Microfluidic laboratories for C. elegans enhance fundamental studies in biology†
Natalia A. Bakhtina
a and
Jan G. Korvink
*ab
aKorvink Laboratory, IMTEK – Department of Microsystems Engineering, University of Freiburg, Georges-Koehler-Allee 103, 79110 Freiburg, Germany. E-mail: natalia.bakhtina@imtek.uni-freiburg.de; Tel: +49 761 203 67477
bFRIAS – Freiburg Institute for Advanced Studies, University of Freiburg, Albertstrasse 19, 79104 Freiburg, Germany. E-mail: korvink@imtek.uni-freiburg.de; Tel: +49 761 203 7436
First published on 27th November 2013
Abstract
The in vivo analysis of a small multicellular organism such as the nematode Caenorhabditis elegans, enables fundamental biomedical and environmental studies of a complete organism under normal physiological conditions. Continuous advancements in photonics, electronics, as well as the material sciences, are paving the way towards miniaturized bioanalytical systems, known as labs-on-a-chip (LOC). These microfluidic technologies facilitate the manipulation and study of nematodes in a precise, real-time, portable, and cost-effective manner, potentially for high throughput operation. In this paper we review all currently available “worm-on-a-chip” miniaturized systems that address the manipulation, detection, and study of the sensory response of C. elegans, and take a close look at their advantages, application challenges, and scientific potential. The paper aims to consolidate recent results of dedicated worm microsystems that target a better understanding of C. elegans.
1. Introduction
The nematode Caenorhabditis elegans is an attractive multicellular animal suitable for investigating a wide spectrum of biological questions from such diverse fields as genetics, cell biology, and neurobiology. Having been established as a model organism by Brenner in 1965, it was the first animal to have its genome completely sequenced in 1998.1 The in vivo analysis of the model multicellular organism C. elegans, allows one to understand the correlation with much more complex biological processes (e.g. human diseases), many of which have been conserved between the worm and humans. This astounding degree of correspondence permits the “connection” of knowledge gained in one area of research with research in another area.The potential usefulness of C. elegans stems from a balance between biological simplicity and complexity, allowing one to address high-level biological questions and providing a unique opportunity to model human ailments, such as Alzheimer's and Parkinson's disease.2,3 Interestingly, C. elegans and human beings share many genes and molecular pathways. For example, 40% of it's genes have a human counterpart, in particular the insulin signalling pathway is one of many that has been preserved between the worm and humans.1,6 C. elegans possesses one of the simplest central nervous systems and by far the best characterized. Despite this fact, the animal exhibits a large variety of physiological behavioural modalities.3 These include: locomotion, foraging, feeding, defecation, egg-laying, dauer larva formation, ageing, sensory responses to touch, smell, taste, gas concentration, temperature, light, ambient osmolarity, electric field, and acoustic waves. Moreover, complex behaviours such as male mating, social behaviour, and even developmental decision making, including learning and memorizing, are typical for the nematode. At the same time, C. elegans is simple enough to be experimentally tractable. The adult hermaphrodite has 302 neurons that fall into 118 morphologically different types. According to the recent literature, males have a larger nervous system with 381 neurons (additional male-specific cells).3 It has a short life cycle (3 days at 25 °C) and lifespan (15–17 days at 25 °C), passes through four larva (L1–L4) stages, and an adult stage. Its small size (1–1.2 mm long and 80 μm wide), transparent body, and preferred food source (Escherichia coli) simplify its maintenance on agar plates or liquid cultures.3 These attractive features provide a means to accelerate studies without involving actual human subjects, and make C. elegans a superb model for human disease investigations. But how does the nervous system function to receive, process, integrate, and interpret sensory signals in order to generate appropriate behaviours? Unfortunately, the correlation between physiology (phenotype) and behaviour in C. elegans is not yet fully understood.
Traditional laboratory techniques to study C. elegans are based on manual manipulation on a Petri dish or a multiwell plate, and permanent immobilization for further investigations by means of glue or anaesthetics.3 Current commercial instrumentations for worm handling, such as the COPAS BIOSORT system (Union Biometrica, Inc.), are expensive and bulky. Apart from large volumes of biochemical agents, skilled staff are necessary for the operation and maintenance of the instruments.
At the same time, microfluidic and micro-electromechanical system (MEMS) devices have become more popular over the past years. Microfluidics devices have several undeniable advantages, enabling to overcome challenges associated with traditional studying techniques. For example, microchannel dimensions matching the microorganism's size facilitate the precise delivery of small amounts of reagent. A comparison between traditional and on-chip manipulation techniques is presented in Table 1. Based on relatively inexpensive polymer-based fabrication techniques, such as polydimethylsiloxane (PDMS) replica moulding, it has become feasible to realize disposable, economic, and biocompatible systems.4 Complex structures adapted to different applications can be easily fabricated in a short time.
| Traditional techniques | On-chip manipulation | |
|---|---|---|
| Functionality | Only one output parameter is monitored per experiment | Multiple parameters are monitored per experiment |
| Manipulation | Manual (possible damage to the worm while handling) | Automatic (reproducible experimental conditions) |
| Sensitivity and precision | Imprecise delivery of external stimuli | High sensitivity and high resolution experiments (precise delivery of small amounts of reagents) |
| Real-time monitoring | Limited | Possible, also for extended lengths of time |
| Integration | Low | High |
| Throughput | Low | High |
| Cost | Expensive (large volumes of reagents) and costly to maintain | Inexpensive, disposable |
| Personnel | Well trained operators are required | Minimally trained personnel can operate the systems, user-friendliness is possible |
Substantial advances in microfluidic techniques and particular research interest in C. elegans have driven the development of numerous microchip systems. Recently, several review articles have been published covering various aspects of miniaturized systems devoted to C. elegans nematode studies.65,76–91,97,99,100 However, to the best of our knowledge, an up-to-date compilation of all available microfluidic approaches has not yet been presented. In this review we present the current state of microfluidic-based platforms, so called “worm-on-a-chip” systems, including their advantages, application challenges, and potential for use in lab-on-a-chip (LOC) platforms. The aim of the review is to systemize approximately 100 publications into four main sections and further subsections according to their intended application, or the end use of the reported assays. Assaying (e.g. behavioural or molecular studies) of C. elegans includes both movement-based and neuronal analysis, yielding a more complete picture of intercellular communication. Since effective assaying of the animal in the microfluidic environment requires knowledge of the neural circuit, we first describe the sensorial abilities of C. elegans. Second, we demonstrate how neuronal and behavioural responses, mediated by congenital perception of environmental changes, are utilized to discover correlations between physiology and behaviour. Microfluidics acts here as a precise microtool to manipulate C. elegans for fundamental studies in biology, chemistry, and physiology.
The structure of this review is as follows: in Section 2 we summarize the worm's sense properties, and its behavioural and neuronal responses to stimuli; in Section 3 we concentrate our attention on different techniques for worm-on-a-chip manipulation, such as culturing, sorting, and immobilization; Section 4 details the most frequently utilized imaging methods, is followed by Section 5, where several analysis techniques (e.g. electrochemical impedance spectroscopy, microsurgery, etc.) are introduced. In addition, we present three tables, listing the relevant sorting, immobilization, and imaging methods that have been reported in the recent literature. These methods are classified according to their reported principles for studying C. elegans on-chip, and for each case the main qualitative and quantitative characteristics are indicated. The listed data have been directly reported in the corresponding publications or have been derived from the presented figures.
Although the review is divided into four main sections, all sections and subsections are interconnected. To simplify a search of publications, we suggest to start with the general overview, which is summarized diagrammatically (Fig. 1). The papers are organised according to five evaluated aspects: measured output data, and method for sorting, immobilization, stimulation, and detection of C. elegans. For example, we associate neuron response, plastic behaviour and electrical signals to the measured output data; direct or indirect force to active or passive sorting; and the application of heat, gas, electric fields, and acoustic waves to physical immobilization. As indicated in the diagram, different output data are sorted according to their corresponding detection method (fluorescent optics, non-fluorescent optics, lensless optics, electrochemical, or a combination thereof).
2. Detecting C. elegans' senses
Major breakthroughs in the fields of neurochemistry and neurobiology have already been made because of the conserved molecular mechanisms that govern many aspects of cellular machinery. Thus, one of the most significant categories of biological research is C. elegans assaying at the scale of its neural circuits.Substantial benefits have been derived from LOC devices in terms of stable environmental control, due to laminar flow conditions, and automatic manipulation by means of image analysis systems. Here, we would like to emphasize the role of the nematode as an integrated network of molecular functions. The application areas of C. elegans sensorial abilities in biology will be extensively discussed in Section 2 and 3. The examples of stimuli (e.g. electric field, gas, etc.) and their link to applications are listed in Table 2 and 3.
| Sorting technique | Principle | C. elegans population | Selectivity | Throughput | Technology | Application | Ref. |
|---|---|---|---|---|---|---|---|
| Electrotaxis | AC, DC, DC pulses | Worm population (L2–L4 stage larvae, adults, and mutants) | Middle | Low | Single PDMS layer | Drug screening, age-synchronization, movement-based behavioural studies | 30–33, 38, 52 and 58 |
| “On-flow” sorting | Self-regulated sample loading | Worm population (larvae and adults) | High | High | Single PDMS layer | Age-synchronization, culturing, stimulation | 21, 37, 45, 50, 55, 68, 101, 102 and 105 |
| “On-valve” sorting | On/off valve switching | Single worm | High | High | Single, two and multi PDMS layers | Stimulation, phenotyping, microsurgery | 28, 47, 51, 53, 56, 104 and 109 |
| Drop encapsulation | Worm encapsulation in a drop | Single worm(L1 larva stage) | Low | High | Single PDMS layer | Drug screening, movement-based behavioural studies | 23 and 49 |
| Immobilization technique | Principle | C. elegans population | Selectivity | Throughput | Technology | Application | Ref. |
|---|---|---|---|---|---|---|---|
| Mechanical force | Microchannel narrowing | Single worm (adults) | High | High | Single, two, and multi PDMS layers | Observation (ageing), drug screening | 11, 13, 14, 16, 17, 20, 22, 24, 28, 32, 35, 36, 37, 46, 47, 51, 56, 59, 60, 63, 64, 67, 68, 94, 95, 98, 102, 104 and 110 |
| Compression | Single worm (L4 stage larvae and adults) | High | High | Two and multi PDMS layers | Stimulation | ||
| Suction flow | Single worm (L4 stage larvae, adults, and mutants) | High | High | Single and multi PDMS layers | Phenotyping | ||
| Combinations | Single worm (adults) | High | High | Two and multi PDMS layers | Microsurgery | ||
| Gel | Thermo-sensitive polymer | Worm population (L1-L4 stage larvae and adults) | High | Low | Two PDMS layers | Culturing, observation | 44, 47 and 96 |
| Temperature | Temperature decrease or increase | Single worm | High | Low | Two PDMS layers or two glass slides | Phenotyping | 28, 53, 68, 93 and 109 |
| Gas | CO2 and N2 | Single worm (L4 stage larvae and adults) | High | Low | Two PDMS layers | Locomotive behaviour | 22 and 35 |
| Electrotaxis | DC, AC | Worm population (L2–L4 stage larvae, adults, mutants) | High | High | Single PDMS layer | Locomotive behaviour, neuronal study | 29–32, 46, 75, 93 and 108 |
| Acoustic waves | Surface acoustic wave | Worm population (adults) | High | High | Single PDMS layer | Locomotive behaviour | 62 |
2.1. Platforms for worm neurobiology studies
C. elegans probes its surrounding environment and moves according to environmental stimuli, including temperature, chemical, electric field, and light trigger signals, which are detected by 24 sensilla organs and various isolated sensor neurons.5 Sensilla neurons function as sensorial tools, with each containing ciliated endings of one or more neurons, and are situated mostly in the head of the worm. Alternatively, oxygen sensing and mechanosensitivity are carried by nonsensilla neurons. Mediated through these neurons, C. elegans generates an attractive or repulsive response.3,15,25Four behavioural paradigms have been established to investigate mechanosensory control: nose touch, gentle body touch, tap, and harsh touch.1 For example, wild-type worms respond by accelerating forward or backward depending on the stimulus placement, while mechanosensory mutants mec-4 and mec-10 fail to respond to a “gentle” touch.9 More importantly, C. elegans can feel mechanical attributes of the surface on which it navigates. It is capable of distinguishing texture (e.g. small, round objects) and depending on the force, “decides” how to move.7–9
A recent paper on nematode muscular force analysis in a microstructured environment has been published by Johari et al.8 Measurements of the PDMS pillar displacement demonstrated a concentration of the nematode's bending force around the middle part of the body while crawling (Fig. 2A). Along the same line, Doll and co-workers have focused their attention on mechanical force sensation with sub-μN force resolution.10 The device revealed an interesting observation: the mechanosensory process was a result of both sensorial ability and the computational power of the worm's nervous system. In contrast to traditional experimental techniques, the system permitted straightforward dynamic force detection of moving nematodes. However, the sensor structure, compared to other platforms,8,11 was relatively complex from a manufacturing standpoint. Going further, Wen et al. succeeded in obtaining a system-level understanding of the worm's motor circuit.12 Using calcium imaging to explore partially constrained animals, they found a previously undescribed role of proprioceptive coupling of the motor neuron circuit and propagating rhythmic activity along the body. This detailed examination provided not only a qualitative, but also a quantitative explanation for gait adaptation to external load.
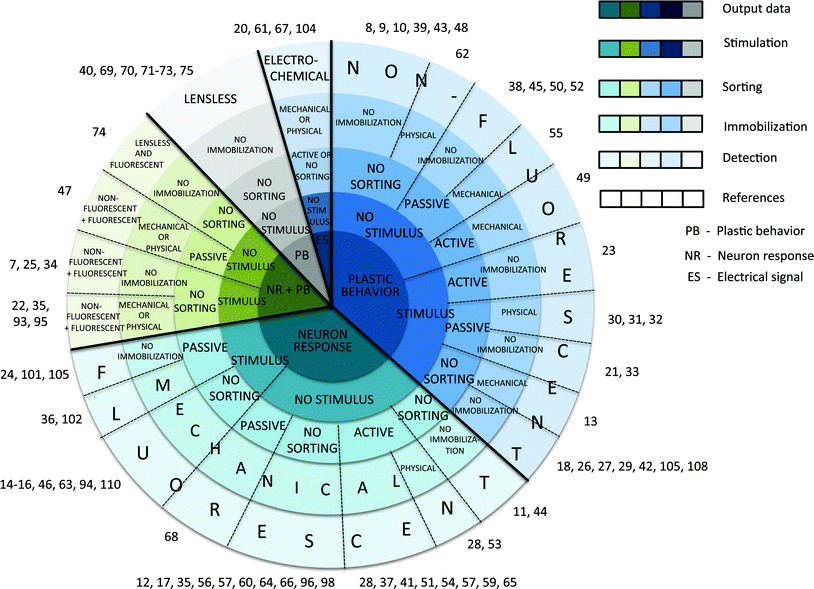
 | ||
| Fig. 2 Microfluidic investigation systems targeting worm behaviour and sensory response. (A) Schematic illustration of a behavioural arena with microposts for worm mechanosensitivity,8 chemosensitivity,20,26 aerotaxis,34 electrosensitivity, and locomotive behaviour7 studies. (B) Schematic illustration of worm stimulation. C. elegans is immobilized in a microchannel and stimuli (chemicals,14,16,46 osmolyte,13,17 etc.) are applied to the worm nose by switching the solution carried by a fluidic inlet. (C) Schematic illustration of a maze assay for behavioural exploration and learning studies.25 | ||
A comprehensive study was recently carried out by McCormick et al., who analyzed behavioural mechanisms in response to temperature change employing step-like spatial and temporal gradients.13 In a two-layer PDMS microfluidic device the temperature gradient inside a channel was regulated manually and monitored by intergrated thermoresistors. The behaviour of semi-restrained C. elegans was quantified in terms of an average head angle such that positive angles corresponded to the head visiting the cooler stream. It was concluded that, for worms having access to two temperature zones, the cooler temperature was preferable. In a different way, Luo et al. directly heated the individuals in a microdroplet array by controlled exposure to infrared laser irradiation and quantified their swimming movements.27
Thermosensitivity also regulates other developmental processes, such as exit from the dauer stage, ageing, and learning. Microfluidics has a potential to meet the challenge of precise environmental control, needed to explain how the information is transmitted by the AFD neuron and integrated in the nervous system.
Much progress has been made to overcome the limitations of precise chemical control in dynamic16,26 and step7,13,14,26,27 delivery methods. A device introduced by McCormick et al., consisting of a single PDMS-layer chip, allowed the reliable supply, via step-like gradients, of a chemosensory stimulant through fluid inlets of a Y-shaped channel.13 Two streams meeting at the point of confluence without mixing exposed only the free moving head of a worm.
It is well known that an anaesthetic is a drug which induces a reversible loss of consciousness. The effect on the worm physiology to a variety of anaesthetics (e.g. tricaine, muscimol, etc.) at different concentrations was successfully examined as a behavioural response by several authors.18,21,22 Two anaesthetic concentrations (sodium azide) were tested by Chung et al. in a highly uniform and precise manner.21 Isolated individually in microfluidic chamber arrays, animals were eventually immobilized by 5 mM and 10 mM sodium azide concentrations at two separate speeds. Another analysis of C. elegans response to anaesthesia was conducted by Carr et al., who presented a new highly sensitive approach for drug resistant screening in a three-step method.18 Firstly, the worm population was characterized prior to drug exposure. Secondly, the animals were guided into a drug well for pre-exposure with 100 μM levamisole (the most common anaesthetic) for real-time locomotion diagnostics. Thirdly, the worms were directed back into a microfluidic channel. Pre- and post-exposure locomotion phenotypes were compared by four dose-generating system parameters (i.e. average velocity, number of worms leaving the drug well, the number of active worms, and time until unresponsiveness). To emphasize the influence of different anaesthetics on subcellular activity, Mondal et al. in 2011 developed a microfluidic platform for studying a contribution of vesicle transport to synaptic growth.22 They imaged such subcellular and cellular processes as pre-synaptic vesicle transport, intraflagellar transport (IFT), dendritic transport, and migration of neuroblasts during early developmental stages of the nematode.
Other important studies include odour sensibility.15,25–27 Using a micropost array (Fig. 2A), Albrecht and Bargmann identified new behavioural responses to attractive odours, where C. elegans was subjected to a liquid borne stimulus in three patterns (temporal pulses, spatial stripes, and a linear concentration gradient).26 To investigate the effect of chemical signaling from hermaphrodites on male behaviour, Chung et al. exposed 48 male worms to hermaphrodite-conditioned media simultaneously. A set of sub-behaviours, observed even after media replacement, was attributed to male sex-specific behaviour (i.e. the tail formed a tight coil-back on a straightened body).21 In contrast to males, hermaphrodites were very rarely affected by male-specific behaviour. Another interesting observation was done by Stirman et al., who inquired into the influence of nicotine on the C. elegans body.36 At first, the worm's muscles were relaxed under blue light illumination, characterized by an increase in body length. Then a rapid contraction of the worm muscles was detected during continuing nicotine stimulation.
The groups of McCormick et al. and Chronis et al. independently evaluated the behavioural mechanism of individual head swing orientations with respect to osmotic gradient.13,17 In terms of worm feedback patterns, established as worm reversal probability, McCormick et al. suggested that both low and high osmolarity level provoked the ASH chemosensory neuron to generate osmotic avoidance.13 A more systematic study of the neuron activity revealed that ASH neurons responded not only to the presentation of a high-osmotic-strength stimulus, but also to the removal of the high-osmolarity solution.17
Neurotransmitters and hormones have widespread effects as chemical regulators for coordinating physiological activity throughout the body of both nematodes and humans.1 For instance, dopamine, as well as serotonin, are implicated in a broad range of physiological functions, including egg laying, pharyngeal pumping, defecation, and locomotion. Another example is 1-methyl-4-phenylpyridinium (MPP+), which plays a key role in Parkinson's disease-like symptoms.6 To verify the effect of MPP + on C. elegans in situ, different MPP + concentrations (0.05 mM–5.0 mM) were tested for co-encapsulation with individual worms23 and to perform single worm stimulation in a time- and dose-dependent manner.24 The experiments provided the evidence that MPP + induced mobility defects in the animal (i.e. slow, titanic, and coiling movements) after treatment and was potentially neurotoxic for dopaminergic neurons. Similar neurotoxins, such as 6-hydroxy dopamine (6-OHDA) and rotenone, caused a significant reduction in speed.108 However, neurotoxin effects could be suppressed by pre-exposing worms to acetaminophen, a well-known neuroprotective compound.
The described experiments could potentially benefit enormously from the use of microfluidic technologies. The level of precision that these research groups have already achieved demonstrates their tremendous potential for the dissection of neuronal function and toxin-induced neurodegeneration. The precise handling and chemical mixing of reagents in nanolitre-volume droplets tremendously decreases stimulus consumption and reaction time. The approach made the investigation of worm behavioural response to chemicals and neurotoxins feasible. Deep molecular functional homology of free-living C. elegans to other parasites (e.g. Brugia malayi) can provide an understanding of the underlying immune response of humans to other nematodes.1 C. elegans can be an effective test-bed for the termination of infections (e.g. malaria) and effective nematicides (a drug against nematodes).
Electrosensitivity has also been employed as a means of guiding the movement of nematodes and for immobilization, known as electrotaxis (Table 2 and 3), aiming to provide a close look at the mechanism of neuronal signalling transduced into behavioural responses. The results showed that microfluidic electrotaxis can identify alterations in dopamine and amphid neuronal signaling based on swimming responses of C. elegans. Thus, abnormalities in neuronal signaling can be identified fairly quickly and reliably.108
In 2004, Gray et al. highlighted the influence of oxygen on the social behaviour of wild-type C. elegans. Placed in a PDMS aerotaxis device with a gas-phase oxygen gradient from 0% to 21% or 21% to 0%, aggregation and bordering behaviours were examined in the animals (Fig. 2A).34 The experiments showed that freely moving adult animals excited a rapid behavioural preference for 5%–12% oxygen, escaping both hypoxia and hyperoxia. Specific soluble guanylate cyclase homologues (GCY-31, GCY-33, GCY-35, and GCY-36), located in URX, AQR, and PQR sensory neurons, activated hypoxia or hyperoxia avoidance and modulated resulting social feeding behaviour.34,63 Gases, including carbon dioxide (CO2) and nitrogen (N2), are also sensed by C. elegans (Table 3).22,35 A gaseous anaesthetic CO2 significantly reduced the anterograde flux of vesicle transport in the animals with modest effect on retrograde velocity when compared to partial loss of mobility through N2.22
One of the theories of aging, considered in the next subsection, posits that this biological process is caused by oxidative damage.1 The availability of mutants that are specifically defective in oxygen sensing and microfluidic devices substantially assist the examination with an improved sensitivity, and increased resolution.102
2.2. Platforms for behaviour studies
In order to establish predictive relationships between the physiology and metabolism of C. elegans in vivo, apart from neuronal response, we need to fully understand its behavioural dynamics. Through the function of sensilla and nonsensilla neurons, mentioned in the previous subsection, it navigates in thermal, chemical, and oxygen gradients. Microfluidic platforms allow precise control of the surrounding environment facilitating, for instance, studies of nematode mechanics or neuromuscular functioning and the comprehensive characterization of C. elegans locomotion by observation.Currently, several research groups have capitalized forces to investigate motor behaviour in C. elegans.7–9,26,38,39 Lockery et al. utilized an agarose-free chamber with microposts (coined artificial soil device) and wave-shaped channels to regulate the waveform and trajectory of crawling in a well-controlled format (Fig. 2A). Their results clearly indicated that crawling does not depend on micropost topographies, and that the mechanism for the generation and propagation of the undulatory wave for crawling is independent of diverse microchannel wavelengths.7 In contrast to Lockery et al., Park et al. and Han et al. discovered that their post array structures and micro-bump array promoted enhanced locomotion.9,38 The narrow spacing between the pillars provided more resistance to the worm to propel itself during movement, hence generating a larger trust force.8 A similar investigation of C. elegans locomotion in modulated sinusoidal channels by Parashar et al. revealed that the nematode is capable of adapting to sinusoidal channels with controlled waveforms (90–225 μm).39 Being relatively simple and easy to fabricate accurately, these microstructured devices provide an incredibly sensitive format to study behavioural response to a wide range of stimuli.26,38 In order to explore both locomotive and physiological responses of the nematode in a confined space, Wang et al. monitored a subcellular distribution of the DAF-16 gene that regulates different stress responses. Their experiments demonstrated an increase of DAF-16 nuclear localization, attributed to crowding stress, in a micro-column array with intervals from 200 μm to 40 μm.11
Investigations of the locomotory movement mainly included observations of nematode behaviour. However, these do not answer the question “How does the circuit create rhythmic patterns?” General locomotion defects could arise due to problems in the neuromuscular system that controls motor responses, such as speed and amplitude of motion. Obviously, further approaches are needed to address this question. For instance, electrophysiological recording from C. elegans neurons allows one to access the pattern-generating activity of the locomotory circuit.20 An alternative experimental technique that permits one to link mechanical stimuli into cellular responses is cell or single synapse ablation (Section 5). Consequently, insight into the mechanism of neurodevelopmental disorders, such as epilepsy, is potentially gained by developing a clear understanding of these mechanisms.
These findings clearly show that movement levels depend on the animal's confinement. Thus, an environment can deliver mechanical stimulation to trigger reduced movement and serve as an alternative to acute immobilization acceptable for imaging.
2.3. Platforms for developmental studies
Since development phenomena are inevitable processes of a biological system, accumulated detailed knowledge about C. elegans significantly contributes to revealing the mechanisms of developmental regulation. In this subsection, learning, social behaviour, memorizing, and ageing, are highlighted. A comprehensive review of neurodevelopmental studies was recently presented by Lionaki et al.106Early in 2005, Zhang et al. used microfluidic mazes (Fig. 2C) to underline that odours, produced by pathogenic bacteria, force animals to avoid them after interacting with the pathogens.25 Neuron diagnosis of pathogen-exposed animals in a four-choice maze configuration concluded that a neurotransmitter (serotonin) directly amplified worm olfactory learning by both transcriptional and post-transcriptional mechanisms. Exposure to pathogenic bacteria activated an expression of serotonin in the ADF chemosensory neurons through the serotonin-gated ion channel (MOD-1) of C. elegans.
Subsequently, Qin et al. examined exploratory behaviour of mutants and wild-type individuals in different configurations of microfluidic mazes.42 To inquire into their tendency to search and explore new environments, T-maze and complex U-maze designs were fabricated. The observations suggested that the mechanism of movement was biased by environmental cues (i.e. olfactory sensing) and were consistent with the mechanism used by C. elegans to move along chemical gradients in chemotaxis. As was mentioned in subsection 2.1, serotonin and dopamine regulate locomotive behaviour.1 For this reason, dopamine-poor C. elegans mutants (cat-2) were stimulated with dopamine in order to enhance their locomotory rate to explore the maze. The lack of significant change of the body bend rate was probably a result of slow adaptation to new environments due to the genetic defect.
Associative learning occurs when animals learn to correlate a stimulus or behaviour with a second temporally associated stimulus.1 By means of microfluidic devices, such as continuous T-mazes, accurate analysis of associative capabilities of freely moving individuals between presence of food (one of the most robust driving mechanisms among animals) and spatial location was guaranteed.42 The experiments evidenced that C. elegans was capable of directional selection on food reward, referred to as conditional response.
Obviously, the mechanisms behind exploration, associative learning, and memorizing, are distinct. But the current state of progress does not provide insight into cellular and molecular complexity of these processes. For example, the role of dopamine in the neural circuit either as a positive or a negative reinforcing signal, and the resulting mechanism of adaptation are not well understood. How does the AFD neuron, responsible for thermotaxis in C. elegans, regulate the memorizing of culturing temperatures and contribute to the behavioural plasticity? More work is required to further study these phenomena.
In 2010, Chokshi et al. presented a microfluidic platform that employed the direct delivery of a chemical odour to the nose of C. elegans, in order to characterize and quantify the effect of ageing.46 The authors detected the age-dependent physiological properties of the ASH chemosensory neuron and hypothesized that it also served in early learning processes, including stimulus detection and recognition. In support of the correlation between age-related changes in phenotypes during organism lifespan, Hulme et al. focused on nematode size and locomotion, pointing out the occurance of an overall decrease in swimming frequency over the lifespan of each animal.45
Several theories exist explaining the phenomena of ageing, such as metabolic rate, caloric intake, ageing program, etc.1 Along with attempts to verify one of the theories, Xian et al. made a quantitative analysis of dietary-induced changes in C. elegans lifespan.105 Their experiments demonstrated that their microfluidic device, so called WormFarm, was able to detect ∼15% average lifespan reductions induced by 2% to 4% glucose that appeared in agreement with the theory.
Obviously, micromanipulations allow one to investigate more closely both physiological and structural differences between old and young cells in vivo. The fact that similar proteins are used for programmed cell death in both nematodes and humans, enables to solve current biological problems.1
3. C. elegans manipulation on a chip
As was emphasized in Section 2, the worm has the capability to discriminate and respond to a diverse range of stimuli.3 Thus, a variety of environmental cues can be applied by devices, embedded in a chip-based microfluidic system, to analyze the interaction of neuronal signaling of the organism and its behavioural response. Not surprisingly, environmental control and manipulation of small live samples pose significant challenges. Transferring traditional neurobiology and behavioural investigation techniques to a microfluidic platform holds promise to overcome these challenges. They are driven by substantial progress in LOC integration of functional components (e.g. valves, detectors, etc.). Nowadays, a wide diversity of microfluidic techniques offer automatic high throughput worm-on-a-chip manipulation. In this section we discuss general manipulation techniques such as culturing,45,47,48 sorting or guiding,18,23,24,28,30–32,38,49–53 worm body orientation,54 and immobilization.46–48 Several reviews are available to guide the design of microfluidic devices for on-chip C. elegans handling.77,79–81,84–86,90,91,97,99,1003.1. Worm culturing
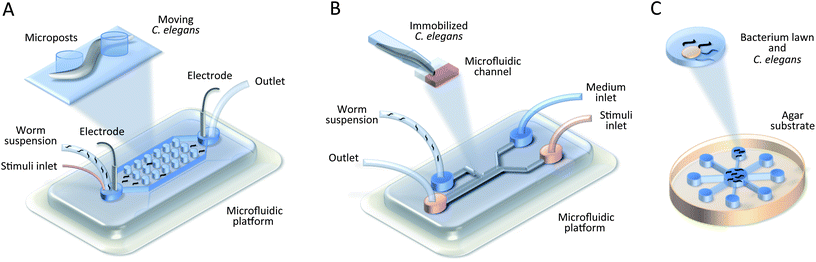
To interpret the underlying metabolic changes and specific developmental processes during nematode ageing, longitudinal experiments over the entire lifespan are necessary. To date, a variety of platforms have been presented by researchers, which enable successful culturing and monitoring of animals in microfluidic reservoirs.43,45,47,105,111 For example, Hulme et al. constructed a chamber array (Fig. 3) for individual long-term observation of C. elegans, from the L4 larval stage until death.45 With a continuous flow of bacteria suspension, they quantified two phenotypes: body size and locomotion. The simple experimental set-up and fabrication process make the device implicitly attractive for downstream applications, however, the design is not applicable to early L1–L3 stages. Thus, a different approach is required to overcome this limitation. Krajniak et al. introduced an age-independent culturing method.47 A hybrid microfluidic device contained eight culture chambers with integrated temperature control layer, which introduced additional constraints for worm population maintenance at the cost of a corresponding increase in overall device complexity.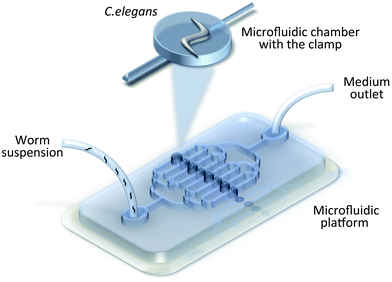 | ||
| Fig. 3 Worm manipulation microfluidic systems. Schematic illustration of the chamber array with clamps for C. elegans loading and culturing.45 | ||
Another integrated microfluidic system for automated culturing and behavioural observation has been demonstrated by Kim et al.48 In this work a centrifuge-based system was capable of automatic nutrient and waste exchange by specified rotational speed and geometry of the compact disc (CD) culturing system. Their results indicated that one incubating chamber on the disk permitted the monitoring of three generations of isogenic living worms. Furthermore, the CD platform requires no mechanical pumping or valving, can be easily adjusted to experimental needs, and can be used for hypergravity environment studies.
3.2. Worm sorting
C. elegans exhibits age-dependent specific neuron and behavioural responses. Therefore, sorting of worm populations or individuals is often required for further diagnostics. A comparison of sorting methods has been presented in Table 2. From the table it can be seen that different sorting approaches have corresponding advantages and disadvantages. Some of the techniques can be coupled with various stimuli, described in Section 2, for particular applications.Typically, worms are exposed to a uniform electric field generated by two electrodes (normally platinum wires) embedded in inlet and outlet reservoirs and connected to external electrical drive circuitry. Such movement-based microfluidic devices allow the differentiation of worms according to locomotive phenotype, for instance, to distinguish adults from larva, or healthy worms from uncoordinated, and to locate individuals defective in electric field sensing. Nevertheless, sorting based on electric field stimulation has several limitations: (1) it is relatively slow because of the need for manual or individual worm loading; (2) the variability of velocities within a population limits its application in high throughput studies (method selectivity is 70–90%); (3) physical sorting depends on the distance worms are allowed to run; (4) it is difficult to implement for large populations; (5) catching worms from microfluidic channels for further separation is not possible.30
In addressing the last aspect, Maniere et al. performed worm sorting in a gel-electrotaxis assay.52 An iterative method enabled isolation of a sub-population, followed by another trace in order to increase sorting resolution. This guiding technique did not allow high throughput (∼40–60 min per trace), however, it characterised the C. elegans population according to locomotive abilities and similar physiological states. Moreover, there was no need for peripheral devices such as pumps or valves, which enabled off-line experiments.
When microfluidic chambers are combined with electrodes to deliver electrical stimuli, both behavioural and neuronal screening can be performed, providing the chance to deduce potential candidates for human muscular disorders.
To meet the requirement of high throughput, Solvas et al. proposed an array of microstructured post “filters” and a network of microfluidic channels. In this system, sorting was accomplished on the basis of age and size in a passive, but extremely high throughput (up to 1200 worms per min) and selective manner (94% of adults with 0.2% larva contamination).50 The device body contained different microstructured, so-called “smart mazes”, and in combination with channel configuration, allowed a large population of adult worms and larvae to be oriented in the desired direction.
In 2007 Rohde et al. created the first “on-valve” two-stage sorting platform with a time response of less than 100 ms.51 On entering a channel, a worm was first captured out of a worm suspension by a single suction channel and later was transferred on an opposite channel wall for subcellular imaging. Depending on the extracted data, the worm could be flushed to either a waste or a sorting outlet by valve actuation. When considering high throughput manipulation, automatic classification of worms (e.g. wild-type from mutants) becomes of high relevance. But unfortunately, multi-step sorting methods require additional image acquisition time, analysis, and valve actuation to synchronize system operation mode.
An alternative concept developed by Chung et al. featured a sequence of valves for automatic sample-positioning and rapid classification based on synaptic characteristics with an overall sorting accuracy of 96.5%.28 Together with real-time rapid image extraction and data processing, the system offered sorting throughput at a rate of 900 worms per hour.
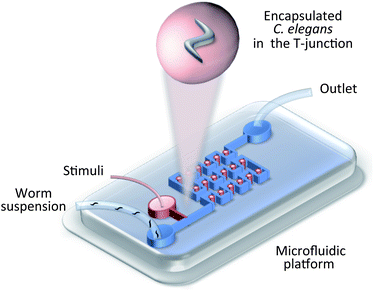 | ||
| Fig. 4 Worm manipulation microfluidic systems. Schematic illustration of droplet-based encapsulation.23 Droplets are continuously generated at the T-junction and are immobilized in the droplet trap array. | ||
3.3. Worm body orientation
For many applications, e.g. drug screening, a specified body orientation of the nematode is required.46 To orient nematodes within microfluidic devices, Caceres and collegues pioneered the use of a curve-shaped channel geometry to passively align worms in a lateral body position with a yield of 84%.54 Being comparatively simple to fabricate (two PDMS layers) and operate (only a pressure source is required to drive valves), the system aided in a full animal body inspection with no reported damage.3.4. Worm immobilization
Because of its high mobility, C. elegans immobilization is critical for further developmental studies (e.g. neurosurgery). The most widely used methods are glueing and anaesthesia.1 The consequent behavioural or physiological perturbations are unpredictable. Moreover, these methods require manual handling and suffer from low throughput and practically no reproducibility, as a consequence, their usage in real-time and long-term monitoring is limited. Immobilization in a microfluidic channel provides a simple well-controlled mechanism for automated handling of worms in a high throughput manner. At the moment, a few techniques, based on different physical principles, have been proposed to immobilize non-anesthetized animals using mechanical force,13,14,17,22,24,28,35,36,46,55,59,60 gel,43,47,96 temperature,28,53,93 gas,35 electric field,29–32 and acoustic waves.62 A comparison of immobilization approaches is listed in Table 3, together with their corresponding characteristics.Micro-arrays of fixed-geometry clamps for C. elegans immobilization, first presented by Hulme et al. in 2007, received wide popularity as a simple method to restrict an individual's motion by employing a single PDMS layer.55 Using a constant pressure difference between the inlet and outlet of the device to drive fluid flow, an array of 128 wedge-shaped microchannels was filled by nematodes with 90% probability. In spite of the fact that viability tests of released worms indicated no significant damage (100% of worms remained alive for up to 9 days), body compression might lead to anatomical changes and modulate physiological processes as aftereffect. In fact, fluorescent calcium imaging by Wen et al. and green fluorescent protein imaging by Mondal et al. showed that immobilizing a portion of the worm can directly override rhythmic activity12 and may cause changes in transport parameters of the touch neuron.22
A year later, researchers utilized a flexible PDMS membrane, that compressed the C. elegans and restricted its motion under an external pressure of 25 psi.35 In most cases, these devices are operated by applying a pressure through a control layer above the main chamber. As a result, the worm is squeezed into the side of microfluidic channel by the deflecting thin membrane.24
Another novel method to fix C. elegans, based on suction flow, was published by Rohde et al.51 A key feature of this device is the vacuum-assisted restraint that aligns the worm along its axis. Unfortunately, suction-reduced movement led to only partial immobilization; and therefore, is limited to genetic screening.
To demonstrate the utility of both immobilization techniques several research groups have combined suction posts with either flexible membranes14,56,59,60 or microchannel narrowing16,20,46,94 for extremely stable immobilization comparable to that of deep anesthesia (Fig. 6B and C).
An alternative technique to immobilize C. elegans employs light-induced sublethal heat, so-called addressable light-induced heat knockdown (ALINK). The technique is based on worm irradiation with a focused laser beam in the presence of electric fields.93 The experimental results indicated that elevated temperatures (31–37 °C) and long recovery times resulting from more operating cycles played a major role in immobilization. However, there are several limitations of this approach. Firstly, only 50% of all worms remained responsive to stimulation before 14 cycles had been applied. Secondly, long recovery times after a few operating cycles resulted. Thirdly, while the immobilization temperature appeared to be harmless, it is known that neural functions of the worm shut down temporarily when a critical temperature of 31 °C is reached.1 All if these factors limit the application of the method.
4. C. elegans imaging techniques
The extraction of meaningful information from a biological organism is a critical consideration in all applications, and this equally applies when using microfluidic technology. Various imaging technologies are used in combination with microfluidic devices for biological and chemical assessment of C. elegans. This combination enables observation of the animal and the extraction of information from e.g. a single neuron. In this section we review the most frequently utilized imaging techniques for C. elegans on-chip applications, which are generalized in Table 4. The techniques can be classified into three major categories: conventional optical, lensless optical, and lensless sensor-less optical detection. Since many of the optical imaging methods have been extensively covered in several reviews,83,87–89 the details will not be discussed in depth.| Imaging technique | Field-of-view | Resolution | Output | Throughput | Technology | Application | Ref. |
|---|---|---|---|---|---|---|---|
| Fluorescent microscopy imaging | Narrow | High | Colour image | Fast | Assembling | Genetic screening, sorting, drug screening | 7, 11, 14–17, 22, 24, 25, 28, 34–37, 41, 44, 46, 47, 53, 54, 56, 57, 60, 63, 66, 68, 93, 94, 95, 96, 98, 101, 102, 105, 107, 108 and 110 |
| Non-fluorescent microscopy imaging | Narrow | High | Colour image | Fast | Assembling | Behavioural phenotyping, monitoring | 7–10, 13, 17, 18, 21–23, 25, 26, 29, 30, 31–35, 38, 39, 42, 43, 45, 47, 49, 50–52, 55, 58, 62, 95, 96, 101, 102, 105 and 107 |
| Contact optofluidic | Wide | High | Colour image | High | Single PDMS layer on a imaging sensor | Monitoring | 70 and 71 |
| Shadow imaging | Wide | Low | Black-and-white image | Fast | Polycarbonate microchambers with polyester film | Monitoring | 69 |
| Digital holography | Wide | High | Colour and monochrome | Slow | Sample plane on printed circuit board (PCB) | Monitoring, neuronal study | 72–75 |
| Micro-electro-fluidic grids | 24 × 24 grids | Low | Pseudo colour, grayscale, and binary image | High | Standard photolithography | Drug screening, electrotaxis | 40 |
4.1. Conventional optical imaging
The transparent body of the nematode offers the immense advantage of obtaining abundant information at the cellular, molecular, and genetic levels. A wide range of conventional optical imaging approaches can be used to monitor C. elegans in microfluidic devices offering several important advantages. Fully automated components, software control, and image processing tools make commercial confocal microscopes extremely versatile for real-time and high-resolution diagnosis. Conventional optical microscopic imaging is the dominant detection technique for worm-on-a-chip observation, which can be classified into non-fluorescent9,10,13,18,21,23,26,27,29–32,38,39,42,43,45,48–50,52,58,62 or fluorescent microscopy,11,12,14–18,22,24,28,33,36,37,41,44,46,53–55,57,60,66,68,92,94 or their various combinations.7,25,34,35,47,74,96,101,102In vivo bright-field microscopy is the most commonly employed method to observe C. elegans behaviour. It has the advantage of being a relatively simple and a label free method, requiring no special preparation of the sample. However, it is restrained to two-dimensions (2D) and is very sensitive to the focus plane position. In the case of differential interface contrast (DIC) microscopy, the phase change of the light incident on the worm can be observed and measured quantitatively.43
In contrast to bright-field imaging, fluorescent microscopy offers high-resolution image acquisition (pixel accuracy >99.9%) and quantification of gene expression. Functional fluorescence imaging methods, such as calcium imaging or green fluorescent protein (GFP) expression, provide a powerful technique for identification of a fluorescently labeled object's response (e.g. signal from a synapse or neuron soma).12,41,46,53,63,103 Using three-dimensional (3D) multi-photon microscopy volume reconstruction, an image deep within the tissues, at multiple time points, can be captured.56,60 The use of these non-invasive imaging techniques is limited mainly by long acquisition times, essential to extract information from a physiologically active nematode, and the probability of phototoxicity in worms.
4.2. Lensless imaging
Even though fluorescence microscopy is a powerful tool for extracting valuable and unique data from biological samples, it is quite expensive and bulky, limiting the miniaturization of chip-based systems. To overcome these limitations, researchers utilize lensless imaging technologies.40,69–74 Compact or on-chip imaging can provide a microscopic image of samples and can be readily integrated with microfluidic devices. This class of techniques includes contact optofluidic imaging,70,71 direct shadow imaging,69 holographic imaging,72–74 and microelectrode grid imaging.40 The schemes are illustrated in Fig. 5. While Zhu et al. put their focus on imaging techniques in general, Wu et al. reviewed lensless microscopy for biological cells and C. elegans in particular.88,89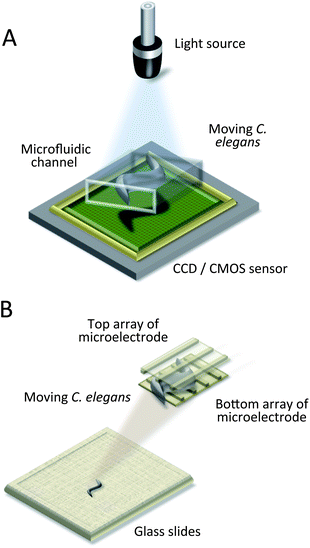 | ||
| Fig. 5 Lensless imaging systems. (A) Schematic drawing of an optofluidic microscope (OFM), and of direct shadow imaging and digital holography systems.69–74 The worm is placed between the light source and sensor, and a video is recorded. (B) Schematic drawing of a micro-electro-fluidic (MEF) grids.40 The worm is detected by the electrical impedance change of the intersection regions of microelectrodes. | ||
In recent years, the development of digital imaging sensors has significantly improved and made the procedure of data processing faster. The combination of digital holography with fluorescence imaging holds a great potential for screening of cellular processes. An inexpensive plastic-based absorption filter and a rhomboid prism have been integrated by Coskun et al. to enable fluorescent excitation of C. elegans in a single platform.74
4.3. Lensless and sensor-less imaging
A completely new approach for real-time monitoring was presented by Liu et al. in 2012.40 In a lensless and an image-sensor-less micro-electro-fluidic (MEF) grid, a moving nematode caused a change in the electrical impedance at the intersection regions of the microelectrode grid, formed by two identical orthogonally arranged arrays of metal lines (Fig. 5B). The authors used a finite element analysis (FEA) approach to simulate the electrical field inside various geometries of the MEF grids for distance optimisation between microelectrode lines, and to evaluate the distribution of the electrical resistance of the MEF grids.Although the read-out frequency of individual detection units enabled high throughput (174 Hz/readout), the spatial resolution (30 μm is the distance between grid lines) and sensitivity were limited by fabrication capabilities. Besides, a complex fabrication process restricted the integration of this approach with other manipulation techniques for the nematode.
5. C. elegans intracellular characterization techniques
On-chip imaging systems and automated data processing have enabled the observation and characterization of key behavioural parameters in vivo at the micrometer and nanometer resolution. However, the approach does not provide a close look at the neuronal signaling and behavioural responses in C. elegans. Techniques for studying and characterization intracellular processes, including dielectrophoresis and electrochemical impedance spectroscopy, can provide high-quality neurogenetic and neuropharmacological data on nematodes. Electrophysiological readouts of neuromuscular function access more information, e.g., effects of drugs on neurons, as well as on muscles.20 In combination with microfluidics and optical image analysis systems, the techniques of microsurgery and microinjection are employed for in vivo neuronal regeneration studies and cell–cell communication by many research groups.56,60,1105.1. Dielectrophoresis and electrochemical impedance spectroscopy
The principle of dielectrophoresis and electrochemical impedance spectroscopy is based on charge transport recordings of free ions moving across an interface between chemical phases in the presence of non-uniform, alternating electric fields. A difference in electric potential can be measured between the applied electrodes, and hence, the electrical properties of a biological sample can be inferred for different frequencies. In contrast to optical techniques, electrochemical methods are label-free, and thus, require no sample pre-processing. Also, these methods are very informative and provide quantitative as well as qualitative information (membrane capacitance, membrane resistance, etc.) Even though electrochemical impedance spectroscopy is not a new technique, there are only few integrated microfluidic “on-chip” systems for C. elegans.20,61,67,104During dielectrophoresis, C. elegans was trapped to investigate the nematode's behavioural response as a function of organism size, electric field intensity, and frequency (Fig. 6A).61 It was concluded that animal on-demand trapping is a result of the electric polarization of the body and not due to electrotaxis. It was also found that the dielectrophoresis (DEP) force is proportional to a worm's size, but that the worm's volume and muscular power did not increase at the same rate in proportion to size.
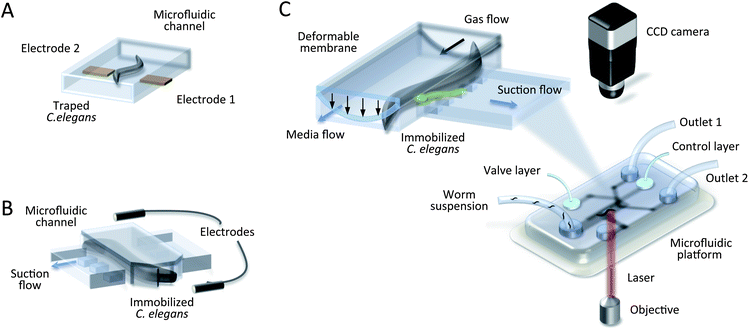 | ||
| Fig. 6 Microfluidic devices for worm study. (A) Schematic drawing of a dielectrophoresis device with a trapped adult worm.61 It is physically immobolised by polarization forces in a non-uniform electric field. (B) Schematic drawing of electrochemical impedance spectroscopy for C. elegans.20 The worm is trapped in the narrowing microchannel with suction flow, and an electropharyngeogram (EPG) is recorded. (C) Schematic drawing of a microsurgical set-up.51,56,60,66 The insert shows a channel cross section where the worm enters a channel and is trapped by a deformable membrane combined with suction flow, enabling laser microsurgery through the cover glass. | ||
An emerging example of electrochemical impedance spectroscopy is a microfluidic device for performing whole-animal chemical screens of neuromuscular function.20,67,104 The screening was accomplished by placing the worm in a physiological saline bath, followed by sucking the animal into a saline filled glass pipette with a microfluidic trap (Fig. 6B).20 The electrical activity from the nematode throat muscles (pharynx) was monitored against a reference electrode using an electric field oscillating at 6 MHz. The recorded electropharyngeogram (EPG) exposed the size and speed of muscle contraction, as well as its depolarization and repolarization. The device was sensitive enough to distinguish wild-type nematodes from mutants. In spite of the fact that this method was capable of automatic real-time monitoring and parallelization (up to 8 worms simultaneously), its usage strongly depended on worm age and body orientation, thus limiting it for high throughput applications. To overcome the limitation of age-dependancy, Hu et al. demonstrated the utility of their microfluidic device, the so called NeuroChip, for smaller size worms (L2 larval stages of C. elegans).104 This experimental approach replaced the need for micromanipulation and a conventional extracellular microelectrode. An additional drug port allowed the rapid chemical application for neuroactive drug screening with a throughput of 12 worms per hour.
One issue associated with impedance spectroscopy is a long EPG recording time, required for subcellular-level stabilization of C. elegans. On the one hand, animal trapping in the microchannel guarantees low noise and reproducible activity monitoring of the nematode pharynx, but on the other hand, this makes electrophysiological readout of actual neuromuscular function impossible.
5.2. Microsurgery
One undeniable advantage of using C. elegans as a neural model is that its response to diverse environmental stimuli, so called cause-and-effect relationships, can be directly studied with laser ablation experiments. Because the nervous system is so well described, the role of an individual neuron in sensory recognition and signalling can be defined once the individual behavioural circuit is characterized.The key issue of this method lies in fluorescent-labeling of individual neurons and their deactivation (death) by a laser nanobeam in the UV wavelength region.103 Afterwards, the resulting phenotypes can be examined. Microfluidics in combination with an optical image analysis system supports this procedure, which requires high precision (Fig. 6C).65 The tremendous potential of femtosecond laser microsurgery has been pointed out by several authors for C. elegans whole-cell ablation68 and single synapse removal in a live neuron.36,37,51,56,57,60,64,66 Yanik's research group has performed precision microsurgical operations to study neural degeneration and regeneration in vivo.51,56,60,66 During microsurgery, C. elegans remains physiologically active, but image processing, acquired at cellular resolution, requires high immobilization stability of the worm. For example, it was observed that upon exposure to sub-milliwatt blue illumination, the worm “panicked” and struggled in the microfluidic channel.37
There are a few concerns which make imaging and microsurgery more challenging. First, many dynamic phenomena are missed because imaging is done before and after neurosurgery. Second, localization of the UV laser to a region in space and time makes synaptic ablation insensitive to the neuron or the muscle side of the synapse. Another limitation of the extracted experimental data is that some neurons might alter their responsiveness when the other neurons are killed (e.g. chemotaxis and thermotaxis share molecular and cellular components).1
5.3. Microinjection
An alternative approach for the in vivo study of the mechanisms underlying intercellular communication in C. elegans is microinjection. Localized chemical stimulation can be delivered to single intestinal cells of the immobilized worms by on-chip microinjection to investigate the propagation of an intercellular calcium wave (ICW) in the intestine essential for coordinating cellular activity.110 Using a micromanipulator, the needle tip containing the first chemical agonist (1 mM TG) followed by 50 mg ml−1 of heparin (an inhibitor for IP3 receptors) was moved into the same focal plane and penetrated into the targeted location. The propagation of ICW was observed by fluorescence imaging of the intestine.This method allows one to study cell–cell communication in the multicellular organism. However, it might influence the precision of imaging subcellular structures and consequent microinjections. As was mentioned in Section 3, immobilization by means of side-wall suction does not provide a complete loss of mobility.
6. Conclusions and perspectives
In this review, we aimed to comprehensively review and organize available in situ “on-a-chip” approaches for C. elegans neuronal, behavioural, and developmental studies. The review highlighted manipulation, imaging, and characterization techniques, their advantages, and their limitations. Although the review covers an interdisciplinary area, we tried to maintain a balance to render the review interesting for both engineers and biologists. We structured the references in a supportive diagram (Fig. 1) and summarized the main relevant characteristics for worm sorting (Table 2), immobilization (Table 3), as well as imaging (Table 4).Obviously, porting C. elegans research into a microfluidic environment provides new impulses for cutting-edge experiments, and will aid in the translation of results for the prevention and treatment of human diseases. By enabling subcellular studies in a whole organism population, microfluidics opens up aspects that would remain hidden from traditional laboratory techniques. From the engineering perspective, considerable advances have been achieved in manufacturing technologies, making the design of novel multi-function microfluidic systems feasible. Numerous experiments can be conducted in parallel on the same chip with fewer reagents, improved sensitivity, and increased resolution. As a result, user-friendly configurations in a compact and cost-effective manner make these systems accessible to the biological and bio-chemical communities.
C. elegans has been the subject of intense study over the past decade. For such a simple organism, it exhibits an extraordinary wide range of behavioural and neuronal responses. Nevertheless, several topics, including worm sensitivity to light,36 acoustic waves,62 and magnetic fields,92 are still less characterized, and, more research is needed. Once well-defined correlations are established between genetic mechanisms and cellular structure in C. elegans, light may be shed on long-standing issues in human molecular biology. Therefore, microfluidic-based studies of C. elegans provides an instrument both to expand and accelerate progress related to the treatment of human diseases and injuries.
To push the frontier of genetic and physiological analysis of the multicellular organism in a microfluidic environment, several challenges have yet to be overcome. PDMS has been the dominant material in these studies, since well defined processing technologies result in easily reproducible results. However, the manufacturing process is limited mainly to single layer devices, and more complex microfluidic devices would require specialized skills to gaurantee fluidic connectivity between multiple fluidic layers. Although conventional photolithography, employed in the integrated circuit (IC) industry, is developing in the direction of lowered fabrication costs, it also has certain disadvantages, such as expensive materials and photolithography masks, and time-consuming processing. Moreover, the manufacturing of 3D structures requires a flat surface to start with and a chemical post-treatment. Thus, further development in the engineering of new materials and fabrication technologies will be required to extend device functionalities to truely 3D applications.
We can expect increased system throughput capabilities in the nearest future, and a combination of several modules into a complete, so called micro total analysis system (microTAS), which should be able to deliver the full potential of automation for biomedical applications. This could play a key role in tracking behavioural and physiological phenotypes of several individuals on a long time scale and in parallel, which is of high relevance for development studies, such as learning, memorizing, and ageing.
We also look forward to new technologies that will open the door to further neuronal studies suitable for freely moving worms. Real-time monitoring could simplify various on-going processes during laser ablation. Thus, this could provide a mechanistic understanding of changes in the behaviour of the animal, and how these changes are caused by physiological or morphological changes in the neural circuitry. Continuous worm-on-a-chip research will most certainly bring about more precise qauntatitive and qualitative in vivo analysis for novel applications.
Acknowledgements
We gratefully acknowledge financial support from the European Research Council (ERC) (contract number 290586 from 1.07.2012) which funded this work.References
- D. L. Riddle, T. Blumenthal, B. J. Meyer and J. R. Priess, C. elegans II, 1997 Search PubMed.
- M. Markaki and N. Tavernarakis, J. Biotechnol., 2010, 5, 1261 CrossRef CAS PubMed.
- D. H. Hall and Z. F. Altun, C. elegans Atlas, 2008 Search PubMed.
- D. C. Duffy, J. C. McDonald, O. J. A. Schueller and G. Whitesides, Anal. Chem., 1998, 70, 4974–4984 CrossRef CAS PubMed.
- J. G. White, E. Southgate, J. N. Thomson and S. Brenner, Philos. Trans. R. Soc., B, 1986, 314, 1–340 CrossRef CAS.
- E. Braungart, M. Gerlach, P. Riederer, R. Baumeister and M. C. Hoener, Neurodegener. Dis., 2004, 1, 175–183 CrossRef CAS PubMed.
- S. R. Lockery, K. J. Lawton, J. C. Doll, S. Faumont, S. M. Coulthard, T. R. Thiele, N. Chronis, K. E. McCormick, M. B. Goodman and B. L. Pruitt, J. Neurophysiol., 2008, 99, 3136–3143 CrossRef CAS PubMed.
- S. Johari, V. Nock, M. M. Alkaisi and W. Wang, Lab Chip, 2013, 13, 1699–1707 RSC.
- S. Park, H. Hwang, S. Nam, F. Martinez, R. H. Austin and W. S. Ryu, PLoS One, 2008, 3, e2550 Search PubMed.
- J. C. Doll, N. Harjee, N. Klejwa, R. Kwon, S. M. Coulthard, B. Petzold, M. B. Goodman and B. L. Pruitt, Lab Chip, 2009, 9, 1449–1454 RSC.
- X. Wang, L. Tang, Y. Xia, L. Hu, X. Feng, W. Du and B. Liu, Integr. Biol., 2013, 5, 728–737 RSC.
- Q. Wen, M. D. Po, E. Hulme, S. Chen, X. Liu, S. W. Kwok, M. Gershow, A. M. Leifer, V. Butler, C. Fang-Yen, T. Kawano, W. R. Schafer, G. Whitesides, M. Wyart, D. B. Chklovskii, M. Zhen and A. D. T. Samuel, Neuron, 2012, 76, 750–761 CrossRef CAS PubMed.
- K. E. McCormick, B. E. Gaertner, M. Sottile, P. C. Phillips and S. R. Lockery, PLoS One, 2011, 6, e25710 CAS.
- Y. Wang, J. Wang, W. Du, X. J. Feng and B. Liu, Anal. Bioanal. Chem., 2011, 399, 3475–3481 CrossRef CAS PubMed.
- S. H. Chalasani, N. Chronis, M. Tsunozaki, J. M. Gray, D. Ramot, M. B. Goodman and C. I. Bargmann, Nature, 2007, 450, 63–70 CrossRef CAS PubMed.
- J. Wang, X. Feng, W. Du and B. Liu, Anal. Chim. Acta, 2011, 701, 23–28 CrossRef CAS PubMed.
- N. Chronis, M. Zimmer and C. Bargmann, Nat. Methods, 2007, 4, 727–731 CrossRef CAS PubMed.
- J. A. Carr, A. Parashar, R. Gibson, A. P. Robertson, R. J. Martin and S. Pandey, Lab Chip, 2011, 11, 2385–2396 RSC.
- Y. Iino and K. Yoshida, J. Neurosci., 2009, 29, 5370–5380 CrossRef CAS PubMed.
- S. R. Lockery, S. E. Hulme, W. M. Roberts, K. J. Robinson, A. Laromaine, T. H. Lindsay, G. M. Whitesides and J. C. Weeks, Lab Chip, 2012, 12, 2211–2220 RSC.
- K. Chung, M. Zhan, J. Srinivasan, P. W. Sternberg, E. Gong, F. C. Schroederd and H. Lu, Lab Chip, 2011, 11, 3689–3697 RSC.
- S. Mondal, S. Ahlawat, K. Rau, V. Venkataraman and S. P. Koushika, Traffic, 2011, 12, 372–385 CrossRef CAS PubMed.
- W. Shi, J. Qin, N. Ye and B. Lin, Lab Chip, 2008, 8, 1432–1435 RSC.
- H. Ma, L. Jiang, W. Shi, J. Qin and B. Lin, Biomicrofluidics, 2009, 3, 044114 Search PubMed.
- Y. Zhang, H. Lu and C. I. Bargmann, Nature, 2005, 438, 179–184 CrossRef CAS PubMed.
- D. R. Albrecht and C. I. Bargmann, Nat. Methods, 2011, 8, 599–605 CrossRef CAS PubMed.
- L. Luo, C. V. Gabel, H. Ha, Y. Zhang and A. D. T. Samuel, J. Neurophysiol., 2008, 99, 2617–2625 CrossRef PubMed.
- K. Chung, M. M. Crane and H. Lu, Nat. Methods, 2008, 5, 637–643 CrossRef CAS PubMed.
- P. Rezai, S. Salam, P. R. Selvaganapathy and B. P. Gupta, Biomicrofluidics, 2011, 5, 044116 CrossRef PubMed.
- P. Rezai, S. Salam, P. R. Selvaganapathy and B. P. Gupta, Lab Chip, 2012, 12, 1831–1840 RSC.
- P. Rezai, A. Siddiqui, S. Salam, P. R. Selvaganapathy and B. P. Gupta, Appl. Phys. Lett., 2010, 96, 153702 Search PubMed.
- P. Rezai, A. Siddiqui, P. R. Selvaganapathy and B. P. Gupta, Lab Chip, 2010, 10, 220–226 RSC.
- P. Rezai, S. Salam, B. P. Gupta, P. R. Selvaganapathy, Proc. 2012 Joint Electrostatics Conference, Cambridge, ON, Canada, June 12–14, 2012 Search PubMed.
- J. M. Gray, D. S. Karow, H. Lu, A. J. Chang, J. S. Chang, R. E. Ellis, M. A. Marletta and C. I. Bargmann, Nature, 2004, 430, 317–322 CrossRef CAS PubMed.
- T. V. Chokshi, A. Ben-Yakar and N. Chronis, Lab Chip, 2009, 9, 151–157 RSC.
- J. N. Stirman, M. Brauner, A. Gottschalk and H. Lu, J. Neurosci. Methods, 2010, 191, 90–93 CrossRef PubMed.
- P. B. Allen, A. E. Sgro, D. L. Chao, B. E. Doepker, J. S. Edgar, K. Shen and D. T. Chiu, J. Neurosci. Methods, 2008, 173, 20–26 CrossRef PubMed.
- B. Han, D. Kim, U. H. Ko and J. H. Shin, Lab Chip, 2012, 12, 4128–4134 RSC.
- A. Parashar, R. Lycke, J. A. Carr and S. Pandey, Biomicrofluidics, 2011, 5, 024112–024119 CrossRef PubMed.
- P. Liu, R. J. Martin and L. Dong, Lab Chip, 2013, 13, 650–661 RSC.
- M. M. Crane, K. Chung and H. Lu, Lab Chip, 2009, 9, 38–40 RSC.
- J. Qin and A. R. Wheeler, Lab Chip, 2007, 7, 186–192 RSC.
- H. Bringmann, J. Neurosci. Methods, 2011, 201, 78–88 CrossRef PubMed.
- J. Schwarz, J. Spies and H. Bringmann, Worm, 2012, 1, 12–14 CrossRef PubMed.
- S. E. Hulme, S. S. Shevkoplyas, A. P. McGuigan, J. Apfeld, W. Fontana and G. M. Whitesides, Lab Chip, 2010, 10, 589–597 RSC.
- T. V. Chokshi, D. Bazopoulou and N. Chronis, Lab Chip, 2010, 10, 2758–2763 RSC.
- J. Krajniak and H. Lu, Lab Chip, 2010, 10, 1862–1868 RSC.
- N. Kim, C. M. Dempsey, J. V. Zoval, J. Sze and M. J. Madou, Sens. Actuators, B, 2007, 122, 511–518 CrossRef CAS PubMed.
- J. Clausell-Tormos, D. Lieber, J. Baret, A. El-Harrak, O. J. Miller, L. Frenz, J. Blouwolff, K. J. Humphry, S. Koester, H. Duan, C. Holtze, D. A. Weitz, A. D. Griffiths and C. A. Merten, Chem. Biol., 2008, 15, 427–437 CrossRef CAS PubMed.
- X. C. i Solvas, F. M. Geier, A. M. Leori, J. G. Bundy, J. B. Edela and A. J. Mello, Chem. Commun., 2011, 47, 9801–9803 RSC.
- C. B. Rohde, F. Zeng, R. Gonzalez-Rubio, M. Angel and M. F. Yanik, Proc. Natl. Acad. Sci. U. S. A., 2007, 104, 13891–13895 CrossRef CAS PubMed.
- X. Maniere, F. Lebois, I. Matic, B. Ladoux, J. D. Meglio and P. Hersen, PLoS One, 2011, 6, e16637 CAS.
- M. M. Crane, J. N. Stirman, C. Ou, P. T. Kurshan, J. M Rehg, K. Shen and H. Lu, Nat. Methods, 2012, 9, 977–980 CrossRef CAS PubMed.
- I. C. Caceres, N. Valmas, M. A. Hilliard and H. Lu, PLoS One, 2012, 7, e35037 CAS.
- S. E. Hulme, S. S. Shevkoplyas, J. Apfeld, W. Fontana and G. M. Whitesides, Lab Chip, 2007, 7, 1515–1523 RSC.
- F. Zeng, C. B. Rohde and M. F. Yanik, Lab Chip, 2008, 8, 653–656 RSC.
- S. X. Guo, F. Bourgeois, T. Chokshi, N. J. Durr, M. A. Hilliard, N. Chronis and A. Ben-Yakar, Nat. Methods, 2008, 5, 531–533 CrossRef CAS PubMed.
- J. Tong, P. Rezai, S. Salam, P. R. Selvaganapathy and B. P. Gupta, J. Visualized Exp., 2013, 75, e50226 Search PubMed.
- C. B. Rohde, C. Gilleland, C. Samara, S. Norton, S. Haggarty and M. F. Yanik, 31st Annual International Conference of the IEEE EMBS, Minneapolis, Minnesota, USA, September 2–6, 2009 Search PubMed.
- C. L. Gilleland, C. B Rohde, F. Zeng and M. F. Yanik, Nat. Protoc., 2010, 5, 1888–1902 CrossRef CAS PubMed.
- H. Chuang, D. Raizen, A. Lamb, N. Dabbish and H. Bau, Lab Chip, 2011, 11, 599–604 RSC.
- X. Ding, S. S. Lin, B. Kiraly, H. Yue, S. Li, I. Chiang, J. Shi, S. J. Benkovic and T. J. Huang, Proc. Natl. Acad. Sci. U. S. A., 2012, 1–5 Search PubMed.
- M. Zimmer, J. M. Gray, N. Pokala, A. J. Chang, D. S. Karow, M. A. Marletta, M. L. Hudson, D. B. Morton, N. Chronis and C. I. Bargmann, Neuron, 2009, 61, 865–879 CrossRef CAS PubMed.
- A. Ben-Yakar and F. Bourgeois, Curr. Opin. Biotechnol., 2009, 20, 100–105 CrossRef CAS PubMed.
- C. Fang-Yen, C. V. Gabel, A. D. T. Samuel, C. I. Bargmann and L. S. Avery, Methods Cell Biol., 2012, 107, 177–206 CAS.
- C. Samara, C. B. Rohde, C. L. Gilleland, S. Norton, S. J. Haggarty and M. F. Yanik, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 18342–18347 CrossRef CAS PubMed.
- C. Hu, V. O'Connor, L. Holden-Dye and H. Morgan, 16th International Conference on Miniaturized Systems for Chemistry and Life Sciences, Okinawa, Japan, October 28–November 1, 2012, 1441–1443 Search PubMed.
- K. Chung and H. Lu, Lab Chip, 2009, 9, 2764–2766 RSC.
- D. Lange, C. W. Storment, C. A. Conley and G. T. A. Kovacs, Sens. Actuators, B, 2005, 107, 904–914 CrossRef CAS PubMed.
- X. Heng, D. Erickson, L. R. Baugh, Z. Yaqoo, P. W. Sternberg, D. Psaltisa and C. Yang, Lab Chip, 2006, 6, 1274–1276 RSC.
- X. Cui, L. M. Lee, X. Heng, W. Zhong, P. W. Sternberg, D. Psaltis and C. Yang, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 10670–10675 CrossRef CAS PubMed.
- S. O. Isikman, I. Sencan, O. Mudanyali, W. Bishara, C. Oztoprak and A. Ozcan, Lab Chip, 2010, 10, 1109–1112 RSC.
- W. Bishara, H. Zhu and A. Ozcan, Opt. Express, 2010, 18, 27499–27510 CrossRef CAS PubMed.
- A. F. Coskun, I. Sencan, T. Su and A. Ozcan, PLoS One, 2011, 6, e15955 CAS.
- S. O. Isikman, W. Bishara, S. Mavandadi, F. W. Yu, S. Feng, R. Lau and A. Ozcan, Proc. Natl. Acad. Sci. U. S. A., 2011, 108, 7296–7301 CrossRef CAS PubMed.
- A. Ben-Yakar, N. Chronis and H. Lu, Curr. Opin. Neurobiol., 2009, 19, 561–567 CrossRef CAS PubMed.
- N. Chronis, Lab Chip, 2010, 10, 432–437 RSC.
- A. M. Taylor and N. L. Jeon, Curr. Opin. Neurobiol., 2010, 20, 640–647 CrossRef CAS PubMed.
- M. M. Crane, K. Chung, J. Stirman and H. Lu, Lab Chip, 2010, 10, 1509–1517 RSC.
- Y. Jian-Ping, Y. Fan, L. Xin-Chun, Y. Yan-Yan and C. Zuan-Guang, Prog. Biochem. Biophys., 2011, 38, 877–883 CrossRef.
- W. Shi, H. Wen, B. Lin and J. Qin, Top. Curr. Chem., 2011, 304, 323–338 CrossRef CAS.
- P. Rezai, S. Salam, P. R. Selvaganapathy and B. P. Gupta, Integrated Microsystems, 2011, ch. 27 Search PubMed.
- U. A. Gurkan, S. Moon, H. Geckil, F. Xu, S. Wang, T. J. Lu and U. Demirci, J. Biotechnol., 2011, 6, 138–149 CrossRef CAS PubMed.
- M. F. Yanik, C. B. Rohde and C. Pardo-Martin, Annu. Rev. Biomed. Eng., 2011, 13, 185–217 CrossRef CAS PubMed.
- X. Xu and S. K. Kim, Nat. Rev. Genet., 2011, 12, 793–801 CrossRef CAS PubMed.
- D. Wlodkowic, K. Khoshmanesh, J. Akagi, D. E. Williams and J. M. Cooper, Cytometry, Part A, 2011, 79, 799–813 CrossRef PubMed.
- C. Yi, Q. Zhang, C. Li, J. Yang, J. Zhao and M. Yang, Anal. Bioanal. Chem., 2006, 384, 1259–1268 CrossRef CAS.
- J. Wu, G. Zheng and L. M. Lee, Lab Chip, 2012, 12, 3566–3575 RSC.
- H. Zhu, S. O. Isikman, O. Mudanyali, A. Greenbaum and A. Ozcan, Lab Chip, 2013, 13, 51–67 RSC.
- A. M. Streets and Y. Huang, Biomicrofluidics, 2013, 7, 011302–011323 CrossRef PubMed.
- W. Hui and Q. JianHua, Sci. China: Chem., 2012, 55, 484–493 CrossRef.
- K. Bessho, S. Yamada, T. Kunitani, T. Nakamura, T. Hashiguchi, Y. Tanimoto, S. Harada, H. Yamamoto and R. Hosono, Experientia, 1995, 51, 284–288 CrossRef CAS.
- H. Chuang, H. Chen, C.-S. Chen and W. Chiu, Lab Chip, 2013, 13, 2980–2989 RSC.
- J. Wang, Z. Li, Z. Xu, L. Hu, X. Feng, M. Chen, W. Du, Z. Wu, Q. Luo, T. Xu and B. Liu, Sens. Actuators, B, 2013, 178, 343–349 CrossRef CAS PubMed.
- J. Yang, Z. Chen, F. Yang, S. Wang and F. Hou, Biomed. Microdevices, 2013, 15, 211–220 CrossRef PubMed.
- J. Krajniak, Y. Hao, H. Y. Mak and H. Lu, Lab Chip, 2013, 13, 2963–2971 RSC.
- V. Sivagnanam and M. A. M. Gijs, Chem. Rev., 2013, 113, 3214–3247 CrossRef CAS PubMed.
- S. Mondal, S. Ahlawat, K. Rao, V. Venkataraman and S. P. Koushika, 14th International Conference on Miniaturized Systems for Chemistry and Life Sciences, Groningen, The Netherlands, 3–7 October, 2010 Search PubMed.
- H. Hwang and H. Lu, Biotechnol. J., 2013, 8, 192–205 CrossRef CAS PubMed.
- A. San-Miguel and H. Lu, C. elegans research community, Wormbook, 2013, Chapter Microfluidics as a Tool for C. elegans Biological Research Search PubMed.
- J. Yang, Z. Chen, P. Ching, Q. Shi and X. Li, Lab Chip, 2013, 13, 3373–3382 RSC.
- H. Wen, W. Shi and J. Qin, Biomed. Microdevices, 2012, 14, 721–728 CrossRef PubMed.
- S. I. C. O. Santos, M. Mathew and P. Loza-Alvarez, Opt. Express, 2010, 18, 364–377 CrossRef CAS PubMed.
- C. Hu, J. Dillon, J. Kearn, C. Murray, V. O'Connor, L. Holden-Dye and H. Morgan, PLoS One, 2013, 8, e64297 Search PubMed.
- B. Xian, J. Shen, W. Chen, N. Sun, N. Qiao, D. Jiang, T. Yu, Y. Men, Z. Pang, M. Kaeberlein, Y. Huang and J. D. Han, Aging Cell, 2013, 12, 398–409 CrossRef CAS PubMed.
- E. Lionaki and N. Tavernarakis, Methods Mol. Biol., 2013, 965, 485–500 Search PubMed.
- J. N. Saldanha, A. Parashar, S. Pandey and J. A. Powell-Coffman, Toxicol. Sci., 2013, 135, 156–168 CrossRef CAS PubMed.
- S. Salam, A. Ansari, S. Amon, P. Rezai, P. R. Selvaganapathy, R. K. Mishra and B. P. Gupta, Worm, 2013, 2, e24558 CrossRef PubMed.
- H. Lee, M. Crane, Y. Zhang and H. Lu, Integr. Biol., 2013, 5, 372–380 RSC.
- X. Zhao, F. Xu, L. Tang, W. Du, X. Feng and B. Liu, Biosens. Bioelectron., 2013, 50, 28–34 CrossRef CAS PubMed.
- J. Jung, M. Nakajima, H. Tajima, Q. Huang and T. Fukuda, J. Micromech. Microeng., 2013, 23, 085008 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c3ra43758b |
| This journal is © The Royal Society of Chemistry 2014 |