Ionic liquid-based optoelectronic sensor arrays for chemical detection†
Waduge Indika S.
Galpothdeniya
a,
Kevin S.
McCarter
b,
Sergio L.
De Rooy
a,
Bishnu P.
Regmi
a,
Susmita
Das
a,
Farhana
Hasan
a,
Attres
Tagge
a and
Isiah M.
Warner
*a
aDepartment of Chemistry, Louisiana State University, Baton Rouge, LA 70803, USA. E-mail: iwarner@lsu.edu; Fax: +1-225-578-3971; Tel: +1-225-578-2829
bDepartment of Experimental Statistics, Louisiana State University, Baton Rouge, LA 70803, USA
First published on 6th January 2014
Abstract
Development of ionic liquid (IL)-based colorimetric sensor arrays for detection and identification of chemicals in both the aqueous and vapor phases is reported. These facile and inexpensive optoelectronic sensors were fabricated by using ionic liquids (ILs) derived from readily available pH indicator dyes. A series of 12 different chemosensory ILs were synthesized by pairing anionic pH indicator dyes with trihexyl(tetradecyl)phosphonium ([P66614]) cation via an ion exchange reaction. The incorporation of the [P66614] cation imparted hydrophobic characteristics to these ILs, and this induced hydrophobicity led to their desired low solubility in aqueous solutions, as well as eliminated the need for a specialized hydrophobic matrix/substrate for immobilization. In this manuscript, four different matrices, i.e. glass microfiber filter papers, cotton threads, silica thin layer chromatography (TLC) plates, and alumina TLC plates, were employed for fabrication of sensor arrays. These sensor arrays were used to analyze pH values of aqueous solutions as well as for detection of acidic and basic vapors. To further prove the applicability of these IL sensor arrays as tools to sense closely related complex materials, the arrays were applied to successful discrimination of aqueous solutions of smoke from three commercially available cigarettes. The digital data generated from these sensor arrays were used in developing predictive models for accurately identifying various analytes. Two approaches were used for developing the models, and two methods were applied for assessing the predictive accuracy of the models. Use of cotton threads as a matrix led to development of a more flexible, low volume, and lightweight array to estimate pH and detect a variety of vapors. These wearable arrays may possibly be incorporated into bandages, sweatbands, diapers, and similar systems. Overall, these IL-based sensor arrays should provide a new research direction in the development of advanced colorimetric sensor arrays for detection and identification of a range of analytes relevant to many different applications.
1. Introduction
Over the last several decades, electronic devices that can mimic human olfactory and gustatory processes have attracted considerable attention amongst researchers.1 These artificial electronic noses and tongues, which are often defined as intelligent chemical array sensor systems,2 are emerging as rapid, low-cost, and sensitive tools for chemical analysis, as compared to traditional more expensive instrumentation such as gas chromatography/mass spectroscopy (GC/MS).1,3–5 Among the current electronic nose and tongue technologies, optoelectronic noses and tongues fabricated by use of an array of different dyes, which change colors based on intermolecular interactions (acid–base, dipole–dipole, and π–π), have shown higher chemical selectivity and sensitivity as compared to other types of sensor arrays.6–9 This advantage has generated intense interest among a number of research groups to develop colorimetric sensor arrays for a wide range of applications.10–13 Among such studies, work conducted by Suslick and co-workers can be identified as quite inspirational. For example, the studies by these authors include detection and identification of chemically diverse analytes using sensor arrays fabricated by immobilization of hydrophobic water-insoluble dyes onto hydrophobic membranes7 or incorporating chemically-sensitive dyes into nanoporous silica microspheres.5,6,14,15 A colorimetric sensor array using reversed phase silica gel plates as a matrix has also been developed by Huang et al.11 for evaluation of freshness of fish. In addition, incorporation of various dyes into inorganic materials such as silica, alumina, and UVM-7 in order to monitor the aging of chicken meat has been reported by Salinas et al.10Fabrication of optoelectronic tongues or colorimetric-taste sensor arrays, which are capable of operating in aqueous environments, is inherently challenging due to interferences from water. For aqueous-phase analysis, the sensor arrays should be relatively hydrophobic in order to avoid dissolution of the arrays. Despite considerable success in this field, current colorimetric sensor arrays are limited with regard to their ability to detect compounds in aqueous samples. The primary reason is that dyes which are used for aqueous-phase analysis should be hydrophobic and thus highly insoluble in water. Therefore, the number of dyes which fulfill this criterion are very limited. Moreover, this approach eliminates the ability to use the same dyes in both optoelectronic noses and tongues. In addition, the matrix/substrate on which the dyes are immobilized is required to be highly hydrophobic. Thus, the requirement of a specialized hydrophobic surface adds an additional constraint to the number of applications for a particular sensor array. Therefore, a proper choice of matrix is also important in successful fabrication of such sensor arrays. We note that there has recently been a burgeoning interest in using cotton threads as a matrix to immobilize dyes or other chemically active species. This interest is primarily due to inherent advantages such as facile, low-cost, low-volume, and excellent materials for displaying colorimetric results.16,17 Natural cotton threads are hydrophobic due to the presence of a surface wax, and hence have low wettability.16,18 Therefore, an expensive pretreatment such as plasma oxidation is required to allow adequate staining or wicking with aqueous dyes.17,19 Otherwise, a specialized hydrophilic cotton thread is required for fabrication, which limits the applicability of the matrix.
As low melting organic salts,20 ionic liquids (ILs), often have virtually no vapor pressure. These compounds are considered excellent alternatives to conventional organic solvents. Favorable properties of these liquid salts include good thermal stability, wide liquid temperature range, considerable ionic conductivity, a broad electrochemical window, and a wide solubility and miscibility range.21,22 In addition, ILs can be designed or tuned for specific applications by altering either the cation or anion. In addition, ILs have recently been demonstrated to be excellent sensing materials for a number of different applications.23–30 For example, Zeng and co-workers23 have utilized a number of room temperature ILs (RTILs) to design a QCM sensor array for detection of organic vapors at high temperature.24 Recently, a few parallel studies on development of sensor arrays based on ILs have also been reported.25,26 Baker and co-workers27 have recently synthesized a class of luminescent ILs using a pyranine anion, and have used these ILs for sensitive detection of alkylamines. Wang and coworkers28,29 have demonstrated the potential use of ILs as fluorescence probes for highly sensitive detection of proteins. Over the last several years, ILs derived from indicator dyes have also been the focus of many studies. For example, Branco and Pina30 have reported a series of photochromic ILs derived from methyl orange, and have showed that the photochromic response of these ILs can be tuned by simply changing the cation. In addition, the sensing performance of IL-modified dyes derived from methyl orange and methyl red toward acids in aqueous and non-aqueous media have been reported by Zhang et al.14
Despite many of the aforementioned outstanding properties of ILs, their use in colorimetric sensor arrays remains largely unexplored. Herein, we outline the development of an IL-based optoelectronic sensor array, and demonstrate its applicability for prediction of values of pH in aqueous solution, detection of acidic and basic vapors, as well as discrimination of complex mixtures such as aqueous samples containing dissolved smoke from various brands of cigarettes. A series of 12 different ILs, all employing [P66614] as the counter ion, were synthesized from anionic dyes via a metathesis reaction. The pairing of [P66614] cation with the dye anions imparted considerable hydrophobicity to the resultant ILs. As a result, these ILs were insoluble in aqueous solution, and thus exhibited reduced interferences from humidity. More importantly, it was found that these ILs do not require a specialized hydrophobic matrix as is normally required for analyses of aqueous samples using this approach. These ILs were immobilized on four commonly available matrices including silica and alumina TLC plates, filter papers, and cotton threads, which allowed successful fabrication of sensor arrays. Furthermore, the sensor arrays fabricated by use of cotton threads as matrices did not require expensive pretreatments or specialized cotton threads. The use of cotton threads as a sensor matrix is a promising approach for preparation of inexpensive wearable sensor arrays. In order to accurately identify the analytes, predictive models were developed using principal component analysis (PCA) and discriminant analysis. The resubstitution and cross-validation methods were used to assess the predictive accuracy of the models.
2. Experimental section
2.1. Materials
All 12 indicator dyes (phenol red (PR), brilliant yellow (BY), bromocresol green (BCG), m-cresol purple (mCP), methyl orange (MO), methyl red (MR), p-xylanol blue (Xyl), bromophenol blue (BPB), thymolphthalein (Thy), phenolphthalein (FFT), chlorophenol red (ClR) and bromothymol blue (BTB)), trihexyl-(tetradecyl)phosphonium chloride [P66614][Cl] (≥95%), ethanol (EtOH), and dichloromethane (DCM) were purchased from Sigma-Aldrich and used as received. The 12 pH indicator dyes that are used in this study can be considered as very common choices for traditional colorimetric sensor arrays.10,31,32 Triply deionized water (18.2 MΩ cm) from an Elga model PURELAB ultra water-filtration system was used for preparation of all buffer systems. Three brands of commercially available cigarettes (Marlboro® red, Crowns®, and Camel® Turkish domestic blend) were purchased from a local grocery store and used as received for smoke identification purposes.2.2. Synthesis and characterization of ILs
The ILs used in these studies were prepared using a previously reported anion exchange procedure.33,34 Briefly, as an example, a mixture of NaMO and P66614Cl with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mole ratio was dissolved in DCM–water (5
1 mole ratio was dissolved in DCM–water (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) mixture, and stirred overnight. Afterwards, the DCM layer was washed with water several times in order to remove the NaCl byproduct. The resultant IL product, [P66614][MO], was obtained after removing DCM and water in vacuo. Other ILs were prepared using a similar procedure where the sodium salt of each indicator dye was coupled with the [P66614] cation to obtain the desired products. Finally, all ILs were characterized using nuclear magnetic resonance (NMR) (Fig. S1†), Fourier transform infrared spectroscopy (FTIR) (Fig. S2a†) and Ultraviolet-visible spectroscopy (UV-Vis) (Fig. S2b†), and electron spray ionization mass spectroscopy (ESI-MS) (Fig. S3†). The resulted ILs showed weak or no fluorescence. Examination of results obtained from ESI-MS, when performed in both positive and negative ion modes, indicated exact masses corresponding to the respective cations and anions present in a given IL, thus confirming formation of the product (Fig. S3-1 through 12†). The presences of [P66614] cation and pH indicator anion in the ILs were further confirmed by 1H NMR and FTIR spectra.†
1 v/v) mixture, and stirred overnight. Afterwards, the DCM layer was washed with water several times in order to remove the NaCl byproduct. The resultant IL product, [P66614][MO], was obtained after removing DCM and water in vacuo. Other ILs were prepared using a similar procedure where the sodium salt of each indicator dye was coupled with the [P66614] cation to obtain the desired products. Finally, all ILs were characterized using nuclear magnetic resonance (NMR) (Fig. S1†), Fourier transform infrared spectroscopy (FTIR) (Fig. S2a†) and Ultraviolet-visible spectroscopy (UV-Vis) (Fig. S2b†), and electron spray ionization mass spectroscopy (ESI-MS) (Fig. S3†). The resulted ILs showed weak or no fluorescence. Examination of results obtained from ESI-MS, when performed in both positive and negative ion modes, indicated exact masses corresponding to the respective cations and anions present in a given IL, thus confirming formation of the product (Fig. S3-1 through 12†). The presences of [P66614] cation and pH indicator anion in the ILs were further confirmed by 1H NMR and FTIR spectra.†
2.3. Preparation of IL sensor arrays using TLC and filter paper matrices
The twelve synthesized ILs were individually dissolved into ethanol to obtain twelve different 1.0 mM solutions of each IL. Each of these solutions was then spotted as a single spot onto a 25 mm × 25 mm matrix surface to obtain a 12-spot IL sensor array, and the resulting sensor array was kept in a desiccator for 24–48 hours. A schematic of the preparation of the sensor array is shown in Scheme 1.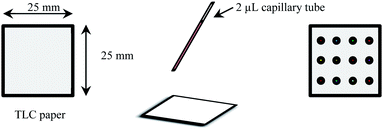 | ||
| Scheme 1 Schematic representation of the preparation of IL sensor array on TLC and filer paper-based matrices. | ||
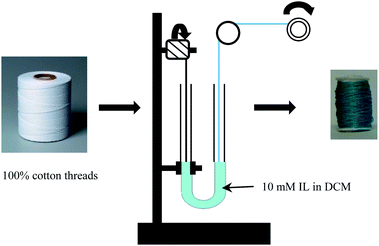
2.4. Preparation of an IL sensor array using cotton thread matrix
In order to stain the natural white cotton threads, 10 mM solution of IL was prepared by dissolving pure IL in DCM. Then 10 mL of each solution was placed in a U-tube and the cotton threads were passed through the tube and wound around a different spool. A schematic representation of this staining procedure is shown in Scheme 2. Afterwards, the IL-stained threads were sewn onto a cotton fabric template using a Singer (model number-7258) sewing machine.2.5. Preparation and analyses of aqueous solutions of cigarette smoke
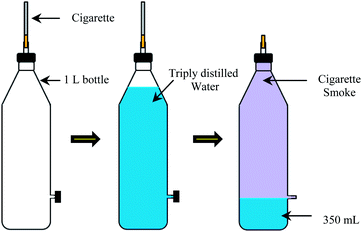
Aqueous solutions of cigarette smoke were freshly prepared using a custom-made device (Scheme 3). This apparatus was fabricated to achieve three main goals: (1) to provide the suction necessary to extract smoke from the cigarette, (2) to collect the cigarette smoke in a closed environment, and (3) to supply an aqueous medium for dissolution of the cigarette smoke. A 1 liter plastic bottle was drilled from the lid and a side, and a one inch rubber tube with a cap was attached to each hole. The hole on the side of the wall was drilled in such a manner as to retain only 350 mL of aqueous layer in the bottle. The bottle was filled with triply deionized water, and the cigarette to be analyzed was fixed into the hole of the lid and sealed by using parafilm. Later, the cigarette smoke was collected after lighting the cigarette followed by releasing the cap from the side-wall outlet. The collected cigarette smoke was dissolved into 350 mL of the residual distilled water by vigorously shaking the bottle for five minutes, and the system allowed to equilibrate for 30 minutes. Finally, the sensor array fabricated by using the filter paper matrix was inserted into a 10 mL portion of the aqueous layer for five minutes in order to obtain color changes that correlate with a particular cigarette brand. In these studies, seven cigarettes of each brand were analyzed.2.6. Development of predictive models for identification of chemical substance
The goal of this study is to use the developed sensor arrays to identify chemical substances accurately and consistently, and as efficiently and as quickly as possible. To accomplish this goal, the information contained on a sensor array can be quantified and processed using a predictive model. Quantitative models can be automated, improving efficiency and timeliness, and their use removes inconsistencies in the prediction process that can result from subjective assessment of qualitative information. In addition, the accuracy of predictive models can be quantified. When more than one potential model is available, such accuracy measures allow for comparisons of models and the selection of the best model from a set of competing models. There are several approaches to quantifying a model's predictive accuracy which are discussed in more detail in the ESI.†To quantify the information contained on a sensor array, the array is optically scanned. The color of each spot on the array is quantified by determining the red, green, and blue (RGB) components comprising that color. Each of these color components is represented by an integer value ranging from 0 to 255. Since a sensor array contains twelve spots, the scanning process generates 36 numeric values each time the array is scanned. Because of potential variation in spot-color intensities from one sensor array to another, an array is scanned twice i.e. before it is exposed to any substance, and then after it has been exposed to a substance to be identified. This results in 72 numeric values for a given array, 36 values taken before exposure and 36 values taken after exposure. The pre-exposure scan provides baseline values by which the post-exposure scan values can be adjusted. Specifically, the pre-exposure RGB component for a given spot is subtracted from the corresponding post-exposure RGB component. This type of adjustment removes the array-to-array variability from the measurement, decreasing the variability in spot intensities for a given analyte and thereby improving the quality and predictive ability of the data. The resulting 36 differences are then used as variables in a predictive model.
The sensor array is the scientifically developed instrument by which measurements are obtained on a chemical substance (i.e. analyte). A predictive model then takes these measurements, and uses them together to generate a prediction about the identity of the analyte. Statistical analysis is used to develop this predictive model, using experimental sensor array data for which the analytes are known. Once the model has been developed, it can be used to identify analytes for which it has been developed by exposing a sensor to the analyte whose identity is unknown and processing the resulting data using the model, generating a prediction.
Discriminant analysis was used to develop statistical models using the sensor array data obtained from these experiments. Two approaches were used in constructing the discriminant models. The first approach utilized as predictor variables all of the original 36 RGB color differences described above. The second approach utilized a smaller number of new variables, constructed from the original 36 variables via PCA. The rationales for considering the two approaches are given in the ESI.† All statistical analyses for this study were generated using version 9.3 of the SAS System.35
3. Results and discussion
The commercially available indicator dyes were modified by incorporating the counter cation, [P66614], which has often been designated as a ‘universal liquifier’ within the ionic liquid community.27 This cation induces hydrophobic properties to the ILs as well as transforms the physical state, of all 12 indicator dyes, from high melting solids to viscous liquids or low melting solids (Fig. S4†).27,34 Because of the resultant hydrophobic characteristics, these ILs are very compatible for detection of analytes dissolved in aqueous medium. In addition, these ILs exhibited reduced interferences from humidity, which has been recognized as an important factor in vapor sensing. In aggregate, this approach allows selection of dyes for sensor arrays without considering hydrophobicity as the selection criterion. In principle, liquids serve as better sensory materials than solids mainly due to the rapid diffusion of analytes.36 Therefore, these IL-based sensors are expected to show higher sensitivity and faster response than a traditional dye-based sensor, which can be attributed to easy accessibility of all dye molecules to the analytes.For sensing applications involving chemosensory pigments, there are basically two primary considerations: (1) analytes must be able to easily access the dyes, and (2) the dye must be immobilized onto a porous or permeable host material in order to avoid leaching of the dye.37 Thus, selecting a host material should not be limited by the pigments of the sensor array. In addition, when designing a facile and inexpensive sensor, the selected host material or matrix should be readily available. The host matrix should also be selected according to the desired application. In this study, we have used [P66614]-based ILs, which are reported to possess good permeability towards gases,38 and are excellent dyes for sensing studies. Additionally, we have used four different matrices including silica and alumina TLC plates, filter papers, and cotton threads depending on the application. Also, these matrices are known to be readily available and relatively inexpensive. Fig. 1 is a schematic representation of the four IL sensor arrays fabricated using these matrices. Moreover, these four materials were found to be effective in immobilizing the ILs without leaching during aqueous-phase analysis.
Compared to a traditional pH paper, pH meter or a colorimetric sensor (which use just a single dye to identify analytes), the discriminatory power of colorimetric sensor arrays are found to be extremely higher.3 In this study, our IL sensor arrays were composed of 12 sensor elements, where each element is a three dimensional vector (RGB). Therefore, each analyte is represented as a 36-dimensional vector. Because of the high dimensionality of our sensor arrays, it is expected to display extraordinarily high levels of chemical discrimination which is extremely useful in the discrimination of closely-related analytes or complex sample mixtures. Compared to the traditional techniques, the data obtained by these colorimetric sensor arrays can be accurately analyzed by use of predictive models which is tremendously important in real time analysis of analyte mixtures.
3.1. Choice of matrix
Our first choice as a host matrix was silica TLC plates. Due to exposed hydroxyl groups, the surface of silica is slightly acidic. Therefore, some of our chemosensory ILs tended to exhibit acidic colors on this blank sensor. As a result, such a sensor would be excellent for detection of basic vapors. In comparison to a silica surface, an alumina TLC matrix has a neutral pH due to the amphoteric properties of alumina. Therefore, ILs deposited on this surface tend to maintain their neutral color, which should make this matrix a good choice for detection over the entire range of pH values. Our third matrix of choice is a glass microfiber filter paper. A filter paper matrix provides greater flexibility over silica and alumina matrices because it can be folded as well as twisted without damaging the matrix. In addition, exploration of paper-based matrices such as filter paper would allow printing of sensor arrays. Finally, we employed cotton threads to develop the sensor arrays. We note that the potential for using these cotton threads as matrices in microfluidic devices has been recently reported.16,17 In general, dewaxing techniques are employed on natural cotton threads in order to produce wettability, and hence to allow staining or wicking of aqueous dyes.16–18 In this study, dewaxing of cotton threads, by expensive pretreatments such as plasma oxidation, was not required in order to dewax the cotton threads due to hydrophobic characteristic of the ILs. The use of threads as matrices for fabricating sensor arrays has several advantages over many other matrices. First, thread is inexpensive, broadly available, and easy-to-handle. In addition, a sensor array fabricated from cotton thread is flexible, lightweight and has very low volume. These properties allow easy transport and storage. As a result of the low volume of thread, a complex sensor array can be fabricated onto a very small volume by use of a sewing machine. Thus, chemosensory IL-stained threads should be applicable to developing wearable sensors. Consequently, potential application areas of such sensors include bandages, sweatbands, headbands, diapers, and other similar matters. Moreover, these arrays can be incorporated into a garment similar to applications using radiation badges. Thus, wearable materials with potential military applications are a distinct possibility.For a successful colorimetric sensor array, the matrix material and dyes should be chemically and physically stable in order to provide reasonable shelf life. The four sensor arrays that we describe in this study have excellent stabilities over a four-week period when covered with aluminum foil and stored in a desiccator. Therefore, due to flexibility in selecting a matrix when using chemosensory ILs, one can select from a variety of very stable matrices with controllable accessibility for a given set of analytes.
3.2. Digital image maps and difference maps
The accumulation of colorimetric sensor array data is usually done by optically scanning the sensor arrays by using a flatbed scanner.32,39 This helps to minimize the effects of variation in imaging or environmental conditions on experimental data. In this study, each sensor array was scanned using an EPSON PERFECTION 2400 scanner before and after exposure to analytes, and two digital image maps were generated to allow removal of edge artifacts. For each spot in the array, color values of RGB were measured before and after digital image mapping, and a difference map was generated for each spot (Fig. 2).40,41 These difference maps have been confirmed to be a convenient methodology to facile and clear representation of a series of color changes which occur after analyte exposure.40,413.3. Estimation of pH values
A pH indicator dye is a halochromic chemical compound that changes colors as a result of protonation or deprotonation depending on the pKa of the dye and the pH value of its surroundings. In other words, it possesses different colors in its protonated and deprotonated forms, and the color at a given pH value depends on the ratio of the two forms. The protonated form of a pH indicator dye can also be defined as a weak acid, while its deprotonated form as a weak base: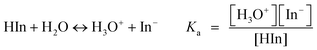 | (1) |
Thus, the effect of pH on the change in color of an indicator dye is readily explained by use of the Henderson–Hasselbalch equation (eqn (2)):
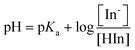 | (2) |
Examination of eqn (2) indicates that the color change of the system at a particular pH value is dependent on the ratio between the protonated and deprotonated forms, and the acid dissociation constant (Ka) of the indicator dye. Therefore, by having different Ka values with different indicator dyes, different color changes over different pH values can be obtained. When selecting a series of indicator dyes for a given sensor array, we have selected dyes with a range of pKa values and colors in order to obtain different color combinations which cover the entire pH range. By using an increased number of ILs with different pKa values in the array, one can further increase the resolution of the pH sensor.
3.4. Aqueous-phase sensing
As noted earlier, the proposed arrays can be used to estimate the pH values of aqueous solutions without the need for specialized hydrophobic matrices. The experimental procedure used for estimating pH, and the differences between the IL-based sensor arrays and the water soluble indicator-based sensor array is displayed in Fig. 3. Note that the regular indicator dye-based sensor array loses much of its integrity and smears over the TLC stripe, while the IL-based sensor array remains stable throughout the experiment. Five different solutions with pH values of 1, 4, 7, 10, and 13 were used in these analyses. The difference maps for pH sensing obtained by using alumina TLC plate, filter paper, and thread-based matrices are shown in Fig. 4. Regardless of the unique individual advantages of each matrix over the other, all three matrices allowed to discriminate between the five pH values with higher degree of variation between each difference map. It is very important to have higher degree of variation between two difference maps of adjacent pH values which enables the sensor arrays to be applied in the discrimination of pH values which are very close. In addition, by using three different matrices, it is clear that matrix specific color changes can be exploited. | ||
| Fig. 4 The difference maps for pH sensing studies by using (A) alumina TLC, (B) filter paper, and (C) cotton thread matrices. | ||
3.5. Vapor-phase sensing
The vapor sensing experiments were performed by exposing the sensor arrays to saturated vapors of different volatile compounds at room temperature, i.e. 298 K. Acidic vapor sensing studies were conducted by using four vapors of acids including hydrochloric acid (HCl), trifluoroacetic acid (CF3COOH), formic acid (HCOOH), and acetic acid (CH3COOH). Basic vapor sensing studies were performed by using six vapors of bases including 1-methylimidazole (1MIm), pyridine, ammonia, diethylamine, triethylamine and dimethylformamide (DMF). Sensor arrays fabricated on alumina and silica TLC plates were used to detect acidic and basic vapors, respectively. These difference maps are shown in Fig. 5. Color changes obtained from silica and alumina TLC plates clearly provide a higher degree of discrimination between closely related groups of chemical vapors, e.g. (1) ammonia, diethylamine, and triethylamine; (2) HCl, CH3COOH, and CF3COOH. Liberty in selecting a matrix, which offers additional number of changes in colors, provides a significant variation in the difference maps, and enables rapid and facile discrimination of vapors. Therefore, this study confirms that the use of alumina matrix for acidic vapor sensing and silica matrix for basic vapor sensing provides better discriminatory power for the IL sensor array. Thread-based sensor arrays were also used to detect CF3COOH and reusability was also tested for that matrix (Fig. 6). After exposing the sensor array to CF3COOH, it was exposed to ammonia, and back again to CF3COOH. This exposure cycle was repeated three times and the difference maps corresponding to the first and last exposure to CF3COOH were compared. Examination of Fig. 6d shows that these two difference maps have very little deviation in RGB levels for most of the IL spots, and this confirms the capability of reusing the thread-based IL sensor arrays. Thus, the thread-based sensor array shows considerable promise for regeneration and reuse. | ||
| Fig. 5 Difference maps of chemosensory IL sensor array. (A) On exposure to acidic vapors by using alumina TLC matrix, and (B) on exposure to basic vapors by using silica TLC matrix. | ||
3.6. Statistical discrimination for identification of pH values and acidic/basic vapors
Our first study involved developing a sensor array and corresponding predictive model to identify 15 chemical samples including acidic and basic vapors, and solutions of varying pH values. Five replicate experiments were performed by exposing the sensor array to each of the 15 chemical samples, for a total of 75 observations. Each sensor array was processed as described above. The resulting dataset contained 36 RGB color difference values measured on each of these 75 observations.Using the first approach described above, a discriminant analysis was performed for the 15 different samples using all 36 variables in the dataset. Because the number of sensor arrays exposed to each substance is small relative to the number of predictor variables used in the model, it was not possible to fit a quadratic discriminant model. The total sample size was large enough to fit a linear discriminant model, however. Using the resubstitution method of assessing the accuracy of the model, the linear discriminant model correctly identified all substances in the dataset. Using the cross-classification method of assessment, the linear discriminant model correctly identified all but one of the substances, for an estimated error rate of 1.33%. In this case, one diethylamine sample was misclassified as triethylamine. The linear discriminant model using all 36 variables does a very good job of utilizing the sensor array data to correctly differentiate among the fifteen substances.
To build a discriminant model using the second approach described above, a PCA was first performed to reduce the dimensionality of the predictor variable space. Fig. 7 shows that the first three principal components account for approximately 80% of the variability in the original 36 variables. In Fig. 8, the observations are plotted with respect to their scores on the first three principal components. From the plot, it appears that the three principal components are adequate in separating many of the analytes although some of the analytes are quite close. Based on Fig. 8, we might expect a discriminant function centered on these principal components would do a good job of distinguishing among most of the fifteen analytes. In an effort to find the simplest model possible, discriminant analysis models were fit using one, two, and three principal components each.
 | ||
| Fig. 7 (A) Scree plot and (B) cumulative proportion of variability accounted for by the principal components obtained from the color change profile of pH, acidic and basic vapor numerical data. | ||
 | ||
| Fig. 8 Plot of the scores for the first three principal components based on the data for the analysis of pH, acidic and basic vapor. | ||
We first fit a model using the first principal component as the only predictor variable in the model. As part of the model-building process, the hypothesis that a linear discriminant model would be adequate was tested. The p-value of the test was less than 0.0001. It was therefore concluded that a linear discriminant model would not adequately fit the data, and a quadratic discriminant model was constructed instead. The accuracy of the model was then assessed. Using the resubstitution method of assessment, the quadratic discriminant model had a resubstitution error rate of 30.7%. Using the cross-classification method, the error rate was 40%.
The next model to be considered used the first two principal components as predictor variables. Again, the hypothesis that a linear discriminant model was adequate was tested. The hypothesis was rejected (p-value < 0.0001), and a quadratic model was constructed. This model had a resubstitution error rate of only 6.7%. However, under cross-classification, the error rate was 34.7%.
The final model for consideration used the first three principal components as predictor variables. A test of the hypothesis that a linear discriminant model was adequate had a p-value of less than 0.0001, and so again a quadratic discriminant model was constructed. For this model, the resubstitution error rate dropped to 0%, but the cross-classification error rate was quite a bit larger at 28%. In order to understand the problem of this model distinguishing between analytes, Table S1† lists the misclassifications under cross-validation by this model. Based on this list, the model appears to have difficulty distinguishing between substances within the following groups: (1) ammonia, diethylamine, and triethylamine; (2) HCl, CH3COOH, and CF3COOH; and (3) DMF and pyridine. In Fig. 8, we see that within each of these three groups, the substances are very close together and in some cases overlap. This explains the difficulty of the model distinguishing between analytes within these groups. In contrast, the model does not have a trouble in distinguishing between the larger groups of substances because of the relatively large amount of separation as seen in the plot. Note that only five training samples for each analyte were available for building the model. If additional training samples were to become available and the model updated, it is possible that we could see an improvement in the model's accuracy. There is no guarantee of that, however. In fact, given the close proximity of some of the groups with respect to these three principal components, it may be that using only three principal components is too great a reduction in the predictor variable space to allow for highly accurate discrimination among these substances.
Based on these results, the first model that uses all 36 of the original predictor variables appears to be the model to use for identifying these 15 analytes. As it stands, the model has very little trouble distinguishing between the substances, even those that were close together in terms of the first three principal components. With the availability of more training samples, the model could be updated and refined, and with the addition of enough training samples a quadratic model could be developed if deemed necessary. Each of these improvements would be expected to improve the accuracy of the model. As a final note on model development for study one, the analyses above demonstrate the importance of evaluating the predictive accuracy of the model. Doing so can illuminate situations where the models may have difficulty. It also provides a basis for comparison and the selection of a best model when more than one is available. What is also critically important is using a method like cross-validation that provides more realistic estimates of the error rates than the resubstitution method.
3.7. Identification of cigarette smoke
One of the greatest advantages of having a colorimetric sensor array over a traditional pH probe is tremendously improved dimensionality, which was obtained by using 12 ILs. Therefore, these sensor arrays could achieve exceptional discrimination among very similar analytes. To demonstrate that the IL sensor arrays can be used to sense complex analytes, and discriminate between closely-related analytes, aqueous smoke solutions prepared from three commercially available cigarette brands (Marlboro® red, Crowns® and Camel® Turkish domestic blend) were analyzed. Cigarette smoke is a very complex mixture of chemical compounds for which more than 4800 compounds have been identified, and many more remain unidentified.42 The chemical composition of the smoke varies widely depending on the brand of cigarette primarily due to the amount and type of tobacco present, and the filter. By using an IL sensor array, these identified and unidentified components of cigarette smoke are used to generate a fingerprint signal which corresponds to the brand of cigarette. After obtaining the pixel differences from the color maps before and after exposure of the IL sensor array, those data were analyzed by use of statistical methods. Overall, the cigarette smoke experiment that discussed in the article can be considered as an ideal example for an application such as environmental monitoring. Also, these IL sensor arrays show promise to be used in military applications, medical diagnosis, water quality analysis, and food safety.3.8. Statistical discrimination of smoke from three brands of cigarettes
Seven replicate experiments were performed by exposing freshly prepared aqueous solutions of smoke from each of the three brands, giving a total of 21 observations. Each of the sensor arrays was processed as described above. The analysis dataset therefore contained 36 RGB color difference values measured on each of 21 observations.The first modeling approach described above, in which the original 36 RGB color difference values are used in the model, could not be applied to this study. This is because the total number of observations is less than the number of predictor variables. As a result, not even a linear discriminant model can be fit. Therefore only the second approach, which uses PCA to reduce the dimensionality of the predictor variable space, was used to develop a discriminant model for this study.
Fig. S5† shows that the first two principal components account for more than 90% of the variability in the original 36 variables. In Fig. 9, the observations are plotted with respect to their scores on the first two principal components. The three different cigarette brands are tightly grouped, and there is a significant separation between them with respect to these two principal components. We therefore expect that a discriminant model based on these principal components will do a good job of distinguishing between cigarette brands.
 | ||
| Fig. 9 PCA score plot for the identification of cigarette smoke of Marlboro® red, Crowns® and Camel® Turkish domestic blend by using filter paper based ionic liquid sensor arrays. | ||
Discriminant models using one and two principal components were fit. For each of these models, a linear discriminant analysis was determined to be adequate (minimum p-value = 0.4270). The models fit very well. The resubstitution and cross-validation error rates were each 0% for both models. Hence, even the model based on only the first principal component was able to correctly identify the brand for every sample, both under resubstitution and under cross-validation. This is not surprising, given the large amount of separation between brands seen in Fig. 9.
4. Conclusions
In summary, we have successfully designed facile, inexpensive, and disposable photonic IL-based sensor arrays that are sensitive to pH values, as well as acidic and basic vapors. We have fabricated four different sensor arrays by using four different matrices, all of which are highly compatible for using in aqueous media. We have also demonstrated that this sensor array allows discrimination between closely related complex mixtures in aqueous medium. Predictive models were used for successful discrimination and identification of a range of analytes. By use of a variety of matrices, we have demonstrated that these chemosensory ILs do not require any specialized hydrophobic matrix for fabrication of a sensor array. Thus, depending on the particular application, a broadly available, low-cost material can be used as a matrix when fabricating a sensor array. We have also successfully demonstrated the fabrication of a wearable personal sensor arrays by using cotton threads. These wearable sensor arrays can be easily fabricated by manipulating IL-stained cotton threads through use of common household tools such as sewing machines. These thread-based IL sensor arrays can be manufactured on a large scale, allowing applications in bandages, sweatbands, diapers, and other similar systems. Although it needs to be fully evaluated, an IL-based sensor is expected to exhibit a higher sensitivity as compared to a traditional dye-based sensor because of easy accessibility of all dye molecules to the analytes. Finally, we believe that by proper choice of ILs and matrices, these arrays can be constructed to sense a wide range of analytes in a variety of applications, e.g. military applications, medical diagnosis, environmental monitoring, water quality analysis, and food safety.Acknowledgements
This material is based upon work supported in part by the National Science Foundation under Grant number CHE-1243916. WIG also acknowledges Dinushi Kankanan Kapuge for technical support. Also, we acknowledge support from an American Chemical Society Division of Analytical Chemistry graduate fellowship sponsored by Eli Lilly and CompanyNotes and references
- P. Gouma, G. Sberveglieri, R. Dutta, J. W. Gardner and E. L. Hines, MRS Bull., 2004, 29, 697 CrossRef.
- J. W. Gardner and P. N. Bartlett, Sens. Actuators, B, 1994, 18, 210 CrossRef.
- J. W. Gardner and P. N. Bartlett, Electronic noses: principles and applications, Oxford University Press, Oxford, New York, 1999 Search PubMed.
- E. Kress-Rogers, Handbook of biosensors and electronic noses: medicine, food, and the environment, CRC Press, Boca Raton, 1997 Search PubMed.
- R. Marks, Handbook of biosensors and biochips, John Wiley & Sons, Chichester, Hoboken, New Jersey, 2007 Search PubMed.
- C. Zhang and K. S. Suslick, J. Am. Chem. Soc., 2005, 127, 11548 CrossRef PubMed.
- K. S. Suslick, MRS Bull., 2004, 29, 720 CrossRef.
- L. A. Feng, C. J. Musto, J. W. Kemling, S. H. Lim, W. X. Zhong and K. S. Suslick, Anal. Chem., 2010, 82, 9433 CrossRef PubMed.
- B. A. Suslick, L. Feng and K. S. Suslick, Anal. Chem., 2010, 82, 2067 CrossRef PubMed.
- Y. Salinas, J. V. Ros-Lis, J.-L. Vivancos, R. Martinez-Manez, M. D. Marcos, S. Aucejo, N. Herranz and I. Lorente, Analyst, 2012, 137, 3635 RSC.
- X. Y. Huang, J. W. Xin and J. W. Zhao, J. Food Eng., 2011, 105, 632 CrossRef PubMed.
- D. Q. Huo, G. P. Zhang, C. J. Hou, J. L. Dong, Y. C. Zhang, Z. Liu, X. G. Luo, H. B. Fa and S. Y. Zhang, Chin. J. Anal. Chem., 2010, 38, 1115 Search PubMed.
- N. A. Rakow and K. S. Suslick, Nature, 2000, 406, 710 CrossRef PubMed.
- Q. Zhang, S. Zhang, S. Liu, X. Ma, L. Lu and Y. Deng, Analyst, 2011, 136, 1302 RSC.
- L. Feng, C. J. Musto, J. W. Kemling, S. H. Lim and K. S. Suslick, Chem. Commun., 2010, 46, 2037 RSC.
- X. Li, J. Tian and W. Shen, ACS Appl. Mater. Interfaces, 2009, 2, 1 Search PubMed.
- M. Reches, K. A. Mirica, R. Dasgupta, M. D. Dickey, M. J. Butte and G. M. Whitesides, ACS Appl. Mater. Interfaces, 2010, 2, 1722 Search PubMed.
- G. L. L. Reniers and C. A. Brebbia, Sustainable chemistry, WIT Press, Southampton; Boston, 2011 Search PubMed.
- C. Tomasino, J. J. Cuomo, C. B. Smith and G. Oehrlein, J. Ind. Text., 1995, 25, 115 CrossRef PubMed.
- J. P. Hallett and T. Welton, Chem. Rev., 2011, 111, 3508 CrossRef PubMed.
- S. Pandey, Anal. Chim. Acta, 2006, 556, 38 CrossRef PubMed.
- N. V. Plechkova and K. R. Seddon, Chem. Soc. Rev., 2008, 37, 123 RSC.
- X. X. Jin, L. Yu, D. Garcia, R. X. Ren and X. Q. Zeng, Anal. Chem., 2006, 78, 6980 CrossRef PubMed.
- L. Yu, D. Garcia, R. Ren and X. Zeng, Chem. Commun., 2005, 2277 RSC.
- R. Toniolo, A. Pizzariello, N. Dossi, S. Lorenzon, O. Abollino and G. Bontempelli, Anal. Chem., 2013, 85, 7241 CrossRef PubMed.
- W. Zhu, W. Li, H. Yang, Y. Jiang, C. Wang, Y. Chen and G. Li, Chem. – Eur. J., 2013, 19, 11603 CrossRef PubMed.
- K. Y. Yung, A. J. Schadock-Hewitt, N. P. Hunter, F. V. Bright and G. A. Baker, Chem. Commun., 2011, 47, 4775 RSC.
- X. W. Chen, J. W. Liu and J. H. Wang, J. Phys. Chem. B, 2011, 115, 1524 CrossRef PubMed.
- Y. Shu, M. Liu, S. Chen, X. Chen and J. Wang, J. Phys. Chem. B, 2011, 115, 12306 CrossRef PubMed.
- L. C. Branco and F. Pina, Chem. Commun., 2009, 6204 RSC.
- K. S. Suslick, MRS Bull., 2004, 29, 720 CrossRef.
- S. H. Lim, L. Feng, J. W. Kemling, C. J. Musto and K. S. Suslick, Nat. Chem., 2009, 1, 562 CrossRef PubMed.
- D. K. Bwambok, H. M. Marwani, V. E. Fernand, S. O. Fakayode, M. Lowry, I. Negulescu, R. M. Strongin and I. M. Warner, Chirality, 2008, 20, 151 CrossRef CAS PubMed.
- R. E. Del Sesto, T. M. McCleskey, A. K. Burrell, G. A. Baker, J. D. Thompson, B. L. Scott, J. S. Wilkes and P. Williams, Chem. Commun., 2008, 447 RSC.
- S. Institute, SAS/STAT 9.3 user's guide, SAS Institute, 2011 Search PubMed.
- C. Liang, C.-Y. Yuan, R. J. Warmack, C. E. Barnes and S. Dai, Anal. Chem., 2002, 74, 2172 CrossRef CAS.
- J. H. Bang, S. H. Lim, E. Park and K. S. Suslick, Langmuir, 2008, 24, 13168 CrossRef CAS PubMed.
- L. Ferguson and P. Scovazzo, Ind. Eng. Chem. Res., 2007, 46, 1369 CrossRef CAS.
- A. Sen, J. D. Albarella, J. R. Carey, P. Kim and W. B. McNamara Iii, Sens. Actuators, B, 2008, 134, 234 CrossRef CAS PubMed.
- K. S. Suslick, N. A. Rakow and A. Sen, Tetrahedron, 2004, 60, 11133 CrossRef CAS PubMed.
- M. C. Janzen, J. B. Ponder, D. P. Bailey, C. K. Ingison and K. S. Suslick, Anal. Chem., 2006, 78, 3591 CrossRef CAS PubMed.
- K. Torikaiu, Y. Uwano, T. Nakamori, W. Tarora and H. Takahashi, Food Chem. Toxicol., 2005, 43, 559 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Approaches to quantifying a model's predictive accuracy, rationales for considering the two approaches in constructing the discriminant models, NMR, FTIR, UV-Vis, ESI-MS, the images for the appearance of the ILs, and the list of misclassifications by discriminant model. See DOI: 10.1039/c3ra47518b |
| This journal is © The Royal Society of Chemistry 2014 |