Synthesis, characterization, and photo- and electro-luminescence of Ir(iii) complexes containing carrier transporting group-substituted β-diketonate ligand†
Abstract
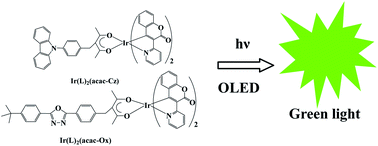
Two new iridium organometallic compounds based on acetylacetone derivative ligands containing carrier-transporting groups, Ir(L)2(acac-Ox) and Ir(L)2(acac-Cz), where L = 3-(pyridin-2-yl)coumarinato N, C4, acac-Ox = 3-(4-(5-(4′-tert-butylphenyl)-1,3,4-oxadiazole)benzyl)-pentane-2,4-dionate and acac-Cz = 3-((4-(9H-carbazol-9-yl)phenyl)methyl)pentane-2,4-dionate, have been successfully synthesized and characterized by elemental analysis, 1H NMR and FT-IR. The structure of the free ligand acac-Ox was established by single-crystal X-ray analysis as a cis-configurational enol of β-diketone. The photophysical properties of the complexes were examined by using UV-vis, photoluminescence spectroscopic analysis. The doped light-emitting devices with a configuration of ITO/MoO3 (2 nm)/NPB (35 nm)/TCTA (5 nm)/CBP:Ir(III) complex (x wt %, 20 nm)/TPBi (40 nm)/LiF (1 nm)/Al (150 nm) were fabricated. The devices based on Ir(L)2(acac-Cz) with a 9 wt% doping concentration showed the best EL efficiency performance, and exhibited green emission with a maximum external quantum efficiency (EQE) of 7.77% and a maximum luminous efficiency of 28.2 cd A−1 at a current density of 2.27 mA cm−2, and a maximum luminance of 6348.7 cd m−2 at 11 V. When the doping concentration is 6 wt%, a maximum brightness of 4230 cd m−2 at 16 V and a maximum current efficiency of 20.33 at 1.23 mA cm−2 and a maximum external quantum efficiency (EQE) of 5.54% were achieved in the devices based on Ir(L)2(acac-Ox). By comparison of the electroluminescent performances of the devices based on Ir(L)2(acac-Ox) and Ir(L)2(acac-Cz), it was shown that the introduction of the hole-transporting group into the ligand improves the performance of Ir(L)2(acac-Cz) doped devices.

 Please wait while we load your content...
Please wait while we load your content...