Preparation of a functionalized core–shell structured polymer by seeded emulsion polymerization and investigation on toughening poly(butylene terephthalate)
Nian
Fu
,
Guohua
Li
,
Qingxin
Zhang
*,
Nongyue
Wang
and
Xiongwei
Qu
*
Institute of Polymer Science and Engineering, School of Chemical Engineering, Hebei University of Technology, Tianjin 300130, People's Republic of China. E-mail: xwqu@hebut.edu.cn; zhqxcn@163.com
First published on 8th November 2013
Abstract
Poly(n-butyl acrylate)/poly(methyl methacrylate-co-methacrylic acid) (PBMMA), a core–shell structured modifier with controlled particle sizes, was prepared, and the toughening effects of PBMMA on poly(butylene terephthalate) (PBT) were investigated. The modifier was prepared at a solid content of 50 wt% by a two-stage sequential emulsion polymerization. Dynamic light scattering (DLS) was used to monitor the particle diameters, which showed that the particles grew without significant secondary nucleation occurring. The morphology was confirmed by means of transmission electron microscopy (TEM). According to the results on the mechanical properties of the PBT/PBMMA blends, a remarkable toughening effect of methacrylic acid (MA) on PBT resin was found. By means of scanning electron microscopy (SEM) observation, the toughening mechanism was proposed to be crazing caused by rubber particles and shear yielding of PBT matrix. The uniform dispersion of rubber particles in PBT matrix was attributed to the good compatibility between PBT and PBMMA modifier.
Introduction
Poly(butylene terephthalate) (PBT) is an important engineering thermoplastic with superior properties, such as high rigidity, solvent resistance, high rates of crystallization, fine insulation and short cycle times in injection molding. It can be used in automotive interiors, electronics and electrical instrumentation, household appliances, communications, and other fields. However, PBT is notch sensitive and will fracture in a brittle way when standard notched specimens are tested.1–7 Toughness is one important parameter to evaluate whether a polymer can be used as an engineering material. So how to improve the toughness of brittle plastics has become an attractive topic. In order to enhance the fracture toughness, the incorporation of rubbery fillers has been proved to be an efficient way.8–11Many investigations on toughening efficiency of a given brittle polymer have focused on varying particles properties such as particle size, particle stiffness, and interfacial adhesion between the particles and matrix.12,13 Among rubbery impact modifiers, core–shell rubber (CSR) particles can be used to toughen PBT. The core–shell particles typically have a core of crosslinked butadiene or acrylic rubber and a shell of grafted chains that may physically or chemically interact with the matrix to ensure desired dispersion and coupling.
With proper CSR particles and suitable processing conditions, PBT can be toughened successfully by CSR core–shell particles even without the presence of any compatibilizers, for example PBT toughened by ABS particles.14,15 However, the properties of PBT/ABS blends obtained from the simple blending method are not stable. In case the compatibilities are improved, the blends may be able to achieve better properties, such as a more stable morphology and a broader processing window. PBT contains hydroxyl (or carboxyl) functional group at the end of the molecular chain, and it can react easily with epoxy ring, anhydride or carboxyl (or hydroxyl). Functionalized polymers have been used for toughening PBT and other engineering plastics such as ethylene propylene diene rubber grafted with glycidyl methacrylate (EPDM-g-GMA),16 styrene–acrylonitrile copolymer (SAN-g-GMA)17 and maleic anhydride containing elastomers such as EPR-g-MAH,18 POE-g-MAH,4et al. However, to the best of our knowledge, few reports are available on methacrylic acid (MA) as functional monomers for toughening PBT.
In this study, a kind of three-layer core–shell structured particles with rubber phase core, rigid phase and functional monomer in shell was prepared. The rubber phase increases the impact strength; meanwhile, the rigid polymer phase offers a route to increasing the modulus. Functional monomer in shell may be chemically bonded to the matrix by reactions of its functional groups with the hydroxyl (or carboxyl) functional group of the PBT. The copolymer formed the shell phase of the impact modifier. The rubber phase is surrounded by rigid polymer, thus, the core–shell modifiers can flow unrestrictedly in PBT matrix. Poly(n-butyl acrylate)/poly(methyl methacrylate-co-methacrylic acid), i.e., PBMMA, is a core–shell structured model with slightly cross-linked poly(BA) rubber phase as a core, rigid PMMA phase as a shell and methacrylic acid (MA) in shell, which is chemically bonded to the matrix by reactions of its carboxyl units with the hydroxyl (or carboxyl) functional groups of the PBT. This article focused on the preparation of acrylic core–shell latex particles from a semi-continuous emulsion polymerization process and the toughening mechanism of these reactive core–shell modifiers on PBT. The notched impact strength of PBT was increased more than 6 times compared with that of pure PBT when the PBMMA particle with 1.8% of MA by weight was used.
Experimental
Materials
The initiators potassium persulfate (KPS) and sodium hydrogensulfite (NaHSO3) were purchased from Tianjin Chemical Agent Co., (Tianjin City, China). The anionic surfactant used in this study was Aerosol MA Series from Cytec, (Heavens City, The Netherlands). Allyl methacrylate (ALMA) was purchased from Tianjiao Chemical of China (Tianjin City, China). All above materials were used without further purification. n-Butyl acrylate (BA), methacrylic acid (MA), and methyl methacrylate (MMA) were purchased from Beijing Dongfang Chemical Co., (Beijing City, China). The BA monomer was freed of inhibitor, which was washed with a 2 wt% of NaOH solution and deionized water until the filtrated water was neutral, dried with CaCl2 overnight, and distilled under reduced pressure. MMA monomer was purified by distillation under reduced pressure before use. The inhibitor in MA was removed just before use by passing through a column of activated aluminum oxide. Hydroquinone was used as an inhibitor of the latexes taken from the emulsion polymerization. Deionized water was used in all the experimental work in this study. PBT resin (1100-211M) was provided by Changchun Industry (Taiwan, ROC). All components were dried for at least 12 h at 80 °C in a vacuum oven to ensure removal of adsorbed water.Semicontinuous emulsion polymerization
Core–shell structured latexes were synthesized as 50% of solid latexes via a two-stage semicontinuous emulsion polymerization. The surfactant (0.3 g), KPS (0.27 g) and water (140 g) were added to a 0.5 L flanged reaction flask under nitrogen, and then heated at about 63 °C. After 30 min, the seed-stage BA monomer (10.0 g, 5 wt% of total monomer) was added to the surfactant solution and then stirred for 10 min, Then NaHSO3 (0.013 g) solution in water (5 g) was added, followed by addition of another batch of NaHSO3 (0.013 g) solution in water (5 g) after 30 min. The seed stage was 60 min. The growth stage involved three layers of preemulsified monomers, which were the first layer of preemulsified monomers of BA (150 g) and ALMA (1.35 g) with surfactant (1.875 g), the second layer of preemulsified monomer of MMA (10 g) with surfactant (0.125 g) and the third layer of preemulsified monomer mixture of MMA (30 g) and MA (3.6 g) with surfactant (0.394 g), respectively. Based on the weight of the monomer, preemulsified monomers of the first two layers were dropped into the flask successively at a constant rate over 1.5 h. At the same time, NaHSO3 (0.104 g) dissolved in distilled water (40.0 g) was added to the reaction flask at a constant rate over 1.5 h. Then, the third layer of preemulsified monomers and NaHSO3 (0.026 g) in water (10.0 g) were added to the reaction flask at a constant rate for 15 min, respectively. After 60 min, the latex was cooled down to room temperature a1nd filtered through a 53 μm sieve to obtain the coagulate products. Based on the above latex preparation, contents of MA were regulated, with similar seed stage procedures as described above. The contents of the first layer and the second layer were as same as above, while the third layer of preemulsified monomer mixture of MA was of 0.2, 0.6, 1.0, 1.4, and 2.0 wt% based on the total weight of BA and MMA monomers.Conversion and particle size measurement of the latexes
At 30 min intervals, samples of the latex (2 mL) were taken and moved into preweighed vials containing 1 mL of hydroquinone solution (2 wt%), which was quenched in an ice bath to prevent further polymerization. Then they were analyzed gravimetrically to determine the instantaneous conversion (based on the monomer fed until the sampling time) and overall conversion (based on the monomer fed in the full emulsion polymerization process). Particles sizes were measured with a fixed 90° scattering angle with dynamic light scattering (DLS) on a Malvern Zetasizer NANO-ZS90 (Worcestershire, UK) online and the cell temperature was controlled at 25 °C. The particle diameters quoted are the mean values of the z-average diameters (dzs) calculated by the cumulate method.Blend preparation
A Φ34 twin-screw extruder (L/D = 28, Nanjing Institute of Extrusion Machinery, China) was employed to prepare PBT/PBMMA blends with the weight ratio of 100/20 at a screw speed of 65 rpm and barrel temperatures 227–235–245–240 °C. The pelletized materials were dried and injection molded into standard specimens in an injection-molding machine (JPH-30, Guangdong Hongli Machine, China).TEM observation
The PBMMA latex morphology was examined by TEM (JEM-2100). The latex was dispersed in deionized water sufficiently with ultrasonic waves before characterization and then prepared by casting of one drop of diluted solution onto a carbon-coated copper grid.Impact and tensile testing
The mechanical testing was performed at 23 °C and 50% of relative humidity, and the samples were climatized into this condition for 48 h before testing. The notched Charpy impact strength was measured with an impact testing machine (ZBC-4) based on National Standard Testing Methods (GB 1043-1993), China. The type of the specimen is 1 and the type of notch is C. The specimen's dimension is 80 × 10 × 4 mm3, with width of notch 2 mm, residual thickness of the gap 2.67 mm, notch at the bottom of the circular arc radius r ≤ 0.1 mm, and impact speed 2.9 m s−1 and energy 4.0 J. The tensile testing was carried out on a universal tensile tester (CMT-6104) according to ASTM D-638 and the type of the specimen is V. The specimen dimensions are as follows: length overall (LO) 63.5 mm, gage length (G) 7.62 mm, width of narrow section (W) 3.18 mm, and the thickness (T) 3.2 mm. The tensile strength was measured at a crosshead speed of 20 mm min−1. The average value of at least six tests was used.Dynamic mechanical analysis
Dynamic mechanical tests were performed at a frequency of 1 Hz by using Tritec-2000 dynamic mechanical analyzer (Triton, UK) under a dual-cantilever beam blending mode. Storage modulus (E′) and loss tangent (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) were measured between −80 and 150 °C with a heating rate of 3 °C min under nitrogen atmosphere.
δ) were measured between −80 and 150 °C with a heating rate of 3 °C min under nitrogen atmosphere.
SEM measurement
The notched Charpy impact-fractured surfaces of PBT/PBMMA blends were observed with a JSM-6490LV scanning electron microscope (SEM, JEOL). The surface was coated with gold and the accelerating voltage was 10 kV.Results and discussion
Preparation of poly (BA/PMMA-co-MA) latexes
Latexes were prepared from seeded emulsion polymerization (i.e., the addition of monomer, initiator, and surfactant to a previously prepared latex), which had the advantage of avoiding the uncertainties of the particle initiator stage and, therefore gave better batch-to-batch reproducibility. Instantaneous and overall conversions were calculated from a mass balance of the reagents in the polymerization by measuring the percentage of solid contents at each sampling time: | (1) |
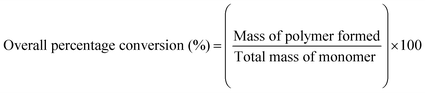 | (2) |
Plots of conversion versus reaction time for each of the latex preparations are similar. A typical plot with 0.9 wt% of crosslinking agent (ALMA) and 1.8 wt% of functional monomer (MA) is shown in Fig. 1(a). Almost all of the polymerizations were observed to proceed at high instantaneous conversion (>90%), indicating that most of the monomers added were polymerized. Final conversions were found to be high (>99%) for all the polymerization, which showed that 1 h of polymerization after the final monomer addition was adequate.
The DLS technique was used to obtain quantitative information about the particle sizes of colloidal systems. In this study, DLS provided a rapid means of monitoring the particle size during both the seed and growth stages of polymerization. So it is possible not only to establish and reproduce a latex system of the known particle diameter but also to determine whether, during the growth stage of polymerizations, the latex particles grew continuously or a secondary nucleation occurred.
Latex particle diameters were determined by DLS and compared with those theoretically calculated from the following equation:
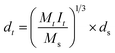 | (3) |
Plots of particle diameter versus reaction time for each of the latex preparations are also similar. Plot with 0.9 wt% of crosslinking agent (ALMA) and 1.8 wt% of functional monomer (MA) is given in Fig. 1(b). Fig. 2 shows the distribution of its final particle size, i.e., polydispersity index, referred as Malvern polydispersity index (M-PI), which was determined to be 0.035. The value of M-PI is very low, indicating a narrow distribution. The good agreement shown between the experimental and theoretical particle diameters for all of the latexes provides strong evidence that the observed particles were grown without significant secondary nucleation and that all of the polymer particles formed were spherical. In addition to that described above the low levels of coagulum (<1.3 wt%), also showed that proper contents of the crosslinking agent and functional monomer were used in the polymerization. The high final overall conversions were achieved with a solid content of 50% and final particle diameters of 330.3 nm. The morphology of PBMMA latex was observed by TEM, as shown in Fig. 3. Mean diameters were calculated to evaluate the morphology of PBMMA latex with an image analyzer. The number-average diameter (Dn) was calculated from the following equation:
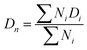 | (4) |
 | ||
| Fig. 2 Particle diameter distribution of the PBMMA latex at the end of the growth stage with 0.9 wt% ALMA at the core layer and 1.8 wt% MA at the out shell layer. | ||
 | ||
| Fig. 3 TEM images of the PBMMA particles with 0.9 wt% ALMA at the core layer and 1.8 wt% MA at the out shell layer (a) scale 100 nm (b) scale 50 nm. | ||
The mean diameter of the PBMMA latex particles calculated from TEM was 300 nm, which is slightly smaller than the mean particle size value measured by DLS. In Fig. 3, it could be seen that the particles consisted of a dark core of poly(BA) and a brighter shell of poly(MMA-co-MA), which indicates obvious core–shell structure of the PBMMA latex. Based on the TEM and DLS analyses, it could be concluded that the final PBMMA latex was spherical particles with narrow distribution.
Table 1 summarized results of the emulsion polymerization process with different functional monomer (MA) levels in the second growth stage. In almost all cases, the final monomer conversions were high, and the particle sizes were pretty close. Therefore, we could see that varying MA contents had no significant effect on the final monomer conversion and the particles' formation in the emulsion polymerization of PBMMA.
| Sample no. | MA content (wt%) | The final conversion (wt%) | Coagulum content (wt%) | The final latex particle size (nm) |
|---|---|---|---|---|
| 1 | 0.2 | 99.41 | 0.98 | 322.3 |
| 2 | 0.4 | 99.37 | 1.07 | 338.5 |
| 3 | 0.8 | 99.58 | 1.03 | 334.2 |
| 4 | 1.6 | 99.75 | 1.25 | 331.7 |
| 5 | 1.8 | 99.86 | 1.30 | 330.3 |
| 6 | 2.0 | 99.78 | 1.10 | 319.1 |
Mechanical properties of PBT/PBMMA blends
It is generally believed that the interfacial adhesion between the dispersed rubber particles and the matrix plays an important role in the toughening of polymers. To increase the interfacial adhesion and to improve the miscibility of polymer blends between the matrix and disperse phase, the reactive compatibilization is very often used.19–22Fig. 4 shows the plots of mechanical properties versus functional monomer (MA) content for PBT/PBMMA blends. According to Wu's classification,23–25 including entanglement density and characteristic ratio of the chain, PBT should be classified as a pseudo ductile polymer. Therefore the notched Charpy impact strength of the pure PBT is only 8.71 kJ m−2. The incorporation of PBMMA core–shell modifiers greatly improved the notched impact strength and a brittle–ductile transition occurred with the increase of MA content, as shown in Fig. 4(a). Meanwhile, the elongations at break of the PBT/PBMMA blends significantly increase with the increase of MA content in PBMMA, as exhibited in Fig. 4(b). The improved toughness clearly indicates that PBMMA acts as an effective toughener for PBT.The interfacial reaction between the carboxyl group of PBMMA and the hydroxyl end groups of the PBT during melt processing improved the rubber particles' dispersity and the stress transfer in the blends. The SEM analysis exhibits the fractured impact surfaces with thin filament connecting and even networking of the dispersed rubber phase, which may contribute to the overall ductility of the high impact strength of the studied blends. Moreover, the phenomenon of stress concentration caused by the addition of PBMMA particles in the PBT matrix could be changed via varying the rubber particles and PBT molecule chains, as well as the effect of interfacial energy transmission. With proper interfacial adhesion between the PBMMA particles and the PBT matrix, PBMMA particles can toughen the PBT matrix by initiating shear bands. Therefore, the ductility of blends became better with the increase of the MA content. However, if the interfacial adhesion is too strong, it would decrease the capacity of initiating the shear bands. When the PBMMA particle with 1.8% of MA by weight is used, as shown in Fig. 4(a), the notched impact strength of the blend reached 65.53 kJ m−2, which increased by 652% compared with that of pure PBT. This led to the significant improvement of the impact toughness of the blends. Meanwhile, the tensile strength slightly changed when the MA content of PBMMA increased Therefore, an improved toughness-stiffness balance may be obtained in PBT/PBMMA blends by introduction of reactive compatibility between the core–shell polymer and the matrix PBT.
SEM microphotographs of fractured surface of the blends
Fig. 5 shows SEM micrographs of the impact fractured surfaces of PBT/PBMMA blends with different MA contents. The surface of pure PBT, as shown in Fig. 5(a), was smooth with many topographical irregularities indicating the homogeneous brittle fracture of the matrix phase. With the increase in MA contents, the voiding is obvious because the functional monomer (MA) was reacted with the carboxyl or hydroxyl end groups of the PBT, as shown in Fig. 5(b)–(d). When there are sufficient reactive groups on the surface of PBT/PBMMA blends, a good compatibilization and dispersion can be achieved. It is expected that this will lead to a decrease in the interfacial tension and the stabilization against coagulation. In Fig. 5(c), the morphology of the optimized blend matrix (1.8 wt% of MA content) reveals evenly distributed fibrillation network structures originated from the bulk of the material. The discontinuous PBMMA-phase in the fibrillated network structures was added to the PBT matrix to enhance the toughness and offer resistance to the fracture process, via energy dissipation mechanism, since poly(BA) is a softer matter with much lower Tg as compared with that of PBT. Thus it may be well comprehended that the mechanism of PBT-phase deformation undergoes a ductile transition behavior with the increase in the MA content. These photomicrographs are quite consistent with the increase in toughness observed by adding PBMMA modifier to the PBT matrix, as shown in Fig. 5(c). On the basis of the above results, it could be concluded that the interfacial adhesion and morphological features are important for toughening PBT and the prepared PBMMA/PBT blend shows potential wide uses in industry. | ||
| Fig. 5 SEM micrographs of fractured surfaces of PBT/PBMMA blends after impact test, where (a) pure PBT and MA content: (b) 1.4%; (c) 1.8%; (d) 2.0%. | ||
Dynamic mechanical properties of PBT/PBMMA blends
Dynamic mechanical analysis (DMA) is a powerful method to study polymer miscibility. To examine the miscibility of PBT/PBMMA blends, a series of PBMMA modifiers with different functional monomer (MA) contents in the shell layers were prepared by semicontinuous emulsion polymerization. The mechanical relaxation process of the pure PBT and PBT/PBMMA blends were measured between −80 °C and 150 °C. The curves in Fig. 6(a) and (b), obtained by DMA measurement, show the variation of storage modulus and loss tangent for PBT and the PBT/PBMMA blends as a function of temperature with different functional monomer (MA) contents in the shell layers. The lower glass-transition temperature (Tg1) corresponds with the Tg of the poly(BA) rubbery core polymer, and the higher temperature (Tg2) belongs to the PBMMA shell polymer and the matrix. The values of the relaxation peaks observed in the blends with different MA contents are listed in Table 2. DMA spectra showed that the internal friction peak of the rubber phase moved to high temperature and the internal friction peak of PBT moved to low temperature. The difference between Tg1 and Tg2 glass transition temperature was also shown in Table 2. As we know,26 if the blend displays two Tg's, at or near the two components, then it is immiscible. On the other hand, if it shows a single transition or two transitions between those of the pure components, then the blend is miscible or partially miscible. So Fig. 6(b) shows that PBT is partially miscible with PBMMA copolymer. Meanwhile, as seen in Table 2, it can be found that the difference (Tg2 − Tg1) between the two (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) loss peaks in low- and high-temperature range becomes smaller. The difference between PBT and PMMA glass transition temperature is the least when MA content was of 1.8 wt%. In this case, the miscibility of PBT/PBMMA blends was the optimum experimental condition, and these results agree with those obtained from the previous mechanical tests.
δ) loss peaks in low- and high-temperature range becomes smaller. The difference between PBT and PMMA glass transition temperature is the least when MA content was of 1.8 wt%. In this case, the miscibility of PBT/PBMMA blends was the optimum experimental condition, and these results agree with those obtained from the previous mechanical tests.
 | ||
Fig. 6 DMA spectra of the PBT/PBMMA blends with different contents of MA in PBMMA: (a) storage modulus and (b) tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ. δ. | ||
| MA (wt%) | T g1 (°C) | T g2 (°C) | T g2 − Tg1 (°C) |
|---|---|---|---|
| 1.4 | −63.2 | 57.6 | 120.8 |
| 1.8 | −49.8 | 49.8 | 99.6 |
| 2.0 | −47.6 | 56.7 | 104.3 |
Conclusions
Poly(BA/PMMA-co-MA), PBMMA, core–shell structured particles with narrow size distribution were successfully prepared via seeded emulsion polymerization. A sharp brittle–ductile transition occurred with the stiffness retained, and the toughness of PBT/PBMMA blends was significantly improved with the addition of the PBMMA modifier. It was suggested that PBMMA was able to act as a compatibilizer in PBT resin based on the tensile properties, impact strength, and two phase morphology of PBT/PBMMA blends. Furthermore, a significant increase in glass-transition temperature of the dispersed poly(BA) phase and a decrease in the continuous PBT phase were observed according to dynamic mechanical analysis, indicating the good compatibility and adhesion between PBT and PBMMA. TEM micrographs showed the obvious core–shell structure, and SEM observation of the fractured surfaces confirmed the plastic flow morphology. The fine control of the phase morphology of multiphase blends was the key approach to impart desired mechanical properties to these materials. The method of employing core–shell particles is a very promising approach to achieving well-compatibilized PBT with high toughness.Acknowledgements
This work was partially supported by Natural Science Foundation of Hebei Province; contract grant number: E2010000107, and National Natural Science Foundation of China; contract grant number: 51073049 and 21001039. The authors would like to thank Prof. Peter A. Lovell (University of Manchester, UK) for many fruitful discussions of this study.References
- S. Sun, Z. Tan, C. Zhou, M. Zhang and H. Zhang, Polym. Compos., 2007, 28, 484–492 CrossRef CAS.
- Z. Yu, C. Yan, A. Dasari, S. Dai, Y. Mai and M. Yang, Macromol. Mater. Eng., 2004, 289, 763–770 CrossRef CAS.
- J. B. Jun, J. G. Park, D. H. Kim and K. D. Suh, Eur. Polym. J., 2001, 37, 597–602 CrossRef CAS.
- A. Arostegui, M. Gaztelumendi and J. Nazabal, Polymer, 2001, 42, 9565–9574 CrossRef CAS.
- S. Sun, Z. Tan, M. Zhang, H. Yang and H. Zhang, Polym. Int., 2006, 55, 834–842 CrossRef CAS.
- Z. Z. Yu, M. S. Yang, S. C. Dai and Y. W. Mai, J. Appl. Polym. Sci., 2004, 93, 1462–1472 CrossRef CAS.
- N. M. Larocca, E. Hage Jr and L. A. Pessan, Polymer, 2004, 45, 5265–5277 CrossRef CAS PubMed.
- S. L. Sun, X. Y. Xu, H. D. Yang and H. X. Zhang, Polymer, 2005, 46, 7632–7643 CrossRef CAS PubMed.
- G. L. Mantovani, L. B. Canto, E. Hage and L. A. Pessan, Macromol. Symp., 2001, 176, 167 CrossRef CAS.
- E. Hage, W. Hale, H. Keskkula and D. R. Paul, Polymer, 1997, 38, 3237–3250 CrossRef CAS.
- A. J. Brady, H. Keskkula and D. R. Paul, Polymer, 1994, 35, 3665–3672 CrossRef CAS.
- C. B. Bucknall and V. L. P. Soares, J. Polym. Sci., Part B: Polym. Phys., 2004, 42, 2168–2180 CrossRef CAS.
- C. B. Bucknall and D. R. Paul, Polymer, 2009, 50, 5539–5548 CrossRef CAS PubMed.
- E. Hage, W. Hale, H. Keskkula and D. R. Paul, Polymer, 1997, 38, 3237–3250 CrossRef CAS.
- S. L. Sun, X. Y. Xu, Z. Y. Tan, C. Zhou, Y. H. Ao, M. Y. Zhang and H. X. Zhang, J. Appl. Polym. Sci., 2006, 102, 5363–5371 CrossRef CAS.
- Q. Shi, P. Stagnaro, C. L. Cai, J. H. Yin, G. Costa and A. Turturro, J. Appl. Polym. Sci., 2008, 110, 3963–3972 CrossRef CAS.
- S. Lumlong, K. Kuboyama, T. Chiba and T. Ougizawa, Macromol. Symp., 2006, 233, 17–25 CrossRef CAS.
- A. Cecer, R. Greco, G. Ragosta and G. Scarinzi, Polymer, 1990, 31, 1239–1244 CrossRef.
- W. Loyens and G. Groeninckx, Macromol. Chem. Phys., 2002, 203, 1702–1714 CrossRef CAS.
- P. Martin, J. Devaux, R. Legras, M. van Gurp and M. van Duin, Polymer, 2001, 42, 2463–2478 CrossRef CAS.
- N. Dayma and B. K. Satapathy, Mater. Des., 2010, 31, 4693–4703 CrossRef CAS PubMed.
- Y. J. Sun, G. H. Hu, M. Lambla and H. K. Kotlar, Polymer, 1996, 37, 4119–4127 CrossRef CAS.
- S. Wu, Polym. Eng. Sci., 1990, 30, 753–761 CAS.
- S. Wu, Polym. Int., 1992, 29, 229–247 CrossRef CAS.
- S. Wu, Polymer, 1985, 26, 1855–1863 CrossRef CAS.
- D. Sudhin and J. L. David, Polymer Compatibilizers, Hanser Publishers, Munich, 1996 Search PubMed.
| This journal is © The Royal Society of Chemistry 2014 |


