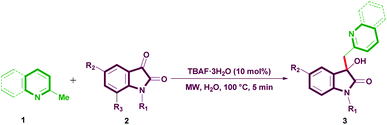
A simple and sustainable tetrabutylammonium fluoride (TBAF)-catalyzed synthesis of azaarene-substituted 3-hydroxy-2-oxindoles through sp3 C–H functionalization†
Kumkum Kumari,
Bharat Kumar Allam and
Krishna Nand Singh*
Department of Chemistry, Faculty of Science, Banaras Hindu University, Varanasi 221005, India. E-mail: knsinghbhu@yahoo.co.in
First published on 16th April 2014
Abstract
A green, practical, and metal-free protocol for direct addition of α-and γ-alkylazaarenes to isatins has been developed via sp3 C–H functionalization in water under controlled microwave radiation. This methodology provides a mild and fast route to biologically important azaarene-substituted 3-hydroxy-2-oxindoles in good to excellent yields.
Introduction
The concept of atom economy has driven chemists to develop more efficient and sustainable methodologies for new bond forming reactions. In this perspective, the direct functionalization of C–H bonds in organic compounds has emerged as a powerful and ideal method for the construction of carbon–carbon and carbon-heteroatom bonds.1 The C–H functionalization logic provides step, atom, and redox economy to advance the organic synthesis.2 The Lewis acid catalyzed activation of the sp3 C–H bond of α-alkylazaarenes has become a focal point of the current research.3 It is well-established that the benzylic C–H bonds of α-alkylpyridines react with electrophiles to form carbon–carbon bonds in nucleophilic fashion.4 However, the substrate scope reported so far is limited to highly reactive polar electrophiles.5 Despite its synthetic utility, only sporadic examples are reported and the functionalization of the sp3 C–H bond in α-alkylquinolines remains less investigated.6 Isatin as a core structure has inspired the development of useful catalytic strategies to give access to interesting molecular architectures with wide biological activities.7 Interest in 3-substituted-3-hydroxy-2-oxindoles has increased rapidly as this core structure is present in a number of potential drug candidates for the treatment of proliferative diseases.8 The direct addition of benzylic C–H bonds of α-alkylazaarenes to isatins for the synthesis of azaarene-substituted 3-hydroxy-2-oxindoles represents the most simple and straightforward method to construct such motifs.9Water is the nature's most amazing gift.10 The use of water as a solvent in organic synthesis is environmentally benign and safe.11 Organocatalysis has emerged and applied rapidly because of its unique advantages.12 The exploitation of organocatalysts that are compatible in aqueous media will provide attractive practical applications.13 However, the use of water as a solvent is still challenging because of the highly insoluble nature of organic molecules in water and the possible reaction of their functional groups with water. Additionally water may weaken the catalytic activity and stereocontrol through interference of the hydrogen bonds and other polar interactions involving catalysts and substrates. Microwave (MW)-assisted synthesis has enriched the rapidly evolving landscape of C–H bond functionalization,14 and results in dramatic rate accelerations, enhanced yields, and cleaner reactions. Therefore, from the point of sustainability and green chemistry, the development of new organocatalytic approaches for reactions in water using controlled MW is highly welcome.
Quaternary ammonium salts are readily available phase transfer catalysts.15,16 Among them, tetraalkylammonium fluorides have been used as a source of naked fluoride ion.17 The nucleophilic affinity of fluoride ion enables the generation of nucleophiles through a deprotonation process.18 Tetrabutylammonium fluoride (TBAF) has been recently explored as a readily available, efficient and water compatible organocatalyst and additive for various organic transformations.19
In light of the above specifics and as a part of our ongoing research interest,20 we report herein the TBAF-catalyzed sp3 C–H functionalization of α/γ-alkylpyridines and α-alkylquinolines in water under controlled MW (Scheme 1). To the best of our knowledge, the findings involve the first time sp3 C–H bond functionalization of α-alkylquinolines for the construction of quinoline-substituted 3-hydroxy-2-oxindoles.
Results and discussion
The optimization of the reaction conditions was carried out by using α-methylquinoline (1b) and N-methylisatin (2a) as model substrates in the presence of TBAF·3H2O as a catalyst under various conditions (Table 1). The screening was initiated using 5 mol% of TBAF·3H2O as a catalyst in pure water at 80 °C, 80 W for 5 min. Gratifyingly, the reaction proceeded to afford selectively the target 3-hydroxy-1-methyl-3-(quinolin-2-ylmethyl)indolin-2-one (3c) in 55% isolated yield (entry 1). Increase of the catalyst concentration to 10 mol% delivered the product in 78% yield under the same conditions (entry 2).| Entry | Catalyst (mol %) | Solvent | T (°C) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: 1b (2 mmol), 2a (2 mmol), 80 W (MW), 5 min.b Isolated yield.c Reaction conducted for 20 min. | ||||
| 1. | TBAF·3H2O (5) | H2O | 80 | 55 |
| 2. | TBAF·3H2O (10) | H2O | 80 | 78 |
| 3. | TBAF·3H2O (10) | H2O | 100 | 90 |
| 4. | TBAF·3H2O (15) | H2O | 100 | 89 |
| 5. | TBAF·3H2O (10) | H2O | 120 | 85 |
| 6. | TBAF·3H2O (10) | — | 100 | 80 |
| 7. | TBAF·3H2O (10) | EtOH | 100 | 47 |
| 8. | TBAF·3H2O (10) | 1,2-Dichloroethane | 100 | 39 |
| 9. | TBAF·3H2O (10) | 1,4-Dioxane | 100 | 60 |
| 10. | TBAF·3H2O (10) | THF | 100 | 80 |
| 11. | TBAF·3H2O (10) | DMSO | 100 | 29 |
| 12. | TBAF·3H2O (10) | Toluene | 100 | 20 |
| 13. | TBAB (10) | H2O | 100 | 69 |
| 14. | TBAI (10) | H2O | 100 | 70 |
| 15. | KF (20) | H2O | 100 | 85 |
| 16. | [Bmim]BF4 (10) | H2O | 100 | 30 |
| 17. | — | H2O | 100 | 20c |
In order to further advance the yield, reaction temperature was raised to 100 °C, which provided the optimum yield (90%, entry 3). Further increase in the catalyst concentration, and temperature did not enhance the yield again (entries 4, 5). Various organic solvents like EtOH, 1,2-dichloroethane, 1,4-dioxane, THF, DMSO, and toluene were also probed for their effect on the reaction yield but did not help (entries 7–12). When the reaction was conducted under solvent-free conditions, a decrease in the product yield was noticed probably due to decomposition of the product (entry 6). To ascertain the role of fluoride ion, tetrabutylammonium salts with different counter ions such as bromide and iodide were also checked, but ended with much lower yields (entries 13–14). When potassium fluoride (20 mol%) was applied as a catalyst under the same set of reaction conditions, it gave rise to 85% product yield (entry 15). The use of neutral ionic liquid [Bmim]BF4 proved rather less effective to deliver the product (entry 16). A control experiment without using TBAF·3H2O, however, delivered the product in 20% yield after 20 min (entry 17).
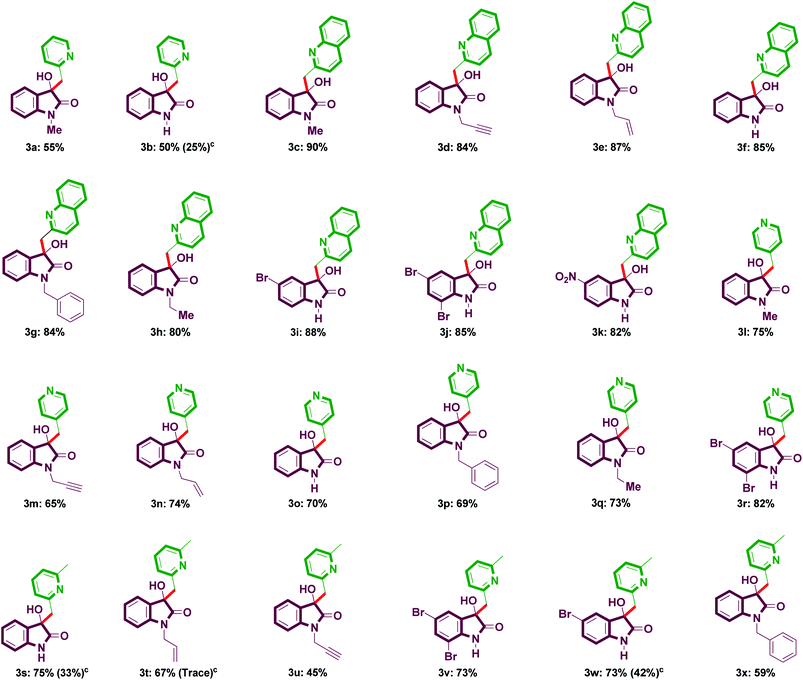
The scope of the optimized procedure was subsequently studied for the reactions of various α/γ-alkylazaarenes with isatins. A number of α/γ-alkylazaarenes viz. α-methylpyridine (1a), α-methylquinoline (1b), γ-methylpyridine (1c), and α,α′-lutidine (1d) were successfully activated and made to react with different isatins such as N-methylisatin (2a), isatin (2b), N-propargylisatin (2c), N-allylisatin (2d), N-benzylisatin (2e), N-ethylisatin (2f), 5-bromoisatin (2g), 5,7-dibromoisatin (2h), and 5-nitroisatin (2i) to provide a diverse range of products 3a–3x in good to excellent yields (Table 2). All the reactions underwent easily with specific product selectivity. Isatins with different substitution patterns participated well in the reaction to deliver the corresponding products. An increase in the yield was noticed for N-substituted isatins in comparison to unsubstituted one. Isatins containing halogen in the aromatic ring also participated well in the reaction. The present catalytic system was also found to be effective in activating the sp3 C–H bond of α-methylquinoline, affording 3-hydroxy-3-(quinolin-2-ylmethyl)indolin-2-ones (3c–3k), although the formation of rather bis(quinolin-2-ylmethyl)indolin-2-one is described in the literature.9a No side products were observed under the present conditions. Interestingly in the case of α,α′-lutidine, only one α-methyl group participated in the functionalization and the corresponding products were isolated exclusively. To confirm the effectiveness of catalytic activity of TBAF·3H2O and to find out the origin of high reaction rates, a model reaction was carried out by using α-methylquinoline (1b) and N-methylisatin (2a) under conventional heating conditions instead of microwave irradiation using the same set of optimized reaction conditions (10 mol% TBAF·3H2O, 100 °C, H2O). To our delight, the reaction went smoothly to deliver the product 3c in 83% yield within 3 h, which clearly demonstrates the efficient catalytic role of TBAF·3H2O in this methodology. However, we may not completely rule out the effect of MW irradiation to help further accelerate the reaction. The product structure of a representative product 1-allyl-3-hydroxy-3-(pyridin-4-ylmethyl)-indolin-2-one (3n) was conclusively confirmed by its single crystal X-ray determination (Fig. 1).
 | ||
| Fig. 1 ORTEP diagram of Product “3n” showing atomic numbering scheme with ellipsoid of 50% probability. | ||
Experimental
General experimental procedure
In a sealed pressure regulation 10 mL pressurized vial were placed α/γ-alkyl azaarene (2 mmol), TBAF·3H2O (10 mol%, 0.2 mmol, 62 mg), isatin (2 mmol), H2O (2 mL), and a teflon coated magnetic stir bar. The vial was closed with a snap on cap, stirred at room temperature for 1 min and then placed into the MW cavity. Microwave irradiation of 80 W at a set temperature of 100 °C was used and the reaction was held under these conditions for 5 min. After completion of the reaction (monitored through TLC), the mixture was cooled to room temperature, poured to a vessel containing distilled water and then extracted with ethyl acetate (2 × 10 mL). The combined organic phase was dried over anhydrous Na2SO4, filtered and concentrated under rotary vacuum evaporator. The resulting crude product was purified using preparative TLC.Conclusions
In conclusion, the work demonstrates a highly efficient, practical, and environmentally benign approach for the sp3 C–H functionalization of α-and γ-alkyl azaarenes catalyzed by a simple water compatible organocatalyst in aqueous media under controlled MW. This study will open a new organocatalytic way for the functionalization of sp3 C–H bonds. The application of this protocol for the functionalization of α-methyl benzothiazoles and α-methyl-1H-benzo[d]imidazoles is presently underway in our laboratory.Acknowledgements
We acknowledge the financial support received from the Department of Biotechnology (DBT), New Delhi. K. K. and B. K. Allam thank Council of Scientific Industrial Research (CSIR), New Delhi for the award of Senior Research Fellowships.Notes and references
- (a) J. Yamaguchi, A. D. Yamaguchi and K. Itami, Angew. Chem., Int. Ed., 2012, 51, 8960–9009 CrossRef CAS PubMed; (b) T. Newhouse and P. S. Baron, Angew. Chem., Int. Ed., 2011, 50, 3362–3374 CrossRef CAS PubMed; (c) W. R. Gutekunst and P. S. Baran, Chem. Soc. Rev., 2011, 40, 1976–1991 RSC; (d) S.-Y. Zhang, F.-M. Zhang and Y.-Q. Tu, Chem. Soc. Rev., 2011, 40, 1937–1939 RSC; (e) L. McMurray, F. O'Hara and M. J. Gaunt, Chem. Soc. Rev., 2011, 40, 1885–1898 RSC; (f) R. Jazzar, J. Hitce, A. Renaudat, J. Sofack-Kreutzer and O. Baudoin, Chem.–Eur. J., 2010, 16, 2654–2672 CrossRef CAS PubMed; (g) T. W. Lyons and M. S. Sanford, Chem. Rev., 2010, 110, 1147–1169 CrossRef CAS PubMed; (h) Y. J. Park, J.-W. Park and C.-H. Jun, Acc. Chem. Res., 2008, 41, 222–234 CrossRef CAS PubMed; (i) G. L. Hamilton and F. D. Toste, Nature, 2009, 459, 917–918 CrossRef CAS PubMed; (j) K. Godula and D. Sames, Science, 2006, 312, 67–72 CrossRef CAS PubMed.
- (a) J. J. Mousseau and A. B. Charette, Acc. Chem. Res., 2013, 46, 412–424 CrossRef CAS PubMed; (b) B. G. Hashiguchi, S. M. Bischof, M. M. Konnick and R. A. Periana, Acc. Chem. Res., 2012, 45, 885–898 CrossRef CAS PubMed; (c) K. M. Engle, T.-S. Mei, M. Wasa and J.-Q. Yu, Acc. Chem. Res., 2012, 45, 788–802 CrossRef CAS PubMed; (d) J. Wencel-Delord, T. Dröge, F. Liu and F. Glorius, Chem. Soc. Rev., 2011, 40, 4740–4761 RSC.
- (a) B.-T. Guan, B. Wang, M. Nishiura and Z. Hou, Angew. Chem., Int. Ed., 2013, 52, 4418–4421 CrossRef CAS PubMed; (b) A. J. Simpson and H. W. Lam, Org. Lett., 2013, 15, 2586–2589 CrossRef CAS PubMed; (c) Y.-G. Zhang, J.-K. Xu, X.-M. Li and S.-K. Tian, Eur. J. Org. Chem., 2013, 3648–3652 CrossRef CAS; (d) J.-j. Jin, D.-c. Wang, H.-y. Niu, S. Wu, G.-r. Qu, Z.-b. Zhang and H.-m. Guo, Tetrahedron, 2013, 69, 6579–6584 CrossRef CAS PubMed; (e) B. Qian, L. Yang and H. Huang, Tetrahedron Lett., 2013, 54, 711–714 CrossRef CAS PubMed; (f) S. A. R. Mulla, M. Y. Pathan and S. S. Chavan, RSC Adv., 2013, 3, 20281–20286 RSC; (g) J.-J. Jin, H.-Y. Niu, G.-R. Qu, H.-M. Guo and J. S. Fossey, RSC Adv., 2012, 2, 5968–5971 RSC; (h) B. Qian, D. Shi, L. Yang and H. Huang, Adv. Synth. Catal., 2012, 354, 2146–2150 CrossRef CAS; (i) S. Duez, A. K. Steib and P. Knochel, Org. Lett., 2012, 14, 1951–1953 CrossRef CAS PubMed; (j) D. Best, S. Kujawa and H. W. Lam, J. Am. Chem. Soc., 2012, 134, 18193–18196 CrossRef CAS PubMed.
- (a) C. A. Ramsden, J. A. Joule, V. V. Zhdankin and A. R. Katritzky, in Handbook of Heterocyclic Chemistry, Elsevier Science & Technology Books, San Diego, 3rd edn, 2010 Search PubMed; (b) V. Rabe, W. Frey, A. Baro, S. Laschat, M. Bauer, H. Bertagnolli, S. Rajagopalan, T. Asthalter, E. Roduner, H. Dilger, T. Glaser and D. Schnieders, Eur. J. Inorg. Chem., 2009, 4660–4674 CrossRef CAS; (c) E. Klingsberg, in The Chemistry of Heterocyclic Compounds, Pyridine and Its Derivatives, Wiley, Hoboken, 2009 Search PubMed; (d) H. Kotsuki, Y. Nakagawa, N. Moriya, H. Tateishi, M. Ochi, T. Suzuki and K. Isobe, Tetrahedron: Asymmetry, 1995, 6, 1165–1174 CrossRef CAS; (e) N. A. Bergman and T. Halvarsson, J. Org. Chem., 1989, 54, 2137–2142 CrossRef CAS.
- (a) Y. Wang, W. Zhao, D. Liu, S. Li, X. Liu, D. Cui and X. Chen, Organometallics, 2012, 31, 4182–4190 CrossRef CAS; (b) R. Niu, J. Xiao, T. Liang and X. Li, Org. Lett., 2012, 14, 676–679 CrossRef CAS PubMed; (c) H. Tsurugi, K. Yamamoto and K. Mashima, J. Am. Chem. Soc., 2011, 133, 732–735 CrossRef CAS PubMed; (d) G. Song, Y. Su, X. Gong, K. Han and X. Li, Org. Lett., 2011, 13, 1968–1971 CrossRef CAS PubMed; (e) B. Qian, P. Xie, Y. Xie and H. Huang, Org. Lett., 2011, 13, 2580–2583 CrossRef CAS PubMed; (f) B. Qian, S. Guo, J. Shao, Q. Zhu, L. Yang, C. Xia and H. Huang, J. Am. Chem. Soc., 2010, 132, 3650–3651 CrossRef CAS PubMed; (g) J. J. Mousseau, A. Larivee and A. B. Charette, Org. Lett., 2008, 10, 1641–1643 CrossRef CAS PubMed; (h) M. L. Hlavinka and J. R. Hagadorn, Organometallics, 2007, 26, 4105–4108 CrossRef CAS; (i) S. Duez, A. K. Steib, S. M. Manolikakes and P. Knochel, Angew. Chem., Int. Ed., 2011, 50, 7686–7690 CrossRef CAS PubMed; (j) F. Sánchez-Sancho and B. Herradón, Heterocycles, 2003, 60, 1843–1854 CrossRef.
- (a) J.-Y. Liu, H.-Y. Niu, S. Wu, G.-R. Qu and H.-M. Guo, Chem. Commun., 2012, 48, 9723–9725 RSC; (b) A. Kumar, L. P. Gupta and M. Kumar, RSC Adv., 2013, 3, 18771–18774 RSC; (c) Y. Obora, S. Ogawa and N. Yamamoto, J. Org. Chem., 2012, 77, 9429–9433 CrossRef CAS PubMed; (d) H. M. Meshram, N. N. Rao, L. C. Rao and N. S. Kumar, Tetrahedron Lett., 2012, 53, 3963–3966 CrossRef CAS PubMed.
- (a) S. Mohammadi, R. Heiran, R. P. Herrera and E. Marqués-López, ChemCatChem, 2013, 5, 2131–2148 CrossRef CAS; (b) G. S. Singh and Z. Y. Desta, Chem. Rev., 2012, 112, 6104–6155 CrossRef CAS PubMed; (c) X.-P. Fu, L. Liu, D. Wang, Y.-J. Chen and C.-J. Li, Green Chem., 2011, 13, 549–553 RSC; (d) C. V. Galliford and K. A. Scheidt, Angew. Chem., Int. Ed., 2007, 46, 8748–8758 CrossRef CAS PubMed; (e) K. C. Nicolaou, S. A. Snyder, N. Giuseppone, X. Huang, M. Bella, M. V. Reddy, P. B. Rao, A. E. Koumbis, P. Giannakakou and A. O'Brate, J. Am. Chem. Soc., 2004, 126, 10174–10182 CrossRef CAS PubMed; (f) K. C. Nicolaou, P. B. Rao, J. Hao, M. V. Reddy, G. Rassias, X. Huang, D. Y.-K. Chen and S. A. Snyder, Angew. Chem., Int. Ed., 2003, 42, 1753–1758 CrossRef CAS PubMed; (g) K. C. Nicolaou, M. Bella, D. Y.-K. Chen, X. Huang, T. Ling and S. A. Snyder, Angew. Chem., Int. Ed., 2002, 41, 3495–3499 CrossRef CAS.
- (a) S. Peddibhotla, Curr. Bioactive Compounds, 2009, 5, 20–38 CrossRef CAS; (b) C. Marti and E. M. Carreira, Eur. J. Org. Chem., 2003, 2003, 2209–2219 CrossRef; (c) Y. Koguchi, J. Kohno, M. Nishio, K. Takahashi, T. Okuda, T. Ohnuki and S. Komatsubara, J. Antibiot., 2000, 53, 105–109 CrossRef CAS.
- (a) R. Niu, J. Xiao, T. Liang and X. Li, Org. Lett., 2012, 14, 676–679 CrossRef CAS PubMed; (b) S. V. N. Vuppalapati and Y. R. Lee, Tetrahedron, 2012, 68, 8286–8292 CrossRef CAS PubMed; (c) M. Raghu, M. Rajasekhar, B. C. O. Reddy, C. S. Reddy and B. V. S. Reddy, Tetrahedron Lett., 2013, 54, 3503–3506 CrossRef CAS PubMed.
- (a) R. N. Butler and A. G. Coyne, Chem. Rev., 2010, 110, 6302–6337 CrossRef CAS PubMed; (b) C.-J. Li and B. M. Trost, Proc. Natl. Acad. Sci., 2008, 105, 13197–13202 CrossRef CAS PubMed; (c) C.-J. Li and L. Chen, Chem. Soc. Rev., 2006, 35, 68–82 RSC; (d) J. E. Klijn and J. B. F. N. Engberts, Nature, 2005, 435, 746–747 CrossRef CAS PubMed; (e) C.-J. Li, Chem. Rev., 2005, 105, 3095–3195 CrossRef CAS PubMed; (f) C.-J. Li, Acc. Chem. Res., 2002, 35, 533–538 CrossRef CAS PubMed; (g) A. J. Kirby, Angew. Chem., Int. Ed., 1996, 35, 706–724 CrossRef.
- (a) C.-J. Li and T. H. Chan, in Comprehensive Organic Reactions in Aqueous Media, Wiley, Hoboken, New Jersey, 2007 Search PubMed; (b) Organic Reactions in Water: Principles, Strategies and Applications, ed. U. M. Lindstrom, Blackwell Publishing, Oxford, 2007 Search PubMed; (c) C.-J. Li and T. H. Chan, in Organic Reactions in Aqueous Media, Wiley, New York, 1997 Search PubMed.
- (a) B. M. Trost and C. S. Brindle, Chem. Soc. Rev., 2010, 39, 1600–1632 RSC; (b) A. Dondoni and A. Massi, Angew. Chem., Int. Ed., 2008, 47, 4638–4660 CrossRef CAS PubMed; (c) P. I. Dalko, in Enantioselective Organocatalysis, Wiley-VCH, Weinheim, 2007 Search PubMed; (d) D. W. C. MacMillan, Nature, 2005, 455, 304–308 CrossRef PubMed; (e) J. Seayad and B. List, Org. Biomol. Chem., 2005, 3, 719–724 RSC.
- Reports on organocatalysis in water: (a) D. K. J. Yeung, T. Gao, J. Huang, S. Sun, H. Guo and J. Wang, Green Chem., 2013, 15, 2384–2388 RSC; (b) F. Zhang, X. Yang, L. Jiang, C. Liang, R. Zhu and H. Li, Green Chem., 2013, 15, 1665–1672 RSC; (c) P. Crisalli and E. T. Kool, J. Org. Chem., 2013, 78, 1184–1189 CrossRef CAS PubMed; (d) S. K. Ghosh, Y. Qiao, B. Ni and A. D. Headley, Org. Biomol. Chem., 2013, 11, 1801–1804 RSC; (e) B. H. Lipshutz and S. Ghorai, Org. Lett., 2012, 14, 422–425 CrossRef CAS PubMed; (f) J. Paradowska, M. Pasternak, B. Gut, B. Gryzło and J. Mlynarski, J. Org. Chem., 2012, 77, 173–187 CrossRef CAS PubMed; (g) Y. Qiao, J. He, B. Ni and A. D. Headley, Adv. Synth. Catal., 2012, 354, 2849–2853 CrossRef CAS; (h) S.-e. Syu, T.-T. Kao and W. Lin, Tetrahedron, 2010, 66, 891–897 CrossRef CAS PubMed; (i) M. Raj and V. K. Singh, Chem. Commun., 2009, 6687–6703 RSC; (j) M. Gruttadauria, F. Giacalone and R. Noto, Adv. Synth. Catal., 2009, 351, 33–57 CrossRef CAS; (k) D. G. Blackmond, A. Armstrong, V. Coombe and A. Wells, Angew. Chem., Int. Ed., 2007, 46, 3798–3800 CrossRef CAS PubMed.
- MW assisted C–H activations: (a) A. Sharama, D. Vacchani and E. V. Der Eycken, Chem.–Eur. J., 2013, 19, 1158–1168 CrossRef PubMed; (b) J. Wencel-Delord, C. Nimphius, F. W. Patureau and F. Glorious, Chem.–Asian J., 2012, 7, 1208–1212 CrossRef CAS PubMed; (c) K. L. Tan, A. Vasudevan, R. G. Bergman, J. A. Ellman and A. J. Souers, Org. Lett., 2003, 5, 2131–2134 CrossRef CAS PubMed.
- (a) J. E. Hein, J. Burés, Y.-H. Lam, M. Hughes, K. N. Houk, A. Armstrong and D. G. Blackmond, Org. Lett., 2011, 13, 5644–5647 CrossRef CAS PubMed; (b) T. Werner, Adv. Synth. Catal., 2009, 351, 1469–1481 CrossRef CAS; (c) M. Lu, D. Zhu, Y. Lu, Y. Hou, B. Tan and G. Zhong, Angew. Chem., Int. Ed., 2008, 47, 10187–10191 CrossRef CAS PubMed; (d) H. Zhao, B. Qin, Z. Liu and Z. Feng, Tetrahedron, 2007, 63, 6822–6826 CrossRef CAS PubMed; (e) J. L. Garcia Ruano, M. Topp, J. López-Cantarero, J. Alemán, M. J. Remuiñán and M. B. Cid, Org. Lett., 2005, 7, 4407–4410 CrossRef CAS PubMed; (f) E. J. Corey and F. Y. Zhang, Angew. Chem., Int. Ed., 1999, 38, 1931–1933 CrossRef CAS; (g) C. M. Starks, J. Am. Chem. Soc., 1971, 93, 195–199 CrossRef CAS.
- (a) J. Novacek and M. Vaser, Eur. J. Org. Chem., 2013, 637–648 CrossRef CAS; (b) D. Enders and T. V. Nguyen, Org. Biomol. Chem., 2012, 10, 5327–5331 RSC; (c) T. Hashimoto and K. Maruoka, Chem. Rev., 2007, 107, 5656–5682 CrossRef CAS PubMed; (d) A. G. Doyle and E. N. Jacobsen, Chem. Rev., 2007, 107, 5713–5743 CrossRef CAS PubMed; (e) M. J. O'Donnell, Acc. Chem. Res., 2004, 37, 506–517 CrossRef CAS PubMed.
- T. Ooi and K. Maruoka, Acc. Chem. Res., 2004, 37, 526–533 CrossRef CAS PubMed.
- J. H. Clark, Chem. Rev., 1980, 80, 429–452 CrossRef CAS.
- (a) N. Singh, B. K. Allam, D. S. Raghuvanshi and K. N. Singh, Adv. Synth. Catal., 2013, 355, 1840–1848 CrossRef CAS; (b) W. Chen, P. Li, T. Miao, L.-G. Meng and L. Wang, Org. Biomol. Chem., 2013, 11, 420–424 RSC; (c) J. Liu, N. Zhang, Y. Yue, D. Wang, Y. Zhang, X. Zhang and K. Zhuo, RSC Adv., 2013, 3, 3865–3868 RSC; (d) C. Bornschein, S. Werkmeister, K. Junge and M. Beller, New J. Chem., 2013, 37, 2061–2065 RSC; (e) J. M. W. Chan, H. Sardon, A. C. Engler, J. M. Garcia and J. L. Hedrick, ACS Macro Lett., 2013, 2, 860–864 CrossRef CAS; (f) M. Revés, A. Lledó, Y. Ji, E. Blasi, A. Riera and X. Verdaguer, Org. Lett., 2012, 14, 3534–3537 CrossRef PubMed; (g) W. Wei, Y. Wang, J. Yin, J. Xue and Y. Li, Org. Lett., 2012, 14, 1158–1161 CrossRef CAS PubMed; (h) Y. Zafrani, L. Yehezkel, M. Goldvaser, D. Marciano, D. Waysbort, E. Gershonov and I. Columbus, Org. Biomol. Chem., 2011, 9, 8445–8451 RSC; (i) D. W. Kim, H.-J. Jeong, S. T. Lim and M.-H. Sohn, Angew. Chem., Int. Ed., 2008, 47, 8404–8406 CrossRef CAS PubMed.
- (a) T. Guntreddi, B. K. Allam and K. N. Singh, RSC Adv., 2013, 3, 9875–9880 RSC; (b) R. Vanjari, B. K. Allam and K. N. Singh, RSC Adv., 2013, 3, 1691–1694 RSC; (c) R. Singh, B. K. Allam, D. S. Raghuvanshi and K. N. Singh, Tetrahedron, 2013, 69, 1038–1042 CrossRef CAS PubMed; (d) R. Singh, D. S. Raghuvanshi and K. N. Singh, Org. Lett., 2013, 15, 4202–4205 CrossRef CAS PubMed; (e) R. Vanjari, T. Guntreddi and K. N. Singh, Org. Lett., 2013, 15, 4908–4911 CrossRef CAS PubMed; (f) D. S. Raghuvanshi, A. K. Gupta and K. N. Singh, Org. Lett., 2012, 14, 4326–4329 CrossRef CAS PubMed; (g) B. K. Allam, Synlett, 2013, 24, 2327–2328 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Detailed experimental procedure and full characterisation data for all the products along with the copies of 1H and 13C spectra. CCDC 980602. For ESI and crystallographic data in CIF or other electronic format See DOI: 10.1039/c3ra47332e |
| This journal is © The Royal Society of Chemistry 2014 |