DOI:
10.1039/C3RA46894A
(Paper)
RSC Adv., 2014,
4, 11723-11731
A novel synthesis of tetra and pentacyclic quinolinopyran tethered pyrazole/coumarin scaffolds via a solid state melt reaction†
Received
21st November 2013
, Accepted 6th January 2014
First published on 9th January 2014
Abstract
A new protocol has been developed for the efficient synthesis of structurally diverse tetra- and pentacyclic quinolinopyran tethered pyrazole/coumarin architectures via a domino Knoevenagel intramolecular hetero-Diels–Alder (IMHDA) strategy using a solid state melt reaction (SSMR) in a highly diastereo and regioselective fashion. Gratifyingly, these heterocyclic frameworks were synthesized under solvent and catalyst free conditions, without the aid of a work-up and column chromatography purification. The generality and functional tolerance of this convergent and environmentally benign method is very attractive.
Introduction
Heterocyclic compounds are pervasive in nature and known to exhibit a variety of highly distinct biological activities1 and thus, have numerous applications in the pharmaceutical industry.2 In general, a longer synthetic sequence is required to synthesize complex heterocyclic compounds. However, the number of steps can be impressively reduced using a domino process. The domino Knoevenagel hetero-Diels–Alder reaction is found to be an effective protocol for the synthesis of complex heterocycles, particularly tetrahydroquinoline, pyrazole and coumarin-tethered scaffolds, which are important as they are the integral part of many natural products with interesting medicinal properties.3,4 Some representative structural motifs5,6 which contain quinoline, pyrano pyrazole and coumarin units are shown in Fig. 1.
 |
| | Fig. 1 Natural products and bioactive molecules embodying the tetrahydroquinoline, pyrazole and coumarin scaffold. | |
Many synthetic chemists are interested in constructing these heterocycles,7 because of their proven biological activities, such as anti-HIV, anticoagulant, antibacterial, antioxidant, anti-inflammatory, antimalarial, antitubercular and molluscicidal activities.
The Baylis–Hillman (BH) reaction is an important carbon–carbon bond forming reaction which has seen immense growth in recent years. It is well documented that Baylis–Hillman adducts have been utilized in the synthesis of a wide variety of natural products and medicinally active components.8
In connection with our ongoing research interest in heterocyclic chemistry,9 we recently reported a novel protocol for the efficient construction of chromenopyran pyrimidinediones9f via a domino Knoevenagel hetero-Diels–Alder reaction sequence through a solid state melt reaction (SSMR). Owing to the biological significance of tetrahydroquinolines,10 we envisaged that the synthesis of quinolinopyranpyrazole/coumarin hybrid molecules is possible to achieve using our solid state melt reaction strategy.
In order to synthesize the key starting material for the execution of our objectives, we have prepared N-tosylated benzyl alcohol (3) from the reaction of O-aminobenzyl alcohol (2) and tosyl chloride with the aid of pyridine as a base. Oxidation of the N-tosylated benzyl alcohol (3) using MnO2 under reflux conditions in DCE as a solvent smoothly led to the N-tosylated aldehyde (4) in good yield (85%). Further treatment of bromo derivatives (5 and 10) of the Baylis–Hillman adducts with N-tosylated aldehyde under the influence of K2CO3 in acetonitrile led to the required precursors10 (6/11) (Scheme 1), which will be very useful in synthesizing the anticipated pyrazole/coumarin tethered tetra/pentacyclic frameworks according to the retrosynthetic strategy shown in Scheme 2.
 |
| | Scheme 1 Synthesis of precursors 6 & 11. | |
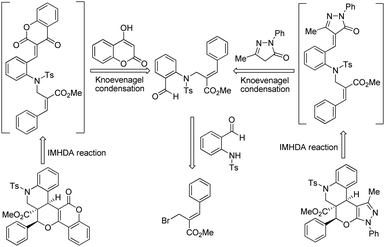 |
| | Scheme 2 Retrosynthetic approach for the synthesis of tetra/pentacyclic quinolinopyranpyrazole/coumarin frameworks. | |
To accomplish our idea towards the construction of these cyclic frameworks, we have utilized the requisite precursor (E)-methyl 2-((N-(2-formylphenyl)-4-methylphenylsulfonamido)methyl)-3-phenylacrylate (6a) by treatment of N-(2-formylphenyl)-4-methylbenzenesulfonamide (4) with methyl (2Z)-2-(bromo methyl)-3-phenylprop-2-enoate (5a). Furthermore, treatment of 6a with 3-methyl-1-phenyl-1H-pyrazol-5(4H)-one (7a) at 180 °C for 1 h in a round bottomed flask successfully afforded the desired product i.e. quinolinopyranpyrazole (9a) (ester moiety in ring junction) in an excellent yield (96%), as shown in Table 1.
Table 1 Synthesis of tetracyclic quinolinopyranpyrazole possessing an ester functionality at an angular position
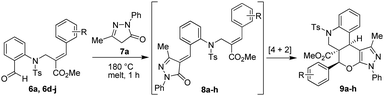
|
| Entry |
R |
Substrate |
Producta,c |
Yieldb (%) |
| All the reactions were carried out using 1 mmol of N-tosylated allyl derivatives (6a, d–j) and 3-methyl-1-phenyl-1H-pyrazol-5(4H)-one (7a) (1 mmol). Isolated yields of the pure product. All the compounds were fully characterized. The structure of the molecule was further confirmed by single-crystal X-ray data. |
| 1 |
H |
6a |
9a |
96 |
| 2 |
4-iPr |
6d |
9b |
92 |
| 3 |
2-OMe |
6e |
9c |
94 |
| 4 |
3,4-OCH2O– |
6f |
9dd |
95 |
| 5 |
4-F |
6g |
9e |
93 |
| 6 |
2-Cl |
6h |
9f |
94 |
| 7 |
3-Cl |
6i |
9g |
95 |
| 8 |
3-Br |
6j |
9h |
96 |
The desired angularly substituted (ester moiety) tetracyclic quinolinopyranpyrazole (9a) was obtained in an excellent yield without using column chromatography purification. The pure product was obtained after washing the crude mass with an ethyl acetate and hexane mixture in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio. Decreasing the reaction temperature (from 180 °C to 140 °C) and time led to the incompletion of the reaction, with the formation of intermediate 8a as well as product 9a.
49 ratio. Decreasing the reaction temperature (from 180 °C to 140 °C) and time led to the incompletion of the reaction, with the formation of intermediate 8a as well as product 9a.
To check the generality of the reaction, we have treated various Baylis–Hillman derivatives (6d–j) with 3-methyl-1-phenyl-1H-pyrazol-5-one (7a), which successfully afforded the desired angularly substituted (ester moiety) tetracyclic quinolinopyranpyrazole products (9b–h) in excellent yields (92–96%). The isolated yields of the pure products 9a–h are summarized in Table 1. All the compounds were characterized by IR, 1H, 13C NMR and mass spectral data.
On the basis of these fruitful results, we examined the reactivity of various Baylis–Hillman derivatives i.e. (Z)-N-(2-cyano-3-arylallyl)-N-(2-formylphenyl)-4-methylbenzenesulfonamides (11a–d,f–h) with pyrazolones (7a/7b), which provided the anticipated tetracyclic quinolinopyranpyrazole products (nitrile moiety in the ring junction) (13a–h) in excellent yields (91–97%). The isolated yields of the pure products 13a–h are summarized in Table 2.
Table 2 Synthesis of tetracyclic quinolinopyranpyrazole possessing a nitrile functionality at an angular position
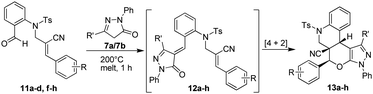
|
| Entry |
R |
Substrate |
R1 |
Producta,c |
Yieldb (%) |
| All the reactions were carried out using 1 mmol of N-tosylated allyl derivatives (11a–d, f–h) and 3-(methyl/phenyl)-1-phenyl-1H-pyrazol-5(4H)-one (7a/7b) (1 mmol). Isolated yields of the pure product. All the compounds were fully characterized. The structure of the molecule was further confirmed by single-crystal X-ray data. |
| 1 |
H |
11a |
Me |
13a |
97 |
| 2 |
2-Me |
11b |
Me |
13b |
92 |
| 3 |
4-Me |
11c |
Me |
13c |
94 |
| 4 |
4-Et |
11d |
Me |
13d |
92 |
| 5 |
4-OMe |
11f |
Me |
13e |
91 |
| 6 |
3,4-diOMe |
11g |
Me |
13f |
93 |
| 7 |
3,4-OCH2O– |
11h |
Me |
13g |
95 |
| 8 |
4-Me |
11c |
Ph |
13hd |
92 |
To further demonstrate the generality of this method, we also decided to utilize 4-hydroxy coumarin in the SSMR strategy using the aforementioned procedure. The Baylis–Hillman derivatives (6a–f,j) were melted with 4-hydroxy-coumarin (14) in a round bottom flask under similar reaction conditions, which successfully led to the desired angularly substituted (ester moiety) pentacyclic quinolinopyrancoumarins (16a–g) in excellent yields (92–98%) (Table 3).
Table 3 Synthesis of pentacyclic quinolinopyrancoumarin possessing an ester functionality at an angular position
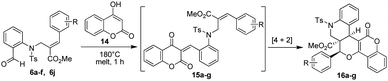
|
| Entry |
R |
Substrate |
Producta,c |
Yieldb (%) |
| All the reactions were carried out using 1 mmol of N-tosylated allyl derivatives (6a–f, j) and 4-hydroxy-2H-chromen-2-one (14) (1 mmol). Isolated yields of the pure product. All the compounds were fully characterized. The structure of the molecule was further confirmed by single-crystal X-ray data. |
| 1 |
H |
6a |
16a |
98 |
| 2 |
2-Me |
6b |
16b |
94 |
| 3 |
4-Me |
6c |
16c |
96 |
| 4 |
4-iPr- |
6d |
16d |
92 |
| 5 |
2-OMe |
6e |
16e |
95 |
| 6 |
3,4-OCH2O |
6f |
16f |
96 |
| 7 |
3-Br |
6j |
16gd |
94 |
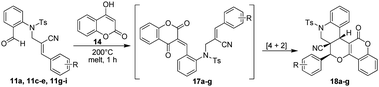
After the successful synthesis of the angularly substituted (ester moiety) pentacyclic quinolinopyrancoumarins (16a–g), we have melted various N-allylated precursors (11a, c–e, g–i) and 4-hydroxy coumarin (14) in a round bottom flask at 200 °C for 1 h, which smoothly afforded the desired angularly substituted pentacyclic quinolinopyrancoumarins (nitrile moiety at angular position) (18a–g) in excellent yields (92–98%). The results are summarized in Table 4. All the compounds are characterized by IR, 1H, 13C NMR and mass spectral data. Based on single-crystal X-ray crystallographic analysis, the structures of compounds 16g and 18d were further confirmed and the ORTEP diagram of the corresponding compounds are shown in Fig. 4 & 5.
Table 4 Synthesis of pentacyclic quinolinopyrancoumarin possessing a nitrile functionality at an angular position
|
| Entry |
R |
Substrate |
Producta,c |
Yieldb (%) |
| All the reactions were carried out using 1 mmol of N-tosylated allyl derivatives (11a, c–e, g–i) and 4-hydroxy-2H-chromen-2-one (14) (1 mmol). Isolated yields of the pure product. All the compounds were fully characterized. The structure of the molecule was further confirmed by single-crystal X-ray data. |
| 1 |
H |
11a |
18a |
98 |
| 2 |
4-Me |
11c |
18b |
94 |
| 3 |
4-Et |
11d |
18c |
96 |
| 4 |
2-OMe |
11e |
18dd |
92 |
| 5 |
3,4-(OMe)2 |
11g |
18e |
95 |
| 6 |
3,4-OCH2O– |
11h |
18f |
96 |
| 7 |
2-Cl |
11i |
18g |
94 |
It is important to describe here that the structure of the compounds (9d, 13h, 16g and 18d) as well as the diastereoselective nature of the reaction was confirmed without ambiguity by X-ray crystallographic analyses, as shown in Fig. 2–5 respectively. The ORTEP diagrams of the following compounds i.e. quinolinopyranpyrazole (9d) and quinolino- pyrancoumarin (16g), show that the relative stereochemistry of the phenyl group and the adjacent ester moiety are in the anti orientation due to the initial trans geometry of the phenyl group and ester moiety present in the double bond at the vicinal position of compounds 6f & 6j. Likewise, the ORTEP diagrams of compounds 13h & 18d (Fig. 3 & 5) show that the phenyl group and adjacent nitrile moiety are in a syn orientation due to the initial cis geometry of the phenyl group and nitrile group tethered with the double bond at the vicinal position of compounds 11c & 11e, confirming the diastereoselectivity of the reaction. Therefore, the trans geometry (aryl and ester groups present in the vicinal positions of compound 6) of the starting material led to the anti products and the cis geometry (aryl and nitrile moieties present in the vicinal positions of compound 11) of the starting material led to the syn product, which clearly shows the high stereoselectivity observed in the reaction.
 |
| | Fig. 2 ORTEP diagram of 9d.11 | |
 |
| | Fig. 3 ORTEP diagram of 13h.11 | |
 |
| | Fig. 4 ORTEP diagram of 16g.11 | |
 |
| | Fig. 5 ORTEP diagram of 18d.11 | |
In conclusion, we have successfully developed a novel and efficient method for the synthesis of tetra- and pentacyclic frameworks containing quinolinopyranpyrazole/coumarin motifs via a domino Knoevenagel intramolecular hetero-Diels–Alder (IMHDA) strategy using a solid state melt reaction (SSMR) in a highly diastereoselective fashion. This reaction leads to a novel class of heterocyclic skeletons, which creates two new rings, and three contiguous stereogenic centers, one of them being an all carbon quaternary center in a single operational step. This reaction also offers other advantages, such as high yields of the products, a short reaction time, clean reactions and no need for a catalyst as well as solvent, which make it a useful and attractive method. Biological screening of these newly synthesized libraries of quinolinopyranpyrazole/coumarin compounds is worth investigating.
Experimental
General
Melting points were recorded on a Superfit (India) capillary melting point apparatus and were uncorrected. IR spectra were recorded on a Perkin Elmer-FTIR spectrometer using solid samples as KBr plates. The compounds 1H NMR (300 MHz, CDCl3) and 13C NMR (75 MHz, 100 MHz, CDCl3) spectra were recorded in deuterochloroform (CDCl3) on a Bruker 300 MHz spectrometer using tetramethylsilane (TMS, δ = 0) as an internal standard at room temperature. Mass spectra were recorded on an Agilent 1200 LC/MS-6110 mass spectrometer. The X-ray diffraction measurements were carried out on a Bruker AXS SMART APEX 2 diffractometer.
Representative procedure for the synthesis of tetracyclic quinolinopyranpyrazoles
A mixture of methyl (2E)-2-{[N-(2-formylphenyl)(4-methylbenzene) sulfonamido]methyl}-3-phenylprop-2-enoate (6a, 0.450 g, 1 mmol) and 3-methyl-1-phenyl-4,5-dihydro-1H-pyrazol-5-one (7a, 0.174 g, 1 mmol) was placed in a round bottom flask and melted at 180 °C for 1 h. After completion of the reaction, as indicated by TLC, the crude product was washed with 5 mL of an ethyl acetate and hexane mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio), which successfully provided the pure product 9a as a colorless solid in a 96% yield (0.58 g).
49 ratio), which successfully provided the pure product 9a as a colorless solid in a 96% yield (0.58 g).
Methyl 16-methyl-8-[(4-methylbenzene)sulfonyl]-11,14-diphenyl-12-oxa-8,14,15-triaza tetracyclo[8.7.0.02,7.013,17] heptadeca-2,4,6,13(17),15-pentaene-10-carboxylate (9a). Yield: 96%; colourless solid; mp: 227–229 °C; 1H NMR (300 MHz, CDCl3): δ 1.44 (s, 3H), 2.13 (s, 3H), 3.43 (s, 3H), 3.91 (d, 1H, J = 14.1 Hz), 4.10 (d, 1H, J = 7.2 Hz), 4.66 (s, 1H), 5.47 (s, 1H), 7.05–7.87 (m, 18H); 13C NMR (75 MHz, CDCl3): δ 13.86, 21.42, 40.45, 42.68, 52.22, 52.61, 84.30, 96.86, 119.65, 123.67, 124.10, 125.59, 126.32, 126.40, 126.53, 128.81, 129.11, 129.42, 130.78, 134.12, 134.52, 135.91, 138.35, 144.14, 145.83, 147.66, 171.52; IR (neat): ν 1735, 1596, 1510 cm−1. MS (m/z): 607 (M+ + 1); elemental analysis for C35H31N3O5S: calculated: C, 69.40; H, 5.16; N, 6.94; found: C, 69.31; H, 5.24; N, 6.86%.
Methyl 16-methyl-8-[(4-methylbenzene)sulfonyl]-14-phenyl-11-[4-(propan-2-yl)phenyl]-12-oxa-8,14,15-triazatetracyclo [8.7.0.02,7.013,17]heptadeca-2(7),3,5,13(17), 15-pentaene-10-carboxylate (9b). Yield: 92%; colourless solid; mp: 227–229 °C; 1H NMR (300 MHz, CDCl3): δ 1.27 (d, J = 6.9 Hz, 6H), 1.50 (s, 3H), 2.19 (s, 3H), 2.94 (sep, 1H, J = 7.2 Hz), 3.37 (s, 3H), 3.89 (d, 1H, J = 12.6 Hz), 4.15 (d, 1H, J = 12.3 Hz), 4.53 (s, 1H), 5.52 (s, 1H), 6.90–7.95 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 13.86, 21.41, 23.82, 23.93, 33.87, 40.41, 42.74, 52.26, 52.56, 84.21, 96.81, 119.64, 123.68, 124.07, 125.53, 126.33, 126.47, 126.84, 128.76, 129.10, 129.41, 130.76, 131.83, 134.10, 135.93, 138.38, 144.13, 145.80, 147.77, 150.17, 171.57; IR (neat): ν 1738, 1592, 1518 cm−1; MS (m/z): 649 (M+ + 1); elemental analysis for C38H37N3O5S: calculated: C 70.46, H 5.76, N 6.49; found: C 70.54, H 5.63, N 6.60%.
Methyl 11-(2-methoxyphenyl)-16-methyl-8-[(4-methylben zene)sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8. 7.0.02,7.013,17]heptadeca-2(7),3,5,13(17),15-pentaene-10-carbo xylate (9c). Yield: 94%; colourless solid; mp: 238–240 °C; 1H NMR (300 MHz, CDCl3): δ 1.54 (s, 3H), 2.22 (s, 3H), 3.37 (s, 3H), 3.79 (s, 3H), 4.04 (d, 1H, J = 12.3 Hz), 4.36 (d, 1H, J = 12.3 Hz), 4.71 (s, 1H), 5.95 (s, 1H), 6.89–7.93 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 13.96, 21.43, 39.31, 43.77, 51.21, 52.46, 55.40, 78.33, 96.97, 110.35, 119.58, 121.05, 123.18, 123.52, 123.99, 125.42, 126.38, 127.29, 127.94, 128.44, 129.05, 129.38, 130.38, 131.10, 134.12, 135.68, 138.45, 144.09, 145.89, 147.95, 156.32, 170.35; IR (neat): ν 1745, 1586, 1520 cm−1; MS (m/z): 637 (M+ + 1); elemental analysis for C36H33N3O6S: calculated: C 68.01, H 5.23, N 6.61; found: C 68.11, H 5.16, N 6.70%.
Methyl 11-(2H-1,3-benzodioxol-5-yl)-16-methyl-8-[(4-methyl benzene)sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0.02,7.013,17]heptadeca-2(7),3,5,13(17),15-pentaene-10-carboxylate (9d). Yield: 95%; colourless solid; mp: 240–242 °C; 1H NMR (300 MHz, CDCl3): δ 1.49 (s, 3H), 2.18 (s, 3H), 3.40 (s, 3H), 3.86 (d, 1H, J = 12.3 Hz), 4.19 (d, 1H, J = 12.3 Hz), 4.50 (s, 1H), 5.44 (s, 1H), 6.03 (d, 2H, J = 4.5 Hz), 6.80–7.95 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 13.86, 21.41, 40.42, 42.69, 52.36, 52.69, 84.16, 96.85, 101.50, 106.88, 108.36, 119.64, 120.51, 123.69, 124.09, 125.60, 126.33, 128.15, 128.80, 129.13, 129.42, 130.72, 134.11, 135.89, 138.33, 144.14, 145.80, 147.61, 148.11, 148.43, 171.54; IR (neat): ν 1785, 1576, 1530 cm−1; MS (m/z): 651 (M+ + 1); elemental analysis for C36H31N3O7S; calculated: C 66.55, H 4.81, N 6.47; found: C 66.63, H 4.70, N 6.58%.
Methyl 11-(4-fluorophenyl)-16-methyl-8-[(4-methylbenzene) sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0. 02,7.013,17]heptadeca-2(7),3,5,13(17),15-pentaene-10-carboxy late (9e). Yield: 93%; colourless solid; mp: 224–226 °C; 1H NMR (300 MHz, CDCl3): δ 1.49 (s, 3H), 2.20 (s, 3H), 3.38 (s, 3H), 3.91 (d, J = 12.3 Hz, 1H), 4.13 (d, J = 12.3 Hz, 1H), 4.52 (s, 1H), 5.53 (s, 1H), 6.91–7.95 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 13.84, 21.43, 40.46, 42.58, 52.20, 52.71, 83.71, 96.84, 115.77, 116.05, 119.68, 123.63, 124.16, 125.70, 126.31, 128.41, 128.52, 128.86, 129.15, 129.44, 130.43, 130.75, 134.10, 135.85, 138.28, 144.19, 145.85, 147.50, 171.54; IR (neat): ν 1792, 1586, 1546 cm−1; MS (m/z): 625 (M+ + 1); elemental analysis for C35H30FN3O5S: calculated: C 67.40, H 4.85, N 6.74; found: C 67.51, H 4.93, N 6.66%.
Methyl 11-(2-chlorophenyl)-16-methyl-8-[(4-methylbenzene) sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0. 02,7.013,17]heptadeca-2(7),3,5,13(17),15-pentaene-10-carboxy late (9f). Yield: 94%; colourless solid; mp: 230–232 °C; 1H NMR (300 MHz, CDCl3): δ 1.49 (s, 3H), 2.21 (s, 3H), 3.39 (s, 3H), 3.89 (d, 1H, J = 12.3 Hz), 4.11 (d, 1H, J = 12.3 Hz), 4.52 (s, 1H), 5.53 (s, 1H), 6.91–7.94 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 13.84, 21.44, 40.50, 42.56, 52.15, 52.76, 83.62, 96.81, 119.67, 123.63, 124.17, 125.73, 126.19, 126.31, 127.96, 128.89, 129.10, 129.16, 129.45, 130.74, 133.04, 134.12, 135.45, 135.84, 138.26, 144.19, 145.85, 147.41, 171.49; IR (neat): ν 1789, 1566, 1556 cm−1. MS (m/z): 641 (M+ + 1); elemental analysis for C35H30ClN3O5S: calculated: C 65.67, H 4.72, N 6.56; found: C 65.78, H 4.63, N 6.63%.
Methyl 11-(3-chlorophenyl)-16-methyl-8-[(4-methylbenzene) sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0. 02,7.013,17]heptadeca-2(7),3,5,13(17),15-pentaene-10-carboxy late (9g). Yield: 95%; colourless solid; mp: 230–232 °C; 1H NMR (300 MHz, CDCl3): δ 1.49 (s, 3H), 2.20 (s, 3H), 3.41 (s, 3H), 3.88 (d, 1H, J = 12.3 Hz), 4.07 (d, 1H, J = 12.6 Hz), 4.53 (s, 1H), 5.54 (s, 1H), 6.91–7.90 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 13.73, 21.32, 40.55, 42.49, 52.06, 52.69, 83.34, 96.69, 119.49, 123.39, 124.18, 125.60, 126.12, 127.83, 128.48, 128.77, 128.98, 129.06, 129.38, 130.64, 132.98, 133.98, 135.24, 135.66, 138.15, 144.16, 145.67, 147.28, 171.17; IR (neat): ν 1758, 1591, 1538 cm−1. MS (m/z): 641 (M+ + 1); elemental analysis for C35H30ClN3O5S: calculated: C 65.67, H 4.72, N 6.56; found: C 65.56, H 4.63, N 6.62%.
Methyl 11-(3-bromophenyl)-16-methyl-8-[(4-methylbenzene) sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0. 02,7.013,17]heptadeca-2(7),3,5,13(17),15-pentaene-10-carboxy late (9h). Yield: 96%; colourless solid; mp: 232–234 °C; 1H NMR (300 MHz, CDCl3): δ 1.49 (s, 3H), 2.20 (s, 3H), 3.42 (s, 3H), 3.87 (d, 1H, J = 12.3 Hz), 4.09 (d, 1H, J = 12.3 Hz), 4.53 (s, 1H), 5.49 (s, 1H), 6.91–7.93 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 13.83, 21.44, 40.39, 42.52, 52.26, 52.83, 83.39, 96.85, 119.71, 122.75, 123.63, 124.21, 124.93, 125.77, 126.22, 126.37, 128.90, 129.19, 129.47, 129.71, 130.56, 130.75, 132.56, 134.10, 135.84, 136.73, 138.23, 144.21, 145.88, 147.31, 171.38; IR (neat): ν 1729, 1578, 1546 cm−1; MS (m/z): 687 (M+ + 2); elemental analysis for C35H30BrN3O5S: calculated: C 61.40, H 4.42, N 6.14; found: C 61.32, H 4.55, N 6.22%.
Representative procedure for the synthesis of tetracyclic quinolinopyranpyrazoles
A mixture of (2Z)-2-{[N-(2-formylphenyl)(4-methylbenzene)sulfonamido]methyl}-3-phenylprop-2-enenitrile (11a), (0.42 g, 1 mmol) and 3-methyl-1-phenyl-1H-pyrazol-5-one (7), (0.17 g, 1 mmol) was placed in a round bottom flask and melted at 200 °C for 1 h. After completion of the reaction, as indicated by TLC, the crude product was washed with 5 mL of an ethyl acetate and hexane mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio) which successfully provided the pure product (13a) as a colorless solid in a 97% (0.56 g) yield.
49 ratio) which successfully provided the pure product (13a) as a colorless solid in a 97% (0.56 g) yield.
16-methyl-8-[(4-methylbenzene)sulfonyl]-11,14-diphenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0.02,7.013,17]heptadeca-2,4,6, 13(17),15-pentaene-10-carbonitrile (13a). Yield: 97%; colourless solid; mp: 223–225 °C; 1H NMR (300 MHz, CDCl3): δ 1.97 (s, 3H), 2.43 (s, 3H), 2.64 (s, 1H), 3.06 (d, 1H, J = 16.2 Hz), 4.59 (d, 1H, J = 15.9 Hz), 4.81 (s, 1H), 7.00–7.92 (m, 18H); 13C NMR (75 MHz, CDCl3): δ 12.35, 21.62, 37.23, 46.03, 50.56, 75.87, 93.80, 120.22, 126.00, 127.31, 127.48, 127.59, 127.92, 128.27, 128.47, 129.07, 129.20, 130.13, 130.22, 130.95, 130.98, 133.15, 136.11, 136.37, 138.08, 144.66, 147.03, 149.85; IR (neat): ν 2228, 1557, 1524 cm−1; MS (m/z): 574 (M+ + 1); elemental analysis for C34H28N4O3S: calculated: C 71.31, H 4.93, N 9.78; found: C 71.40, H 4.85, N 9.86%.
16-Methyl-8-[(4-methylbenzene)sulfonyl]-11-(2-methylphe nyl)-14-phenyl-12-oxa-8,14,15–triazatetracyclo [8.7. 0.02,7. 013,17]heptadeca-2,4,6,13(17),15-pentaene-10-carbonitrile (13b). Yield: 92%; colourless solid; mp: 228–230 °C; 1H NMR (300 MHz, CDCl3): δ 1.86 (s, 3H), 1.97 (s, 3H), 2.43 (s, 3H), 2.64 (s, 1H), 3.06 (d, 1H, J = 15.9 Hz), 4.60 (d, 1H, J = 16.2 Hz), 4.81 (s, 1H), 6.97–7.94 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 12.34, 19.27, 21.61, 37.25, 46.03, 50.57, 93.80, 118.90, 120.23, 120.33, 126.00, 127.32, 127.47, 127.59, 127.91, 128.25, 128.47, 128.69, 129.07, 129.19, 129.91, 130.13, 130.21, 130.95, 133.15, 136.11, 136.38, 138.08, 144.65, 147.04, 149.85; IR (neat): ν 2258, 1537, 1544 cm−1; MS (m/z): 588 (M+ + 1); elemental analysis for C35H30N4O3S: calculated: C 71.65; H, 5.15; N, 9.55; found: C 71.76; H, 5.24; N, 9.63%.
16-Methyl-8-[(4-methylbenzene)sulfonyl]-11-(4-methylphen yl)-14-phenyl-12-oxa-8,14,15–triazatetracyclo [8.7.0.02,7. 013,17]heptadeca-2,4,6,13(17),15-pentaene-10-carbonitrile (13c). Yield: 94%; colourless solid; mp: 229–231 °C; 1H NMR (300 MHz, CDCl3): δ 1.96 (s, 3H), 2.40 (s, 3H), 2.41 (s, 3H), 2.73 (s, 1H), 3.20 (d, 1H, J = 15.6 Hz), 4.44-4.49 (m, 2H), 7.00–7.73 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 12.46, 21.30, 21.60, 36.81, 45.92, 51.33, 80.52, 93.11, 120.22, 125.96, 127.36, 128.05, 128.33, 129.02, 129.07, 129.62, 129.74, 130.12, 132.57, 136.47, 137.14, 138.12, 140.68, 144.56, 147.05, 149.56; IR (neat): ν 2246, 1528, 1536 cm−1; MS (m/z): 588 (M+ + 1); elemental analysis for C35H30N4O3S: calculated: C 71.65; H, 5.15; N, 9.55; found: C 71.76; H, 5.08; N, 9.61%.
16-Methyl-8-[(4-methylbenzene) sulfonyl]-11-(4-ethylphenyl) −14-phenyl-12-oxa-8,14,15-triazatetracyclo[8.7.0.02,7.013,17] heptadeca-2,4,6,13(17),15-pentaene-10-carbonitrile (13d). Yield: 92%; colourless solid; mp: 228–230 °C; 1H NMR (300 MHz, CDCl3): δ 1.27 (t, 3H, J = 7.5 Hz), 1.96 (s, 3H), 2.42 (s, 3H), 2.66–2.72 (m, 3H), 3.21 (d, 1H, J = 15.6 Hz), 4.45–4.50 (m, 2H), 7.00–7.73 (m, 17H); 13C NMR (CDCl3, 75 MHz): δ 12.44, 15.27, 21.59, 28.64, 36.82, 45.96, 51.34, 80.55, 93.10, 120.26, 125.97, 127.36, 128.04, 128.35, 128.39, 128.56, 129.01, 129.06, 129.80, 130.11, 132.59, 136.50, 137.15, 138.13, 144.56, 146.86, 147.04, 149.58; IR (neat): ν 2264, 1567, 1524 cm−1; MS (m/z): 602 (M+ + 1); elemental analysis for C36H32N4O3S: calculated: C 71.98; H, 5.37; N, 9.33; found: C 71.86; H, 5.44; N, 9.42%.
11-(4-Methoxyphenyl)-16-methyl-8-[(4-methylbenzene) sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0.02,7.013,17]heptadeca-2,4,6,13(17),15-pentaene-10-carbonitrile (13e). Yield: 91%; colourless solid; mp: 224–226 °C; 1H NMR (300 MHz, CDCl3): δ 1.96 (s, 3H), 2.42 (s, 3H), 2.64 (s, 1H), 3.41 (d, 1H, J = 15.3 Hz), 3.59 (s, 3H), 4.51 (d, 1H, J = 15.6 Hz), 5.18 (s, 1H), 6.86–7.84 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 12.41, 21.60, 36.79, 46.69, 50.01, 55.41, 72.65, 93.43, 110.60, 120.22, 121.41, 121.76, 125.90, 127.32, 127.47, 127.97, 128.33, 128.85, 129.04, 130.11, 131.20, 132.96, 136.50, 136.86, 138.17, 144.48, 147.03, 149.79, 156.08; IR (neat): ν 2289, 1597, 1532 cm−1; MS (m/z): 604 (M+ + 1); elemental analysis for C35H30N4O4S: calculated: C 69.75; H, 5.02; N, 9.30; found: C 69.86; H, 5.11; N, 9.24%.
11-(3,4-Dimethoxyphenyl)-16-methyl-8-[(4-methylbenzene) sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetracyclo [8.7.0. 02,7.013,17]heptadeca-2,4,6,13(17),15-pentaene-10-carbonitrile (13f). Yield: 93%; colourless solid; mp: 218–220 °C; 1H NMR (300 MHz, CDCl3): δ 1.97 (s, 3H), 2.42 (s, 3H), 2.76 (s, 1H), 3.19 (d, 1H, J = 15.6 Hz), 3.91 (s, 6H), 4.43–4.51 (m, 2H), 6.83–7.73 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 12.49, 21.61, 36.84, 45.82, 51.38, 56.01, 56.18, 60.40, 80.56, 93.16, 110.78, 110.84, 120.11, 121.58, 124.84, 125.95, 127.25, 127.37, 128.01, 128.36, 129.00, 129.10, 130.11, 132.49, 136.47, 137.15, 138.20, 144.59, 147.06, 149.61, 150.77; IR (neat): ν 2268, 1581, 1529 cm−1; MS (m/z): 634 (M+ + 1); elemental analysis for C36H32N4O5S: calculated: C 68.34; H, 5.10; N, 8.85; found: C 68.25; H, 5.17; N, 8.78%.
11-(2H-1,3-Benzodioxol-5-Yl)-16-methyl-8-[(4-methyl benze ne)sulfonyl]-14-phenyl-12-oxa-8,14,15-triazatetra cyclo[8.7. 0.02,7.013,17]heptadeca-2,4,6,13(17),15-pentaene-10-carbo nitrile (13g). Yield: 95%; colourless solid; mp: 217–219 °C; 1H NMR (300 MHz, CDCl3): δ 1.96 (s, 3H), 2.41 (s, 3H), 2.74 (s, 1H), 3.21 (d, 1H, J = 15.9 Hz), 4.39 (s, 1H), 4.52 (d, 1H, J = 15.6 Hz), 6.04 (s, 2H), 6.81–7.73 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 12.46, 21.61, 36.86, 45.88, 51.29, 80.52, 84.15, 93.11, 101.74, 108.28, 108.37, 120.28, 122.90, 126.03, 126.13, 127.35, 128.06, 128.32, 129.05, 129.10, 130.13, 132.48, 136.47, 137.11, 138.08, 144.59, 147.05, 148.53, 149.46; IR (neat): ν 2259, 1578, 1543 cm−1; MS (m/z): 618 (M+ + 1); elemental analysis for C35H30N4O5S: calculated: C 68.17; H, 4.58; N, 9.09; found: C 68.10; H, 4.46; N, 9.17%.
8-[(4-Methylbenzene)sulfonyl]-11-(4-methylphenyl)-14,16-diphenyl-12-oxa-8,14,15-triazatetracyclo[8.7.0.02,7.013,17] heptadeca-2(7),3,5,13(17),15-pentaene-10-carbonitrile (13h). Yield: 92%; colourless solid; mp: 246–248 °C; 1H NMR (300 MHz, CDCl3): δ 2.15 (s, 3H), 2.42 (s, 3H), 3.28 (d, 1H, J = 15.6 Hz), 3.40 (s, 1H), 4.55–4.60 (m, 2H), 7.02–7.82 (m, 22H); 13C NMR (75 MHz, CDCl3): δ 21.29, 21.41, 38.79, 46.35, 51.37, 80.25, 91.96, 120.29, 120.75, 126.14, 126.37, 127.12, 127.69, 128.13, 128.39, 128.43, 129.08, 129.17, 129.61, 129.69, 129.80, 130.37, 132.10, 132.63, 136.74, 137.12, 138.16, 140.77, 144.67, 148.08, 150.00; IR (neat): ν 2234, 1542, 1531 cm−1. MS (m/z): 650 (M+ + 1); elemental analysis for C40H32N4O3S: calculated: C, 74.05; H, 4.97; N, 8.64; found: C 74.12, H 4.90, N 8.71%.
General procedure for the synthesis of pentacyclic quinolinopyrancoumarins
A well ground mixture of methyl (2E)-2-{[N-(2-formylphenyl)(4-methylbenzene) sulfonamido]methyl}-3-phenylprop-2-enoate (6a), (0.45 g, 1 mmol) and 4-hydroxy coumarin (14), (0.16 g, 1 mmol) was placed in a round bottom flask and melted at 180 °C for 1 h. After completion of the reaction, as indicated by TLC, the crude product was washed with 5 mL of an ethyl acetate and hexane mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio) which successfully provided the pure product (16a) as a colorless solid in a 98% (0.58 g) yield.
49 ratio) which successfully provided the pure product (16a) as a colorless solid in a 98% (0.58 g) yield.
Methyl 9-[(4-methylbenzene)sulfonyl]-22-oxo-12-phenyl-13,21-dioxa-9-azapentacyclo [12.8.0.02,11.03,8.015,20]docosa-1(14),3,5,7,15,17,19-heptaene-11-carboxylate (16a). Yield: 98%; colourless solid; mp: 237–239 °C; 1H NMR (300 MHz, CDCl3): δ 2.17 (s, 3H), 3.39 (s, 3H), 3.90 (d, 1H, J = 12.6 Hz), 4.10 (d, 1H, J = 12.3 Hz), 4.52 (s, 1H), 5.50 (s, 1H), 6.66–7.80 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 21.69, 41.40, 43.29, 52.79, 52.84, 81.81, 102.31, 114.64, 116.29, 122.78, 124.01, 124.61, 126.36, 127.37, 128.68, 128.93, 129.16, 129.53, 132.03, 133.87, 134.14, 134.83, 135.55, 143.41, 152.40, 158.69, 159.54, 170.87; IR (neat): ν 1729, 1613, 1567 cm−1; MS (m/z): 595 (M+ + 1); elemental analysis for C34H27NO7S: calculated: C 68.79; H, 4.58; N, 2.36; found: C 68.71; H, 4.66; N, 2.28%.
Methyl 9-[(4-methylbenzene)sulfonyl]-12-(2-methylphenyl)-22-oxo-13,21-dioxa-9-azapentacyclo[12.8.0.02,11. 03,8.015,20] docosa-1(14),3,5,7,15,17,19-heptaene-11-carboxylate (16b). Yield: 94%; colourless solid; mp: 228–230 °C; 1H NMR (300 MHz, CDCl3): δ 2.18 (s, 3H), 2.31 (s, 3H), 3.31 (s, 3H), 4.08 (d, 1H, J = 12.9 Hz), 4.49 (d, 1H, J = 12.6 Hz), 4.60 (s, 1H), 5.73 (s, 1H), 6.66–7.81 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 19.37, 21.69, 41.95, 44.05, 51.96, 52.84, 78.97, 102.44, 114.65, 116.24, 122.87, 123.89, 123.94, 124.59, 126.39, 126.63, 127.24, 127.56, 128.61, 129.16, 129.51, 131.30, 131.96, 132.32, 134.05, 134.70, 135.24, 136.23, 143.42, 152.37, 159.25, 159.65, 170.55; IR (neat): ν 1719, 1623, 1567 cm−1; MS (m/z): 609 (M+ + 1); elemental analysis for C35H29NO7S: calculated: C 69.18; H, 4.81; N, 2.30; found: C 69.10; H, 4.69; N, 2.22%.
Methyl 9-[(4-methylbenzene)sulfonyl]-12-(4-methylphenyl)-22-oxo-13, 21-dioxa-9-azapentacyclo[12.8.0.02,11.03,8.015,20] docosa-1(14),3,5,7,15,17,19-heptaene-11-carboxylate (16c). Yield: 96%; colourless solid; mp: 230–232 °C; 1H NMR (300 MHz, CDCl3): δ 2.20 (s, 3H), 2.42 (s, 3H), 3.42 (s, 3H), 3.91 (d, 1H, J = 11.7 Hz), 4.13 (d, 1H, J = 11.7 Hz), 4.53 (s, 1H), 5.50 (s, 1H), 6.70–7.81 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 21.26, 21.69, 41.42, 43.33, 52.78, 52.88, 81.81, 102.26, 114.69, 116.27, 122.80, 123.97, 124.59, 126.27, 126.36, 127.40, 128.67, 129.15, 129.59, 131.13, 131.98, 133.86, 134.85, 135.56, 139.50, 143.38, 152.40, 158.79, 159.59, 170.93; IR (neat): ν 1717, 1621, 1569 cm−1; MS (m/z): 609 (M+ + 1); elemental analysis for C35H29NO7S: calculated: C 69.18; H, 4.81; N, 2.30; found: C 69.25; H, 4.90; N, 2.21%.
Methyl 9-[(4-methylbenzene)sulfonyl]-12-(4-(propan-2-yl) phenyl)-22-oxo-13,21-dioxa-9-azapentacyclo [12.8.0.02,11. 03,8.015,20]docosa-1(14),3,5,7,15,17,19-heptaene-11-carboxy late (16d). Yield: 92%; colourless solid; mp: 224–226 °C; 1H NMR (300 MHz, CDCl3): δ 1.29 (d, 6H, J = 6.6 Hz), 2.18 (s, 3H), 2.96 (sep, 1H, J = 6 Hz), 3.40 (s, 3H), 3.89 (d, 1H, J = 12.9 Hz), 4.13 (d, 1H, J = 12.9 Hz), 4.51 (s, 1H), 5.50 (s, 1H), 6.66–8.01 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 21.69, 23.84, 23.92, 33.89, 41.37, 43.33, 52.75, 52.84, 81.76, 102.24, 114.70, 116.27, 117.54, 122.81, 123.95, 124.00, 124.59, 126.35, 126.38, 126.96, 127.39, 128.67, 129.14, 129.53, 131.45, 131.96, 133.84, 143.37, 150.34, 152.41, 158.76, 159.59, 170.93; IR (neat): ν 1721, 1619, 1572 cm−1; MS (m/z): 637 (M+ + 1); elemental analysis for C37H33NO7S: calculated: C 69.90; H, 5.23; N, 2.20; found: C 69.99; H, 5.14; N, 2.31%.
Methyl 9-[(4-methylbenzene)sulfonyl]-12-(2-methoxy phenyl)-22-oxo-13,21-dioxa-9-azapentacyclo[12.8.0.02,11.03,8. 015,20] docosa-1(14),3,5,7,15,17,19-heptaene-11-carboxylate (16e). Yield: 95%; colourless solid; mp: 221–223 °C; 1H NMR (300 MHz, CDCl3): δ 2.18 (s, 3H), 3.41 (s, 2H), 3.93 (s, 6H), 4.51 (s, 1H), 5.46 (s, 1H), 6.67–7.78 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 21.67, 41.49, 43.55, 53.11, 55.98, 56.12, 81.72, 102.35, 109.75, 111.30, 114.68, 116.32, 119.03, 122.72, 123.70, 124.02, 124.57, 126.35, 127.34, 128.69, 129.17, 132.02, 133.79, 134.97, 135.69, 143.43, 149.12, 149.89, 152.40, 158.85, 171.10; IR (neat): ν 1729, 1631, 1570 cm−1; MS (m/z): 625 (M+ + 1); elemental analysis for C35H29NO8S: calculated: C 67.40; H, 4.69; N, 2.25; found: C 67.30; H, 4.76; N, 2.37%.
Methyl 12-(2H-1,3-benzodioxol-5-yl)-9-[(4-methylbenzene) sulfonyl]-22-oxo-13,21-dioxa-9-azapentacyclo [12.8.0.02,11. 03,8.015,20]docosa-1(14),3,5,7,15,17,19-heptaene-11-carboxy late (16f). Yield: 96%; colourless solid; mp: 226–228 °C; 1H NMR (300 MHz, CDCl3): δ 2.19 (s, 3H), 3.44 (s, 3H), 3.86 (d, 1H, J = 12.6 Hz), 4.17 (d, 1H, J = 12.6 Hz), 4.51 (s, 1H), 5.44 (s, 1H), 6.06 (d, 2H, J = 2.7 Hz), 6.68–7.81 (m, 15H); 13C NMR (75 MHz, CDCl3): δ 21.69, 41.43, 43.26, 52.87, 52.97, 81.70, 101.58, 102.29, 106.69, 108.52, 114.58, 116.29, 120.32, 122.78, 124.02, 124.63, 126.37, 127.31, 127.73, 128.72, 129.16, 132.05, 133.81, 134.79, 135.51, 143.41, 148.24, 148.53, 152.37, 158.69, 159.58, 170.90; IR (neat): ν 1737, 1641, 1589 cm−1; MS (m/z): 639 (M+ + 1); elemental analysis for C35H27NO9S: calculated: C 65.93; H, 4.27; N, 2.20; found: 65.83; H, 4.33; N, 2.14%.
Methyl 9-[(4-methylbenzene)sulfonyl]-12-(3-bromophenyl)-22-oxo-13,21-dioxa-9-azapentacyclo[12.8.0.02,11.03,8.015,20] docosa-1(14),3,5,7,15,17,19-heptaene-11-carboxylate (16g). Yield: 94%; colourless solid; mp: 232–234 °C; 1H NMR (300 MHz, CDCl3): δ 2.19 (s, 3H), 3.45 (s, 3H), 3.87 (d, 1H, J = 12.9 Hz), 4.06 (d, 1H, J = 12.6 Hz), 4.52 (s, 1H), 5.48 (s, 1H), 6.68–7.80 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 21.69, 41.31, 43.22, 52.96, 53.00, 80.99, 102.49, 114.48, 116.35, 122.72, 122.97, 123.91, 124.10, 124.74, 124.86, 126.41, 127.22, 128.79, 129.21, 129.47, 130.57, 132.15, 132.68, 133.79, 134.83, 135.57, 136.39, 143.50, 152.39, 158.48, 159.44, 170.79; IR (neat): ν 1728, 1614, 1569 cm−1; MS (m/z): 675 (M+ + 2); elemental analysis for C34H26BrNO7S: calculated: C 60.72; H, 3.90; N, 2.08; found: C 60.63; H, 3.81; N, 2.17%.
General procedure for the synthesis of pentacyclic quinolinopyrancoumarins
A mixture of (2Z)-2-{[N-(2-formylphenyl)(4-methylbenzene)sulfonamido] methyl}-3-phenylprop-2-enenitrile (11a), (0.42 g, 1 mmol) and and 4-hydroxy coumarin (14), (0.16 g, 1 mmol) was placed in a round bottom flask and melted at 200 °C for 1 h. After completion of the reaction, as indicated by TLC, the crude product was washed with 5 mL of an ethyl acetate and hexane mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio) which successfully provided the pure product (18a) as a colorless solid in a 98% (0.55 g) yield.
49 ratio) which successfully provided the pure product (18a) as a colorless solid in a 98% (0.55 g) yield.
9-[(4-Methylbenzene)sulfonyl]-22-oxo-12-phenyl-13,21-dioxa -9-azapentacyclo [12.8. 0.02,11.03,8.015,20]docosa-1(14), 3,5,7,15,17,19-heptaene-11-carbonitrile (18a). Yield: 98%; colourless solid; mp: 226–228 °C; 1H NMR (300 MHz, CDCl3): δ 2.41 (s, 3H), 3.15 (s, 1H), 3.43 (d, 1H, J = 15.3 Hz), 4.46 (d, 1H, J = 15.6 Hz), 4.61 (s, 1H), 6.96–7.78 (m, 17H); 13C NMR (75 MHz, CDCl3): δ 21.70, 37.70, 43.91, 50.28, 78.69, 98.46, 114.21, 116.88, 119.15, 122.98, 124.35, 126.94, 127.84, 128.37, 128.71, 129.03, 129.19, 130.57, 130.70, 130.98, 132.12, 133.02, 135.64, 136.45, 145.27, 153.03, 160.72, 161.32; IR (neat): ν 2241, 1738, 1569 cm−1; MS (m/z): 562 (M+ + 1); elemental analysis for C33H24N2O5S: calculated: C 70.70; H, 4.31; N, 5.00; found: C 70.62; H, 4.25; N, 5.11%.
9-[(4-Methylbenzene) sulfonyl]-12-(4-methylphenyl)-22-oxo-13,21-dioxa-9-azapentacyclo [12.8.0.02,11.03,8.015,20]docosa-1(14),3,5,7,15,17,19-heptaene-11-carbonitrile (18b). Yield: 94%; colourless solid; mp: 220–222 °C; 1H NMR (300 MHz, CDCl3): δ 2.41 (s, 3H), 2.42 (s, 3H), 3.13 (s, 1H), 3.41 (d, 1H, J = 15.6 Hz), 4.46 (d, 1H, J = 15.6 Hz), 4.57 (s, 1H), 6.94–7.77 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 21.32, 21.69, 37.73, 43.95, 50.36, 78.64, 98.41, 114.26, 116.85, 119.27, 122.99, 124.31, 126.94, 127.80, 128.26, 128.69, 128.98, 129.13, 129.83, 130.56, 131.07, 132.96, 135.69, 136.48, 140.92, 145.23, 153.03, 160.80, 161.35; IR (neat): ν 2253, 1729, 1558 cm−1; MS (m/z): 576 (M+ + 1); elemental analysis for C34H26N2O5S: calculated: C 71.06; H, 4.56; N, 4.87; found: C 71.13; H, 4.44; N, 4.78%.
9-[(4-Methylbenzene) sulfonyl]-12-(4-ethylphenyl)-22-oxo-13,21-dioxa-9-azapentacyclo [12.8.0.02,11.03,8.015,20]docosa-1(14),3,5,7,15,17,19-heptaene-11-carbonitrile (18c). Yield: 96%; colourless solid; mp: 228–230 °C; 1H NMR (300 MHz, CDCl3): δ 1.31 (t, 3H, J = 7.5 Hz), 2.43 (s, 3H), 2.74 (q, 2H, J = 6.9 Hz), 3.13 (s, 1H), 3.43 (d, 1H, J = 15.3 Hz), 4.49 (d, 1H, J = 15.6 Hz), 4.59 (s, 1H), 6.97–7.79 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 15.35, 21.69, 28.68, 37.72, 44.00, 50.37, 78.68, 98.37, 114.27, 116.85, 119.30, 123.01, 124.29, 126.94, 127.03, 127.83, 128.34, 128.66, 128.98, 129.31, 130.56, 131.12, 132.95, 135.70, 136.50, 145.24, 147.13, 153.04, 160.81, 161.34; IR (neat): ν 2229, 1739, 1542 cm−1; MS (m/z): 590 (M+ + 1); elemental analysis for C35H28N2O5S: calculated: C 71.41; H, 4.79; N, 4.76; found: C 71.48; H, 4.88; N, 4.89%.
12-(2-Methoxyphenyl)-9-[(4-methylbenzene)sulfonyl]-22-oxo-13,21-dioxa-9-azapentacyclo[12.8.0.02,11.03,8.015,20] doco sa-1(14),3,5,7,15,17,19-heptaene-11-carbonitrile (18d). Yield: 92%; colourless solid; mp: 239–241 °C; 1H NMR (300 MHz, CDCl3): δ 2.42 (s, 3H), 2.98 (s, 1H), 3.52 (d, 1H, J = 15.3 Hz), 3.64 (s, 3H), 4.51 (d, 1H, J = 15.3 Hz), 5.25 (s, 1H), 6.93–7.95 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 21.69, 37.81, 44.92, 49.24, 55.47, 71.16, 98.52, 110.72, 114.37, 116.83, 119.32, 120.87, 121.68, 122.93, 124.24, 126.85, 127.49, 127.87, 128.59, 128.82, 130.58, 131.44, 131.85, 132.85, 135.70, 136.31, 145.19, 153.03, 156.33, 161.04, 161.30; IR (neat): ν 2239, 1730, 1529 cm−1; MS (m/z): 592 (M+ + 1); elemental analysis for C34H26N2O6S: calculated: C 69.14; H, 4.44; N, 4.74; found: C 69.25; H, 4.53; N, 4.87%.
12-(3,4-Dimethoxyphenyl)-9-[(4-methylbenzene)sulfonyl]-22-oxo-13,21-dioxa-9-azapentacyclo [12.8.0.02,11.03,8.015,20] docosa-1(14),3,5,7,15,17,19-heptaene-11-carbonitrile (18e). Yield: 95%; colourless solid; mp: 238–240 °C; 1H NMR (300 MHz, CDCl3): δ 2.42 (s, 3H), 3.25 (s, 1H), 3.46 (d, 1H, J = 15.3 Hz), 3.93 (s, 6H), 4.44 (d, 1H, J = 15.0 Hz), 4.59 (s, 1H), 6.89–7.78 (m, 15H); 13C NMR (75 MHz, CDCl3): δ 21.68, 37.79, 43.64, 50.36, 56.16, 60.39, 78.61, 98.60, 110.69, 110.99, 114.29, 116.87, 119.48, 121.52, 123.01, 124.35, 126.40126.98, 127.59, 128.79, 128.93, 130.52, 130.61, 132.99, 135.65, 136.37, 145.22, 149.67, 150.92, 153.03, 160.78, 161.44; IR (neat): ν 2241, 1728, 1538 cm−1; MS (m/z): 622 (M+ + 1); elemental analysis for C35H28N2O7S: calculated: C 67.73; H, 4.55; N, 4.51; found: C 67.80; H, 4.42; N, 4.62%.
9-(Benzenesulfonyl)-12-(2H-1,3-benzodioxol-5-Yl)-22-oxo-13,21-dioxa-9-azapentacyclo [12.8.0.02,11.03,8.015,20]docosa-1(14),3,5,7,15,17,19-heptaene-11-carbonitrile (18f). Yield: 96%; colourless solid; mp: 225–227 °C; 1H NMR (300 MHz, CDCl3): δ 2.41 (s, 3H), 3.14 (s, 1H), 3.43 (d, 1H, J = 15.3 Hz), 4.47 (s, 1H), 4.52 (s, 1H), 6.06 (s, 2H), 6.85–7.77 (m, 15H); 13C NMR (75 MHz, CDCl3): δ 21.70, 37.81, 43.88, 50.30, 78.64, 98.46, 101.82, 108.13, 108.49, 114.19, 116.87, 119.26, 122.91, 122.98, 124.34, 125.61, 126.94, 127.80, 128.68, 129.01, 130.57, 130.94, 133.01, 135.68, 136.42, 145.25, 148.60, 149.60, 153.03, 160.72, 161.32; IR (neat): ν 2240, 1719, 1539 cm−1; MS (m/z): 606 (M+ + 1); elemental analysis for C34H24N2O7S: calculated: C 67.54; H, 4.00; N, 4.63; found: C 67.66; H, 4.10; N, 4.52%.
9-(Benzenesulfonyl)-12-(2-chlorophenyl)-22-oxo-13,21-dioxa-9-azapentacyclo [12.8.0.02,11.03,8.015,20]docosa-1(14),3,5,7,15,17,19-heptaene-11-carbonitrile (18g). Yield: 94%; colourless solid; mp: 238–240 °C; 1H NMR (300 MHz, CDCl3): δ 2.43 (s, 3H), 2.97 (s, 1H), 3.46 (d, 1H, J = 15.3 Hz), 4.59 (d, 1H, J = 15.3 Hz), 5.26 (s, 1H), 6.94–7.94 (m, 16H); 13C NMR (75 MHz, CDCl3): δ 21.71, 38.05, 44.95, 49.25, 73.77, 98.71, 114.10, 116.91, 119.11, 122.86, 124.33, 126.83, 126.92, 128.13, 128.16, 128.23, 128.50, 129.16, 129.45, 130.03, 130.27, 130.64, 131.69, 133.05, 133.82, 135.73, 136.17, 145.28, 153.04, 160.72, 161.02; IR (neat): ν 2241, 1711, 1539 cm−1; MS (m/z): 596 (M+ + 1); elemental analysis for C33H23ClN2O5S: calculated: C 66.61; H, 3.90; N, 4.71; found: C 66.69; H, 3.81; N, 4.65%.
Acknowledgements
We thank DST-SERB for the financial support. We also thank DST-FIST for the NMR facility. D.K. thanks CSIR for his SRF.
Notes and references
-
(a) R. P. Maskey, M. Shaaban, I. Grün-Wollny and H. Laatsch, J. Nat. Prod., 2004, 67, 1131–1134 CrossRef CAS PubMed;
(b) T. M. Potewar, M. K. Kathiravan, A. S. Chothec and K. V. Srinivasana, Eur. J. Chem., 2011, 2, 235–237 CrossRef CAS;
(c) K. A. Milinkevich and M. J. Kurth, Synlett, 2009, 3019–3023 CAS;
(d) S. Dadiboyena, E. J. Valente and A. T. Hamme, Tetrahedron Lett., 2009, 50, 291–294 CrossRef CAS PubMed;
(e) D. Shen, M. Shu and K. T. Chapman, Org. Lett., 2000, 2, 2789–2792 CrossRef CAS PubMed;
(f) A. A. Bekhit, O. A. El-Sayed, E. Aboulmagd and J. Y. Park, Eur. J. Med. Chem., 2004, 39, 249–255 CrossRef CAS PubMed;
(g) V. B. Bojinov and I. K. Garchav, Org. Lett., 2003, 5, 2185–2187 CrossRef CAS PubMed;
(h) A. Kumar, S. Srivastava, G. Gupta, V. Chaturvedi, S. Sinha and R. Srivastava, ACS Comb. Sci., 2011, 13, 65–71 CrossRef CAS PubMed;
(i) B. Eftekhari-Sis, M. Zirak and A. Akbari, Chem. Rev., 2013, 113, 2958–3043 CrossRef CAS PubMed;
(j) C. Despotopoulou, L. Klier and P. Knochel, Org. Lett., 2009, 11, 3326–3329 CrossRef CAS PubMed;
(k) G. Bez and C.-G. Zhao, Org. Lett., 2003, 5, 4991–4993 CrossRef CAS PubMed;
(l) M. Periasamy, G. Srinivas and P. Bharathi, J. Org. Chem., 1999, 64, 4204–4205 CrossRef CAS.
-
(a) W. He, M. R. Meyers, B. Hanney, A. P. Spada, G. Bilder, H. Galzcinski, D. Amin, S. Needle, K. Page, Z. Jayyosi and M. H. Perrone, Bioorg. Med. Chem. Lett., 2003, 13, 3097–3100 CrossRef CAS;
(b) A. R. Katritzky, M. Wang, S. Zhang and M. V. Voronkov, J. Org. Chem., 2001, 66, 6787–6791 CrossRef CAS PubMed;
(c) C. R. W. Guimaraes, D. L. Boger and W. L. Jorgensen, J. Am. Chem. Soc., 2005, 127, 17377–17384 CrossRef CAS PubMed;
(d) M. Wolf and J. L. Diebold, US Pat., 3,647,800, 1972;
(e) M. Wolf and J. L. Diebold, US Pat., 3,637,706, 1972;
(f) M. Wolf and J. L. Diebold, US Pat., 3,674,790, 1972.
-
(a) F. Yang, L. Zheng, J. Xiang, Q. Dang and X. Bai, J. Comb. Chem., 2010, 12, 476–481 CrossRef CAS PubMed;
(b) L. Kim, S. G. Kim, J. Choi and G. H. Lee, Tetrahedron, 2008, 64, 664–671 CrossRef PubMed;
(c) L. F. Tietze and A. Modi, Med. Res. Rev., 2000, 20, 304–322 CrossRef CAS;
(d) L. F. Tietze, C. Ott, H. Geibler and F. Haunert, Eur. J. Org. Chem., 2001, 1625–1630 CrossRef CAS;
(e) L. F. Tietze and A. Steinmetz, Angew. Chem., Int. Ed. Engl., 1996, 35, 651–652 CrossRef CAS;
(f) L. F. Tietze, Chem. Rev., 1996, 96, 115 CrossRef CAS PubMed;
(g) R. A. A. Foster and M. C. Willis, Chem. Soc. Rev., 2013, 42, 63–76 RSC.
-
(a) L. F. Tietze, J. Bachmann, J. Wichmann, Y. Zhou and T. Rasche, Liebigs Ann./Recl., 1997, 881–886 CrossRef CAS;
(b) D. A. Evans, J. S. Johnson and E. J. Olhava, J. Am. Chem. Soc., 2000, 122, 1635–1649 CrossRef CAS;
(c) M. Toyota, C. Komori and M. Ihara, J. Org. Chem., 2000, 65, 7110–7113 CrossRef CAS PubMed;
(d) B. Bear and K. J. Shea, Org. Lett., 2001, 3, 723–726 CrossRef CAS PubMed;
(e) M. Prakesch, U. Sharma, M. Sharma, S. Khadem, D. M. Leek and P. Arya, J. Comb. Chem., 2006, 8, 715–734 CrossRef CAS PubMed;
(f) D. Catarzi, L. Cecchi, V. Colotta and G. Filacchioni, J. Med. Chem., 1995, 38, 1330–1336 CrossRef CAS;
(g) S. Fustero, M. Sanchez-Rosello, P. Barrio and A. Simon-Fuentes, Chem. Rev., 2011, 111, 6984–7034 CrossRef CAS PubMed;
(h) S. Bondock, W. Khalifa and A. A. Fadda, Eur. J. Med. Chem., 2011, 46, 2555–2561 CrossRef CAS PubMed;
(i) N. Isambert and R. Lavilla, Chem.–Eur. J., 2008, 14, 8444–8454 CrossRef CAS PubMed.
-
(a) Y. Miura, N. Hayashi, S. Yokoshima and T. Fukuyama, J. Am. Chem. Soc., 2012, 134, 11995–11997 CrossRef CAS PubMed;
(b) C. Z. Ding, J. T. Hunt, C. Ricca and V. Manne, Bioorg. Med. Chem. Lett., 2000, 10, 273–275 CrossRef CAS;
(c) M. Radi, V. Bernardo, B. Bechi, D. Castagnolo, M. Pagano and M. Botta, Tetrahedron Lett., 2009, 50, 6572–6575 CrossRef CAS PubMed.
-
(a) D. A. Powell and R. A. Batey, Org. Lett., 2002, 4, 2913–2916 CrossRef CAS PubMed;
(b) H. Ishii, T. Ishikawa, M. Murota, Y. Aoki and T. Harayama, J. Chem. Soc., Perkin Trans. 1, 1993, 1019–1022 RSC;
(c) H. Ishii, T. Ishikawa and J. Haginiwa, Yakugaku Zasshi, 1977, 97, 890–900 CAS;
(d) T. T. Y. Lee, Y. Kashiwada, L. Huang, J. Snider, M. Cosentino and K. H. Lee, Bioorg. Med. Chem., 1994, 2, 1051–1056 CrossRef CAS.
-
(a) G. Sabitha, N. Fatima, E. V. Reddy and J. S. Yadav, Adv. Synth. Catal., 2005, 347, 1353–1355 CrossRef CAS;
(b) Y. R. Lee and T. V. Hung, Tetrahedron, 2008, 64, 7338–7346 CrossRef CAS PubMed;
(c) B. Baruah and P. J. Bhuyan, Tetrahedron, 2009, 65, 7099–7104 CrossRef CAS PubMed;
(d) M. Radi, V. Bernardo, B. Bechi, D. Castagnolo, M. Pagano and M. Botta, Tetrahedron Lett., 2009, 50, 6572–6575 CrossRef CAS PubMed;
(e) E. Ceulemans, M. Voets, S. Emmers, K. Uytterhoeven, L. V. Meervelt and W. Dehaen, Tetrahedron, 2002, 58, 531–544 CrossRef CAS;
(f) K. C. Majumdar, A. Taher and R. K. Nandi, Tetrahedron, 2012, 68, 5693–5718 CrossRef CAS PubMed;
(g) J. N. Tan, M. Li and Y. Gu, Green Chem., 2010, 12, 908–914 RSC;
(h) J. S. Yadav, B. V. S. Reddy, V. Naveenkumar, R. S. Rao and K. Nagaiah, Synthesis, 2004, 1783–1788 CrossRef CAS PubMed;
(i) M. J. Khoshkholgh, M. Lotfi, S. Balalaie and F. Rominger, Tetrahedron, 2009, 65, 4228–4234 CrossRef CAS PubMed;
(j) Z. Dong, J. Feng, W. Cao, X. Liu, L. Lin and X. Feng, Tetrahedron Lett., 2011, 52, 3433–3436 CrossRef CAS PubMed.
-
(a) D. Basavaiah and G. Veeraraghavaiah, Chem. Soc. Rev., 2012, 41, 68–78 RSC;
(b) T. Y. Liu, M. Xie and Y. C. Chen, Chem. Soc. Rev., 2012, 41, 4101–4112 RSC;
(c) D. Basavaiah, B. S. Reddy and S. S. Badsara, Chem. Rev., 2010, 110, 5447–5674 CrossRef CAS PubMed;
(d) V. Singh and S. Batra, Tetrahedron, 2008, 64, 4511–4574 CrossRef CAS PubMed;
(e) Y. Wei and M. Shi, Chem. Rev., 2013, 113, 6659–6690 CrossRef CAS PubMed.
-
(a) M. Bakthadoss, D. Kannan and G. Sivakumar, Synthesis, 2012, 44, 793–797 CrossRef CAS PubMed;
(b) M. Bakthadoss, J. Srinivasan and R. Selvakumar, Synthesis, 2012, 5, 755–766 CrossRef PubMed;
(c) M. Bakthadoss and G. Murugan, Eur. J. Org. Chem., 2010, 5825–5830 CrossRef CAS;
(d) M. Bakthadoss, N. Sivakumar and A. Devaraj, Synthesis, 2011, 611–618 CrossRef CAS PubMed;
(e) M. Bakthadoss and N. Sivakumar, Synlett, 2011, 1296–1302 CrossRef CAS PubMed;
(f) M. Bakthadoss, G. Sivakumar and D. Kannan, Org. Lett., 2009, 11, 4466–4469 CrossRef CAS PubMed;
(g) M. Bakthadoss and N. Sivakumar, Synlett, 2009, 1014–1018 CrossRef CAS PubMed.
- V. Sridharan, P. A. Suryavanshi and J. C. Menendez, Chem. Rev., 2011, 111, 7157–7259 CrossRef CAS PubMed.
- Structures were confirmed by single-crystal X-ray data.†.
Footnote |
| † Electronic supplementary information (ESI) available: Copies of the 1H and 13C spectra of all the new compounds. CCDC 970849, 970851, 902987 and 902984. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c3ra46894a |
|
| This journal is © The Royal Society of Chemistry 2014 |
Click here to see how this site uses Cookies. View our privacy policy here. 

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio. Decreasing the reaction temperature (from 180 °C to 140 °C) and time led to the incompletion of the reaction, with the formation of intermediate 8a as well as product 9a.
49 ratio. Decreasing the reaction temperature (from 180 °C to 140 °C) and time led to the incompletion of the reaction, with the formation of intermediate 8a as well as product 9a.



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio), which successfully provided the pure product 9a as a colorless solid in a 96% yield (0.58 g).
49 ratio), which successfully provided the pure product 9a as a colorless solid in a 96% yield (0.58 g).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio) which successfully provided the pure product (13a) as a colorless solid in a 97% (0.56 g) yield.
49 ratio) which successfully provided the pure product (13a) as a colorless solid in a 97% (0.56 g) yield.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio) which successfully provided the pure product (16a) as a colorless solid in a 98% (0.58 g) yield.
49 ratio) which successfully provided the pure product (16a) as a colorless solid in a 98% (0.58 g) yield.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 ratio) which successfully provided the pure product (18a) as a colorless solid in a 98% (0.55 g) yield.
49 ratio) which successfully provided the pure product (18a) as a colorless solid in a 98% (0.55 g) yield.





