Highly selective direct reductive amidation of nitroarenes with carboxylic acids using cobalt(II) phthalocyanine/PMHS†‡
Vishal Kumar,
Manoranjan Kumar,
Sushila Sharma and
Neeraj Kumar*
Natural Plant Products Division, CSIR-Institute of Himalayan Bioresource Technology, Palampur, H. P., India 176001. E-mail: neerajnpp@rediffmail.com; neeraj@ihbt.res.in; Fax: +91-1894-230433; Tel: +91-1894-230426
First published on 17th February 2014
Abstract
Cobalt phthalocyanine has been employed for highly chemoselective direct reductive amidation of nitroarenes with non-activated carboxylic acids using polymethylhydrosiloxane as an inexpensive, stable and environmentally benign reducing agent. A large range of functional groups such as amide, ester, nitrile, halogen, lactone, hydroxy, alkene and heterocycles were well tolerated.
Introduction
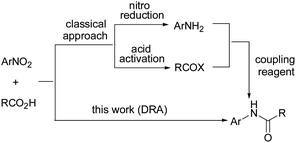
Amides constitute the key functional part of various natural products, polymers, proteins and pharmaceuticals.1,2 Therefore, amide bond synthesis is one of the most important transformations in organic chemistry. Typically, amide synthesis is carried out by coupling of carboxylic acids and amines using either a coupling reagent3 or by prior conversion of the carboxylic acid into a derivative (Scheme 1).4 However, direct amidation of non-activated carboxylic acids is more economic, and highly desirable for commercial purposes. On the other hand, amines required for amide synthesis are usually prepared by reduction of corresponding nitro compounds, for which various methods including catalytic hydrogenation,5 sodium borohydride/catalyst,6 hydrazine/catalyst7 and a variety of other catalytic systems8 have been reported. These methods mostly involved the use of expensive metal catalysts and moisture sensitive reagents. Recently, we have utilized metal phthalocyanines (MPcs) of relatively cheap metals as catalyst for highly chemoselective reduction of nitroarenes with hydrazine hydrate as reducing agent.9 Nevertheless, being mild and environmentally safe, organosilanes are advantageous over hydrazine hydrate and other reducing agents. Amongst silane based reducing agents, polymethylhydrosiloxane (PMHS) attracts great attention due to its low cost, and stable and environment friendly nature. Also, PMHS is waste of silicon industry and it gets converted into polysiliconates during hydrosilylation reactions, which are precursors of porous derivatives useful for absorbent properties.10 Silanes have been used extensively as reducing agents, however, very few reports are focused on nitro reduction albeit most of these methods utilized high loading of costly metal catalysts or toxic ligands.11Scrutinizing direct reductive amidation (DRA) of nitroarenes led to very few reports such as Mo(CO)6, thioacetate mediated acetamidation, Zn/AcOH, Pt nanowire etc.12 Most of these methods have one or more drawbacks like use of stoichiometric amount of metal catalyst, activated carboxylic acid derivatives or difficult to handle H2 as reducing agent.
In our recent report on direct reductive amination of carbonyl compounds using CoPc/Ph2SiH2 system in ethanol, the reduction of nitro group was not observed, however, the reaction of 2-carboxybenzaldehyde and amine resulted in reductive amination followed by intramolecular amidation of secondary amine with CO2H group.13 Inspired by this result and our reports on MPcs catalyzed reduction of nitroarenes,9 we explored MPcs for direct reductive amidation of nitroarenes with carboxylic acids using hydrosilanes as reducing agent. Here, in the present work we disclose CoPc catalyzed direct reductive amidation of nitroarenes with carboxylic acids using polymethylhydrosiloxane (PMHS) as an inexpensive, stable and environment friendly10,14 reducing agent.
Results and discussion
For the optimization of reaction conditions, the direct reductive amidation of nitrobenzene with acetic acid was carried out by using different catalysts, hydrogen sources and solvents under varying temperature conditions. Amongst different catalysts, CoPc was found to be the most active at 100 °C (Table 1, entry 4), whereas relatively lower yield was obtained with FePc, tBu4CoPc and NiPc (Table 1, entries 3, 5 and 6). In the presence of CuPc, the desired product was not observed and most of the substrate was converted to azoxybenzene (Table 1, entry 7). No reaction was observed with cobalt salts, such as CoCl2·6H2O and CoSO4·7H2O (Table 1, entries 1 and 2). The use of carboxylic acid itself as reaction medium afforded best result and 2.0 mL amount was optimized for complete reaction of 1.0 mmol of substrate (Table 1, entries 4, 14 and 15). Different proportions of mixture of AcOH and H2O as reaction medium were also tested. A gradual decrease in yield of product was observed with decrease in concentration of AcOH (Table 1, entries 16–18). The investigation of different reducing agents revealed PMHS as the best hydrogen source giving 91% yield of the desired product (Table 1, entries 4, 8–11). As expected, no reaction was observed without catalyst or reducing agent (Table 1, entries 12 and 13).| Entry | Catalyst | Solvent | Reducing agent | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: nitrobenzene (1.0 mmol), catalyst (1 mol%), reducing agent (4.0 mmol), solvent (2 mL) at 100 °C for 12 h.b Yield of isolated product, nd = not detected.c Azoxybenzene was formed in 62% yield.d Reaction was carried out with 0.5 mL of AcOH.e Reaction was carried out with 1.0 mL of AcOH. | ||||
| 1 | CoCl2·6H2O | AcOH | PMHS | nd |
| 2 | CoSO4·7H2O | AcOH | PMHS | nd |
| 3 | FePc | AcOH | PMHS | 37 |
| 4 | CoPc | AcOH | PMHS | 91 |
| 5 | tBu4CoPc | AcOH | PMHS | 79 |
| 6 | NiPc | AcOH | PMHS | 80 |
| 7 | CuPc | AcOH | PMHS | ndc |
| 8 | CoPc | AcOH | PhSiH3 | 60 |
| 9 | CoPc | AcOH | Ph2SiH2 | 80 |
| 10 | CoPc | AcOH | Ph3SiH | nd |
| 11 | CoPc | AcOH | Et3SiH | nd |
| 12 | — | AcOH | PMHS | nd |
| 13 | CoPc | AcOH | — | nd |
| 14d | CoPc | AcOH | PMHS | 36 |
| 15e | CoPc | AcOH | PMHS | 45 |
| 16 | CoPc | AcOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1 H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
PMHS | nd |
| 17 | CoPc | AcOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1 H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
PMHS | 54 |
| 18 | CoPc | AcOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (3 H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
PMHS | 81 |
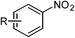
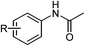
Furthermore, to explore the scope of the developed catalytic method, the reductive amidation of structurally diverse nitroarenes with acetic acid was carried out. The reaction of halogen substituted nitroarenes proceeded smoothly without dehalogenation and the desired products were obtained in excellent yield (Table 2, entries 2–4), overcoming the drawback of dehalogenation as observed in earlier reported methods such as catalytic hydrogenation,5 Pd(OAc)2/PMHS11b,c and S8/mild base.15 In the reaction of methyl or hydroxy substituted nitroarenes, the desired amides were obtained in very good yields (Table 2, entries 5 and 6). N-Acylation is generally accompanied by O-acylation, but, under present reaction conditions –OH group remained unaffected. Substrates containing sensitive functional groups such as ester, nitrile, keto, amide and lactone were found to give the desired products in good to excellent yield with high chemoselectivity (Table 2, entries 7–10 and 15). Various heterocyclic nitroarenes were successfully converted to corresponding amides without affecting heterocyclic ring (Table 2, entries 16–18).
| Entry | Substrate | Product | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: nitroarene (1 mmol), PMHS (4.0 mmol) and AcOH (2.0 mL) at 100 °C for 12 h.b For entries 1, 7, 14, 16, 20 and 21, average isolated yields of three experiments are reported (SD < 2%), and for rest of the entries, isolated yield of signal experiment is given.c Yield in parenthesis is of isolated product on carrying out the reaction with 1 g of substrate.d Quantity of PMHS used was 8.0 mmol.e Direduction product was obtained in 14% yield.f Monoreduction product was obtained in 22% yield.g Corresponding amine was obtained in 25% yield.h Corresponding amine was obtained in 18% yield. | |||
 |
 |
||
| 1 | R = H | 91 | |
| 2 | R = 4-F | 94 | |
| 3 | R = 4-Cl | 88 (82)c | |
| 4 | R = 4-Br | 93 (83)c | |
| 5 | R = 4-CH3 | 78 | |
| 6 | R = 4-OH | 85 | |
| 7 | R = 3-CO2CH3 | 68 | |
| 8 | R = 3-CN | 61 (54)c | |
| 9 | R = 4-COCH3 | 92 (80)c | |
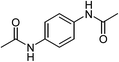
| 10 | R = 4-NHCOCH3 | 78 | |
| 11 |  |
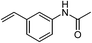 |
70 |
| 12d | 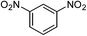 |
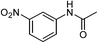 |
67e |
| 13d | 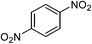 |
 |
63f |
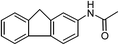
| 14 | 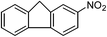 |
 |
52 |
| 15 | 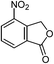 |
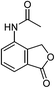 |
84 (71)c |
| 16 | 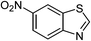 |
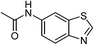 |
90 |
| 17 | 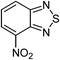 |
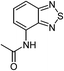 |
80 |
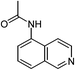
| 18 | 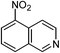 |
 |
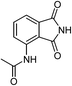
52g |
| 19 | 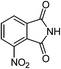 |
 |
64h |
| 20 | 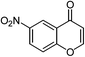 |
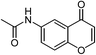 |
86 |
| 21 |  |
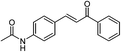 |
81 (70)c |
One of the most important finding of the present method was the tolerance of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond (highly susceptible to reduction).5 In case of 3-nitrostyrene the desired product was obtained in good yield (Table 2, entry 11). The reductive amidation of dinitro compounds resulted in mixture of products. The reaction of 1,3-dinitrobenzene yielded monoamide as the major product, whereas in case of 1,4-dinitrobenzene, the product corresponding to the reduction of both nitro groups was obtained in major quantity (Table 2, entries 12 and 13). 2-Nitrofluorene was selectively (>99%) reduced to the corresponding amide with moderate yield (Table 2, entry 14). Another major finding of the present method was the high selectivity in case of substrates with α,β-unsaturated carbonyl group giving the corresponding amides in very good yields (Table 2, entries 20 and 21). The direct reductive amidation of rarely studied substrate 4-nitrophthalimide gave the desired product in good yield with high selectivity (Table 2, entry 19). Further, the scope of the method was extended to other carboxylic acids (Table 3). The CoPc catalyst showed good to excellent activity with formic acid, acetic acid, propionic acid and butyric acid and good yields of the desired amides were obtained (Table 3, entries 1–4).
C bond (highly susceptible to reduction).5 In case of 3-nitrostyrene the desired product was obtained in good yield (Table 2, entry 11). The reductive amidation of dinitro compounds resulted in mixture of products. The reaction of 1,3-dinitrobenzene yielded monoamide as the major product, whereas in case of 1,4-dinitrobenzene, the product corresponding to the reduction of both nitro groups was obtained in major quantity (Table 2, entries 12 and 13). 2-Nitrofluorene was selectively (>99%) reduced to the corresponding amide with moderate yield (Table 2, entry 14). Another major finding of the present method was the high selectivity in case of substrates with α,β-unsaturated carbonyl group giving the corresponding amides in very good yields (Table 2, entries 20 and 21). The direct reductive amidation of rarely studied substrate 4-nitrophthalimide gave the desired product in good yield with high selectivity (Table 2, entry 19). Further, the scope of the method was extended to other carboxylic acids (Table 3). The CoPc catalyst showed good to excellent activity with formic acid, acetic acid, propionic acid and butyric acid and good yields of the desired amides were obtained (Table 3, entries 1–4).
| Entry | Carboxylic acid | Product | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: nitroarene (1 mmol), PMHS (4.0 mmol) and carboxylic acid (2.0 mL) at 100 °C for 12 h.b For entry 2 average isolated yield of three experiments is reported (SD = 1.8%), and for rest of the entries, isolated yield of signal experiment is given. | |||
| 1 | HCOOH | 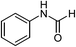 |
83 |
| 2 | CH3COOH | 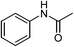 |
91 |
| 3 | C2H5COOH | 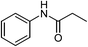 |
89 |
| 4 | C3H7COOH |  |
78 |
When the method was applied on larger scales (up to gram scale), with some important substrates containing functional groups such as chloro, bromo, cyano and ketone (Table 2, entries 3, 4, 8, 9, 15 and 21) good isolated yields were obtained in all the cases.
As far as mechanism of the reaction is concerned, it was proposed in our previous report on CoPc/N2H4·H2O catalyzed reduction of nitroarenes that the electron transfer process between N2H4·H2O and CoPc leads to H2 generation.9a In the present case, no change in oxidation state (as monitored by UV-Vis spectroscopy) and color of CoPc was observed, which ruled out the involvement of electron transfer process for the activation of PMHS. Since, hydrosilanes are known to get activated by Lewis as well as Bronsted acids,13a,14 the involvement of AcOH in PMHS activation was anticipated. To confirm this, two separate reactions of PMHS with AcOH and CoPc were performed and after 12 h the reaction mixtures were analyzed by 1H NMR. The appearance of a new peak at δ 2.10 corresponding to Si–OAc and decrease in the intensity of peak at δ 4.70 (Si–H) in former case and no reaction in the latter case clearly showed the activation of PMHS by AcOH (for details see ESI‡). It was further supported by the fact that nitro reduction was not observed in the presence of other solvents (see ESI, Table S1‡). In order to further assure the requirement of acidic medium, the model reaction was carried out in the presence of mineral acids (HCl and HBr) in different solvents using stoichiometric amounts of acetic acid. The desired amide was obtained, but in low yield (22%) using ethylene glycol as solvent (1 M HCl) and 3.0 equivalents of acetic acid (for details see ESI, Table S2‡), indicating the requirement of carboxylic acid in excess. However, the exact role of CoPc has not been established, some kind of interaction between CoPc and nitro substrate to form a transition state at elevated temperature (100 °C) was anticipated, as no reaction took place in the absence of CoPc. Further, to determine which pathway is predominant between two classical pathways for nitro reduction, the reductive amidation of two intermediates phenylhydroxylamine and azobenzene was carried out under present reaction conditions. It was observed that phenylhydroxylamine gave desired amide in 76% yield in shorter time (after 3 h) while, the reduction of azobenzene was not complete even after 12 h and desired amide was obtained only in 50% yield. These results along with the observation of azoxybenzene intermediate in model reaction (by ESI-QTOF-MS), clearly suggested the involvement of both the pathways with hydroxylamine route (direct route) being the predominant way. On the basis of these observations, the mechanism of the reaction is proposed in Fig. 1.
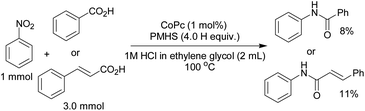
Further, the method was tested for the reaction of nitrobenzene with solid acids like benzoic acid and cinnamic acid in ethylene glycol (1 M HCl). But, the corresponding products were obtained in very low yields (Scheme 2).
The recyclability of the catalytic system (CoPc + AcOH) was checked in the model reaction by re-adding nitrobenzene (1.0 mmol) and PMHS (4.0H equiv.) to the reaction mixture after first consumption of the starting material. The catalytic system was found to be recyclable up to three cycles without any significant effect on yield (Table 4).
Conclusions
In conclusion, an efficient method employing CoPc as catalyst has been developed for highly chemoselective reductive amidation of nitroarenes with carboxylic acids. The merits of the current catalytic method include (i) low catalyst loading (1 mol%) (ii) high isolated yields (iii) inexpensive, stable and environmentally benign reducing agent (PMHS) (iv) no requirement of pre-activation of carboxylic acids. Further studies on understanding the mechanism of the reaction and applications of MPcs for other reductive amidation processes are being carried out.Experimental section
General experimental procedure for reductive amidation of nitroarenes with carboxylic acids: to a stirred suspension of CoPc (0.01 mmol) in carboxylic acid (2 mL) were added nitroarene (1.0 mmol) and PMHS (4.0H equiv.) at room temperature and then the temperature was raised to 100 °C. On completion of the reaction (as monitored by TLC), reaction mixture was dried under vacuum and the desired product was purified by column chromatography over silica-gel (60–120 mesh) using n-hexane/ethyl acetate.Acknowledgements
Authors are grateful to Dr P. S. Ahuja, Director of the Institute for encouragement and support. Mr VK, Mr MK and Ms. SS are thankful to UGC for granting research fellowships. The financial support from CSIR (CSC-0108) is also acknowledged.Notes and references
- (a) T. Cupido, J. Tulla-Puche, J. Spengler and F. Albericio, Curr. Opin. Drug Discovery Dev., 2007, 10, 768 CAS; (b) J. W. Bode, Curr. Opin. Drug Discovery Dev., 2006, 9, 765 CAS; (c) J. M. Humphrey and A. R. Chamberlin, Chem. Rev., 1997, 97, 2243 CrossRef CAS PubMed.
- (a) F. Albericio, Curr. Opin. Chem. Biol., 2004, 8, 211 CrossRef CAS PubMed; (b) G. S. Singh, Tetrahedron, 2003, 59, 7631 CrossRef CAS.
- S. Y. Han and Y. A. Kim, Tetrahedron, 2004, 60, 2447 CrossRef CAS PubMed.
- C. A. G. N. Montalbetti and V. Falque, Tetrahedron, 2005, 61, 10827 CrossRef CAS PubMed.
- (a) M. Takasaki, Y. Motoyama, K. Higashi, S. H. Yoon, I. Mochida and H. Nagashima, Org. Lett., 2008, 10, 1601 CrossRef CAS PubMed; (b) M. L. Kakshmi, R. Chakravati, U. Pal, B. Screedhar and S. Bhargava, Adv. Synth. Catal., 2008, 350, 822 CrossRef; (c) M. L. Kantam, T. Byopadhyay, A. Rahman, N. M. Reddy and B. M. Choudary, J. Mol. Catal. A: Chem., 1998, 133, 293 CrossRef CAS.
- (a) A. Rahman and S. B. Jonnalagadda, Catal. Lett., 2008, 123, 264 CrossRef CAS; (b) I. Pogoreli, M. Filipan-Litvi, S. Merka, G. Ljubi, I. Cepane and M. Litvi, J. Mol. Catal. A: Chem., 2007, 274, 202 CrossRef PubMed; (c) B. Zeynizadeh and D. Setamdideh, Synth. Commun., 2006, 36, 2699 CrossRef CAS; (d) K. P. Chary, S. R. Ram and D. S. Iyengar, Synlett, 2000, 683 CAS; (e) P. D. Ren, S. F. Pan, T. W. Dong and S. H. Wu, Synth. Commun., 1995, 25, 3799 CrossRef CAS.
- (a) Q. Shi, R. Lu, L. Lu, X. Fu and D. Zhao, Adv. Synth. Catal., 2007, 349, 1877 CrossRef CAS; (b) Q. Shi, R. Lu, K. Jin, Z. Zhang and D. Zhao, Green Chem., 2006, 8, 868 RSC; (c) M. Kumarraja and K. Pitchumani, Appl. Catal., A, 2004, 265, 135 CrossRef CAS PubMed; (d) A. Vass, J. Dudas, J. Toth and R. S. Varma, Tetrahedron Lett., 2001, 42, 5347 CrossRef CAS; (e) R. V. Jagadeesh, G. Wienhofer, F. A. Westerhaus, A. E. Surkus, M. M. Pohl, H. Junge, K. Junge and M. Beller, Chem. Commun., 2011, 47, 10972 RSC.
- (a) H. U. Blaser, H. Steiner and M. Studer, ChemCatChem, 2009, 1, 210 CrossRef CAS; (b) I. Sorribes, G. Wienhofer, C. Vicent, K. Junge, R. Llusar and M. Beller, Angew. Chem., Int. Ed., 2012, 51, 7794 CrossRef CAS PubMed; (c) G. Wienhöfer, I. Sorribes, A. Boddien, F. Westerhaus, K. Junge, H. Junge, R. Llusar and M. Beller, J. Am. Chem. Soc., 2011, 133, 12875 CrossRef PubMed; (d) N. García, P. García-García, M. A. Fernández-Rodríguez, R. Rubio, M. R. Pedrosa, F. J. Arnáiz and R. Sanz, Adv. Synth. Catal., 2012, 354, 321 CrossRef.
- (a) U. Sharma, P. Kumar, N. Kumar, V. Kumar and B. Singh, Adv. Synth. Catal., 2010, 352, 1834 CrossRef CAS; (b) U. Sharma, P. K. Verma, N. Kumar, V. Kumar, M. Bala and B. Singh, Chem. – Eur. J., 2011, 17, 5903 CrossRef CAS PubMed; (c) U. Sharma, N. Kumar, P. K. Verma, V. Kumar and B. Singh, Green Chem., 2012, 14, 2289 RSC.
- H. Mimoun and S. A. Firminich, WO 00/37540 A1 , 2000.
- (a) K. Junge, B. Webdt, N. Shaikh and M. Beller, Chem. Commun., 2010, 46, 1769 RSC; (b) R. J. Rahaim, Jr and R. E. Maleczka, Jr, Org. Lett., 2005, 7, 5087 CrossRef PubMed; (c) R. J. Rahaim, Jr and R. E. Maleczka, Jr, Synthesis, 2006, 3316 Search PubMed; (d) R. G. de Noronha, C. C. Romao and A. C. Fernandes, J. Org. Chem., 2009, 74, 6960 CrossRef CAS PubMed; (e) L. Pehlivan, E. Métay, S. Laval, W. Dayoub, P. Demonchaux, G. Mignani and M. Lemaire, Tetrahedron Lett., 2010, 51, 1939 CrossRef CAS PubMed.
- (a) A. Bhattacharya, V. C. Purohit, V. Suarez, R. Tichkule, G. Parmer and F. Rinaldi, Tetrahedron Lett., 2006, 47, 1861 CrossRef CAS PubMed; (b) A. E. Wahba, J. Peng and M. T. Hamann, Tetrahedron Lett., 2009, 50, 3901 CrossRef CAS PubMed; (c) M. Li, L. Hu, X. Cao, H. Hong, J. Lu and H. Gu, Chem. – Eur. J., 2011, 17, 2763 CrossRef CAS PubMed; (d) T. L. Ho, J. Org. Chem., 1977, 42, 3755 CrossRef CAS.
- (a) V. Kumar, U. Sharma, P. K. Verma, N. Kumar and B. Singh, Adv. Synth. Catal., 2012, 354, 870 CrossRef CAS; (b) V. Kumar, U. Sharma, B. Singh and N. Kumar, Aust. J. Chem., 2012, 65, 1594 CrossRef CAS.
- V. Kumar, S. Sharma, U. Sharma, B. Singh and N. Kumar, Green Chem., 2012, 14, 3410 RSC.
- M. A. McLaughlin and D. M. Barnes, Tetrahedron Lett., 2006, 47, 9095 CrossRef CAS PubMed.
Footnotes |
| † IHBT communication no. 3461. |
| ‡ Electronic supplementary information (ESI) available: For detailed experimental procedures and 1H and 13C NMR spectra. See DOI: 10.1039/c3ra46619a |
| This journal is © The Royal Society of Chemistry 2014 |