Substituent effect on fluorophores instead of ionophores: its implication in highly selective fluorescent probes for Zn2+ over Cd2+†
Abstract
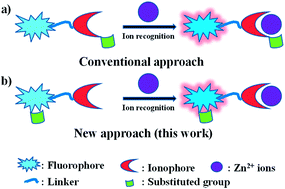
Three fluorescent molecules with different substituent modification on 5-position showing different extents of fluorescent response to Zn2+ and distinct discriminating effect of RZn/RCd have been readily synthesized. The addition of a dithiol group induces the discriminating effect of RZn/RCd to be obviously improved from twice to 32 times.

 Please wait while we load your content...
Please wait while we load your content...