A study on the catalytic activity and theoretical modeling of a novel dual acidic mesoporous silica†
Majid Vafaeezadeh and
Alireza Fattahi*
Department of Chemistry, Sharif University of Technology, Tehran, Iran. E-mail: fattahi@sharif.edu; Fax: +98 21 66029165; Tel: +98 21 66005718
First published on 25th March 2014
Abstract
A novel mesoporous silica-functionalized dual Brønsted acidic species has been introduced as an efficient catalyst for solvent-free esterification of fatty acids with ethanol. The structure of the catalyst has been characterized by FT-IR spectroscopy, thermal gravimetric analysis (TGA), TEM and N2 adsorption–desorption. TGA of catalyst 1 showed no weight loss before 200 °C, indicating a high degree of hydrophobicity of the surface of the mesoporous silica. TEM images and nitrogen adsorption–desorption showed no noticeable changes to the structure of the catalyst before and after acid treatment. pH metric analysis was performed for the catalyst to determine the loading of the acidic sites. The structure of the catalyst was modeled by mimicking the surface of functionalized silica gel in the form of a cage-like cluster. Various conformers from the proposed structures were selected and optimized at the B3LYP/6-311++G** level of calculation. Natural bond orbital (NBO) analysis was performed to investigate the nature of hydrogen bonding of the catalyst in more detail. Based on the data gained from the optimized structures of the catalyst, a mechanism was proposed for the esterification reaction.
Introduction
Synthesis of fatty acid methyl/ethyl esters known as biodiesel has attracted special attention in the last decade1 as they are a good replacement of fossil fuels to diminish the global energy crisis. The traditional route to biodiesel production is transesterification of triglycerides with a short-chain alcohol (e.g., methanol or ethanol) using sodium hydroxide as a base catalyst.2 However, sodium hydroxide is a corrosive reagent and hence an additional neutralization step is required which leads to production of a high volume of salty waste. Furthermore, the residual free fatty acid (in the case of less expensive feedstock such as waste oil) is saponified by the homogeneous alkali catalyst, which increases the separation cost. In this regard, several approaches based on metal oxides (such as CaO and MgO) have been developed as heterogeneous solid catalysts.3 However, despite promising improvements, the leaching of the active sites and low surface area are among the drawbacks of these protocols which remain unresolved. Moreover, in some cases to gain sufficient biodiesel products the applied metal oxide should be mixed with strong bases such as MgO–KOH and MgO–NaOH systems.3dAcid catalyst esterification is a promising way to solve the drawbacks of the mentioned traditional base catalyst reactions. However, using homogeneous acid catalysts such as H2SO4, H3PO4, and HCl for esterification reaction have many drawbacks such as releasing hazardous wastes and difficulties in catalyst separation. It has been shown that various esterification reactions can be efficiently performed with heterogeneous acid catalysts as good replacements of the traditional hazardous and corrosive homogeneous acid catalysts.4 Esterification of the fatty acid with mesoporous silica–sulfonic acid improves reaction efficiency, catalyst separation, and reusability.1a,5 In comparison to carbon-based sulfonic acid catalyst, SiO2-based sulfonic acids have some benefits such as high loading of acidic centers, thermal and hydrothermal stability of the silica backbone, and avoiding usage of concentrated sulfuric acid and elevated temperature during the catalyst preparation.
However, the major drawback of these types of catalysts is that the loading of organic groups are typically low in the organically modified mesoporous silica. In the case of supported sulfonic acid this phenomena causes some technical problems especially in the solvent-free reactions such as inefficient mixing of reactant(s) and catalysts. To the best of our knowledge, only few methods are developed to design heterogeneous acid catalysts with enhanced loading of acidic sites.6 In this regard, Karimi and co-worker introduced that SBA-15 propylsulfonic acid-confined Brønsted acidic ionic liquid can efficiently catalyze some organic transformations at room temperature without using special methodologies.7 The authors proposed that the existence of a synergistic effect between the grafted sulfonic acid groups and hydrogensulfate counter anion of the supported ionic liquid accompanied by hydrophobic nature of the catalyst is the main reason of the extraordinary activity of the catalyst. Other successful examples of the synthesis of dual Brønsted acidic catalysts were reported by Luo and co-workers.8
They showed that both amorphous silica gel and silica coated magnetic nanoparticles functionalized with dual Brønsted acidic ionic liquid can catalyze some multicomponent reactions under solvent-free conditions.8 Mesoporous silica with two Brønsted acidic sites has also been synthesized and applied for efficient thioacetalization of various carbonyl compounds in water.9 The structure of this catalyst was theoretically studied to gain useful insight into the orientation of organic groups which were attached to the surface of the catalyst. In the current work, we wish to report detailed studies on the direct esterification of fatty acid with a novel organically modified mesoporous silica gel containing dual Brønsted acidic species. The method for preparation of the catalyst is simple and can be perform with commercially available starting materials. In this method fatty acid esterification has been performed without using any additional water-removal step (such as azeotropic distillation) and using molecular sieves or concentrated sulfuric acid. Moreover, we have reported novel theoretical modeling techniques to study the structure of the catalyst for better determining and introducing the nature of interactions of active sites with high level of calculations. In this regards, a cage-like cluster of the silica gel which was functionalized with organic group has been selected to mimic the surface of the catalyst.
Results and discussion
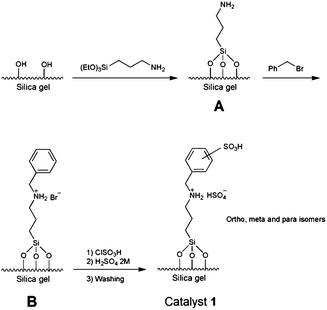
As stated in the experimental section, catalyst 1 was simply prepared by grafting of the commercially available amine precursor onto the high surface area silica gel (Scheme 1, A). This prepared material easily reacts with benzyl bromide to afford surface-bonded ammonium bromide specie (B). Chlorosulfonic acid treatment of the material10 followed by acidification and washing afforded catalyst 1 as a dry and slightly cream powder (Scheme 1).To investigate the structure of the catalyst and intermediates, comparative FT-IR spectra were performed for pure silica, material A and catalyst 1. As shown in Fig. 1, the top spectrum is for pure silica, while the next two are for intermediate A and the final catalyst 1. Intermediate A and catalyst 1 show important differences relative to the pure silica.
The bands at 2852 and 2923 cm−1 in the spectra of the intermediate A belong to C–H and N–H stretching frequencies in the surface-bonded aminopropyl functionalities. In the case of catalyst 1, another band at 3041 cm−1 can be interpreted as C–H stretching frequency of benzyl group moiety. The band at 582 cm−1 with relatively high intensity belongs to the S–O frequency of –SO3H group. The double band frequency (S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of –SO3H group seems to be overlapped with the broad band of the pure silica gel around the 1100 cm−1. It is worth to note that the bands around 1470 and 1740 cm−1 in both A and catalyst 1 are attributed to the N–H bending frequencies of the surface-bonded amine and/or ammonium functionalities.
O) of –SO3H group seems to be overlapped with the broad band of the pure silica gel around the 1100 cm−1. It is worth to note that the bands around 1470 and 1740 cm−1 in both A and catalyst 1 are attributed to the N–H bending frequencies of the surface-bonded amine and/or ammonium functionalities.
Silica-based catalysts inherently tend to absorb humidity and/or water molecules which participate in the reaction media in the form solvent, impurity or generated as by-product. In the case of silica supported acid catalyst this phenomenon is more important than others.
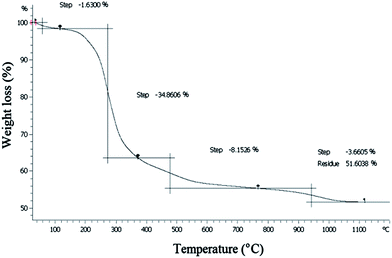
As previously studied by van Grieken et al., “acid solids are subjected to poisoning with water in reactions where this highly polar molecule is involved”.11 Typically, the presence of water in the silica-based catalyst is explained as a presence of a sharp weight loss which usually occurs around 100 °C in the thermal gravimetric analysis (TGA) of the catalyst.7b However, as shown in Fig. 2 no detectable weight loss could be assigned up to 200 °C in the thermogram of the catalyst 1. This observation can be interpreted via the fact that the surface of the mesoporous silica modified with organic groups has high degree of hydrophobicity. This hydrophobicity has two important effects in the esterification reaction: (i) the functionalized silica gel does not have tendency to absorb water and thus no significant deactivation of catalyst 1 occurs during the reaction (ii) it facilitates mass transfer of organic starting material and simultaneously expels out water by-product in the esterification reaction.7a,12
TEM images of catalyst before (material B) and after acid treatment (catalyst 1) are shown in Fig. 3. As shown, no noticeable changes have been made for the structure of the catalyst.
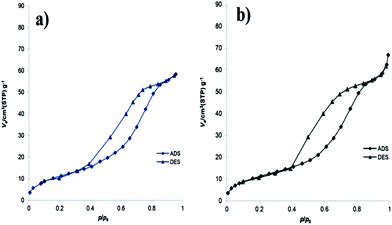
For further investigation about the structure of the catalyst before and after acid treatment, nitrogen adsorption–desorption analysis was performed for material B and catalyst 1 (Fig. 4). The results show the type IV adsorption–desorption isotherm over the range P/P0 = 0.4–0.8 with H2 hysteresis loop. The BET (Brunauer–Emmett–Teller) surface area of the catalyst 1 was found to be 43 m2 g−1. The value of the mean pore diameter of catalyst 1 was derived from the BJH (Barrett–Joyner–Halenda) average pore diameter analysis and was calculated to be 6.2 nm.
Ion exchange pH analysis was performed for catalyst 1 with 1 M solution of NaCl according to the known literature procedures.7a,13 The result of experiment indicated that the loading of catalyst 1 is approximately 1.12 mmol H+ g−1.
The performance of catalyst 1 was initially investigated for esterification of the 3-phenylpropionic acid with ethanol. Our preliminary investigation showed that an isolated yield of 54% of the ester product was obtained when 10 mol% of the catalyst 1 was used at room temperature for 48 h (Table 1, entry 1). When the mentioned reaction was performed at 78 °C (in which 3-phenylpropionic acid and the fatty acids are molten), it produced the corresponding ester in 91% yield (Table 1, entry 2).
| Entry | Substrate | Catalyst | Solvent | Yielda (%) |
|---|---|---|---|---|
| a Yields refer to isolated pure products in the presence of 10 mol% of proton equivalent of a weighted sample of catalyst 1 at 78 °C for 12 h and 2 mL ethanol, unless otherwise stated.b The reaction was performed at room temperature for 48 h.c 150 mg of material A and B was used as catalyst.d 1.5 mL of solvent was used in each case.e 20 mol% of catalyst was used. | ||||
| 1 | 3-Phenylpropionic acid | 1 | — | 54b |
| 2 | 3-Phenylpropionic acid | 1 | — | 91 |
| 3 | Lauric acid | 1 | — | 86 |
| 4 | Palmitic acid | 1 | — | 85 |
| 5 | Stearic acid | 1 | — | 80 |
| 6 | 3-Phenylpropionic acid | A | — | —c |
| 7 | 3-Phenylpropionic acid | B | — | 38c |
| 8 | 3-Phenylpropionic acid | 1 | H2O | 50d |
| 9 | 3-Phenylpropionic acid | 1 | H2O | 82e |
| 10 | 3-Phenylpropionic acid | 1 | CH3CN | 24d |
| 11 | 3-Phenylpropionic acid | 1 | Toluene | 19d |
Encouraged by these results, we then managed esterification of some available fatty acid with ethanol. When 10 mol% of catalyst 1 was used at 78 °C (Table 1, entries 3–5), the fatty acid ethyl ester (FAES) known as biodiesel was obtained in high yield. The reaction of 3-phenylpropionic acid with ethanol and material B as catalyst at the optimized reaction condition gave 38% ester yield (Table 1, entry 7) owing to the potential catalytic activity of ammonium salts for esterification reaction.12a,14
To investigate the effect of solvent, the reaction was performed in water, toluene and acetonitrile (Table 1, entries 8–11). Our investigation showed that the esterification reaction can proceed well in water (Table 1, entries 8 and 9). In this regard we found that high yield (82%) of ester was obtained at 78 °C after 12 h by increasing the amount of catalyst to 20 mol%. In this condition, the supported organic groups provide hydrophobic environment in water.15 A plausible explanation for significant catalytic activity in water is related to the differences in polarities between the applied solvents and the surface of the catalyst. In the case of water as the highest polar solvent, it acts as a driving force to push organic substrate into the hydrophobic channels. Simultaneously, the back reaction (ester hydrolysis) could not occur due to the fact that water cannot diffuse into the hydrophobic environment of the catalyst. The net effect is shifting the reaction to completion even in the presence of water as solvent (Fig. 5).
 | ||
| Fig. 5 Schematic illustration of shifting the reaction equilibrium for ester synthesis with catalyst 1. | ||
The reusability of catalyst 1 was investigated for esterification of 3-phenylpropionic acid with ethanol at the optimized reaction condition (Fig. 6). In this regard, after the first run of reaction, the mixture has been filtrated and the catalyst was recovered. After drying, nearly 95% of the initial weighted catalyst was recovered. The recovered material was subjected to another reaction with identical conditions. The results indicate that catalyst 1 exhibits almost similar catalytic activity for at least four times without appreciable loss of efficiency.
Computational methods are fruitful ways to study various behavior of the catalytic reactions such reaction mechanism, intermediates and structures of catalysts in molecular dimension.16 To gain more realistic insight about the structure of the catalyst we performed some computational modeling by mimicking the surface of the silica.
Silica-based catalysts are common class of catalyst with broad catalytic applications. Modeling the surface of silica gel is a challenging step for computational study. Recently, Shanks and co-worker performed interesting mechanistic study for acetic acid esterification by using simple fixed-Si(OMe)3 groups to model the surface of silica gel.17 Using the relatively large cage-like structure of silicon–oxygen sequence is a useful way to perform a more accurate mimicking the surface of silica gel and thus to obtain more reliable data.18 In the present study, a 6-edge cage-like cluster9,19 of silica (Si4O6(OH)) was selected to model the surface of silica gel. The initial structures were built by attaching the organic groups of catalyst to the surface of silica gel.9 In preparation of catalyst 1, material B undergo one step sulfonation of benzene ring. This electrophilic substitution reaction can occur in the three positions (ortho, meta and para) of the benzene ring according to values of the negative charge of carbon atoms in the ring. Exploring the orientation of such electrophilic substitution can be predicted by means of computational modeling. In this regard, the structure of material B was optimized at B3LYP/6-311++G** level of calculation.9,20 Then the values of Mulliken and NBO charges21 were extracted for carbon atoms of benzene ring of the optimized structures (Fig. 7).
 | ||
| Fig. 7 The values of Mulliken charge (black) and NBO charge (red) of carbon atoms in benzene ring of the optimized structure of material B. | ||
The values of Mulliken charge (black) show the preference of the electrophilic substitution (sulfonation) reaction to the meta position of benzene ring in material B.
However, the corresponding data derived from NBO charge (red) did not show significant difference in orientation to the sulfonation of benzene ring in material B (Fig. 7). With these promising results, we then tried to investigate the structure of the catalyst 1 considering the occurrence of sulfonation reaction of material B at three positions (ortho, meta and para). The structures of ortho (O), meta (M), and para (P) substitute were built with Spartan software. Conformer search was performed for each category using Merck Molecular Force Field. Then the conformers with the lowest energy were optimized at B3LYP/6-311++G** level. The lowest energy conformers of each geometry of the substituted benzene ring (O1, M1 and P1) are shown in Fig. 8.22 By comparing the energetic of the optimized structures (Table 2), we found that in the optimized structure, a cooperativity exists between the sulfonic acid head groups (–SO3H) and HSO4− moieties. As previously proposed7a the cooperativity factor is an effective parameter which significantly improves the catalytic activity.
| Structure | Total energya | Charge transfer | rH⋯Ob | E(2) | ΔqCT |
|---|---|---|---|---|---|
| a The total energies are reported in atomic unit.b rH⋯O is the length of H bonds that are reported in Å. | |||||
| O1 | −3380.203564 | lp O40 → σ*H47–O46 | 1.438 | 72.09 | 0.777 |
| lp O48 → σ*H25–N23 | 1.756 | 23.51 | 0.472 | ||
| lp O42 → σ*H24–N23 | 1.666 | 32.93 | 0.588 | ||
| M1 | −3380.199109 | lp O42 → σ*H47–O46 | 1.662 | 24.50 | 0.477 |
| lp O40 → σ*H25–N23 | 1.510 | 64.17 | 0.719 | ||
| lp O13 → σ*H39–O38 | 1.852 | 10.87 | 0.218 | ||
| P1 | −3380.192690 | lp O42 → σ*H47–O46 | 1.761 | 16.06 | 0.358 |
| lp O40 → σ*H24–N23 | 1.800 | 20.69 | 0.511 | ||
| lp O41 → σ*H25–N23 | 1.892 | 13.15 | 0.388 | ||
The experimental and theoretical data about the structure of the silica-functionalized sulfonic acids and the nature of the intra-structural interactions are rare.9 As shown in Fig. 8, the optimized structures of catalyst 1 mainly comprises intra-structural hydrogen bonds (H bond). The lengths of H bonds of all optimized structures are listed in Table 2.
To better explore the nature of H bonds, NBO analysis was performed for the global minimum energy geometries (the geometry with the lowest energy) in O1, M1 and P1 of the proposed structures (Table 2). In the NBO analysis, the interaction between the electron pairs of the H bond acceptor atoms and the antibonding orbitals of the H bond donor is investigated as a characteristic of H bonding. The second order perturbation stabilization energy, E(2), is related to the delocalization of electrons in a donor–acceptor pair and is calculated by perturbation theory.23 The higher value of E(2) indicates a higher delocalization trend of an electron from donor to acceptor orbitals, and thus a stronger interaction. The selected values of E(2) for donor–acceptor (lp–σ*) interactions are listed in Table 2. It can be deduced from these data that the highest value of E(2) (72.09) is attributed to the shortest H bond (1.438 Å) which exists in O1. Typically, formation of H bond leads to charge transfer from donor sites to the acceptors. The values of charge transfer (ΔqCT) can be calculated from the NBO analysis. These values are known as a criterion of the H bond strength. As shown in Table 2, shorter H bonds generally have higher ΔqCT values.
Since both E(2) and ΔqCT values are responsible for the strength of H bonds, therefore a relationship between these parameters should exist (Fig. 9). According to Fig. 9, this linear correlation between the values of E(2) and ΔqCT (which were derived from the optimized structures of O1, M1 and P1) is observed.
 | ||
| Fig. 9 Correlation between the values of second-order perturbation energies E(2) (kilocalorie per mole) and charge transfer (ΔqCT). | ||
Based on the structural data gained from the optimized structures of catalyst 1, a mechanism was proposed for synthesis of FAES with ethanol (Fig. 10). The presence of sulfonic acid moiety (–SO3H) and HSO4− in its close proximity proposed that both of these species can simultaneously participate in the reaction. In this mechanism, a non-adsorbed ethanol molecule reacts with an adsorbed carboxylic acid molecule.4a,17,24 Despite the Brønsted acidity, the role of HSO4− is activation of ethanol via H bond assistance.
Conclusion
In summary, we designed a novel dual Brønsted acidic mesoporous silica with commercially available starting material for esterification reaction. The high loading acidic sites accompanied by hydrophobic nature of the catalyst facilitate esterification reaction in high yield without using special methodology. The results showed that the reaction can also be performed in water as an environmentally benign and cheap solvent. The catalyst has been simply separated from the reaction mixture and used for at least four reaction runs without noticeable loss of activity. The structure of the catalyst was investigated using a computational modeling technique. Mulliken and NBO charges were calculated to explore the orientation of electrophilic substitution sulfonation on the surface of the catalyst. For the first time, the presence of synergistic effect between sulfonic acid specie (–SO3H) and HSO4− moiety on the surface of the dual Brønsted acidic catalyst was theoretically explored and proved. The useful structural data derived from the optimized geometries of the catalyst 1 helped us propose a reasonable mechanism for esterification. In this mechanism, it was proposed that the reaction occurs on a single acidic site of the mesoporous silica. The cooperation of –SO3H and HSO4− species of catalyst 1 was directly involved in the mechanism of esterification. We believe that this modeling method gives more realistic insight and useful structural information about the catalyst in comparison with simple paper-drawing demonstrations. Further computational investigations about the reaction mechanism are being carried out in our laboratories.Experimental
Preparation of aminopropyl silica
Silica gel Davisil™ (CAS number 112926-00-8) grade 635 (average pore diameter 60 Å, pore volume 0.75 cm3 g−1, surface area 480 m2 g−1) was purchased from Sigma Aldrich. First, the silica gel was activated by refluxing in 6 M hydrochloric acid for 24 h. Then it was washed thoroughly with the deionized water to adjust the pH of the solution to 6–7 and dried at 100 °C for overnight. The activated silica gel (5.00 g) was mixed with 10 mmol of (3-aminopropyl)triethoxysilane in dry toluene for 24 h. Then, the solid materials were filtered off and washed with hot toluene for 24 h in a continuous extraction apparatus (Soxhlet) and dried in an oven at 90 °C for 12 h to give aminopropyl silica as a white solid.Preparation of dual Brønsted acidic mesoporous silica (catalyst 1)
A mixture of 2.0 g aminopropyl silica and 5 mmol benzyl bromide was refluxed in toluene for 24 h. Then, the solid was filtrated and washed with dry toluene (2 × 50 mL) and dried at 90 °C. Chlorosulfonic acid (10 mmol) was added to the solution of the resulted material in 50 mL of dry chloroform and the mixture was refluxed for 48 h.10 Then, the resulted material was filtrated and the residual chlorosulfonic acid was carefully washed by absolute ethanol. Finally, the material was subjected to 2 molar solution of H2SO4 at room temperature. After 12 h, the material was filtrated and washed with diethyl ether and dichloromethane to afford catalyst 1.pH analysis of the catalyst25
0.50 g of catalyst 1 was added to an aqueous solution of NaCl (25 mL, 1 M). The resulting mixture was stirred for 2 h after which the pH of the solution decreased to 1.65. This is approximately equal to a loading of 1.12 mmol H+ g−1 of the catalyst 1.General procedure for fatty acid esterification
In a 10 mL round-bottom flask equipped with a condenser, fatty acid (2 mmol), ethanol (2 mL) and catalyst 1 (10 mol %) were added and heated to reflux. The reaction progress was monitored by TLC (thin layer chromatography) and gas chromatography. After the completion of the reaction, the slurry was filtrated and the catalyst was washed with n-hexane (50 mL) and recovered. The residual of the remained fatty acid in organic phase was quenched with 5% aqueous solution of NaHCO3 (2 × 3 mL). Then, the organic phase was dried over Na2SO4. The corresponding FAES in high yield was afforded following evaporation of the n-hexane under reduced pressure.Computational modeling
Computational modeling was used to study the structure of catalyst 1. The structure of the catalyst studied in this work comprises an organic group anchored on the surface with a Si atom of the initial (3-aminopropyl) triethoxysilane precursor. For the purpose of modeling the catalyst, 6-edge cage-like cluster of the silicon–oxygen arrays which bears organic group of catalyst 1 was selected. Dangling bonds of the surface were saturated by hydrogen atoms.26 Conformer search was performed for all proposed structure using Merck Molecular Force Field (MMFF) in Spartan software.27 The lowest energy conformers were selected and optimized at B3LYP/6-311++G** (ref. 20) to find global minimum energy geometries for each proposed structure. Natural bond orbital (NBO) analysis23 was performed for the optimized structures at the same level of calculation.Notes and references
- (a) J. A. Melero, J. Iglesias and G. Morales, Green Chem., 2009, 11, 1285 RSC; (b) J. A. Melero, L. F. Bautista, G. Morales, J. Iglesias and D. Briones, Energy Fuels, 2009, 23, 539 CrossRef CAS; (c) E. Lotero, Y. Liu, D. E. Lopez, K. Suwannakarn, D. A. Bruce and J. G. Goodwin Jr, Ind. Eng. Chem. Res., 2005, 44, 5353 CrossRef CAS; (d) K. Suwannakarn, E. Lotero, K. Ngaosuwan and J. G. Goodwin Jr, Ind. Eng. Chem. Res., 2009, 48, 2810 CrossRef CAS.
- F. Maa and M. A. Hanna, Bioresour. Technol., 1999, 70, 1 CrossRef.
- (a) M. Kouzu, T. Kasuno, M. Tajika, Y. Sugimoto, S. Yamanaka and J. Hidaka, Fuel, 2008, 87, 2798 CrossRef CAS PubMed; (b) S. Yan, H. Lu and B. Liang, Energy Fuels, 2008, 22, 646 CrossRef CAS; (c) S. D. T. Barros, A. V. Coelho, E. R. Lachter, R. A. S. San Gil, K. Dahmouche, M. I. Pais da Silva and A. L. F. Souza, Renewable Energy, 2013, 50, 585 CrossRef CAS PubMed; (d) M. Manríquez-Ramírez, R. Gómez, J. G. Hernández-Cortez, A. Zúniga-Moreno, C. M. Reza-San Germán and S. O. Flores-Valle, Catal. Today, 2013, 212, 23 CrossRef PubMed.
- (a) K. Suwannakarn, E. Lotero and J. G. Goodwin Jr, Ind. Eng. Chem. Res., 2007, 46, 7050 CrossRef CAS; (b) K. Suwannakarn, E. Lotero and J. G. Goodwin Jr, Catal. Lett., 2007, 114, 122 CrossRef CAS PubMed.
- J. A. Melero, L. F. Bautista, G. Morales, J. Iglesias and R. Sánchez-Vázquez, Chem. Eng. J., 2010, 161, 323 CrossRef CAS PubMed.
- (a) D. Margolese, J. A. Melero, S. C. Christiansen, B. F. Chmelka and G. D. Stucky, Chem. Mater., 2000, 12, 2448 CrossRef CAS; (b) J. A. Melero, G. D. Stucky, R. van Grieken and G. Morales, J. Mater. Chem., 2002, 12, 1664 RSC.
- (a) B. Karimi and M. Vafaeezadeh, Chem. Commun., 2012, 48, 3327 RSC; (b) B. Karimi and M. Vafaeezadeh, RSC Adv., 2013, 3, 23207 RSC.
- (a) Q. Zhang, J. Luo and Y. Wei, Green Chem., 2010, 12, 2246 RSC; (b) Q. Zhang, H. Su, J. Luo and Y. Wei, Green Chem., 2012, 14, 201 RSC.
- M. Vafaeezadeh, Z. Bahrami Dizicheh and M. Mahmoodi Hashemi, Catal. Commun., 2013, 41, 96 CrossRef CAS PubMed.
- (a) M. Chidambaram, S. Selvakumar, T. Tamil Selvi and A. P. Singh, J. Mol. Catal. A: Chem., 2006, 245, 69 CrossRef CAS PubMed; (b) M. Karaki, A. Karout, J. Toufaily, F. Rataboul, N. Essayem and B. Lebeau, J. Catal., 2013, 305, 204 CrossRef CAS PubMed.
- R. van Grieken, J. A. Melero and G. Morales, J. Mol. Catal. A: Chem., 2006, 256, 29 CrossRef CAS PubMed.
- (a) C. Tsai, H. Chen, S. M. Althaus, K. Mao, T. Kobayashi, M. Pruski and V. S.-Y. Lin, ACS Catal., 2011, 1, 365 CrossRef; (b) I. K. Mbaraka and B. H. Shanks, J. Catal., 2005, 229, 336 CrossRef PubMed.
- B. Karimi and D. Zareyee, Org. Lett., 2008, 10, 3989 CrossRef CAS PubMed.
- T. Funatomi, K. Wakasugi, T. Misaki and Y. Tanabe, Green Chem., 2006, 8, 1022 RSC.
- Y. Gu, C. Ogawa, J. Kobayashi, Y. Mori and S. Kobayashi, Angew. Chem., Int. Ed., 2006, 118, 7375 CrossRef PubMed.
- R. A. van Santen and M. Neurock, Molecular Heterogeneous Catalysis: A Conceptual and Computational Approach, WILEY–VCH Verlag GmbH & Co. KGaA, Weinheim, 2006 Search PubMed.
- S. Miao and B. H. Shanks, J. Catal., 2011, 279, 136 CrossRef CAS PubMed.
- (a) J. Ambati, H. Saiyed and S. E. Rankin, Phys. Chem. Chem. Phys., 2012, 14, 6617 RSC; (b) L. A. Montero-Cabrera, Y. Perez-Badell and M. J. Mora-Fonz, J. Phys. Chem. A, 2008, 112, 2880 CrossRef CAS PubMed; (c) H. Staube, I. Del Rosal, L. Maron, F. Kleitz and F. Fontaine, J. Phys. Chem. C, 2012, 116, 25919 CrossRef.
- (a) J. P. Blitz, R. E. Diebel, C. A. Deakyne, J. M. Christensen and V. M. Gun'ko, J. Phys. Chem. B, 2005, 109, 5667 CrossRef CAS PubMed; (b) K. Xiang, R. Pandey, U. C. Pernisz and C. J. Freeman, J. Phys. Chem. B, 1998, 102, 8704 CrossRef CAS; (c) R. Franco, A. K. Kandalam, R. Pandey and U. C. Pernisz, J. Phys. Chem. B, 2002, 106, 1709 CrossRef CAS.
- For examples of coincident between experimental and computational results which used B3LYP to study H bonds see; (a) M. Vafaeezadeh and A. Fattahi, J. Phys. Org. Chem., 2014, 27, 163 CrossRef CAS PubMed; (b) Z. Tian, A. Fattahi, L. Lis and S. R. Kass, J. Am. Chem. Soc., 2009, 131, 16984 CrossRef CAS PubMed; (c) A. Shokri, A. Abedin, A. Fattahi and S. R. Kass, J. Am. Chem. Soc., 2012, 134, 10646 CrossRef CAS PubMed.
- The data of NBO charge were derived from the NBO analysis of the optimized structure of the material B. For reliability of applied level of calculation for NBO analysis see: M. Shakourian-Fard, A. Fattahi and A. Bayat, J. Phys. Chem. A, 2012, 116, 5436 CrossRef CAS PubMed.
- See ESI† for other geometries (O2,3, M2,3 and P2,3) which optimized at B3LYP/6-311++G** and their total energy values. Worthy to note that O1, M1 and P1 are global minimum energy structures..
- A. E. Reed, L. A. Curtiss and F. Weinhold, Chem. Rev., 1988, 88, 899 CrossRef CAS.
- (a) Y. Liu, E. Lotero and J. G. Goodwin Jr, J. Catal., 2006, 242, 278 CrossRef CAS PubMed; (b) S. R. Kirumakki, N. Nagaraju and S. Narayanan, Appl. Catal., A, 2004, 273, 1 CrossRef CAS PubMed.
- M. Vafaeezadeh, M. Mahmoodi Hashemi and M. Shakourian-Fard, Catal. Commun., 2012, 26, 54 CrossRef CAS PubMed.
- P. Sautet and F. Delbecq, Chem. Rev., 2010, 110, 1788 CrossRef CAS PubMed.
- Spartan ‘10 V102’, Wavefunction, Inc., Irvine, CA, 2010 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: O2,3, M2,3 and P2,3 which optimized at B3LYP/6-311++G** and their total energy values, 1H NMR spectra of the esters and XYZ coordinates of the optimized structures. See DOI: 10.1039/c3ra47638c |
| This journal is © The Royal Society of Chemistry 2014 |