A nanocomposite of carbon quantum dots and TiO2 nanotube arrays: enhancing photoelectrochemical and photocatalytic properties†
Mingxuan
Sun
a,
Xiaoqing
Ma
a,
Xi
Chen
a,
Yujun
Sun
a,
Xiaoli
Cui
*a and
Yuehe
Lin
b
aDepartment of Materials Science, Fudan University, Shanghai 200433, China. E-mail: xiaolicui@fudan.edu.cn; Fax: +86-21-65642397; Tel: +86-21-65642397
bSchool of Mechanical and Materials Engineering, Washington State University, Pullman, WA 99164-2920, USA
First published on 6th November 2013
Abstract
A novel nanocomposite of carbon quantum dots (CQDs) and TiO2 nanotubes was fabricated and its enhanced photocatalytic and photoelectrochemical properties were demonstrated. Carbon quantum dots were obtained by electrochemical-etching graphite electrodes and TiO2 nanotubes arrays were prepared by anodization methods. Subsequently, CQDs were assembled on the surface of vertically aligned TiO2 nanotube arrays (CQDs/TiO2 nanotubes) and the as-prepared samples were characterized by field-emission scanning electron microscopy, X-ray diffraction, UV-vis diffuse reflectance spectroscopy, photoelectrochemical and photocatalytic measurements. XPS measurement shows the presence of carbon species which come from CQDs. A red shift of light absorption edge and more absorption in the visible light region were observed for the resulting samples from the UV-vis diffuse reflectance spectra. An enhanced photocurrent and photopotential were demonstrated for the CQDs sensitized TiO2 nanotubes under visible light irradiation and the photocurrent density was 2.7 times larger than that of pristine TiO2 nanotubes. A solar cell was fabricated for further verifying the sensitization of CQDs over TiO2 nanotubes. Moreover, the photocatalytic activity of CQDs/TiO2 nanotubes towards the degradation of methylene blue was demonstrated and about 14% enhanced degradation efficiency was obtained with the presence of CQDs. This work developed a simple method to fabricate CQDs and demonstrated the introduction of CQDs to be a new approach for improving the utilization of visible light for TiO2 nanotube arrays.
1. Introduction
Titanium dioxide (TiO2) is one of the most important semiconductors due to its low cost, high stability and nontoxicity. Recently, highly ordered TiO2 nanotube arrays have been extensively studied in photocatalysis,1,2 dye- or quantum dot-sensitized solar cells3–6 and so on.7–9 The vertically oriented TiO2 nanotube arrays obtained by Ti anodization exhibited good performance when used as a photoelectrode, due to the unique nanostructure that facilitates the separation of photoexcited charges and has higher charge collection efficiency.10 However, the wide band gap (3.2 eV for anatase and 3.0 eV for rutile) restricts the response of TiO2 to ultraviolet light, which is less than 5% of solar light. Thus, tuning the visible response of TiO2 has generated considerable scientific interest.11,12As a promising solution to enhance the utilization of solar energy, sensitizing TiO2 with dye,13 polymers,14,15 or semiconductor quantum dots (QDs) such as CdS,16,17 CdSe,18,19 PbS20,21 and CdTe22,23 has been widely investigated to extend the absorption range of TiO2. Particularly, many studies demonstrate that depositing QDs onto TiO2 nanotubes can effectively improve the photoelectrochemical and photocatalytic activity of the TiO2 nanotubes electrode in the visible region.14–21 The unique properties of QDs over dye molecules are their strong photoresponse in the visible region, tunable optical properties and band gaps controlled by sizes. However, the environmental hazards and strict synthesis conditions often limit their practical applications.
Recently, carbon quantum dots have attracted much attention due to the properties of facile synthesis, nontoxicity and stable photoluminescence. A variety of methods have been developed to prepare carbon quantum dots, including ammonium citrate salts,24,25 laser ablation of a carbon target,26 electrochemical oxidation of graphite,27–29 chemical acid oxidation of soots,30 supported synthesis31 and microwave heating.32 Among the available methods, the electrochemical method has advantages for its low cost, easy manipulation, and high output. Kang et al.28 synthesized carbon quantum dots using graphite rods as both anode and cathode with a direct current intensity of 10–200 mA cm−2. Some researches focused on CQDs-sensitized nanocomposites were also carried out. Li et al.28 prepared the efficient nanocomposite photocatalyst for the degradation of methylene blue. Zhang et al.33 fabricated Fe2O3/CQDs nanocomposites and illustrated the roles of CQDs for high photocatalytic activity. Mirtchev et al.34 used CQDs as sensitizers for nanocrystalline TiO2 solar cell and measured its power conversion efficiency.
Combining TiO2 with carbonaceous nanomaterials is being increasingly investigated as a promising means in tuning the behavior of TiO2.35 In an earlier paper, we demonstrated the sensitizing effect of graphene oxide on the photoelectrochemical and photocatalytic properties of the TiO2 nanotube arrays under visible light.36 In this work, we synthesized carbon quantum dots and TiO2 nanotubes nanocomposite by immersing TiO2 nanotubes in CQDs solution via an impregnation method. Enhanced visible light absorption and photoelectrochemical response were demonstrated. This is the first time to demonstrate the improved photoelectrochemical and photocatalytic behavior of CQDs/TiO2 nanotube composites.
2. Experimental section
2.1. Preparation of TiO2 nanotubes
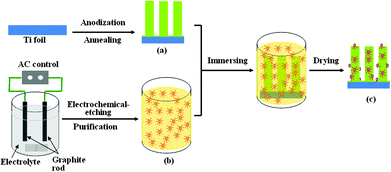
TiO2 nanotube arrays were fabricated on a Ti substrate by electrochemical anodization as described elsewhere.4,37 Briefly, the titanium sheets (99.6% purity) were first chemically etched by dipping in a mixture solution of HF (40%), HNO3 (65%) and deionized water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 in volume) for 20 s, and rinsed by deionized water. Then, the anodic oxidation was performed with the polished Ti sheets as the working electrode and a graphite sheet as the counter electrode in a 0.5 wt% HF aqueous solution for 40 min at 20 V using a DC power supply. Finally, the as-prepared samples were rinsed in deionized water and dried with an air stream.
5 in volume) for 20 s, and rinsed by deionized water. Then, the anodic oxidation was performed with the polished Ti sheets as the working electrode and a graphite sheet as the counter electrode in a 0.5 wt% HF aqueous solution for 40 min at 20 V using a DC power supply. Finally, the as-prepared samples were rinsed in deionized water and dried with an air stream.
2.2. Preparation of CQDs
Carbon quantum dots were synthesized by a one-step electrochemical-etching process using a modified Kang's method.28 Typically, graphite rods (diameter about 0.6 cm) were employed as both anode and cathode. The electrolyte was composed of 150 mL ethanol–H2O (vol% = 99.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5) with 0.5 g NaOH dissolved in it. The electrochemical oxidation was carried out at an alternate voltage in the range of 100–150 V for 10 h. After the completion of the reaction, a suitable amount of anhydrous MgSO4 was added to the crude CQDs solution, followed by a magnetic stirring for 1 h, and then stand for 24 h to remove the salts and water. Afterwards, the as-obtained solid salts were removed by centrifugation. Ethanol was evaporated under reduced pressure and CQDs powders were obtained. As far as we are aware, this is the first report on the fabrication of CQDs by electrochemical-etching process at an alternate voltage.
0.5) with 0.5 g NaOH dissolved in it. The electrochemical oxidation was carried out at an alternate voltage in the range of 100–150 V for 10 h. After the completion of the reaction, a suitable amount of anhydrous MgSO4 was added to the crude CQDs solution, followed by a magnetic stirring for 1 h, and then stand for 24 h to remove the salts and water. Afterwards, the as-obtained solid salts were removed by centrifugation. Ethanol was evaporated under reduced pressure and CQDs powders were obtained. As far as we are aware, this is the first report on the fabrication of CQDs by electrochemical-etching process at an alternate voltage.
2.3. Fabrication of CQDs/TiO2 nanotube composites
The nanocomposites of CQDs/TiO2 were synthesized by assembling CQDs on the surface of self-organized TiO2 nanotube arrays via an impregnation-assembling method. The obtained TiO2 nanotubes were annealed at 450 °C for 2 h under ambient condition and cooled down to 60 °C, then immersed in ethanol solution of 5 mg mL−1 CQDs for 3, 6, 12 h at room temperature, respectively, and dried with a nitrogen stream. The composite was denoted as CQDs/TiO2. The synthesis procedures of CQDs, TiO2 nanotubes and CQDs/TiO2 are illustrated in Scheme 1.2.4. Characterizations
X-ray diffraction (XRD) of the CQDs/TiO2 was carried out on a Bruker D8 advanced diffractometer. TEM images were acquired by using a JEM-2010FEF transmission electron microscope operating at an acceleration voltage of 200 kV. Fourier transform infrared spectra (FT-IR, Nicolet Nexus 470, Nicolet) were recorded to analyze functional groups of CQDs. The morphology and microstructure of the CQDs/TiO2 nanotubes were characterized by field-emission scanning electron microscopy (FE-SEM, S-4800, Hitachi). A RBD upgraded PHI-5000C ESCA system (Perkin Elmer) with Al/Mg–K radiation was used to measure X-ray photoelectron spectroscopy (XPS) at pass energy of 93.9 eV, the binding energies were calibrated based on the containment carbon (C 1s 284.6 eV). The UV-vis diffuse reflectance spectra (DRS) were recorded by UV-vis spectrophotometer (Cary-50 Scan).2.5. Photoelectrochemical and photocatalytic measurements
The photoelectrochemical measurements were performed on a CHI 660 electrochemical workstation (Shanghai Chenhua Instruments, China) in a three-electrode system, with CQDs/TiO2, Pt foil and Ag/AgCl as the working, counter and reference electrode, respectively. The working electrode was illuminated with a 500 W xenon lamp (CHF-XM35, Trusttech Co., Ltd. Beijing), from which infrared wavelengths were removed by a quartz water filter and wavelengths below 420 nm were cut off by an optical filter. The visible light intensity was 80 mW cm−2 (Oriel 70260).The photovoltaic performance was investigated via carbon quantum dots sensitized solar cells. The CQDs sensitized photoanode and ITO with a little Pt counter electrode were assembled to solar cell by sandwiching redox electrolyte solution. The redox electrolyte composed of 0.05 M I2, 0.1 M LiI, 0.1 M guanidinium thiocyanate, 0.1 M N-methylbenzimidazole and 0.5 M 4-tert-butylpyridine in 3-methoxypropionitrile was injected via the small hole on the counter electrode using syringe. The performance of the CQDs-sensitized solar cells was tested by recording the current density voltage (J–V) curves with a Keithley 2420 source meter under illumination of simulated AM 1.5 solar light from a solar simulator (Oriel sol3A class AAA solar simulator, model 94023A and an AM 1.5 filter).
The photocatalytic degradation of methylene blue (MB, C16H18N3S) was evaluated under visible light irradiation at room temperature. Typically, CQDs/TiO2 (1.5 × 2.0 cm) immersed in 15 mL methyl blue (5 mg L−1) was first kept in the dark for 2 h to achieve the adsorption/desorption equilibrium. A 500 W xenon lamp (CHF-XM35, Trusttech Co., Ltd. Beijing) with an optical filter to cut off wavelength below 420 nm was used to provide visible light with intensity of 180 mW cm−2. Once visible light illumination began, the concentration change of MB was analyzed at 20 min intervals for 100 min. The UV-vis adsorption spectrum of MB solution was collected with an UV-visible spectrophotometer in the wavelength range of 400–800 nm.
The electrochemical impedance spectra (EIS) were recorded over 100 kHz–100 mHz with a potentionstat/galvanostat (EG&G, Princeton Applied Research, Model 273A) and a two-phase lock-in analyzer (Model 5210). The resultant electrode served as the working electrode, with a platinum sheet as the counter electrode and a Ag/AgCl electrode as the reference electrode.
3. Results and discussion
3.1. Sample characterizations
Figure 1(A) displays a transmission electron microscopy image of carbon quantum dots synthesized at an alternate voltage of 100 V, which reveals that the as-synthesized CQDs with a spherical shape are uniform and monodisperse. This is consistent with the ref. 28. It can be seen that the obtained samples were composed of a mixture of different sized carbon quantum dots (<4 nm) by the electrochemical-etching of graphite rods.The pristine graphite powders obtained by gridding graphite rod were used for the comparison in the structure measurement. The XPS spectra of C 1s for CQDs and graphite powders are shown in Fig. 1(B). The appearance of the new peak (located at about 288.8 eV) for carbon quantum dots correspond to the oxidation groups (C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), indicating successful electrochemical etching of graphite and the formation of hydrophilic groups. The O/C atomic percentage was 0.53 for carbon quantum dots, whereas the value was 0.05 for graphite (Fig. S1†). The remarkably increase of O/C atomic percentage from graphite to CQDs further demonstrated the presence of abundant oxygenic functional groups in carbon quantum dots. Thus, we can deduce that the sp2-hybridized C
O), indicating successful electrochemical etching of graphite and the formation of hydrophilic groups. The O/C atomic percentage was 0.53 for carbon quantum dots, whereas the value was 0.05 for graphite (Fig. S1†). The remarkably increase of O/C atomic percentage from graphite to CQDs further demonstrated the presence of abundant oxygenic functional groups in carbon quantum dots. Thus, we can deduce that the sp2-hybridized C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C in aromatic ring in graphite were partially decomposed into oxygen functional groups in carbon quantum dots.
C in aromatic ring in graphite were partially decomposed into oxygen functional groups in carbon quantum dots.
Fourier transform infrared spectra were also recorded to analyze functional groups of CQDs (Fig. S2†). Peaks located at 3432 cm−1 (hydroxyl OH), 2910 cm−1 (C–H), 1776 cm−1 (carboxy C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1614 cm−1 (aromatic C
O), 1614 cm−1 (aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1443 cm−1 (aromatic C
C), 1443 cm−1 (aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1081 cm−1 (epoxide/ether C–O–C), 871, 695 and 610 cm−1 (aromatic Ar–H) were observed. It can be further confirmed the formation of oxygen functional groups during the electrochemical-etching process. All of above confirmed the successful synthesis of carbon quantum dots.
C), 1081 cm−1 (epoxide/ether C–O–C), 871, 695 and 610 cm−1 (aromatic Ar–H) were observed. It can be further confirmed the formation of oxygen functional groups during the electrochemical-etching process. All of above confirmed the successful synthesis of carbon quantum dots.
XRD was employed to characterize the crystal phase of the CQDs/TiO2 nanotube arrays, as illustrated in Fig. 2(A). All the diffraction peaks are ascribed to TiO2 and Ti substrate. The peak at 25.1° corresponding to the (101) plane diffraction of anatase TiO2 indicated that the samples were in anatase form after annealing at 450 °C for 2 h. The average crystallite size was estimated to be around 35.0 nm according to Scherrer formula. Raman spectra further confirmed the formation of anatase TiO2 nanotube arrays (Fig. S3†). The bands at 144 cm−1, 398 cm−1, 515 cm−1 and 636 cm−1 can be assigned to the Eg, B1g, A1g + B1g and Eg modes of anatase TiO2, respectively.38 However, no obvious carbonous peak for CQDs was detected in XRD patterns and no D or G bands characteristic of graphitic carbon can be observed in Raman spectra, which is possibly because of the small assembled amount of CQDs on TiO2 nanotubes.
 | ||
| Fig. 2 XRD patterns of the as-prepared CQDs/TiO2 nanotube arrays (A), top and cross-section (the insert) view FE-SEM images of CQDs/TiO2 nanocomposites (B). | ||
The morphology of resulted CQDs/TiO2 nanotube arrays was characterized by field emission scanning electron microcopy images. As shown in Fig. 2(B) and S4,† highly ordered and vertically aligned nanotube arrays were formed on Ti substrates with a single tube inner diameter of approximately 90 nm and wall thickness of around 19 nm. The nanotube arrays provide unidirectional channels for the charge transport. From the cross section, it can be observed that the nanotube arrays are short in length around 500 nm. No obvious CQDs particles can be observed from the FE-SEM images, possibly because of the small size and amount of assembled CQDs particles.
Figure 3(A) illustrates the XPS spectra of CQDs/TiO2 which exhibited the presence of Ti, O and C element. The peaks with binding energies of 458.6 eV and 464.3 eV are assigned to Ti 2p3/2 and Ti 2p1/2 for Ti(IV) of the surface titania, respectively, which confirmed the presence of TiO2 (Fig. 3(B)). In order to study the binding stages of C 1s, the XPS of C 1s core level was also provided in Fig. 3(C). A single strong peak at 284.6 eV is attributed to the elemental carbon, and the weak shoulder at 288.1 eV corresponds to the carbonate species, which is similar to the XPS spectras of C 1s CQDs above. The content of oxygen and titanium can be detected to be 37.8% and 11.7% for CQDs/TiO2, respectively, in which the O/Ti atomic ratio is 3.2, far higher than the stoichiometry of TiO2 (2.0). For comparison, XPS for pure TiO2 without CQDs was carried out and the content of oxygen and titanium was 46.02% and 19.04%. The atomic percentage of O/Ti was calculated to be 2.4, slightly higher than 2.0. Compared to TiO2, the higher content of oxygen in CQDs/TiO2 nanocomposites may be ascribed to CQDs. The XPS results confirmed the successful fabrication of CQDs/TiO2 nanocomposites.
3.2. Photoelectrochemical performance
Fig. 4(A) presents the transient photocurrent densities of TiO2 and CQDs/TiO2 samples under intermittent visible light illumination with a bias of 0 V (vs. Ag/AgCl). The electrodes show relatively good reproducibility and stability as the illumination was turned on and off. All the samples showed anodic photocurrent as soon as light on, which is a typical character for n-type TiO2 semiconductor. The influence of impregnation-assembling time to the photocurrent response was shown from curves b to d in Fig. 4(A). The photocurrent density was 0.37 μA cm−2 for the sample immersed for 3 h. Further prolonging the immersing time from 6 to 12 h, the photocurrent densities increased from 0.43 to 0.51 μA cm−2. Enhanced photocurrent was observed with the increasing of impregnation-assembling time, which was possibly related to the increasing amount of CQDs on the surface of TiO2 nanotube arrays. The highest photocurrent density of 0.51 μA cm−2 was detected for the CQDs/TiO2 electrode, while it was 0.19 μA cm−2 for the pristine TiO2 electrode. The photocurrent density increased about 2.7 times with the introduction of CQDs on the surface of TiO2 nanotube arrays. A higher transient photocurrent density was also obtained for CQDs/TiO2 electrode compared to TiO2 electrode under UV-vis light irradiation (Fig. S5†). The results above are in conformity with the results of UV-vis diffuse reflectance absorption spectra, which further demonstrated CQDs can be used as a promising sensitizer to enhance the visible light response of TiO2. The remarkably enhanced photocurrent may be ascribed to the enhanced broad light absorption and the retarding electron–hole recombination due to the introduction of CQDs to the surface of TiO2 nanotubes.Figure 4(B) shows the transient open-circuit potential of the TiO2 and CQDs/TiO2 nanotube electrodes under the intermittent illumination of visible light. It's noticed that the CQDs/TiO2 electrode exhibited a photopotential of 0.075 V, which was larger than that of pristine TiO2 electrode (0.065 V). An improved photopotential for CQDs/TiO2 electrode was also observed under UV-vis light illumination (Fig. S6†). The measured potential reflects the kinetic behavior at TiO2 nanotube arrays. The open-circuit potential under illumination is more negative than that in the dark, holes are preferentially transferred to solution, the electrode behaving as a photoanode. The results are consistent with the photocurrent measurements. Enhanced open-circuit potential implied the increasing of carriers in CQDs/TiO2 electrodes, indicating a faster separation and transport rates of electrons and holes. The photopotential of TiO2 nanotubes increased with the presence of CQDs, thus also demonstrating the sensitizing effects of CQDs on the photoelectrochemical properties of TiO2.
In order to further verify the influence of CQDs to the TiO2 nanotubes, a solar cells based on CQDs-sensitized TiO2 nanotubes electrodes were fabricated and their photovoltaic performances were investigated. The obtained J–V curve was similar to that of Mirtchev's report.34 The related parameters were illustrated in Fig. S7,† which provided the values of open-circuit photovoltage (Voc), short-circuit photocurrent densities (Jsc) and fill factor (FF) were 0.58 V, 0.020 mA cm−2 and 0.35 for CQDs sensitized solar cells, respectively. Furthermore, an energy conversion efficiency of 0.0041% was achieved, which was relative low for the solar cells. However, the result indicated that CQDs were feasible to be used as a sensitizer to tuning the behavior of TiO2 nanotubes. Better performance is expected through the optimization of electrolyte and electrodes.
3.3. Photocatalytic degradation of MB
The photocatalytic activity of TiO2 and CQDs/TiO2 was evaluated by measuring the decomposition rate of MB under visible light irradiation (λ > 420 nm). Fig. 5(A) and (B) were illustrative optical absorption spectra of MB solution at each irradiated time interval over TiO2 and CQDs/TiO2 under visible light irradiation for 100 min. Weakened intensity of the absorption indicated that MB was gradually degraded under visible light irradiation. Fig. 5(C) displays TiO2 and CQDs/TiO2 nanocomposite induced photocatalytic degradation of MB under visible light illumination. The normalized concentration changes (C/C0) of MB during the degradation process were proportional to the normalized maximum absorbance value (A/A0), therefore, the relative concentration of MB at a given time interval was derived from the absorption value at 664 nm in the absorption profile. Under visible light illumination, ∼35% and ∼21% of the initial MB were decomposed by CQDs/TiO2 and TiO2 nanotube arrays after 100 min, respectively. CQDs/TiO2 exhibited higher efficiency than TiO2 nanotube arrays by ca. 14%. The photostability of this composite material was also investigated under visible light irradiation. As shown in Fig. 5(D), CQDs/TiO2 could be reused one or two times without significant decrease in activity after being washed with ethanol and activated at 80 °C for 1 h. The photodecomposition ratio of MB was about 10.1%, 9.3% and 8.8% after 30 min over the composite material used for once, twice and three times, respectively. It was clear from Fig. 5 that CQDs/TiO2 nanocomposite showed an enhanced photocatalytic activity throughout the illumination compared with TiO2 nanotubes. Thus, CQDs were further demonstrated to be able to improve the visible light photocatalytic activity of TiO2 nanotubes.3.4. Mechanism discussion
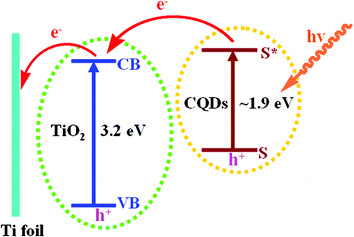
The light absorption range of the semiconductor plays an important role for its application in the utilization of solar energy. The UV-vis diffuse reflectance absorption spectra of TiO2 and CQDs/TiO2 nanotube arrays are shown in Fig. 6. A red shift of the absorption edge is observed for CQDs/TiO2 compared with the pristine TiO2. Moreover, the CQDs/TiO2 shows an enhanced absorbance in both ultraviolet and visible light region. The variation should be due to the existence of CQDs, which exhibited an optical absorption edge of 648 nm in the visible light region (the inset). The optical band gap energy (Eg) was estimated to be 1.9 eV according to the formula: Eg = 1240/λ, where λ is the optical absorption edge. The red shift of the absorption edge and the enhanced visible light absorption are potent to explain the improved the visible light photoelectrochemical and photocatalytic response of CQDs/TiO2 nanocomposite. A proposed model for the sensitization of CQDs on TiO2 nanotube arrays was illustrated in Scheme 2.39 When illuminated by visible light, CQDs excite electrons and holes. The conduction band of TiO2 nanotubes is more positive than that of CQDs, resulting in a local electric field. As a result, the excited electrons can quickly transfer from CQDs to TiO2 conduction band. The tubular structure of TiO2 is helpful for separating and transferring generated electrons to the Ti substrate foil, which contributed to the increasing photocurrent density. | ||
| Fig. 6 The UV-vis diffuse reflectance absorption spectra of TiO2 (a) and CQDs/TiO2 nanocomposites (b). The inset shows the UV-vis diffuse reflectance absorption spectra of CQDs. | ||
Electrochemical impedance spectroscopy provides a powerful method for the study of charge transfer and recombined process. Typical EIS Nyquist plots for CQDs/TiO2 electrode were presented in Fig. 7. It is observed that the semicircle diameter in the plot was reduced with the introduction of CQDs, which indicated a decrease in the interface resistance and the charge transfer resistance on the surface. EIS data were also analyzed in terms of an equivalent circuit model. Table 1 showed the results obtained by fitting the Nyquist plots with the equivalent circuit as the inset in Fig. 7. Here, Rs is electrolyte resistance; Rf and Cf are resistance and capacitance of the film; Rct is charge transfer resistance and Cdl is the double layer capacitance. As a simple comparison, the value of Rf, for CQDs/TiO2, was smaller than that of TiO2. This showed that the electrode resistivity for CQDs/TiO2 was lower. In addition, the values of Rct were 289.2 and 359.8 Ω cm−2 for CQDs/TiO2 and TiO2, respectively, which indicated the charge transfer resistance decreased in the presence of CQDs. It can be concluded that CQDs were helpful to the charge transfer from the EIS results. CQDs are attached on the surface of TiO2 nanotubes. Thus, the charge transfer across the interface between the semiconductor and solution becomes easy. Overall, CQDs may act as sensitizer, and transporter in the CQDs/TiO2 nanotubes arrays, and thereby improved photoelectrochemical properties, photopotential and higher photocatalysis performance would be achieved compared with TiO2 nanotubes.
| Sample | R s (Ω cm−2) | C f (F cm−2) | R f (Ω cm−2) | C dl (F cm−2) | R ct (Ω cm−2) |
|---|---|---|---|---|---|
| CQDs/TiO2 | 41.04 | 3.77 × 10−6 | 1.75 × 104 | 5.99 × 10−8 | 289.2 |
| TiO2 | 41.02 | 3.50 × 10−6 | 5.21 × 104 | 5.33 × 10−8 | 359.8 |
4. Conclusions
In this work, we have successfully synthesized CQDs via an electrochemical-etching technique at alternative voltage. CQDs/TiO2 nanocomposites were prepared by an assembling method with immersing TiO2 nanotubes in CQDs solutions. A red shift in the optical absorption edge and enhanced absorbance was confirmed for CQDs/TiO2 throughout the UV-vis region. Improved photocurrent and photopotential was observed after the modification of CQDs on the surface of TiO2 nanotubes. The photocurrent densities of CQDs/TiO2 electrode were 2.7 times larger than that of pristine TiO2 under visible light illumination. A solar cell was successfully assembled and further verified the sensitization of CQDs over TiO2 nanotubes. The degradation efficiency of TiO2 nanotubes towards MB aqueous solution was enhanced by about 14% with the presence of CQDs. This work provides a novel and simple strategy to prepare CQDs and demonstrated the introduction of CQDs to be a new method for improving the visible light response of TiO2 nanotube arrays.Acknowledgements
This work was supported by the Natural Science Foundation of China (no. 21273047) and the National Basic Research Program of China (no. 2012CB934300, 2011CB933300). We also appreciate the referee's very valuable comments, which have greatly improved the quality of the manuscript.References
- X. B. Chen, S. H. Shen, L. J. Guo and S. S. Mao, Chem. Rev., 2010, 110, 6503–6570 CrossRef CAS PubMed.
- L. K. Tan, M. K. Kumar, W. W. An and H. Go, ACS Appl. Mater. Interfaces, 2010, 2, 498–503 CAS.
- A. Hagfeldt, G. Boschloo, L. Sun, L. Kloo and H. Pettersson, Chem. Rev., 2010, 110, 6595–6663 CrossRef CAS PubMed.
- M. X. Sun and X. L. Cui, Electrochem. Commun., 2012, 20, 133–136 CrossRef CAS PubMed.
- M. X. Sun and X. L. Cui, J. Power Sources, 2013, 223, 74–78 CrossRef CAS PubMed.
- W. T. Sun, Y. Yu, H. Y. Pan, X. F. Gao, Q. Chen and L. M. Peng, J. Am. Chem. Soc., 2008, 130, 1124–1125 CrossRef CAS PubMed.
- M. Salari, S. H. Aboutalebi, K. Konstantinov and H. K. Liu, Phys. Chem. Chem. Phys., 2011, 13, 5038–5041 RSC.
- Y. Liu, J. Li, B. Zhou, H. Chen, Z. Wang and W. Cai, Chem. Commun., 2011, 47, 10314–10316 RSC.
- P. Roy, S. Berger and P. Schmuki, Angew. Chem., Int. Ed., 2011, 50, 2904–2939 CrossRef CAS PubMed.
- K. Zhu, N. R. Neale, A. Miedaner and A. J. Frank, Nano Lett., 2007, 7, 69–74 CrossRef CAS PubMed.
- P. V. Kamat, J. Phys. Chem. C, 2012, 116, 11849–11851 CAS.
- M. X. Sun, X. Y. Zhang, J. Li, X. L. Cui, D. L. Sun and Y. H. Lin, Electrochem. Commun., 2012, 16, 26–29 CrossRef CAS PubMed.
- W. Zhao, Y. Sun and F. N. Castellano, J. Am. Chem. Soc., 2008, 130, 12566–12567 CrossRef CAS PubMed.
- J. K. Mwaura, X. Zhao, H. Jiang, K. S. Schanze and J. R. Reynolds, Chem. Mater., 2006, 18, 6109–6111 CrossRef CAS.
- P. Gorbovyi, A. Uklein, S. Tieng, O. Brinza, M. Traore, K. Chhor, L. Museur and A. Kanaev, Nanoscale, 2011, 3, 1807–1812 RSC.
- H. Y. Wang, G. M. Wang, Y. C. Ling, M. Lepert, C. C. Wang, J. Z. Zhang and Y. Li, Nanoscale, 2012, 4, 1463–1466 RSC.
- D. R. Baker and P. V. Kamat, Adv. Funct. Mater., 2009, 19, 805–811 CrossRef CAS.
- X. Yan, X. Cui, B. Li and L. S. Li, Nano Lett., 2010, 10, 1869–1873 CrossRef CAS PubMed.
- B. Mukherjee, W. Wilson and V. Subramanian, Nanoscale, 2013, 5, 269–274 RSC.
- V. Gupta, N. Chaudhary, R. Srivastava, G. D. Sharma, R. Bhardwaj and S. Chand, J. Am. Chem. Soc., 2011, 133, 9960–9963 CrossRef CAS PubMed.
- L. Etgar, T. Moehl, S. Gabriel, S. G. Hickey, A. Eychmüller and M. Grätzel, ACS Nano, 2012, 6, 3092–3099 CrossRef CAS PubMed.
- X. F. Gao, H. B. Li, W. T. Sun, Q. Chen, F. Q. Tang and L. M. Peng, J. Phys. Chem. C, 2009, 113, 7531–7535 CAS.
- J. A. Seabold, K. Shankar, R. H. T. Wilke, M. Paulose, O. K. Varghese, C. A. Grimes and K. S. Choi, Chem. Mater., 2008, 20, 5266–5273 CrossRef CAS.
- A. B. Bourlinos, A. Stassinopoulos, D. Anglos, R. Zboril, M. Karakassides and E. P. Giannelis, Small, 2008, 4, 455–458 CrossRef CAS PubMed.
- F. Wang, Z. Xie, H. Zhang, C. Y. Liu and Y. G. Zhang, Adv. Funct. Mater., 2011, 21, 1027–1031 CrossRef CAS.
- L. Cao, X. Wang, M. J. Meziani, F. S. Lu, H. F. Wang, P. J. G. Luo, Y. Lin, B. A. Harruff, L. M. Veca, D. Murray, S. Y. Xie and Y. P. Sun, J. Am. Chem. Soc., 2007, 129, 11318–11319 CrossRef CAS PubMed.
- L. Y. Zheng, Y. W. Chi, Y. Q. Dong, J. P. Lin and B. B. Wang, J. Am. Chem. Soc., 2009, 131, 4564–4565 CrossRef CAS PubMed.
- H. T. Li, X. D. He, Z. H. Kang, H. Huang, Y. Liu, J. L. Liu, S. Y. Lian, C. H. A. Tsang, X. B. Yang and S. T. Lee, Angew. Chem., Int. Ed., 2010, 49, 4430–4434 CrossRef CAS PubMed.
- L. Bao, Z. L. Zhang, Z. Q. Tian, L. Zhang, C. Liu, Y. Lin, B. P. Qi and D. W. Pang, Adv. Mater., 2011, 23, 5801–5806 CrossRef CAS PubMed.
- S. C. Ray, A. Saha, N. R. Jana and R. Sarkar, J. Phys. Chem. C, 2009, 113, 18546–18551 CAS.
- R. L. Liu, D. Q. Wu, S. H. Liu, K. Koynov, W. Knoll and Q. Li, Angew. Chem., 2009, 121, 4668–4671 CrossRef.
- H. Zhu, X. L. Wang, Y. L. Li, Z. J. Wang, F. Yang and X. R. Yang, Chem. Commun., 2009, 5118–5120 RSC.
- H. Zhang, H. Ming, S. Lian, H. Huang, H. Li, L. Zhang, Y. Liu, Z. Kang and S.-T. Lee, Dalton Trans., 2011, 40, 10822–10825 RSC.
- P. Mirtchev, E. J. Henderson, N. Soheilnia, C. M. Yip and G. A. Ozin, J. Mater. Chem., 2012, 22, 1265–1269 RSC.
- R. Leary and A. Westwood, Carbon, 2011, 49, 741–772 CrossRef CAS PubMed.
- P. Song, X. Y. Zhang, M. X. Sun, X. L. Cui and Y. H. Lin, Nanoscale, 2012, 4, 1800–1804 RSC.
- X. L. Cui, Z. Z. Li, Y. C. Yang, W. Zhang and Q. F. Wang, Electroanalysis, 2008, 20, 970–975 CrossRef CAS.
- H. Wang, X. Quan, H. T. Yu and S. Chen, Carbon, 2008, 46, 1126–1132 CrossRef CAS PubMed.
- Y. Q. Zhang, D. K. Ma, Y. G. Zhang, W. Chen and S. M. Huang, Nano Energy, 2013, 2, 545–552 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Fig. S1–S7. See DOI: 10.1039/c3ra45474f |
| This journal is © The Royal Society of Chemistry 2014 |