+ ions, the heavy atom effect and the energy transfer from L to the Tm3+ ions in the process of an f–f transition may cause the fluorescence quenching.34 The red shift might arise from the coordination effect between L and the Tm3+ ions. The action ‘hot spot’ was the O atoms of the carboxyl group, which was the electron-acceptor unit of L. The interactions may influence the electron distribution of L, which increased the electron cloud density of the carboxyl group and further increased the strength of the D–A pair, the direct result of which was the red-shift of the fluorescence.
1 at this moment), the emission showed a significant red shift to 470 nm with a quenched intensity. In the presence of the Tm3+ ions, the heavy atom effect and the energy transfer from L to the Tm3+ ions in the process of an f–f transition may cause the fluorescence quenching.34 The red shift might arise from the coordination effect between L and the Tm3+ ions. The action ‘hot spot’ was the O atoms of the carboxyl group, which was the electron-acceptor unit of L. The interactions may influence the electron distribution of L, which increased the electron cloud density of the carboxyl group and further increased the strength of the D–A pair, the direct result of which was the red-shift of the fluorescence.
 | ||
| Fig. 7 The fluorescence spectra of L–Tm (a), L–Yb (b) with different concentrations of RE metal ions. | ||
As the usage of Tm3+ increased, the fluorescence quenched regularly. Along with this quenched emission, a more interesting phenomenon appeared. When the concentration increased to 2 × 10−5 M (the ratio of L to Tm3+ was 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the fluorescence emission showed a wide peak, which centred at 470 nm with a shoulder peak at 513 nm. To further increase the concentration, the shoulder peak became more dominant. When the concentration increased to 2 × 10−3 M (the ratio of L to Tm3+ was 1
1), the fluorescence emission showed a wide peak, which centred at 470 nm with a shoulder peak at 513 nm. To further increase the concentration, the shoulder peak became more dominant. When the concentration increased to 2 × 10−3 M (the ratio of L to Tm3+ was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), the fluorescence band mainly centred at 513 nm. After this point, the emission band and the fluorescence intensity did not change any more.
2), the fluorescence band mainly centred at 513 nm. After this point, the emission band and the fluorescence intensity did not change any more.
The emission band at 513 nm can be considered as the inherent fluorescent emission of the L–Tm complex. When a small amount of Tm3+ was used, either the proportion of the L–Tm complex was very small in the solution or the L–Tm complex had not yet been generated. Thus, the fluorescence showed only a red shift emission. On increasing the usage of the Tm3+ ions, a strong coordination ability emerged and the inherent fluorescent emission at 513 nm appeared. When the concentration of the added Tm3+ ions increased to 2 × 10−3 M, the ratio of Tm3+ to L was 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in this condition. At this point, all of the L molecules may be coordinated with the Tm3+ ions according to the results mentioned in the SEM analysis. After this point, little change could be observed in the emission band and the fluorescence intensity. The results further confirmed that the optimum ratio of Tm3+ to L was 2
1 in this condition. At this point, all of the L molecules may be coordinated with the Tm3+ ions according to the results mentioned in the SEM analysis. After this point, little change could be observed in the emission band and the fluorescence intensity. The results further confirmed that the optimum ratio of Tm3+ to L was 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in this study. To achieve the perfect coordination of the Tm atom, some of the water molecules may be involved in the coordination effect.
1 in this study. To achieve the perfect coordination of the Tm atom, some of the water molecules may be involved in the coordination effect.
The fluorescence properties of the L–Yb nanohybrid were similar to those that occurred for L–Tm. With the increasing concentration of Yb3+, the emission was gradual quenched. When the concentration was 2 × 10−6 M (the ratio of L to Yb3+ was 500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), there appeared an emission band centred at 467 nm, which red shifted for about 17 nm compared to that of the L–EtOH solution. When the concentration increased to 8 × 10−5 M (the ratio of L to Yb3+ was 12.5
1), there appeared an emission band centred at 467 nm, which red shifted for about 17 nm compared to that of the L–EtOH solution. When the concentration increased to 8 × 10−5 M (the ratio of L to Yb3+ was 12.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the emission band at 467 nm disappeared, a new band centred at 515 nm appeared and the fluorescence intensity reached the minimum. Furthermore, the emission band and the fluorescence intensity did not change any more when the concentration continually increased.
1), the emission band at 467 nm disappeared, a new band centred at 515 nm appeared and the fluorescence intensity reached the minimum. Furthermore, the emission band and the fluorescence intensity did not change any more when the concentration continually increased.
In terms of the mechanism, the driving forces for the formation of L–RE probably related to several kinds of forces, such as the RE ions induced changing of the intermolecular interactions and the corresponding crystal growth process, because X-ray powder diffraction (XRD) patterns of the three samples (Fig. S5†) showed slight variations, such as the marked parts. Based on the results, the possible conversion process was illustrated in Scheme 2. In this process, when the RE ions were introduced, the metal ions influenced the intermolecular interactions of the L molecules and further changed the crystal growth mode of them. When the usage of the RE ions increased, the trend of change increased to result in L–RE nanohybrids.35 Moreover, different RE ions had a different influencing model. Thus, the crystal growth can be controlled by altering the species of the RE ion, which resulted in crystals with a different size and morphology. On the basis of the above analysis and SEM results, the possible conversion process may be as follows.
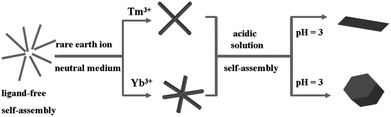 | ||
| Scheme 2 The possible formation mechanisms of L and the RE metal ions nano/submicrocrystals with multiform morphologies and size. | ||
When Tm3+ was used, there was a tendency of the L molecules to grow along four different directions and aggregate to form the cross-morphology. In the acidic conditions, the molecular interaction of the carboxylic group and the Tm3+ was restrained due to protonation, resulting in the receded force and the formation of unordered rectangular structures of nanoplates. As the concentration increased, the force of a particular direction enhanced to grow into a large size sheet-like nanostructure finally.
Similarly, the Yb3+ ions can induce L molecules grow along six different directions to form a hexagonal structure. The as-prepared hexagonal particles could serve as seeds to guide the subsequent growth of L–Yb to form the branched structures. To understand the formation process of the unique L–Yb architecture, intermediates of the product were traced. The existence of considerable hexagonal flakes was observed as typically shown in Fig. 5c. These hexagonal flakes were believed to be the gem of the six-branched L–Yb structures. The branched structure was formed from the crystalline growth of the tiny particles along six high-surface-energy angle sections of the hexagonal sheet.
4. Conclusion
In this work, the self-aggregation of the fluorophore-triphenylamine (L) nanostructures was studied in different conditions, including different solvents, different acidities of the solution, and under guide with the RE ions Tm3+ and/or Yb3+. The results revealed that the controlled synthesis of self-assembled organic materials can be realized through changing the growth environment, which can also optimize the optical properties. This type of controlled synthesis method also opens up the possibility of utilizing organic nanomaterials for a multitude of applications in bio- and chemical sensing and nanoelectronics.Acknowledgements
This work was supported by the NSFC (no: 21101001, 21271004, 51372003), the 211 project of AnHui University and the Natural Science Foundation of Anhui Province (1208085MB22).Notes and references
- Y. S. Zhao, A. D. Peng, H. B. Fu, Y. M. Ma and J. N. Yao, Adv. Mater., 2008, 20, 1661–1665 CrossRef CAS.
- H. B. Fu, B. H. Loo, D. B. Xiao, R. M. Xie, X. H. Ji, J. N. Yao, B. W. Zhang and L. Q. Zhang, Angew. Chem., Int. Ed., 2002, 41, 962–965 CrossRef CAS.
- D. H. Kim, D. Y. Lee, H. S. Lee, W. H. Lee, Y. H. Kim, J. I. Han and K. Cho, Adv. Mater., 2007, 19, 678–682 CrossRef CAS.
- J. G. Yu, F. R. F. Fan, S. L. Pan, V. M. Lynch, K. M. Omer and A. J. Bard, J. Am. Chem. Soc., 2008, 130, 7196–7197 CrossRef CAS PubMed.
- K. Takazawa, Y. Kitahama, Y. Kimura and G. Kido, Nano Lett., 2005, 5, 1293–1296 CrossRef CAS.
- A. L. Briseno, S. C. B. Mannsfeld, X. M. Lu, Y. J. Xiong, S. A. Jenekhe, Z. N. Bao and Y. N. Xia, Nano Lett., 2007, 7, 668–675 CrossRef CAS PubMed.
- Y. B. Wang, H. B. Fu, A. D. Peng, Y. S. Zhao, J. S. Ma, Y. Ma and J. J. Yao, Chem. Commun., 2007, 1623–1625 RSC.
- A. P. H. J. Schenning and E. W. Meijer, Chem. Commun., 2005, 3245–3258 RSC.
- D. W. P. M. Löwik, I. O. Shklyarevskiy, L. Ruizendaal, P. C. M. Christianen, J. C. Maan and J. C. M. van Hest, Adv. Mater., 2007, 19, 1191–1195 CrossRef.
- H. Katagi, H. Kasai, S. Okada, H. Oikawa, H. Matsuda and H. Nakanishi, J. Macromol. Sci., Part A: Pure Appl. Chem., 1997, 34, 2013–2024 CrossRef.
- H. Nakanishi and H. Katagi, Supramol. Sci., 1998, 5, 289–295 CrossRef CAS.
- T. Onodera, T. Oshikiri, H. Katagi, H. Kasai, S. Okada, H. Oikawa, M. Terauchi, M. Tanaka and H. Nakanishi, J. Cryst. Growth, 2001, 229, 586–590 CrossRef CAS.
- X. L. Qiao and Z. J. Zhang, J. Photochem. Photobiol., A, 2007, 190, 15–21 CrossRef CAS PubMed.
- G. Jia, K. Liu, Y. H. Zheng, Y. H. Song, M. Yang and H. P. You, J. Phys. Chem. C, 2009, 113, 6050–6055 CAS.
- J. Yang, C. X. Li, Z. W. Quan, C. M. Zhang, P. P. Yang, Y. Y. Li, C. C. Yu and J. Lin, J. Phys. Chem. C, 2008, 112, 12777–12785 CAS.
- S. Q. Ma, X. S. Wang, D. Q. Yuan and H. C. Zhou, Angew. Chem., Int. Ed., 2008, 47, 4130–4133 CrossRef CAS PubMed.
- B. L. Chen, L. B. Wang, F. Zapata, G. D. Qian and E. B. Lobkovsky, J. Am. Chem. Soc., 2008, 130, 6718–6719 CrossRef CAS PubMed.
- K. M. L. Taylor, J. D. Rocca, R. C. Huxford and W. B. Lin, Chem. Commun., 2010, 46, 5832–5849 RSC.
- R. Si, Y. W. Zhang, L. P. You and C. H. Yan, Angew. Chem., Int. Ed., 2005, 44, 3256–3260 CrossRef CAS PubMed.
- Y. W. Zhang, X. Sun, R. Si, L. P. You and C. H. Yan, J. Am. Chem. Soc., 2005, 127, 3260–3261 CrossRef CAS PubMed.
- Y. Li, F. K. Zheng, X. Liu, W. Q. Zou, G. Cong, C. Z. Lu and J. S. Huang, Inorg. Chem., 2006, 45, 6308–6316 CrossRef CAS PubMed.
- Y. B. Song, C. A. Di, X. D. Yang, S. P. Li, W. Xu, Y. Q. Liu, L. M. Yang, Z. G. Shuai, D. Q. Zhang and D. B. Zhu, J. Am. Chem. Soc., 2006, 128, 15940–15941 CrossRef CAS PubMed.
- Y. T. Tao, Q. Wang, L. Ao, C. Zhong, C. L. Yang, J. G. Qin and D. G. Ma, J. Phys. Chem. C, 2010, 114, 601–609 CAS.
- M. Faccini, M. Balakrishnan, M. B. J. Diemeer, Z. P. Hu, K. Clays, I. Asselberghs, A. Leinse, A. Driessen, D. N. Reinhoudt and W. Verboom, J. Mater. Chem., 2008, 18, 2141–2149 RSC.
- R. S. Moog, D. D. Kim, J. J. Oberle and S. G. Ostrowski, J. Phys. Chem. A, 2004, 108, 9294–9301 CrossRef CAS.
- D. Gudeika, A. Michaleviciute, J. V. Grazulevicius, R. Lygaitis, S. Grigalevicius, V. Jankauskas, A. Miasojedovas, S. Jursenas and G. Sini, J. Phys. Chem. C, 2012, 116, 14811–14819 CAS.
- G. M. Sheldrick, SHELXTL V.51 Software Reference Manual, Bruker AXS, Inc., Madison, Wisconsin, USA, 1997 Search PubMed.
- M. J. G. W. T. Frisch, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision B. 01, Gaussian, Inc., Wallingford CT, 2009 Search PubMed.
- L. J. Prins, D. N. Reinhoudt and P. Timmerman, Angew. Chem., Int. Ed., 2001, 40, 2382–2426 CrossRef CAS.
- T. Y. Wang, J. Jiang, Y. Liu, Z. B. Li and M. H. Liu, Langmuir, 2010, 26, 18694–18700 CrossRef CAS PubMed.
- T. Imae, Y. Takahashi and H. Muramatsu, J. Am. Chem. Soc., 1992, 114, 3414–3419 CrossRef CAS.
- X. L. Tang, W. M. Liu, J. S. Wu, C. S. Lee, J. J. You and P. F. Wang, J. Org. Chem., 2010, 75, 7273–7278 CrossRef CAS PubMed.
- (a) H. D. Ju, X. T. Tao, Y. Wan, J. H. Shi, J. X. Yang, Q. Xin, D. C. Zou and M. H. Jiang, Chem. Phys. Lett., 2006, 432, 321–325 CrossRef CAS PubMed; (b) Z. Zheng, Q. Zhang, Z. P. Yu, M. D. Yang, H. P. Zhou, J. Y. Wu and Y. P. Tian, J. Mater. Chem. C, 2013, 1, 822–830 RSC.
- S. Bano, A. Mohd, A. A. P. Khan and K. S. Siddiqi, J. Chem. Eng. Data, 2010, 55, 5759–5765 CrossRef CAS.
- J. Liu, B. Xu, C. Song, H. D. Luo, X. Zou, L. X. Han and X. B. Yu, CrystEngComm, 2012, 14, 2936–2943 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: selected bond lengths [Å] and angles [°] for L; the packing diagram of L; SEM image of L nanostructures prepared in pH = 4; SEM image of nanostructures of L with the excess concentration of Tm3+; SEM image of hexagonal prisms nanostructures when the Yb3+ was added to acidic aqueous solution of pH = 3. CCDC number 976845. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c3ra47893a |
| This journal is © The Royal Society of Chemistry 2014 |