Etched titania nanoplates by both HF and HAc and the photocatalytic activities for degradation of pollutants†
Mengjiao Xu,
Fei Teng*,
Juan Xu,
Tianyun Lu and
Mindong Chen
Jiangsu Key Laboratory of Atmospheric Environment Monitoring and Pollution Control, School of Environmental Sciences and Engineering, Nanjing University of Information Sciences and Engineering, Nanjing 210044, China. E-mail: tfwd@163.com; Fax: +86-25-58731090; Tel: +86-25-58731090
First published on 14th January 2014
Abstract
A HF/HAc-containing system has been developed to synthesize uniform anatase TiO2 nanoplates with controllable thickness. The interesting etch figures caused by misorientations on the {001} facets of anatase TiO2 nanoplates are found, leading to the obvious transformation from square edges to round ones. Remarkably, the typical nanoplates exhibit a 6 times higher activity than commercial Degussa P25 for the RhB degradation, due to the thin thickness and highly exposed {001} facets.
Since the first report by Fujishima,1 TiO2 has been extensively investigated because of its high photocatalytic activity, nontoxicity, low cost and stability.2 It has been reported that anatase is the active phase among TiO2 polymorphs,3,4 and {001} facets of anatase TiO2 are more reactive than {101} ones for photocatalytic degradation of organic chemicals.5 Since the properties of photocatalysts are strongly dependent on their shapes and sizes,3–9 a significant progress has been made on the controllable synthesis of TiO2.10–16 For example, TiO2 nanocubes with the {100} and {001} facets exposed have been synthesized by using ionic liquid as capping agent13 and the large TiO2 nanosheets have been prepared with the assistance of PVP.15 It is generally accepted that photogenerated carriers over thin nanosheets are easy to migrate to the surfaces from bulk phase due to the short path, which favors for surface reaction.16,17 It still remains a big challenge to synthesize anatase TiO2 nanoplates with controllable thickness in order to further improve the activity. Recently, Yang et al. and Wang et al. has reported that HF can stabilize the {001} facets of anatase TiO2 at low concentrations, but can selectively etch {001} facets at high concentrations.18,19 To the best of our knowledge, however, there are fairly fewer reports about the etch phenomenon on anatase TiO2 caused by misorientations under acidic conditions.
Herein, we report a template- and surfactant-free hydrothermal approach to synthesize the uniform anatase TiO2 nanoplates with controllable size, in which etch figures and pits are observed obviously. In our experiments, titanium butoxide (TB) is used as the precursor. Acetic acid (HAc) and HF serve as the hinder agent for the hydrolysis of TB and the capping agent to stabilize {001} facets, respectively.6 Anatase TiO2 nanoplates with different thicknesses can be prepared through altering the molar ratio of HAc/H2O and reaction temperature. It is found that etch figures on nanoplates lead to the shape evolution from square to round nanoplates. Experimental details are given in (ESI†).
Fig. S1† gives the typical XRD pattern of the sample synthesized at 180 °C for 24 h, confirming the formation of phase-pure anatase TiO2 (JCPDS no. 21-1272, space group: I41/amd (141)).20
Fig. 1A and B show that the as-prepared TiO2 sample is uniform nanoplates with round edges, which are 600 nm large and 100 nm thick with an aspect ratio of 6. Its single-crystal nature can be confirmed by the typical selected area electron diffraction (Fig. 1C). It is determined that both (200) and (020) planes have the same lattice spacing of 1.9 Å and their interfacial angle is 90°. The lattice spacing is 0.35 nm, corresponding to the {101} facets of TiO2 (Fig. 1D). This suggests that the nanoplates preferentially grow along the [101] direction. We can determine that both top and bottom surfaces of TiO2 nanoplates are {001} facets and the lateral surfaces are bounded by {101} facets.6
Fig. 2 and S2† show the influence of reaction temperature on the samples. It is found that the nanoplates almost have the same sizes at 160–200 °C. Comparing the samples obtained at 160 and 180 °C, nevertheless, their lateral sides are obviously different (Fig. 2A and B vs. Fig. 1B). The former nanoplate seems multi-layered, whereas the latter seems thick layered. We hold that due to the high surface energy of {001} facets, the single-layer nanoplates have a high tendency to stack along the c-axis through an oriented attachment mechanism, instead of extending or growth along the ab-plane.15,21 Typically, oriented attachment is easy to occur on high energy facets and involves spontaneous self-organization of the adjacent particles, in which these particles share a common crystallographic orientation.22 In Fig. 2C and D, the nanoplates obtained at 140 °C are 180 nm large and 50 nm thick, which are smaller than those (600 nm large and 100 nm thick) at 160 °C. Furthermore, it is worth mentioning that etch figures and caves can be clearly observed on the nanoplates, marked by red frames in Fig. 2C and D. When crystals grow by oriented attachment at crystallographical facets, local small misorientations will bring on the dislocation and steps.22 Generally, the dislocations and steps are easier to dissolve than others parts. As a result, the gaps and caves can form, along with the expansion of etch figures. In fact, the etch figure and cave phenomena have also occurred at high temperatures (Fig. S3†). It seems that the phenomena at 160 and 180 °C are not more obvious than those at 140 °C, which is due to the formation of the multilayers or thick layers at 160 and 180 °C. The fact that the square plates at 160 °C change into the round ones at 180 °C may be also attributed to the preferential corrosion of etch figures, which needs intensive investigation in future.
 | ||
| Fig. 2 TEM images of the TiO2 nanoplates synthesized at different temperatures for 24 h in 70H2O/100HAc/2HF/1TB system: (A and B) 160 °C and (C and D) 140 °C. | ||
To investigate the effect of HAc molecules on the hydrolysis of TB, the molar ratio of HAc to H2O is changed while others conditions are kept the same, shown in Fig. 3 and Table S1 of ESI.† When the H2O/HAc molar ratio is 50/100, TiO2 nanoplates with the square edges form, which are 2.5 μm large and 200 nm thick with an aspect ratio of 12.5 (Fig. 3A). At 60/100, TiO2 nanoplates with the coarse edges are 1 μm large and 100 nm thick with an aspect ratio of 10, whose edges are etched obviously (Fig. 3B). At 70/100, TiO2 nanoplates with the round edges are obtained, which are 600 nm large and 100 nm thick with an aspect ratio of 6 (Fig. 1A). At 100/100, TiO2 square nanoplates are 200 nm large and 50 nm thick with an aspect ratio of 4 (the inset of Fig. 3C). It is clear that the size and thickness of nanoplates decrease with increasing the molar ratio of H2O/HAc. With increasing the amount of water, the hydrolysis and condensation processes of TB are accelerated. A large amount of water favors for the rapid formation of numerous anatase TiO2 nuclei, which conduces to the formation of small crystals.13 Too much water, nevertheless, would result in severe agglomeration of the nanoparticles, as shown in Fig. S4.† Finally, the sizes of the TiO2 nanoplates are summarized in Tables S2 and S3 (ESI†)
 | ||
| Fig. 3 SEM images of the as-prepared samples at 180 °C for 24 h in a xH2O/100HAc/2HF/1TB system: (A) x = 50, (B) x = 60, and (C) x = 100 (the inset of TEM image). | ||
Furthermore, our contrast experiment shows that the titania nanoparticles, instead of nanoplates, can be only obtained without adding HAc or HF, as shown in Fig. S5.† It has been documented that at high concentrations, HF can selectively etch the {001} facets of anatase TiO2.18,19 The etching reaction may take place as follows (1):
| TiOF2 + 4HF → H2TiF6 + H2O | (1) |
In our study, it seems that the {101} facets, instead of the {001} facets, are significantly etched (Fig. 3B). Generally, crystal corners or edges dissolved fast due to their more crystal defects. Thus, it looks like the {101} facets (i.e. the edges) are etched, which also favor to the transformation of large and square nanoplates to small and round ones.
It is found that the anatase TiO2 nanoplates assembled to 3D flowers, when the molar ratios of HF/TB are changed from 2/1 to 4/1, as shown in Fig. S2A and S6 (seeing ESI†). It seems that at a high concentration, HF can promote the formation of flowers. The plausible mechanism can be described as follows. The {001} facets of anatase TiO2 are greatly stabilized by HF, precluding their fast growth. Preferential growth of the lateral {101} facets leads to the formation of large ultrathin nanoplates with the exposed {001} facets. These thin nanoplates are highly flexible and readily self-organize into hierarchical architecture by dipole–dipole interactions and oriented attachment.23,24 Han et al.16 have reported that the size of TiO2 nanosheets varied with the amount of HF in a HAc-free system. Herein, we could only conjecture that in the presence of HAc, HF at a high concentration act as an effective revulsant for the assembly of nanoplates. Fig. S7† describes the formation mechanism of the typical TiO2 samples. In our study, the nanoplates can not only stack along the c-axis to form a multi-layer nanoplate, but also can form a flower-like architecture through an oriented attachment mechanism. A further dissolving of etching figures on nanoplates may lead to the evolution from square nanoplates to round ones. It is noted that the real mechanism is still unclear, and needs further investigation.
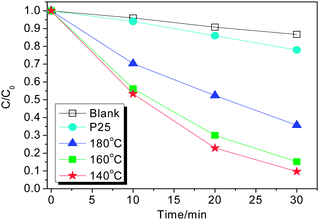
Fig. 4 presents the RhB degradation activities for the typical TiO2 samples under ultraviolet light irradiation (λ < 400 nm). It is exciting that the degradation activity of the sample synthesized at 160 °C is 6 times higher than that of Degussa P25 TiO2. Since the BET area (21.0 m2 g−1) of the TiO2 nanoplates is smaller than that (50 m2 g−1) of Degussa P25 TiO2. The as-prepared TiO2 nanoplates mainly expose {001} facets, but Degussa P25 TiO2 contains 80% anatase and 20% rutile phases and is mainly expose {101} facets.5–7 Therefore, we hold that the exposed active {001} facets mainly contribute to the improved activity of TiO2 nanoplates. In addition, the activity of the sample obtained at 160 °C is 1.8 times higher than that at 180 °C. The exposed percentages of {001} facets have been calculated to be 75% for both samples. The BET areas and thickness of nanoplates are mainly considered. On the one hand, the former has a higher BET area than that of the latter (21.0 vs.12.6 m2 g−1). The larger surface area could provide more active sites and favor for the adsorption of reaction molecules. On the other hand, the former nanoplates seem to consist of at least four thin layers attached loosely, meaning that the thickness of the single-layer nanoplate is thinner than 25 nm. But the latter nanoplates are 100 nm thick attached closely or grown into one thick layer. The thinner layer favors for transportation of photogenerated carriers from bulk phase to the surfaces due to the short path, which also favors for photocatalytic reaction.17 It should be noted that others factors (e.g. surface defects, surface reactivity, etc.) could affect the photocatalytic performance.25,26 After being etched, the oxygen vacancy probably are produced, which could affect the photocatalytic activity. At present time, nevertheless, we cannot obtain the information about oxygen deficiency due to the limited research conditions, which needs further research.
In conclusion, the thickness and the exposed facets of TiO2 nanoplates can be controlled easily in the presence of both HAc and HF, leading to the greatly improved photocatalytic activity. The etch figures caused by misorientations obviously appear in anatase TiO2 nanoplates, leading to the transformation from square edges to round ones.
Acknowledgements
This work is financially supported by National Science Foundation of China (21377060 and 21103049), Six Talent Climax Foundation of Jiangsu (20100292), Jiangsu Science Foundation of China (BK2012862), Jiangsu Province of Academic Scientific Research Industrialization Projects (JHB2012-10 and JH10-17), The Project of Foreign Culture and Education expert (N0502001003), Jiangsu province of Key Environmental protection projects (2012028), Teaching Reform Project to Enhance the Practice Innovation of NUIST-2013 (N1885013014), Support Program for Undergraduate Dissertation of NUIST-2013 (N1085002008), A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and Jiangsu Province Innovation Platform for Superiority Subject of Environmental Science and Engineering, “333” Outstanding Youth Scientist Foundation of Jiangsu (2011–2015), and the Project Sponsored by SRF for ROCS, SEM (2013S002).Notes and references
- A. Fujishima and K. Honda, Nature, 1972, 238, 37 CrossRef CAS.
- G. Liu, L. Z. Wang, H. G. Yang, H. M. Cheng and G. Q. Lu, J. Mater. Chem., 2010, 20, 831 RSC.
- R. D. Shannon and J. A. Pask, J. Am. Ceram. Soc., 1965, 48, 391 CrossRef CAS.
- U. Diebold, Surf. Sci. Rep., 2003, 48, 53 CrossRef CAS.
- H. G. Yang, C. H. Sun, S. Z. Qiao, J. Zou, G. Liu, S. C. Smith, H. M. Cheng and G. Q. Lu, Nature, 2008, 453, 638 CrossRef CAS PubMed.
- Z. Ding, G. Q. Lu and P. F. Greenfield, J. Phys. Chem. B, 2000, 104, 4815 CrossRef CAS.
- J. C. Yu, L. Zhang, Z. Zheng and J. Zhao, Chem. Mater., 2003, 15, 2280 CrossRef CAS.
- T. V. D. Meulen, A. Mattson and L. J. Osterlund, J. Catal., 2007, 251, 131 CrossRef PubMed.
- T. Nonoyama, T. Kinoshita and M. Higuchi, J. Am. Chem. Soc., 2012, 134, 8841 CrossRef CAS PubMed.
- B. Koo, J. Park, Y. K. Kim, S. H. Choi, Y. E. Sung and T. Hyeon, J. Phys. Chem. B, 2006, 110, 24318 CrossRef CAS PubMed.
- C. T. Dinh, T. D. Nguyen, F. Kleitz and T. O. Do, ACS Nano, 2009, 3, 3737 CrossRef CAS PubMed.
- K. L. Ding, Z. J. Miao, B. J. Hu, G. M. An, Z. Y. Sun, B. X. Han and Z. M. Liu, Langmuir, 2010, 26, 5129 CrossRef CAS PubMed.
- X. W. Zhao, W. Z. Jin, J. G. Cai, J. F. Ye, Z. H. Li, Y. R. Ma, J. L. Xie and L. M. Qi, Adv. Funct. Mater., 2011, 21, 3554 CrossRef CAS.
- T. R. Gordon, M. Cargnello, T. Paik, F. Mangolini, R. T. Weber, P. Fornasiero and C. B. Murray, J. Am. Chem. Soc., 2012, 134, 6751 CrossRef CAS PubMed.
- J. S. Chen, J. Liu, S. Z. Qiao, R. Xu and X. W. Lou, Chem. Commun., 2011, 47, 10443 RSC.
- X. G. Han, Q. Kuang, M. S. Jin, Z. X. Xie and L. S. Zheng, J. Am. Chem. Soc., 2009, 131, 3152 CrossRef CAS PubMed.
- Y. W. Jun, J. S. Choi and J. Cheon, Angew. Chem., Int. Ed., 2006, 45, 3414 CrossRef CAS PubMed.
- X. H. Yang, H. G. Yang and C. Z. Li, Chem.–Eur. J., 2011, 17, 6615 CrossRef CAS PubMed.
- Y. Wang, H. M. Zhang, Y. H. Han, P. R. Liu, X. D. Yao and H. J. Zhao, Chem. Commun., 2011, 47, 2829 RSC.
- O. O. Prieto-Mahaney, N. Murakami, R. Abe and B. Ohtani, Chem. Lett., 2009, 38, 238 CrossRef CAS.
- W. Zhou, M. Yao, L. Guo, Y. M. Li, J. H. Li and S. H. Yang, J. Am. Chem. Soc., 2009, 131, 2959 CrossRef CAS PubMed.
- R. L. Penn and J. F. Banfield, Science, 1998, 281, 969 CrossRef CAS.
- J. S. Chen, Y. L. Tang, C. M. Li, Y. L. Cheah, D. Luan, S. Madhavi, F. Y. C. Boey, L. A. Archer and X. W. Lou, J. Am. Chem. Soc., 2010, 132, 6124 CrossRef CAS PubMed.
- J. G. Yu and L. F. Qi, J. Hazard. Mater., 2009, 169, 221 CrossRef CAS PubMed.
- R. Long and N. J. English, J. Phys. Chem. C, 2009, 113(21), 9423 CAS.
- R. Long, N. J. English and Y. Dai, J. Phys. Chem. C, 2009, 113(40), 17464 CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c3ra44580a |
| This journal is © The Royal Society of Chemistry 2014 |