Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Serum ferritin is an important inflammatory disease marker, as it is mainly a leakage product from damaged cells
Douglas B.
Kell
*a and
Etheresia
Pretorius
*b
aSchool of Chemistry and The Manchester Institute of Biotechnology, The University of Manchester, 131, Princess St, Manchester M1 7DN, Lancs, UK. E-mail: dbk@manchester.ac.uk; Tel: +44 (0)161 306 4492
bDepartment of Physiology, Faculty of Health Sciences, University of Pretoria, Arcadia 0007, South Africa. E-mail: resia.pretorius@up.ac.za; Tel: +27 12 420 2864
First published on 24th January 2014
Abstract
"Serum ferritin" presents a paradox, as the iron storage protein ferritin is not synthesised in serum yet is to be found there. Serum ferritin is also a well known inflammatory marker, but it is unclear whether serum ferritin reflects or causes inflammation, or whether it is involved in an inflammatory cycle. We argue here that serum ferritin arises from damaged cells, and is thus a marker of cellular damage. The protein in serum ferritin is considered benign, but it has lost (i.e. dumped) most of its normal complement of iron which when unliganded is highly toxic. The facts that serum ferritin levels can correlate with both disease and with body iron stores are thus expected on simple chemical kinetic grounds. Serum ferritin levels also correlate with other phenotypic readouts such as erythrocyte morphology. Overall, this systems approach serves to explain a number of apparent paradoxes of serum ferritin, including (i) why it correlates with biomarkers of cell damage, (ii) why it correlates with biomarkers of hydroxyl radical formation (and oxidative stress) and (iii) therefore why it correlates with the presence and/or severity of numerous diseases. This leads to suggestions for how one might exploit the corollaries of the recognition that serum ferritin levels mainly represent a consequence of cell stress and damage.
Introduction
In mammals (in contrast, for instance, to some functions in insects1–4), ferritin is supposed to be a cellular means of storing iron,5 not of transporting it, yet serum ferritin levels are widely measured as indicators of iron status. However, the soluble transferrin receptor (sTfR)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log ferritin ratio (sTfR Index) probably provides a better estimate of body iron over a wide range of normal and depleted iron stores.6–9 This is because serum ferritin levels can be raised significantly in response to inflammation and/or a variety of diseases (see later). "Serum ferritin" thus presents something of a paradox. Taking a systems approach, we develop and summarise the view that "serum ferritin" actually originates from damaged cells (and thus reflects cellular damage), that it contains some iron but has lost or liberated most of its normal content, and that since the protein part of ferritin is assumed to be benign, that it is this (initially) free iron that correlates with and is causative of disease. The rest of this analytical and synthetic review summarises the wide-ranging evidence for this. We necessarily start by reviewing iron metabolism from a systems point of view (Fig. 1).
log ferritin ratio (sTfR Index) probably provides a better estimate of body iron over a wide range of normal and depleted iron stores.6–9 This is because serum ferritin levels can be raised significantly in response to inflammation and/or a variety of diseases (see later). "Serum ferritin" thus presents something of a paradox. Taking a systems approach, we develop and summarise the view that "serum ferritin" actually originates from damaged cells (and thus reflects cellular damage), that it contains some iron but has lost or liberated most of its normal content, and that since the protein part of ferritin is assumed to be benign, that it is this (initially) free iron that correlates with and is causative of disease. The rest of this analytical and synthetic review summarises the wide-ranging evidence for this. We necessarily start by reviewing iron metabolism from a systems point of view (Fig. 1).
A systems biology overview of human iron metabolism
A starting point for systems biology is the creation of the network (mathematically a ‘graph’) of interacting partners (e.g.ref. 10–14). To this end, a number of recent genomic-level or systems biology reviews have summarised the chief features of human iron metabolism (e.g.ref. 15–19). (Systems genetics analyses are also available.20–23) For the present purposes, aimed at seeking the ‘function’ of human serum ferritin (SF), we shall take a particularly high level view, and assume that the body has a very restricted number of compartments. Fig. 2, updated from ref. 15 shows essentially just three: intestinal tissue, peripheral tissue and blood/serum, and (see also ref. 24, 25 and cf.ref. 26) these will be quite sufficient.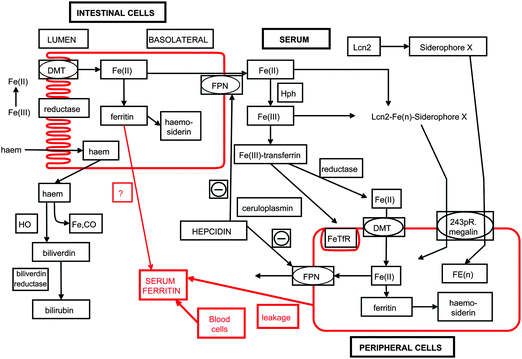 | ||
| Fig. 2 A high-level, three-compartment overview of iron metabolism (based on15) and the means by which we consider that ferritin appears in serum by leakage from peripheral (and possibly intestinal) cells. BR biliverdin reductase, DMT1 divalent metal transporter1, HO haem oxygenase, Hph hephaestin, TfR transferrin receptor, Lcn2 lipocalin2, also known as Neutrophil gelatinase-associated lipocalin. Diagrams rendered by Dr Steve O'Hagan. | ||
Thus, as is well known, ferric salts and ions are poorly water soluble (hence the need for siderophores – better known in microbiology27–30), and much of the complex (redox) chemistry of iron in the body is designed to deal with this. In addition to its existence in divalent and trivalent states, iron is also capable of being liganded in up to 6 places (4 equatorial, 2 polar), and this liganding is necessary to stop its otherwise exceptional reactivity, specifically the production of the very damaging hydroxyl radical that reacts in nanoseconds with the nearest biological substances15,17via the Fenton reaction31–35 of H2O2 and Fe(II). This may be coupled to the re-reduction of Fe(III) to Fe(II) by superoxide in the Haber–Weiss reaction,31–35 such that unliganded (or poorly liganded) iron moieties are catalytic and thus especially dangerous. Thus, while iron is vital for living processes, there is an exceptionally important need to sequester iron in a suitably liganded form, and cellular ferritin is a major means of doing this.36
Leaving aside haem, and also nutrient-derived ferritins,37,38 iron is absorbed in the intestine as ferrous ions and transported in the serum bound (in the ferric form) to transferrin, where it can enter peripheral tissues via suitable receptors, being re-reduced in the process. Ferrous iron is incorporated into ferritin, simultaneously being oxidised at a di-iron centre39 to ferric iron. Thus, importantly, ferritin is made in cells (including intestinal cells), and not in serum. We also note the evidence for the presence of ferritin within erythrocytes,40–54 the largest volume fraction of serum.55 In nucleated cells, ferritin resides mainly in the cytoplasm, but there are nuclear56–61 and mitochondrial62–64 forms (not considered here, as our focus is serum ferritin). An overview of cellular iron metabolism is given in Fig. 3.
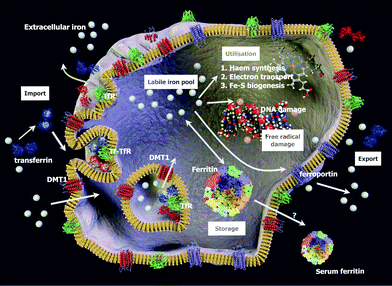 | ||
| Fig. 3 Some relevant aspects of cellular iron metabolism, including ferritin and its possible loss to serum. The figure is not to scale, and is based in part on.67 Membrane protein concentrations shown are lower (for clarity) than those in real cell membranes.458 Diagram rendered by Dr Steve O'Hagan. | ||
Although there are bacterial (and other) ferritins that have only 12 subunits,65 human ferritins consist of 24 subunits of a light (L) and heavy (H) chain arranged by self-assembly in a tetracosameric, octahedral cage with 4-3-2 symmetry (e.g.ref. 5, 66–70). In humans, the molecular masses of the two chains are 19 (173 amino acids) and 21 kDa (183 amino acids), respectively,61 and the subunits are structurally interchangeable,71 even between mammalian species.72 The heavy subunit is primarily responsible for the ferroxidase activity of the ferritin complex,39 whereas the light subunit (L also standing for Lacks catalysis73) facilitates the storage of iron into the ferritin core.61 Many X-ray structures are known.74 Broadly, each subunit consists of a 4-helix bundle, and their self-assembly (whether iron is present or not) is energetically extremely favourable – the melting or denaturation temperature of the 24mer cage is some 40°C greater than that of an individual subunit.75
Iron loading mechanism of ferritin
The main features of the typical 24-subunit ferritin architecture (shown as an all-H-chain variant) are given in Fig. 4. Human ferritin is some 12 nm diameter overall, with a 2 nm thick protein shell and a hollow internal 8 nm diameter cavity capable of holding up to 4500 iron atoms. Ferrous ions can diffuse into (and out of) the core via the eight, hydrophilic ∼4 Å × 15–20 Å channels located at the 3-fold symmetry axis,70,73,76–82 where they are oxidised by dioxygen (or H2O2 if present) at a di-iron catalytic site to form Fe(III)2–O products that then form the Fe2O3·H2O mineral core.78,83,84 Other materials such as phosphate may also serve as counterions.82,85 Ferritin Fe3+O nucleation channels open onto the internal surfaces of ferritin protein cages at the four-fold symmetry axes of the ferritin protein cage.82 The six channels located at the 4-fold axis of the protein are hydrophobic; their function does not seem to be known with any certainty, but they may permit entry of dioxygen and/or H2O2.77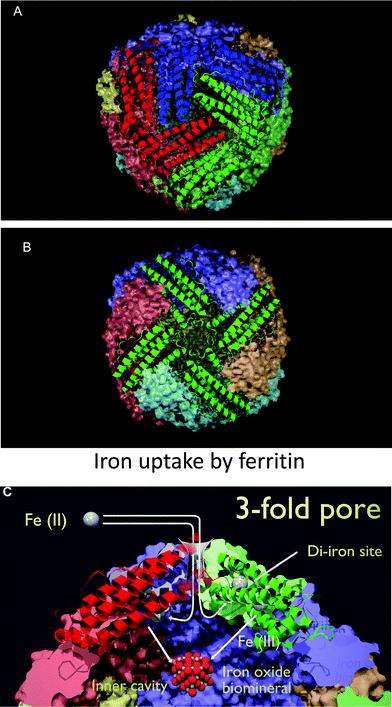 | ||
| Fig. 4 The architecture of a human ferritin, rendered from PDB structure 1FHA (all-H-chain variant). (A) A view down one of the hydrophilic channels representing the 3-fold axis of symmetry through which iron enters the ferroxidase site en route to the core. (B) A view down the hydrophobic channels representing the 4-fold axis of symmetry (whose function is unknown). (C) Entry of Fe2+ into ferritin via a hydrophilic channel, and conversion at a di-iron site to Fe3+, based loosely on a diagram in73 – note that for clarity the iron atoms are not drawn to scale. Diagrams rendered by Dr Steve O'Hagan. | ||
It is not quite so clear how (after storage as Fe(III) in the ferritin core) Fe(II) exits the channels81,86 to become available to cells, nor how the physiological (in vivo) reductant reaches the potential site of reduction inside the small channels. It is not clear even what the physiological reductant is,87 though NADH and FMN have been reported to serve,82,88 as have superoxide89 and other materials.81
How much iron in cellular/tissue ferritin?
The number of iron atoms/ferritin cage is said to average 1000–1500 normally,73 governed more by iron availability than anything else, with a maximum of 4500 iron atoms normally being quoted (e.g., ref. 90–92, and attained for iron overload conditions or when loaded artificially in vitro). Direct observation also leads to a mode value of ∼1500 in a liver biopsy from a patient with hereditary haemochromatosis.93What kind of ferritin in which tissues?
As mentioned, from a structural point of view in terms of forming the 24mer nanocage, ferritin H and L forms are interchangeable.71 Similarly, as expected, ferritin is expressed in most tissues. Thus, human protein atlas expression data for the light chain http://www.proteinatlas.org/ENSG00000087086/normal show it mainly in CNS, bone marrow, spleen, liver, kidney, lung and adipocytes. Expression of the heavy chain is broadly similar http://www.proteinatlas.org/ENSG00000167996/normal save that it is also highly expressed in breast, uterus, testis, prostate and thyroid tissue. In terms of the actual stoichiometries of L:H in ferritin molecules in different tissues (which also affects the ordering or crystallinity of the mineral core73,87) there is rather less information, and variations in this may be causative of disease.94,95 Clearly, for a 24-subunit molecule with two kinds of subunits, one can build 25 canonical ‘isoferritins’.74 Liver and spleen ferritin is mainly the L subunit while heart and brain ferritin is mainly the H subunit. Serum ferritin is mainly in the L form,5,96 consistent with the view that it typically originates in the liver.97 The same (i.e. mainly the L form) is presumably true for erythrocyte ferritin, in that this is what the usual ELISA tests for serum ferritin are designed to detect.Natural degradation of ferritin
The exact circumstances under which ferritin is normally degraded in vivo (if it is intact) are not entirely clear, but what is clear is that there is a fundamental conceptual problem, in that if the only part degraded is the protein the result is the damaging liberation of unliganded iron. Certainly, as expected for normal cellular degradation, the proteasome is involved,38,98 but there is also a major lysosomal degradation pathway.38,99–103 We note too that overexpression can lead to the formation of ferritin inclusion bodies.104As well as proteolytic degradation, there are other means of ferritin removal. Thus, haemosiderin is an insoluble material formed from damaged ferritin (ferritin with exposed and potentially chemically reactive mineral sites), commonly appearing under conditions of iron overload and often reflecting a poorer disease prognosis (e.g.ref. 71, 105–112). (Note that another insoluble cellular degradation cluster – lipofuscin (e.g.ref. 113–116) – is different, as it does not contain haemosiderin.) However, the insoluble substance neuromelanin (e.g.ref. 115, 117–119) may contain ferritin or ferritin-like material.120–122 The question of what happens to haemosiderin seems rather poorly understood, but in contrast to ferritin it is not normally seen (nor at least measured) in serum;123,124 since it is composed of large, insoluble aggregates it is possibly not surprising that it does not leak from cells. Overall, however, it seems that we have comparatively little information on the important question of what happens to its iron content when the protein part of the ferritin molecule either leaves the intracellular environment or is degraded.
In what form is serum ferritin measured?
As mentioned previously, ferritin has an H and L form that are structurally interchangeable. Serum (L-)ferritin is usually measured with antibodies; only rarely is its iron content measured as well. Mass spectrometric methods, that can measure both protein and internal materials, may thus be expected to become the methods of choice.125–128 When such measurements are done, serum ferritin is usually found to contain some iron, but nothing like its full complement.91,92,97,129,130 This implies that it has lost it, whether during or after effluxing from the cells in which it originates.87Is the protein component of serum ferritin benign or toxic?
This question arises because if the iron has escaped and now (say) the inside of the ferritin is exposed in the serum it might have effects that the intact protein does not (given that the intact protein is extremely stable to thermal unfolding75). There is some fragmentary evidence that serum ferritin itself may have apoptotic and other actions on cells.68,131,132 However, at present it is rather difficult to answer the question of how benign the protein-only form of ferritin (i.e. apoferritin) actually is, since serum ferritin does always tend to contain at least some iron, which can be released and is then not at all benign. When the iron is varied systematically, it is iron-loaded ferritin that is the more toxic,133 with apoferritin in fact being protective.133–137 An important piece of evidence comes from the fact that homozygous ferritin knockout mice are embryo-lethal138 but that heterozygous Fth+/− mice are fairly normal save that they have greatly increased levels of serum ferritin but unchanged serum iron.139 This shows us, importantly, (i) that iron and ferritin can be regulated independently, and (ii) that excess ferritin protein is not of itself toxic in vivo (see also ref. 140). Hereditary hyperferritinemia-cataract syndrome is another disease in which serum ferritin is high but there is no evidence of systemic iron overload.141–146 However, as well as (sometimes) being a marker of liver iron stores, serum ferritin is also an inflammatory marker, and there is often a considerable correlation between disease status and the serum ferritin protein level as measured using antibodies (which do not distinguish ferritins with varying iron content).Serum ferritin can be a marker of iron stores but is also an inflammatory biomarker
What matters from the point of view of mammalian biology is both the total amount of iron and its speciation. While iron is necessary in every metabolising tissue, a substantial amount of iron is held in the liver, so ‘liver iron stores’ are often taken as the gold standard. Traditionally, these were measured in a biopsy, although this is not something that can be done with any frequency. Fortunately non-invasive measurement and imaging methods, e.g. neutron-stimulated emission controlled tomography,147 SQUID-biosusceptometry129,148 and (in particular) MRI (e.g.ref. 149–158), also widely used for brain imaging (e.g.ref. 159–161), are coming through. In some cases, where there is no inflammation and/or if a specific iron-related disease state is known, liver iron content can correlate with serum ferritin (e.g.ref. 162 and 163), but more often the correlation is poor (e.g.ref. 129, 157, 164–171). This is more or less inevitable when serum ferritin levels can be affected by two largely independent causes, viz. iron status and inflammatory status. Thus, as mentioned above, serum ferritin alone is falling out of favour as a marker of iron status, with serum (‘soluble’) transferrin receptor (sTfR) being seen as much more useful, since sTfR may be used to distinguish the anaemia of chronic disease from iron-deficiency anaemia.172 In particular, the "sTfR Index" (the sTfR/log ferritin ratio when both are measured in μg L−1) is now considered to provide an estimate of body iron over a wide range of normal and depleted iron stores,6–9 and again is thus better for discriminating iron deficiency anaemia from the anaemia of chronic disease9,173–175 (cf.ref. 176).In consequence, and especially in countries where inflammatory diseases are highly prevalent, it would seem that serum ferritin may in general be a better marker of inflammation than of iron status.
Some diseases in which serum ferritin levels correlate with the presence or severity of disease
One of us has previously listed a great many (inflammatory) diseases in which iron dysregulation clearly plays a major role (e.g.ref. 15 and 17), but did not there distinguish serum ferritin explicitly. It is therefore helpful to set down some of the studies in which serum ferritin is known to associate with disease and/or disease severity, and this is done in Table 1.| Disease or syndrome | Selected references |
|---|---|
| Acute respiratory distress syndrome | 181–184 |
| Amyotrophic lateral sclerosis | 185–189 |
| Atherosclerosis | 96, 190–200 |
| Cancer | 201–214 |
| Cirrhosis of the liver | 215–217 |
| Coronary artery disease | 218–221 |
| Diabetes mellitus, type 2 | 221–249 |
| Hypertension | 250–254 |
| Metabolic syndrome | 235, 236, 252, 255–272 |
| Multiple sclerosis | 273–276 |
| Myocardial infarction | 277–285 |
| Non-alcoholic fatty liver disease | 260, 262, 264, 270, 286–301 |
| Preeclampsia | 302–306 |
| Rheumatoid arthritis | 307–314 |
| Sepsis/SIRS | 315–318 |
| Stroke | 319–330 |
| Systemic lupus erythematosus | 274, 331–342 |
There can be very little doubt that high serum ferritin levels accompany a great many diseases, and the corollary of this is that iron-induced hydroxyl radical formation leading to oxidative damage is likely to be a contributory factor in all of them. In addition, there are other useful phenotypic readouts that change with serum ferritin, and the next section describes one.
Some morphological and related readouts of haematological changes associated with inflammatory diseases
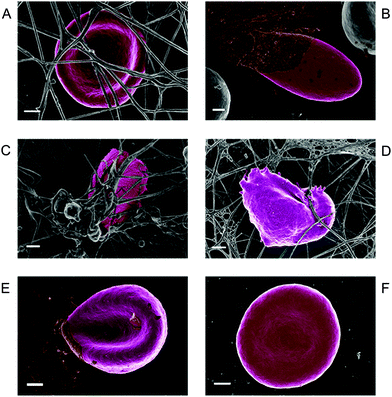
While not the entire focus of this review, we highlight two other accompaniments to the unliganded iron caused by its loss from ferritin, namely morphological changes to both fibrin and erythrocytes. Thus, we have recently been developing the idea that many of the consequences of unliganded iron can be observed directly, by changes in properties such as erythrocyte (RBC) morphology and deformability and the nature and morphology of fibrin fibres generated in the presence of thrombin (as is observed in a number of diseases343–346). When thrombin is added to healthy whole blood, the RBCs will keep their typical discoid shape while fibrin fibres will form over and around the RBCs (such a typical healthy RBC (from an individual with a serum ferritin of 19 ng·mL−1), surrounded by fibrin is shown in Fig. 5A). However, in inflammatory conditions, where iron overload is present, the RBCs lose their typical discoid shape, while the fibrin network forms a dense matted layer. This was previously noted in RBCs of hereditary haemochromatosis, pro-thrombin mutation and antiphospholipid syndrome with increased serum ferritin levels and in high serum ferritin levels in Alzheimer's disease.347–351Fig. 5B–D show examples of RBCs and fibrin in these conditions. The corollary is clear, namely that these kinds of changes should be observable in cases where we see high serum ferritin, and some examples have already been published.In the presence of iron, the already compromised RBCs are entrapped in the pathological fibrin masses. Iron plays an important role in the change of a netlike fibrin layer to a matted mass. We previously showed that healthy fibrin can be changed to resemble this matted appearance, when physiological levels of iron are added to plasma.352 Such matted fibrin morphology was also previously noted in type II diabetes, thrombotic ischemic stroke and systemic lupus erythematosus. Here the compromised RBCs twist around the fibres and this may cause a tight and rigid clot that might be particularly resistant to fibrinolysis.353–355
As well as undergoing a shape change, the RBC membranes, in the presence of iron overload, also lose their elastic ability (deformability). This was noted in Alzheimer's Disease individuals with iron overload, where their RBCs have a decreased membrane elasticity.347 A changed RBC membrane roughness was also noted in diabetes.356
Further, RBC shape and membrane changes have been noted in smokers and in individuals with Chronic Obstructive Pulmonary Disorder (COPD).357,358 Both conditions are known to cause a general inflammatory state in the user as well as increased serum ferritin levels,359 and this may aid in the developing of the changed RBC deformability.
RBCs are extremely adaptable cells, particularly due to their rheological properties that force them to deform and reform under shear forces when they travel through narrow capillaries, while in the presence of high (poorly liganded) iron levels, they lose this deformability. By contrast, diseased RBCs can regain their discoid shape when selected chelators are added.350 Here we show how an RBC from a HH individual can return to the typical discoid shape after the addition of physiological levels of the iron chelator Desferal (Fig. 5E and F). This may have profound clinical implications under conditions where iron overload is present.
Thus, this unliganded iron affects (negatively) at least three things that can each contribute to vascular woes: erythrocyte morphology, erythrocyte deformability and fibrin structure/morphology.
Chelation for the reversal of iron-induced effects
The recognition that these changes can be reversed by known iron chelators leads to the recognition of a further prediction: that disease severity may be decreased through the use of iron chelators that may be pharmacological or nutritional. For the former, three iron chelators have been approved for clinical use (e.g.ref. 15, 360–364), viz. desferal/deferoxamine/desferrioxamine,365 L1/deferiprone366–368 and deferasirox.369–372 From the nutritional point of view, there is considerable evidence that many of the benefits of polyphenolic antioxidants (such as are found in coloured, and especially purple, fruits) derive from their ability to chelate unliganded iron (see e.g.ref. 17, 373–380).Chemical kinetic basis of the relation of serum ferritin to liver iron stores and with disease
Many dozens of references indicate that in normal humans (without overt inflammation) serum ferritin levels are more or less closely related to body iron stores (e.g. in the liver) as judged by magnetic resonance imaging, biopsy or repeated phlebotomies. A selection of such references includes.163,169,381–385Since there is normally a decent correlation between body iron stores and serum ferritin, a series of simple (even first order) reactions in which cells release ferritin can account for this (Fig. 6). The question arises as to the nature of this ‘release’.
Ferritin transfer from cells to serum in humans: less active secretion, more simply leakage from damaged cells
Partly because a fraction of serum ferritin is glycosylated, as judged more or less solely by its ability to bind to concanavalin A (not a very specific assay), it is occasionally stated that ferritin is ‘secreted’ (e.g.ref. 382, 386 and 387), implying a controlled process, but without – so far as we are aware – any actual evidence for secretion rather than leakage being the mechanism in vivo. Indeed when ferritin is genuinely secreted, as it is for instance in insects,3,4,388 it has suitable leader (secretion signal) sequences, and mammalian ferritins do not.This said, in cell cultures, there is some (scant) evidence for a comparatively small amount of regulated secretion,389 and one paper states that secretion can be decreased by brefeldin, an inhibitor of Golgi processes.390 This secreted form is said to be mainly the more acidic H form131 and is glycosylated. We note that both SCARA5 and the transferrin receptor can act as receptors for serum ferritin,68,391,392 as can TIM-2 in mice,393 that can in some circumstances be taken up into cells.394 There is also evidence for active secretion (of a non-glycosylated form) in mice.395 Overall, however, there is not as yet any real evidence for regulated or active secretion in humans in vivo, such that the origin of serum ferritin must indeed largely, if not entirely, be seen as cellular damage. A number of analyses in the literature are consistent with this, and the following four sections pertain.
Relative lack of homeostasis of serum ferritin
The ‘normal range’ of a biochemical concentration is a body fluid is usually taken as the middle 95 percentiles. Somewhat like the Gini indices of economics,396 it is then possible to assess the ratio of particular percentiles, which gives an indication of the spread of these among populations. We shall call this ratio (of the 2.5th and 97.5th percentile) the 95 percentile ratio or 95PR. A small spread implies a tighter degree of regulation or control. The large normal range of serum ferritin (18–350 ng mL−1) relative to other biochemical variables (http://www.globalrph.com/labs_def.htm#Ferritin_), with a 95PR of nearly 20, implies that it is not the subject of homeostasis, i.e. that its appearance is not regulated. One might also comment on the very low normal concentrations of serum ferritin (up to say 350 ng mL−1 in men, up to say 150 ng mL−1 in women) relative to say transferrin (1.88–3.41 mg mL−1) (http://www.globalrph.com/labs_t.htm) or fibrinogen (2–4 mg mL−1).Association between serum ferritin and biomarkers of liver damage
As stated by Theil:70 "serum ferritin likely originates from cell leakage". The figure in67 implies a similar role. Similarly, Hubel305 points out correlations between serum aspartate aminotransferase (a marker of hepatocellular damage) and SF,397 which again implies that serum ferritin originates from cellular damage. Many other authors (e.g.ref. 87, 91, 129, 288, 382 and 398) take a similar view. Serum alanine aminotransferase is another well known marker of liver damage that correlates with serum ferritin,93,215,257,287–289,399–407 consistent with the view that serum ferritin is indeed a marker of damaged cells. In this regard, it is worth noting that the rate of cell turnover, and especially liver cell turnover/regeneration, can be very high (e.g.ref. 408–411).Correlation of serum ferritin with other markers of oxidative stress and hydroxyl radical formation
Since intracellular ferritin is a means of storing iron safely,412 and indeed its synthesis is increased in response to oxidative stress,413–416 one should not necessarily expect serum ferritin to be related to biomarkers reflecting hydroxyl radical formation via the Fenton reaction, that is catalysed by unliganded iron. However, in a similar vein to the liver damage above, serum ferritin levels do correlate with serum markers of hydroxyl radical formation such as 8-hydroxydeoxyguanosine,17,417–424 27-hydroxycholesterol,425 4-hydroxynonenal,131,290 isoprostanes,426,427 and malondialdehyde.406,428–436 Given that only unliganded iron can do this, the easiest interpretation of such data is that the serum ferritin has lost its iron and that it is this unliganded iron that catalyses hydroxyl radical formation and thus the production of these markers. An extensive food processing literature also documents this loss of iron from ferritin in muscle foods (e.g.ref. 437–439), where the consequent lipid oxidation is a major issue in causing rancid tastes, and where metal chelators decrease it.440,441Correlation of platelet microparticles with serum ferritin – further evidence for the cell damage hypothesis
As mentioned, a considerable number of papers note the presence of ferritin in erythrocytes, the largest cellular compartment in blood.40,43–50,53,54 In RBCs, one of the more notable cell death mechanisms is eryptosis, a suicidal death of erythrocytes; this is characterized by erythrocyte shrinkage, blebbing, and phospholipid scrambling of the cell membrane. There is limited evidence that eryptosis occurs in iron overload conditions like β-thalassemia.442 It is noteworthy that erythrocyte-derived microparticles are also often observable in the blood of patients with diseases associated with high serum ferritin levels (Table 1).443–453 These microparticles are circulating fragments derived from blebbing and shedding of cell membranes through several mechanisms that include activation, apoptosis (in nucleated cells) and cell damage.444,454 These microparticles are well-known in cardiovascular, neoplastic, and inflammatory diseases and this again implies a correlation between cellular damage and serum ferritin. Cell damage also releases both phospholipids and DNA, and (in a similar vein) ferritin levels are also raised in diseases in which antibodies to such molecules are also present (e.g.ref. 455–457).Summarising remarks
Although serum ferritin is widely seen as an inflammatory biomarker, our understanding of its role as an intracellular iron storage protein gives no explanation of why it should even exist in serum. The view summarised here is that serum ferritin leaks from damaged cells, losing most of its iron on the way, and leaving that iron in an unliganded form that can impact negatively on health. This unliganded iron can of course stimulate further cell damage.17 This overall view serves straightforwardly to explain the following, known observations.(1) Serum ferritin exists, despite the fact that ferritin is not synthesised in the serum.
(2) Serum ferritin lacks most of the iron it contained when intracellular.
(3) The intracellular ferritin must have ‘dumped’ its unliganded iron somewhere, where it can participate in Haber–Weiss and Fenton reactions, creating hydroxyl radicals and consequent further cellular damage.
(4) The serum ferritin protein is itself considered benign.139
(5) Yet the level of serum ferritin correlates with numerous inflammatory and degenerative diseases.
Quo vadis (where next)? A perspective for future work
We consider the summary presented here rather persuasive, as it has considerable explanatory power in terms of accounting for the nature and consequences of serum ferritin, and providing corollaries of the fact that it has largely ‘lost’ its iron that are borne out by evidence. It also leads us to note some of the experiments that need to be done. First, we need to understand much better the state of both cellular and serum ferritin in terms both of its subunit composition and the nature and extent of its iron content. We also need to understand better the different cellular and tissue distributions of the variously loaded forms, and we certainly need to determine the toxicity displayed, or protection afforded, by the different forms of well characterised ferritins under different circumstances. Far from implying that serum ferritin is a poor biomarker, it leads us rather to suggest that we need to follow it (and its sequelae) more carefully and longitudinally during the development or otherwise of various diseases, and to test how well its changes reflect therapeutic benefits to disease progression. Only then will we determine its true utility, whether alone or in combination with other biomarkers.Acknowledgements
We thank Dr Steve O'Hagan for considerable assistance with a number of the figures, and Janette Bester for the preparation of the samples for SEM analysis.References
- A. Mehta, A. Deshpande and F. Missirlis, Genetic screening for novel Drosophila mutants with discrepancies in iron metabolism, Biochem. Soc. Trans., 2008, 36, 1313–1316 CrossRef CAS PubMed.
- F. Missirlis, S. Kosmidis, T. Brody, M. Mavrakis, S. Holmberg, W. F. Odenwald, E. M. Skoulakis and T. A. Rouault, Homeostatic mechanisms for iron storage revealed by genetic manipulations and live imaging of Drosophila ferritin, Genetics, 2007, 177, 89–100 CrossRef CAS PubMed.
- D. Q. D. Pham and J. J. Winzerling, Insect ferritins: Typical or atypical?, Biochim. Biophys. Acta, 2010, 1800, 824–833 CrossRef CAS PubMed.
- X. Tang and B. Zhou, Ferritin is the key to dietary iron absorption and tissue iron detoxification in Drosophila melanogaster, FASEB J., 2013, 27, 288–298 CrossRef CAS PubMed.
- P. Arosio, R. Ingrassia and P. Cavadini, Ferritins: A family of molecules for iron storage, antioxidation and more, Biochim. Biophys. Acta, 2009, 1790, 589–599 CrossRef CAS PubMed.
- B. S. Skikne, C. H. Flowers and J. D. Cook, Serum transferrin receptor: a quantitative measure of tissue iron deficiency, Blood, 1990, 75, 1870–1876 CAS.
- Y. Beguin, Soluble transferrin receptor for the evaluation of erythropoiesis and iron status, Clin. Chim. Acta, 2003, 329, 9–22 CrossRef CAS.
- B. S. Skikne, Serum transferrin receptor, Am. J. Hematol., 2008, 83, 872–874 CrossRef CAS PubMed.
- B. S. Skikne, K. Punnonen, P. H. Caldron, M. T. Bennett, M. Rehu, G. H. Gasior, J. S. Chamberlin, L. A. Sullivan, K. R. Bray and P. C. Southwick, Improved differential diagnosis of anemia of chronic disease and iron deficiency anemia: A prospective multicenter evaluation of soluble transferrin receptor and the sTfR/log ferritin index, Am. J. Hematol., 2011, 86, 923–927 CrossRef CAS PubMed.
- E. Klipp, R. Herwig, A. Kowald, C. Wierling and H. Lehrach, Systems biology in practice: concepts, implementation and clinical application, Wiley/VCH, Berlin, 2005 Search PubMed.
- D. B. Kell and J. D. Knowles, The role of modeling in systems biology, in System modeling in cellular biology: from concepts to nuts and bolts, ed. Z. Szallasi, J. Stelling and V. Periwal, MIT Press, Cambridge, 2006, pp. 3–18 Search PubMed.
- D. B. Kell, Metabolomics, modelling and machine learning in systems biology: towards an understanding of the languages of cells. The 2005 Theodor Bücher lecture, FEBS J., 2006, 273, 873–894 CrossRef CAS PubMed.
- B. Ø. Palsson, Systems biology: properties of reconstructed networks, Cambridge University Press, Cambridge, 2006 Search PubMed.
- U. Alon, An introduction to systems biology: design principles of biological circuits, Chapman and Hall/CRC, London, 2006 Search PubMed.
- D. B. Kell, Iron behaving badly: inappropriate iron chelation as a major contributor to the aetiology of vascular and other progressive inflammatory and degenerative diseases, BMC Med. Genomics, 2009, 2, 2 CrossRef PubMed.
- V. Hower, P. Mendes, F. M. Torti, R. Laubenbacher, S. Akman, V. Shulaev and S. V. Torti, A general map of iron metabolism and tissue-specific subnetworks, Mol. Biosyst., 2009, 5, 422–443 RSC.
- D. B. Kell, Towards a unifying, systems biology understanding of large-scale cellular death and destruction caused by poorly liganded iron: Parkinson's, Huntington's, Alzheimer's, prions, bactericides, chemical toxicology and others as examples, Arch. Toxicol., 2010, 577, 825–889 CrossRef PubMed.
- C. Funke, S. A. Schneider, D. Berg and D. B. Kell, Genetics and iron in the systems biology of Parkinson's disease and some related disorders, Neurochem. Int., 2013, 62, 637–652 CrossRef CAS PubMed.
- S. Mitchell and P. Mendes, A Computational Model of Liver Iron Metabolism, http://arxiv.org/pdf/1308.5826v1.pdf, 2013.
- L. C. Jellen, J. L. Beard and B. C. Jones, Systems genetics analysis of iron regulation in the brain, Biochimie, 2009, 91, 1255–1259 CrossRef CAS PubMed.
- B. C. Jones, J. L. Beard, J. N. Gibson, E. L. Unger, R. P. Allen, K. A. McCarthy and C. J. Earley, Systems genetic analysis of peripheral iron parameters in the mouse, Am. J. Physiol.: Regul., Integr. Comp. Physiol., 2007, 293, R116–R124 CrossRef CAS PubMed.
- D. Hwang, I. Y. Lee, H. Yoo, N. Gehlenborg, J. H. Cho, B. Petritis, D. Baxter, R. Pitstick, R. Young, D. Spicer, N. D. Price, J. G. Hohmann, S. J. Dearmond, G. A. Carlson and L. E. Hood, A systems approach to prion disease, Mol. Syst. Biol., 2009, 5, 252 CrossRef PubMed.
- L. Yin, E. L. Unger, L. C. Jellen, C. J. Earley, R. P. Allen, A. Tomaszewicz, J. C. Fleet and B. C. Jones, Systems genetic analysis of multivariate response to iron deficiency in mice, Am. J. Physiol.: Regul., Integr. Comp. Physiol., 2012, 302, R1282–R1296 CrossRef CAS PubMed.
- C. Berzuini, P. C. Franzone, M. Stefanelli and C. Viganotti, Iron kinetics: modelling and parameter estimation in normal and anemic states, Comput. Biomed. Res., 1978, 11, 209–227 CrossRef CAS.
- P. C. Franzone, A. Paganuzzi and M. Stefanelli, A mathematical model of iron metabolism, J. Math. Biol., 1982, 15, 173–201 CrossRef CAS.
- T. J. S. Lopes, T. Luganskaja, M. Vujić Spasić, M. W. Hentze, M. U. Muckenthaler, K. Schümann and J. G. Reich, Systems analysis of iron metabolism: the network of iron pools and fluxes, BMC Syst. Biol., 2010, 4, 112 CrossRef PubMed.
- G. Winkelmann, Ecology of siderophores with special reference to the fungi, Biometals, 2007, 20, 379–392 CrossRef CAS PubMed.
- M. Sandy and A. Butler, Microbial iron acquisition: marine and terrestrial siderophores, Chem. Rev., 2009, 109, 4580–4595 CrossRef CAS PubMed.
- R. C. Hider and X. Kong, Chemistry and biology of siderophores, Nat. Prod. Rep., 2010, 27, 637–657 RSC.
- M. Miethke, Molecular strategies of microbial iron assimilation: from high-affinity complexes to cofactor assembly systems, Metallomics, 2013, 5, 15–28 RSC.
- S. Akatsuka, Y. Yamashita, H. Ohara, Y. T. Liu, M. Izumiya, K. Abe, M. Ochiai, L. Jiang, H. Nagai, Y. Okazaki, H. Murakami, Y. Sekido, E. Arai, Y. Kanai, O. Hino, T. Takahashi, H. Nakagama and S. Toyokuni, Fenton reaction induced cancer in wild type rats recapitulates genomic alterations observed in human cancer, PLoS One, 2012, 7, e43403 CAS.
- S. Goldstein, D. Meyerstein and G. Czapski, The Fenton reagents, Free Radical Biol. Med., 1993, 15, 435–445 CrossRef CAS.
- S. Toyokuni, Iron and carcinogenesis: from Fenton reaction to target genes, Redox Rep., 2002, 7, 189–197 CrossRef CAS PubMed.
- P. Wardman and L. P. Candeias, Fenton chemistry: An introduction, Radiat. Res., 1996, 145, 523–531 CrossRef CAS.
- C. C. Winterbourn, Toxicity of iron and hydrogen peroxide: the Fenton reaction, Toxicol. Lett., 1995, 82–83, 969–974 CrossRef CAS.
- X. Liu and E. C. Theil, Ferritins: dynamic management of biological iron and oxygen chemistry, Acc. Chem. Res., 2005, 38, 167–175 CrossRef CAS PubMed.
- E. C. Theil, H. Chen, C. Miranda, H. Janser, B. Elsenhans, M. T. Nunez, F. Pizarro and K. Schumann, Absorption of iron from ferritin is independent of heme iron and ferrous salts in women and rat intestinal segments, J. Nutr., 2012, 142, 478–483 CrossRef CAS PubMed.
- M. C. Linder, Mobilization of stored iron in mammals: a review, Nutrients, 2013, 5, 4022–4050 CrossRef CAS PubMed.
- K. H. Ebrahimi, E. Bill, P. L. Hagedoorn and W. R. Hagen, The catalytic center of ferritin regulates iron storage via Fe(II)-Fe(III) displacement, Nat. Chem. Biol., 2012, 8, 941–948 CrossRef PubMed.
- F. S. Porter, Erythrocyte ferritin, Pediatr. Res., 1973, 7, 954–957 CrossRef CAS PubMed.
- E. R. Bauminger, S. G. Cohen, S. Ofer and E. A. Rachmilewitz, Quantitative studies of ferritinlike iron in erythrocytes of thalassemia, sickle-cell anemia, and hemoglobin Hammersmith with Mössbauer spectroscopy, Proc. Natl. Acad. Sci. U. S. A., 1979, 76, 939–943 CrossRef CAS.
- A. Jacobs, S. W. Peters, E. R. Bauminger, J. Eikelboom, S. Ofer and E. A. Rachmilewitz, Ferritin concentration in normal and abnormal erythrocytes measured by immunoradiometric assay with antibodies to heart and spleen ferritin and Mössbauer spectroscopy, Br. J. Haematol., 1981, 49, 201–207 CrossRef CAS.
- M. B. Van der Weyden, H. Fong, L. Hallam and M. J. Breidahl, A rapid and simple assay for human erythrocyte ferritin, Clin. Chim. Acta, 1983, 127, 397–401 CrossRef CAS.
- M. B. Van Der Weyden, H. Fong, H. H. Salem, R. G. Batey and F. J. Dudley, Erythrocyte ferritin content in idiopathic haemochromatosis and alcoholic liver disease with iron overload, BMJ, 1983, 286, 752–754 CrossRef CAS.
- L. Muylle, F. L. Van de Vyver and P. P. Blockx, Erythrocyte ferritin content in idiopathic haemochromatosis and alcoholic liver disease with iron overload, BMJ, 1983, 286, 2064–2065 CrossRef CAS PubMed.
- A. Piperno, M. T. Taddei, M. Sampietro, S. Fargion, P. Arosio and G. Fiorelli, Erythrocyte ferritin in thalassemia syndromes, Acta Haematol., 1984, 71, 251–256 CrossRef CAS PubMed.
- H. H. Bodemann, A. Rieger, K. J. Bross, H. Schroter-Urban and G. W. Lohr, Erythrocyte and plasma ferritin in normal subjects, blood donors and iron deficiency anemia patients, Blut, 1984, 48, 131–137 CrossRef CAS.
- A. Piperno, M. Sampietro, M. T. Taddei and G. Fiorelli, Factors affecting erythrocyte ferritin content in thalassaemia intermedia, Br. J. Haematol., 1984, 56, 173–174 CrossRef CAS.
- S. W. Peters, S. J. May and A. Jacobs, Erythrocyte ferritin concentration in patients with myelodysplastic syndromes, J. Clin. Pathol., 1985, 38, 113–114 CrossRef CAS.
- M. K. Cruickshank, J. Ninness, A. Curtis, R. M. Barr, P. R. Flanagan, C. N. Ghent and L. S. Valberg, Usefulness of erythrocyte ferritin analysis in hereditary hemochromatosis, CMAJ, 1987, 136, 1259–1264 CAS.
- M. I. Oshtrakh and V. A. Semionkin, Mössbauer study of red blood cells from patients with erythremia, FEBS Lett., 1989, 257, 41–44 CrossRef CAS.
- E. R. Bauminger, E. Fibach, A. M. Konijn, S. Ofer and E. A. Rachmilewitz, Mössbauer studies of iron uptake, ferritin and hemoglobin synthesis and denaturation in erythroid cell cultures, Hyperfine Interact., 1991, 66, 11–23 CrossRef CAS.
- V. Christopoulou, A. Varsou, A. Travlou and G. Drivas, Erythrocyte ferritin in patients with chronic renal failure and heterozygous beta-thalassemia, Nephron, 2002, 91, 463–467 CrossRef CAS PubMed.
- C. Novembrino, A. Porcella, D. Conte, A. F. de Vecchi, G. Buccianti, S. Lonati, L. Duca, A. Ciani and F. Bamonti-Catena, Erythrocyte ferritin concentration: analytical performance of the immunoenzymatic IMx-Ferritin (Abbott) assay, Clin. Chem. Lab. Med., 2005, 43, 449–453 CrossRef CAS PubMed.
- H. Beving, L. E. G. Eriksson, C. L. Davey and D. B. Kell, Dielectric properties of human blood and erythrocytes at radio frequencies (0.2-10 MHz): dependence on medium composition, Eur. Biophys. J., 1994, 23, 207–215 CrossRef CAS.
- C. Cai, A. Ching, C. Lagace and T. Linsenmayer, Nuclear ferritin-mediated protection of corneal epithelial cells from oxidative damage to DNA, Dev. Dyn., 2008, 237, 2676–2683 CrossRef CAS PubMed.
- N. Surguladze, K. M. Thompson, J. L. Beard, J. R. Connor and M. G. Fried, Interactions and reactions of ferritin with DNA, J. Biol. Chem., 2004, 279, 14694–14702 CrossRef CAS PubMed.
- N. Surguladze, S. Patton, A. Cozzi, M. G. Fried and J. R. Connor, Characterization of nuclear ferritin and mechanism of translocation, Biochem. J., 2005, 388, 731–740 CrossRef CAS PubMed.
- M. V. Nurminskaya, C. J. Talbot, D. I. Nurminsky, K. E. Beazley and T. F. Linsenmayer, Nuclear ferritin: a ferritoid-ferritin complex in corneal epithelial cells, Invest. Ophthalmol. Visual Sci., 2009, 50, 3655–3661 Search PubMed.
- H. L. Storr, B. Kind, D. A. Parfitt, J. P. Chapple, M. Lorenz, K. Koehler, A. Huebner and A. J. Clark, Deficiency of ferritin heavy-chain nuclear import in triple A syndrome implies nuclear oxidative damage as the primary disease mechanism, Mol. Endocrinol., 2009, 23, 2086–2094 CrossRef CAS PubMed.
- A. A. Alkhateeb and J. R. Connor, Nuclear ferritin: A new role for ferritin in cell biology, Biochim. Biophys. Acta, 2010, 1800, 793–797 CrossRef CAS PubMed.
- P. Arosio and S. Levi, Cytosolic and mitochondrial ferritins in the regulation of cellular iron homeostasis and oxidative damage, Biochim. Biophys. Acta, 2010, 1800, 783–792 CrossRef CAS PubMed.
- A. Campanella, E. Rovelli, P. Santambrogio, A. Cozzi, F. Taroni and S. Levi, Mitochondrial ferritin limits oxidative damage regulating mitochondrial iron availability: hypothesis for a protective role in Friedreich ataxia, Hum. Mol. Genet., 2009, 18, 1–11 CrossRef CAS PubMed.
- W. S. Wu, Y. S. Zhao, Z. H. Shi, S. Y. Chang, G. J. Nie, X. L. Duan, S. M. Zhao, Q. Wu, Z. L. Yang, B. L. Zhao and Y. Z. Chang, Mitochondrial ferritin attenuates beta-amyloid-induced neurotoxicity: reduction in oxidative damage through the Erk/P38 mitogen-activated protein kinase pathways, Antioxid. Redox Signaling, 2013, 18, 158–169 CrossRef CAS PubMed.
- S. C. Andrews, The Ferritin-like superfamily: Evolution of the biological iron storeman from a rubrerythrin-like ancestor, Biochim. Biophys. Acta, 2010, 1800, 691–705 CrossRef CAS PubMed.
- K. Orino and K. Watanabe, Molecular, physiological and clinical aspects of the iron storage protein ferritin, Vet. J., 2008, 178, 191–201 CrossRef CAS PubMed.
- M. A. Knovich, J. A. Storey, L. G. Coffman, S. V. Torti and F. M. Torti, Ferritin for the clinician, Blood Rev., 2009, 23, 95–104 CrossRef CAS PubMed.
- W. Wang, M. A. Knovich, L. G. Coffman, F. M. Torti and S. V. Torti, Serum ferritin: Past, present and future, Biochim. Biophys. Acta, 2010, 1800, 760–769 CrossRef CAS PubMed.
- R. K. Watt, The many faces of the octahedral ferritin protein, Biometals, 2011, 24, 489–500 CrossRef CAS PubMed.
- E. C. Theil, Ferritin: The Protein Nanocage and Iron Biomineral in Health and in Disease, Inorg. Chem., 2013, 52, 12223–12233 CrossRef CAS PubMed.
- P. M. Harrison and P. Arosio, Ferritins: Molecular properties, iron storage function and cellular regulation, Biochim. Biophys. Acta, 1996, 1275, 161–203 CrossRef.
- P. Rucker, F. M. Torti and S. V. Torti, Role of H and L subunits in mouse ferritin, J. Biol. Chem., 1996, 271, 33352–33357 CrossRef CAS PubMed.
- E. C. Theil, Ferritin protein nanocages use ion channels, catalytic sites, and nucleation channels to manage iron/oxygen chemistry, Curr. Opin. Chem. Biol., 2011, 15, 304–311 CrossRef CAS PubMed.
- R. R. Crichton and J. P. Declercq, X-ray structures of ferritins and related proteins, Biochim. Biophys. Acta, 2010, 1800, 706–718 CrossRef CAS PubMed.
- D. J. E. Huard, K. M. Kane and F. A. Tezcan, Re-engineering protein interfaces yields copper-inducible ferritin cage assembly, Nat. Chem. Biol., 2013, 9, 169–176 CrossRef CAS PubMed.
- F. Bou-Abdallah, G. Zhao, G. Biasiotto, M. Poli, P. Arosio and N. D. Chasteen, Facilitated diffusion of iron(II) and dioxygen substrates into human H-chain ferritin. A fluorescence and absorbance study employing the ferroxidase center substitution Y34W, J. Am. Chem. Soc., 2008, 130, 17801–17811 CrossRef CAS PubMed.
- F. Bou-Abdallah, The iron redox and hydrolysis chemistry of the ferritins, Biochim. Biophys. Acta, 2010, 1800, 719–731 CrossRef CAS PubMed.
- T. Tosha, H. L. Ng, O. Bhattasali, T. Alber and E. C. Theil, Moving Metal Ions through Ferritin-Protein Nanocages from Three-Fold Pores to Catalytic Sites, J. Am. Chem. Soc., 2010, 132, 14562–14569 CrossRef CAS PubMed.
- P. Turano, D. Lalli, I. C. Felli, E. C. Theil and I. Bertini, NMR reveals pathway for ferric mineral precursors to the central cavity of ferritin, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 545–550 CrossRef CAS PubMed.
- I. Bertini, D. Lalli, S. Mangani, C. Pozzi, C. Rosa, E. C. Theil and P. Turano, Structural Insights into the Ferroxidase Site of Ferritins from Higher Eukaryotes, J. Am. Chem. Soc., 2012, 134, 6169–6176 CrossRef CAS PubMed.
- F. Carmona, Ò. Palacios, N. Gálvez, R. Cuesta, S. Atrian, M. Capdevila and J. M. Domínguez-Vera, Ferritin iron uptake and release in the presence of metals and metalloproteins: Chemical implications in the brain, Coord. Chem. Rev., 2013, 257, 2752–2764 CrossRef CAS PubMed.
- E. C. Theil, R. K. Behera and T. Tosha, Ferritins for Chemistry and for Life, Coord. Chem. Rev., 2013, 257, 579–586 CrossRef CAS PubMed.
- T. Tosha, R. K. Behera and E. C. Theil, Ferritin ion channel disorder inhibits Fe(II)/O2 reactivity at distant sites, Inorg. Chem., 2012, 51, 11406–11411 CrossRef CAS PubMed.
- R. K. Watt, A unified model for ferritin iron loading by the catalytic center: implications for controlling “free iron” during oxidative stress, ChemBioChem, 2013, 14, 415–419 CrossRef CAS PubMed.
- R. K. Watt, R. J. Hilton and D. M. Graff, Oxido-reduction is not the only mechanism allowing ions to traverse the ferritin protein shell, Biochim. Biophys. Acta, 2010, 1800, 745–759 CrossRef CAS PubMed.
- M. R. Hasan, T. Tosha and E. C. Theil, Ferritin Contains Less Iron (Fe-59) in Cells When the Protein Pores Are Unfolded by Mutation, J. Biol. Chem., 2008, 283, 31394–31400 CrossRef CAS PubMed.
- J. M. Domínguez-Vera, B. Fernández and N. Gálvez, Native and synthetic ferritins for nanobiomedical applications: recent advances and new perspectives, Future Med. Chem., 2010, 2, 609–618 CrossRef PubMed.
- G. Melman, F. Bou-Abdallah, E. Vane, P. Maura, P. Arosio and A. Melman, Iron release from ferritin by flavin nucleotides, Biochim. Biophys. Acta, 2013, 1830, 4669–4674 CrossRef CAS PubMed.
- F. Bou-Abdallah, J. McNally, X. X. Liu and A. Melman, Oxygen catalyzed mobilization of iron from ferritin by iron(III) chelate ligands, Chem. Commun., 2011, 47, 731–733 RSC.
- F. M. Torti and S. V. Torti, Regulation of ferritin genes and protein, Blood, 2002, 99, 3505–3516 CrossRef CAS PubMed.
- H. Yamanishi, S. Iyama, Y. Yamaguchi, Y. Kanakura and Y. Iwatani, Relation between iron content of serum ferritin and clinical status factors extracted by factor analysis in patients with hyperferritinemia, Clin. Biochem., 2002, 35, 523–529 CrossRef CAS.
- T. Konz, E. Añón Alvarez, M. Montes-Bayon and A. Sanz-Medel, Antibody labeling and elemental mass spectrometry (inductively coupled plasma-mass spectrometry) using isotope dilution for highly sensitive ferritin determination and iron-ferritin ratio measurements, Anal. Chem., 2013, 85, 8334–8340 CrossRef CAS PubMed.
- Y. H. Pan, K. Sader, J. J. Powell, A. Bleloch, M. Gass, J. Trinick, A. Warley, A. Li, R. Brydson and A. Brown, 3D morphology of the human hepatic ferritin mineral core: new evidence for a subunit structure revealed by single particle analysis of HAADF-STEM images, J. Struct. Biol., 2009, 166, 22–31 CrossRef CAS PubMed.
- J. Dobson, Magnetic iron compounds in neurological disorders, Ann. N. Y. Acad. Sci., 2004, 1012, 183–192 CrossRef CAS PubMed.
- J. Gałązka-Friedman, Iron as a risk factor in neurological diseases, Hyperfine Interact., 2008, 182, 31–44 CrossRef.
- D. G. Meyers, The iron hypothesis – does iron cause atherosclerosis?, Clin. Cardiol., 1996, 19, 925–929 CrossRef CAS.
- P. Arosio, M. Yokota and J. W. Drysdale, Characterization of Serum Ferritin in Iron Overload – Possible Identity to Natural Apoferritin, Br. J. Haematol., 1977, 36, 199–207 CrossRef CAS.
- M. Rudeck, T. Volk, N. Sitte and T. Grune, Ferritin oxidation in vitro: implication of iron release and degradation by the 20S proteasome, IUBMB Life, 2000, 49, 451–456 CrossRef CAS PubMed.
- T. Z. Kidane, E. Sauble and M. C. Linder, Release of iron from ferritin requires lysosomal activity, Am. J. Physiol., 2006, 291, C445–C455 CrossRef CAS PubMed.
- Y. Zhang, M. Mikhael, D. Xu, Y. Li, S. Soe-Lin, B. Ning, W. Li, G. Nie, Y. Zhao and P. Ponka, Lysosomal proteolysis is the primary degradation pathway for cytosolic ferritin and cytosolic ferritin degradation is necessary for iron exit, Antioxid. Redox Signaling, 2010, 13, 999–1009 CrossRef CAS PubMed.
- T. Asano, M. Komatsu, Y. Yamaguchi-Iwai, F. Ishikawa, N. Mizushima and K. Iwai, Distinct mechanisms of ferritin delivery to lysosomes in iron-depleted and iron-replete cells, Mol. Cell. Biol., 2011, 31, 2040–2052 CrossRef CAS PubMed.
- T. C. Iancu, Ultrastructural aspects of iron storage, transport and metabolism, J. Neural Transm., 2011, 118, 329–335 CrossRef CAS PubMed.
- A. Terman and T. Kurz, Lysosomal Iron, Iron Chelation, and Cell Death, Antioxid. Redox Signaling, 2013, 18, 888–898 CrossRef CAS PubMed.
- R. Vidal, L. Miravalle, X. Gao, A. G. Barbeito, M. A. Baraibar, S. K. Hekmatyar, M. Widel, N. Bansal, M. B. Delisle and B. Ghetti, Expression of a mutant form of the ferritin light chain gene induces neurodegeneration and iron overload in transgenic mice, J. Neurosci., 2008, 28, 60–67 CrossRef CAS PubMed.
- E. Miyazaki, J. Kato, M. Kobune, K. Okumura, K. Sasaki, N. Shintani, P. Arosio and Y. Niitsu, Denatured H-ferritin subunit is a major constituent of haemosiderin in the liver of patients with iron overload, Gut, 2002, 50, 413–419 CrossRef CAS.
- P. Zamboni, M. Izzo, L. Fogato, S. Carandina and V. Lanzara, Urine hemosiderin: a novel marker to assess the severity of chronic venous disease, J. Vasc. Surg., 2003, 37, 132–136 CrossRef PubMed.
- C. Quintana, S. Bellefqih, J. Y. Laval, J. L. Guerquin-Kern, T. D. Wu, J. Avila, I. Ferrer, R. Arranz and C. Patino, Study of the localization of iron, ferritin, and hemosiderin in Alzheimer's disease hippocampus by analytical microscopy at the subcellular level, J. Struct. Biol., 2006, 153, 42–54 CrossRef CAS PubMed.
- C. Quintana, About the presence of hemosiderin in the hippocampus of Alzheimer patients, J. Alzheimer's Dis., 2007, 12, 157–160 CAS.
- P. Zamboni, S. Lanzara, F. Mascoli, A. Caggiati and A. Liboni, Inflammation in venous disease, Int. Angiol., 2008, 27, 361–369 CAS.
- F. Maldonado, J. G. Parambil, E. S. Yi, P. A. Decker and J. H. Ryu, Haemosiderin-laden macrophages in the bronchoalveolar lavage fluid of patients with diffuse alveolar damage, Eur. Respir. J., 2009, 33, 1361–1366 CrossRef CAS PubMed.
- H. L. Persson and L. K. Vainikka, Lysosomal iron in pulmonary alveolar proteinosis: a case report, Eur. Respir. J., 2009, 33, 673–679 CrossRef CAS PubMed.
- N. Sakalihasan and J. B. Michel, Functional imaging of atherosclerosis to advance vascular biology, Eur. J. Vasc. Endovasc. Surg., 2009, 37, 728–734 CrossRef CAS PubMed.
- A. Terman and U. T. Brunk, Lipofuscin, Int. J. Biochem. Cell Biol., 2004, 36, 1400–1404 CrossRef CAS PubMed.
- T. Jung, N. Bader and T. Grune, Lipofuscin: formation, distribution, and metabolic consequences, Ann. N. Y. Acad. Sci., 2007, 1119, 97–111 CrossRef CAS PubMed.
- K. L. Double, V. N. Dedov, H. Fedorow, E. Kettle, G. M. Halliday, B. Garner and U. T. Brunk, The comparative biology of neuromelanin and lipofuscin in the human brain, Cell. Mol. Life Sci., 2008, 65, 1669–1682 CrossRef CAS PubMed.
- A. Höhn, T. Jung, S. Grimm and T. Grune, Lipofuscin-bound iron is a major intracellular source of oxidants: role in senescent cells, Free Radical Biol. Med., 2010, 48, 1100–1108 CrossRef PubMed.
- M. Gerlach, A. X. Trautwein, L. Zecca, M. B. H. Youdim and P. Riederer, Mössbauer Spectroscopic Studies of Purified Human Neuromelanin Isolated from the Substantia-Nigra, J. Neurochem., 1995, 65, 923–926 CrossRef CAS.
- K. L. Double, M. Gerlach, V. Schunemann, A. X. Trautwein, L. Zecca, M. Gallorini, M. B. Youdim, P. Riederer and D. Ben-Shachar, Iron-binding characteristics of neuromelanin of the human substantia nigra, Biochem. Pharmacol., 2003, 66, 489–494 CrossRef CAS.
- M. Gerlach, K. L. Double, D. Ben-Shachar, L. Zecca, M. B. Youdim and P. Riederer, Neuromelanin and its interaction with iron as a potential risk factor for dopaminergic neurodegeneration underlying Parkinson's disease, Neurotoxic Res., 2003, 5, 35–44 CrossRef.
- L. Zecca, M. Gallorini, V. Schunemann, A. X. Trautwein, M. Gerlach, P. Riederer, P. Vezzoni and D. Tampellini, Iron, neuromelanin and ferritin content in the substantia nigra of normal subjects at different ages: consequences for iron storage and neurodegenerative processes, J. Neurochem., 2001, 76, 1766–1773 CrossRef CAS.
- S. Bohic, K. Murphy, W. Paulus, P. Cloetens, M. Salome, J. Susini and K. Double, Intracellular Chemical Imaging of the Developmental Phases of Human Neuromelanin Using Synchrotron X-ray Microspectroscopy, Anal. Chem., 2008, 80, 9557–9566 CrossRef CAS PubMed.
- F. Tribl, E. Asan, T. Arzberger, T. Tatschner, E. Langenfeld, H. E. Meyer, G. Bringmann, P. Riederer, M. Gerlach and K. Marcus, Identification of L-ferritin in neuromelanin granules of the human substantia nigra: a targeted proteomics approach, Mol. Cell. Proteomics, 2009, 8, 1832–1838 CAS.
- H. Saito, A. Tomita, H. Ohashi, H. Maeda, H. Hayashi and T. Naoe, Determination of ferritin and hemosiderin iron in patients with normal iron stores and iron overload by serum ferritin kinetics, Nagoya J. Med. Sci., 2012, 74, 39–49 CAS.
- H. Saito, H. Hayashi, A. Tomita, H. Ohashi, H. Maeda and T. Naoe, Increasing and Decreasing Phases of Ferritin and Hemosiderin Iron Determined by Serum Ferritin Kinetics, Nagoya J. Med. Sci., 2013, 75, 213–223 Search PubMed.
- G. Ricolleau, C. Charbonnel, L. Lode, D. Loussouarn, M. P. Joalland, R. Bogumil, S. Jourdain, S. Minvielle, M. Campone, R. Déporte-Fety, L. Campion and P. Jézéquel, Surface-enhanced laser desorption/ionization time of flight mass spectrometry protein profiling identifies ubiquitin and ferritin light chain as prognostic biomarkers in node-negative breast cancer tumors, Proteomics, 2006, 6, 1963–1975 CrossRef CAS PubMed.
- M. E. del Castillo Busto, M. Montes-Bayón and A. Sanz-Medel, The potential of mass spectrometry to study iron-containing proteins used in clinical diagnosis, Anal. Chim. Acta, 2009, 634, 1–14 CrossRef PubMed.
- M. Hoppler, C. Zeder and T. Walczyk, Quantification of Ferritin-Bound Iron in Plant Samples by Isotope Tagging and Species-Specific Isotope Dilution Mass Spectrometry, Anal. Chem., 2009, 81, 7368–7372 CrossRef CAS PubMed.
- H. Q. Huang, X. H. Hu, X. P. Fang, T. M. Cao and B. Kong, Characteristics of H and L Subunits with Mass Spectrometry, Electrophoresis and Transmission Electron Microscopy in Liver Ferritin of Dasyatis Akajei, Chin. J. Anal. Chem., 2009, 37, 631–636 CAS.
- P. Nielsen, U. Günther, M. Dürken, R. Fischer and J. Düllmann, Serum ferritin iron in iron overload and liver damage: Correlation to body iron stores and diagnostic relevance, J. Lab. Clin. Med., 2000, 135, 413–418 CrossRef CAS PubMed.
- K. Watanabe, Y. Yamashita, K. Ohgawara, M. Sekiguchi, N. Satake, K. Orino and S. Yamamoto, Iron content of rat serum ferritin, J. Vet. Med. Sci., 2001, 63, 587–589 CrossRef CAS.
- N. Bresgen, H. Jaksch, H. Lacher, I. Ohlenschlager, K. Uchida and P. M. Eckl, Iron-mediated oxidative stress plays an essential role in ferritin-induced cell death, Free Radical Biol. Med., 2010, 48, 1347–1357 CrossRef CAS PubMed.
- A. A. Alkhateeb, B. Han and J. R. Connor, Ferritin stimulates breast cancer cells through an iron-independent mechanism and is localized within tumor-associated macrophages, Breast Cancer Res. Treat., 2013, 137, 733–744 CrossRef CAS PubMed.
- T. Kurz, B. Gustafsson and U. T. Brunk, Cell sensitivity to oxidative stress is influenced by ferritin autophagy, Free Radical Biol. Med., 2011, 50, 1647–1658 CrossRef CAS PubMed.
- B. Garner, K. Roberg and U. T. Brunk, Endogenous ferritin protects cells with iron-laden lysosomes against oxidative stress, Free Radical Res., 1998, 29, 103–114 CrossRef CAS.
- B. Garner, W. Li, K. Roberg and U. T. Brunk, On the cytoprotective role of ferritin in macrophages and its ability to enhance lysosomal stability, Free Radical Res., 1997, 27, 487–500 CrossRef CAS.
- H. L. Persson, K. J. Nilsson and U. T. Brunk, Novel cellular defenses against iron and oxidation: ferritin and autophagocytosis preserve lysosomal stability in airway epithelium, Redox Rep., 2001, 6, 57–63 CrossRef CAS PubMed.
- T. Kurz, J. W. Eaton and U. T. Brunk, The role of lysosomes in iron metabolism and recycling, Int. J. Biochem. Cell Biol., 2011, 43, 1686–1697 CrossRef CAS PubMed.
- C. Ferreira, D. Bucchini, M. E. Martin, S. Levi, P. Arosio, B. Grandchamp and C. Beaumont, Early embryonic lethality of H ferritin gene deletion in mice, J. Biol. Chem., 2000, 275, 3021–3024 CrossRef CAS PubMed.
- C. Ferreira, P. Santambrogio, M. E. Martin, V. Andrieu, G. Feldmann, D. Henin and C. Beaumont, H ferritin knockout mice: a model of hyperferritinemia in the absence of iron overload, Blood, 2001, 98, 525–532 CrossRef CAS PubMed.
- J. t. Wilkinson, X. Di, K. Schönig, J. L. Buss, N. D. Kock, J. M. Cline, T. L. Saunders, H. Bujard, S. V. Torti and F. M. Torti, Tissue-specific expression of ferritin H regulates cellular iron homoeostasis in vivo, Biochem. J., 2006, 395, 501–507 CrossRef CAS PubMed.
- G. Hetet, I. Devaux, N. Soufir, B. Grandchamp and C. Beaumont, Molecular analyses of patients with hyperferritinemia and normal serum iron values reveal both L ferritin IRE and 3 new ferroportin (SLC11A3) mutations, Blood, 2003, 102, 1904–1910 CrossRef CAS PubMed.
- K. P. Burdon, S. Sharma, C. S. Chen, D. P. Dimasi, D. A. Mackey and J. E. Craig, A novel deletion in the FTL gene causes hereditary hyperferritinemia cataract syndrome (HHCS) by alteration of the transcription start site, Hum. Mutat., 2007, 28, 742 CrossRef PubMed.
- C. Kannengiesser, A. M. Jouanolle, G. Hetet, A. Mosser, F. Muzeau, D. Henry, E. Bardou-Jacquet, M. Mornet, P. Brissot, Y. Deugnier, B. Grandchamp and C. Beaumont, A new missense mutation in the L ferritin coding sequence associated with elevated levels of glycosylated ferritin in serum and absence of iron overload, Haematologica, 2009, 94, 335–339 CrossRef CAS PubMed.
- J. Álvarez-Coca-Gonzalez, M. I. Moreno-Carralero, J. Martinez-Pérez, M. Méndez, M. García-Ros and M. J. Morán-Jiménez, The hereditary hyperferritinemia-cataract syndrome: a family study, Eur. J. Pediatr., 2010, 169, 1553–1555 CrossRef PubMed.
- C. Beaumont, Miscellaneous Iron-Related Disorders, in Iron Physiology and Pathophysiology in Humans, ed. G. J. Anderson and G. D. McLaren, 2012, pp. 417–439 Search PubMed.
- S. Luscieti, G. Tolle, J. Aranda, C. B. Campos, F. Risse, É. Morán, M. U. Muckenthaler and M. Sánchez, Novel mutations in the ferritin-L iron-responsive element that only mildly impair IRP binding cause hereditary hyperferritinaemia cataract syndrome, Orphanet. J. Rare Dis., 2013, 8, 30 CrossRef PubMed.
- G. A. Agasthya, B. C. Harrawood, J. P. Shah and A. J. Kapadia, Sensitivity analysis for liver iron measurement through neutron stimulated emission computed tomography: a Monte Carlo study in GEANT4, Phys. Med. Biol., 2012, 57, 113–126 CrossRef CAS PubMed.
- P. Nielsen, R. Engelhardt, J. Dullmann and R. Fischer, Non-invasive liver iron quantification by SQUID-biosusceptometry and serum ferritin iron as new diagnostic parameters in hereditary hemochromatosis, Blood Cells, Mol., Dis., 2002, 29, 451–458 CrossRef CAS.
- A. Castiella, J. M. Alústiza, J. I. Emparanza, E. M. Zapata, B. Costero and M. I. Díez, Liver iron concentration quantification by MRI: are recommended protocols accurate enough for clinical practice?, Eur. Radiol., 2010, 21, 137–141 CrossRef PubMed.
- M. I. Argyropoulou and L. Astrakas, MRI evaluation of tissue iron burden in patients with beta-thalassaemia major, Pediatr. Radiol., 2007, 37, 1191–1200 CrossRef PubMed ; quiz 1308-1199.
- O. Dereure, N. Jumez, D. Bessis, B. Gallix and B. Guillot, Measurement of liver iron content by magnetic resonance imaging in 20 patients with overt porphyria cutanea tarda before phlebotomy therapy: a prospective study, Acta Derm.-Venereol., 2008, 88, 341–345 Search PubMed.
- K. M. Musallam, M. D. Cappellini, J. C. Wood, I. Motta, G. Graziadei, H. Tamim and A. T. Taher, Elevated liver iron concentration is a marker of increased morbidity in patients with beta thalassemia intermedia, Haematologica, 2011, 96, 1605–1612 CrossRef CAS PubMed.
- K. M. Musallam, M. D. Cappellini and A. T. Taher, Evaluation of the 5mg/g liver iron concentration threshold and its association with morbidity in patients with beta-thalassemia intermedia, Blood Cells, Mol., Dis., 2013, 51, 35–38 CrossRef CAS PubMed.
- V. Positano, B. Salani, A. Pepe, M. F. Santarelli, D. De Marchi, A. Ramazzotti, B. Favilli, E. Cracolici, M. Midiri, P. Cianciulli, M. Lombardi and L. Landini, Improved T2* assessment in liver iron overload by magnetic resonance imaging, Magn. Reson. Imaging, 2009, 27, 188–197 CrossRef PubMed.
- C. Rose, P. Vandevenne, E. Bourgeois, N. Cambier and O. Ernst, Liver iron content assessment by routine and simple magnetic resonance imaging procedure in highly transfused patients, Eur. J. Haematol., 2006, 77, 145–149 CrossRef CAS PubMed.
- K. Tziomalos and V. Perifanis, Liver iron content determination by magnetic resonance imaging, World J. Gastroenterol., 2010, 16, 1587–1597 CrossRef CAS.
- S. Tony, S. Daar, M. Elshinawy, S. Al-Zadjaly, M. Al-Khabori and Y. Wali, T2* MRI in regularly transfused children with thalassemia intermedia: serum ferritin does not reflect liver iron stores, Pediatr. Hematol. Oncol., 2012, 29, 579–584 CrossRef CAS PubMed.
- K. Ziv, G. Meir, A. Harmelin, E. Shimoni, E. Klein and M. Neeman, Ferritin as a reporter gene for MRI: chronic liver over expression of H-ferritin during dietary iron supplementation and aging, NMR Biomed., 2010, 23, 523–531 CrossRef CAS PubMed.
- E. M. Haacke, N. Y. Cheng, M. J. House, Q. Liu, J. Neelavalli, R. J. Ogg, A. Khan, M. Ayaz, W. Kirsch and A. Obenaus, Imaging iron stores in the brain using magnetic resonance imaging, Magn. Reson. Imaging, 2005, 23, 1–25 CrossRef CAS PubMed.
- W. Kirsch, G. McAuley, B. Holshouser, F. Petersen, M. Ayaz, H. V. Vinters, C. Dickson, E. M. Haacke, W. Britt Iii, J. Larsen, I. Kim, C. Mueller, M. Schrag and D. Kido, Serial susceptibility weighted MRI measures brain iron and microbleeds in dementia, J. Alzheimer's Dis., 2009, 17, 599–609 CAS.
- W. Zheng, H. Nichol, S. Liu, Y. C. Cheng and E. M. Haacke, Measuring iron in the brain using quantitative susceptibility mapping and X-ray fluorescence imaging, NeuroImage, 2013, 78C, 68–74 CrossRef PubMed.
- M. J. Kim, D. G. Mitchell, K. Ito, H. W. Hann, Y. N. Park and P. N. Kim, Hepatic iron deposition on MR imaging in patients with chronic liver disease: correlation with serial serum ferritin concentration, Abdom. Imaging, 2001, 26, 149–156 CrossRef CAS.
- A. W. Olthof, P. E. Sijens, H. G. Kreeftenberg, P. Kappert, R. Irwan, E. J. van der Jagt and M. Oudkerk, Correlation between serum ferritin levels and liver iron concentration determined by MR imaging: impact of hematologic disease and inflammation, Magn. Reson. Imaging, 2007, 25, 228–231 CrossRef CAS PubMed.
- O. G. Papakonstantinou, T. G. Maris, V. Kostaridou, A. D. Gouliamos, G. K. Koutoulas, A. E. Kalovidouris, G. B. Papavassiliou, G. Kordas, C. Kattamis and L. J. Vlahos, et al., Assessment of liver iron overload by T2-quantitative magnetic resonance imaging: correlation of T2-QMRI measurements with serum ferritin concentration and histologic grading of siderosis, Magn. Reson. Imaging, 1995, 13, 967–977 CrossRef CAS.
- P. D. Jensen, F. T. Jensen, T. Christensen and J. Ellegaard, Evaluation of transfusional iron overload before and during iron chelation by magnetic resonance imaging of the liver and determination of serum ferritin in adult non-thalassaemic patients, Br. J. Haematol., 1995, 89, 880–889 CrossRef CAS.
- P. D. Jensen, F. T. Jensen, T. Christensen, H. Eiskjaer, U. Baandrup and J. L. Nielsen, Evaluation of myocardial iron by magnetic resonance imaging during iron chelation therapy with deferrioxamine: indication of close relation between myocardial iron content and chelatable iron pool, Blood, 2003, 101, 4632–4639 CrossRef CAS PubMed.
- P. D. Jensen, F. T. Jensen, T. Christensen, J. L. Nielsen and J. Ellegaard, Relationship between hepatocellular injury and transfusional iron overload prior to and during iron chelation with desferrioxamine: a study in adult patients with acquired anemias, Blood, 2003, 101, 91–96 CrossRef CAS PubMed.
- Z. Pakbaz, R. Fischer, E. Fung, P. Nielsen, P. Harmatz and E. Vichinsky, Serum ferritin underestimates liver iron concentration in transfusion independent thalassemia patients as compared to regularly transfused thalassemia and sickle cell patients, Pediatr. Blood Cancer, 2007, 49, 329–332 Search PubMed.
- A. Taher, F. El Rassi, H. Isma'eel, S. Koussa, A. Inati and M. D. Cappellini, Correlation of liver iron concentration determined by R2 magnetic resonance imaging with serum ferritin in patients with thalassemia intermedia, Haematologica, 2008, 93, 1584–1586 CrossRef CAS PubMed.
- A. Kolnagou, K. Natsiopoulos, M. Kleanthous, A. Ioannou and G. J. Kontoghiorghes, Liver iron and serum ferritin levels are misleading for estimating cardiac, pancreatic, splenic and total body iron load in thalassemia patients: factors influencing the heterogenic distribution of excess storage iron in organs as identified by MRI T2*, Toxicol. Mech. Methods, 2013, 23, 48–56 CrossRef CAS PubMed.
- D. A. Tsitsikas, R. Nzouakou, V. Ameen, B. Sirigireddy and R. J. Amos, Comparison of Serial Serum Ferritin Measurements and Liver Iron Concentration Assessed by MRI in Adult Transfused Patients with Sickle Cell Disease, Eur. J. Haematol., 2014, 92, 164–167 CrossRef CAS PubMed.
- B. J. Ferguson, B. S. Skikne, K. M. Simpson, R. D. Baynes and J. D. Cook, Serum Transferrin Receptor Distinguishes the Anemia of Chronic Disease from Iron-Deficiency Anemia, J. Lab. Clin. Med., 1992, 119, 385–390 CAS.
- K. Punnonen, K. Irjala and A. Rajamäki, Serum transferrin receptor and its ratio to serum ferritin in the diagnosis of iron deficiency, Blood, 1997, 89, 1052–1057 CAS.
- J. D. Cook, Diagnosis and management of iron-deficiency anaemia, Best Pract. Res., Clin. Haematol., 2005, 18, 319–332 CrossRef CAS PubMed.
- E. Hanif, M. Ayyub, M. Anwar, W. Ali and M. Bashir, Evaluation of serum transferrin receptor concentration in diagnosing and differentiating iron deficiency anaemia from anaemia of chronic disorders, J. Pak. Med. Assoc., 2005, 55, 13–16 Search PubMed.
- E. Joosten, R. Van Loon, J. Billen, N. Blanckaert, R. Fabri and W. Pelemans, Serum transferrin receptor in the evaluation of the iron status in elderly hospitalized patients with anemia, Am. J. Hematol., 2002, 69, 1–6 CrossRef CAS PubMed.
- A. Skoumalová and J. Hort, Blood markers of oxidative stress in Alzheimer’s disease, J. Cell. Mol. Med., 2012, 16, 2291–2300 CrossRef PubMed.
- K. Henriksen, S. E. O’Bryant, H. Hampel, J. Q. Trojanowski, T. J. Montine, A. Jeromin, K. Blennow, A. Lönneborg, T. Wyss-Coray, H. Soares, C. Bazenet, M. Sjögren, W. Hu, S. Lovestone, M. A. Karsdal and M. W. Weiner, The future of blood-based biomarkers for Alzheimer’s disease, Alzheimer's Dementia, 2014, 10, 115–131 CrossRef PubMed.
- M. Schrag, C. Mueller, M. Zabel, A. Crofton, W. M. Kirsch, O. Ghribi, R. Squitti and G. Perry, Oxidative stress in blood in Alzheimer’s disease and mild cognitive impairment: a meta-analysis, Neurobiol. Dis., 2013, 59, 100–110 CrossRef CAS PubMed.
- K. Ikeda, Y. Nakamura, T. Kiyozuka, J. Aoyagi, T. Hirayama, R. Nagata, H. Ito, K. Iwamoto, K. Murata, Y. Yoshii, K. Kawabe and Y. Iwasaki, Serological profiles of urate, paraoxonase-1, ferritin and lipid in Parkinson’s disease: changes linked to disease progression, Neurodegener. Dis., 2011, 8, 252–258 CrossRef CAS PubMed.
- K. G. Connelly, M. Moss, P. E. Parsons, E. E. Moore, F. A. Moore, P. C. Giclas, P. A. Seligman and J. E. Repine, Serum ferritin as a predictor of the acute respiratory distress syndrome, Am. J. Respir. Crit. Care Med., 1997, 155, 21–25 CrossRef CAS PubMed.
- R. A. Sharkey, S. C. Donnelly, K. G. Connelly, C. E. Robertson, C. Haslett and J. E. Repine, Initial serum ferritin levels in patients with multiple trauma and the subsequent development of acute respiratory distress syndrome, Am. J. Respir. Crit. Care Med., 1999, 159, 1506–1509 CrossRef CAS PubMed.
- A. L. Lagan, G. J. Quinlan, S. Mumby, D. D. Melley, P. Goldstraw, G. J. Bellingan, M. R. Hill, D. Briggs, P. Pantelidis, R. M. du Bois, K. I. Welsh and T. W. Evans, Variation in iron homeostasis genes between patients with ARDS and healthy control subjects, Chest, 2008, 133, 1302–1311 CrossRef CAS PubMed.
- Y. Y. Park, Ischemia/reperfusion Lung Injury Increases Serum Ferritin and Heme Oxygenase-1 in Rats, Korean J. Physiol. Pharmacol., 2009, 13, 181–187 CrossRef CAS PubMed.
- E. F. Goodall, M. S. Haque and K. E. Morrison, Increased serum ferritin levels in amyotrophic lateral sclerosis (ALS) patients, J. Neurol., 2008, 255, 1652–1656 CrossRef CAS PubMed.
- M. Qureshi, R. H. Brown Jr., J. T. Rogers and M. E. Cudkowicz, Serum ferritin and metal levels as risk factors for amyotrophic lateral sclerosis, Open Neurol. J., 2008, 2, 51–54 CrossRef CAS PubMed.
- K. Ikeda, T. Hirayama, T. Takazawa, K. Kawabe and Y. Iwasaki, Relationships between Disease Progression and Serum Levels of Lipid, Urate, Creatinine and Ferritin in Japanese Patients with Amyotrophic Lateral Sclerosis: A Cross-Sectional Study, Intern. Med., 2012, 51, 1501–1508 CrossRef CAS.
- Y. Nadjar, P. Gordon, P. Corcia, G. Bensimon, L. Pieroni, V. Meininger and F. Salachas, Elevated serum ferritin is associated with reduced survival in amyotrophic lateral sclerosis, PLoS One, 2012, 7, e45034 CAS.
- X. W. Su, Z. Simmons, R. M. Mitchell, L. Kong, H. E. Stephens and J. R. Connor, Biomarker-Based Predictive Models for Prognosis in Amyotrophic Lateral Sclerosis, JAMA Neurol., 2013, 70, 1505–1511 Search PubMed.
- S. Kiechl, J. Willeit, G. Egger, W. Poewe and F. Oberhollenzer, Body iron stores and the risk of carotid atherosclerosis: prospective results from the Bruneck study, Circulation, 1997, 96, 3300–3307 CrossRef CAS.
- S. A. You, S. R. Archacki, G. Angheloiu, C. S. Moravec, S. Rao, M. Kinter, E. J. Topol and Q. Wang, Proteomic approach to coronary atherosclerosis shows ferritin light chain as a significant marker: evidence consistent with iron hypothesis in atherosclerosis, Physiol. Genomics, 2003, 13, 25–30 CAS.
- B. Wolff, H. Volzke, J. Ludemann, D. Robinson, D. Vogelgesang, A. Staudt, C. Kessler, J. B. Dahm, U. John and S. B. Felix, Association between high serum ferritin levels and carotid atherosclerosis in the study of health in Pomerania (SHIP), Stroke, 2004, 35, 453–457 CrossRef CAS PubMed.
- K. A. Reis, G. Guz, H. Ozdemir, Y. Erten, V. Atalay, Z. Bicik, Z. N. Ozkurt, M. Bali and S. Sindel, Intravenous iron therapy as a possible risk factor for atherosclerosis in end-stage renal disease, Int. Heart J., 2005, 46, 255–264 CrossRef CAS.
- S. A. You and Q. Wang, Ferritin in atherosclerosis, Clin. Chim. Acta, 2005, 357, 1–16 CrossRef CAS PubMed.
- J. J. M. Marx, A. E. R. Kartikasari and N. A. Georgiou, Can iron chelators influence the progression of atherosclerosis?, Hemoglobin, 2008, 32, 123–134 CrossRef CAS PubMed.
- J. L. Sullivan, Iron in arterial plaque: A modifiable risk factor for atherosclerosis, Biochim. Biophys. Acta, 2009, 1790, 718–723 CrossRef CAS PubMed.
- N. Ahluwalia, A. Genoux, J. Ferrieres, B. Perret, M. Carayol, L. Drouet and J. B. Ruidavets, Iron status is associated with carotid atherosclerotic plaques in middle-aged adults, J. Nutr., 2010, 140, 812–816 CrossRef CAS PubMed.
- R. G. DePalma, V. W. Hayes, B. K. Chow, G. Shamayeva, P. E. May and L. R. Zacharski, Ferritin levels, inflammatory biomarkers, and mortality in peripheral arterial disease: A substudy of the Iron (Fe) and Atherosclerosis Study (FeAST) Trial, J. Vasc. Surg., 2010, 51, 1498–1503 CrossRef PubMed.
- P. Syrovatka, P. Kraml, K. Hulikova, L. Fialova, M. Vejrazka, J. Crkovska, J. Potockova and M. Andel, Iron stores are associated with asymptomatic atherosclerosis in healthy men of primary prevention, Eur. J. Clin. Invest., 2011, 41, 846–853 CrossRef CAS PubMed.
- L. R. Zacharski, R. G. Depalma, G. Shamayeva and B. K. Chow, The Statin-Iron Nexus: Anti-Inflammatory Intervention for Arterial Disease Prevention, Am. J. Public Health, 2013, 103, e105–e112 CrossRef PubMed.
- J. T. Hazard and J. W. Drysdale, Ferritinaemia in cancer, Nature, 1977, 265, 755–756 CrossRef CAS.
- B. M. Jones, M. Worwood and A. Jacobs, Serum ferritin in patients with cancer: determination with antibodies to HeLa cell and spleen ferritin, Clin. Chim. Acta, 1980, 106, 203–214 CrossRef CAS.
- A. Jacobs, Serum ferritin and malignant tumours, Med. Oncol. Tumor Pharmacother., 1984, 1, 149–156 CAS.
- J. H. Silber, A. E. Evans and M. Fridman, Models to Predict Outcome from Childhood Neuroblastoma – the Role of Serum Ferritin and Tumor Histology, Cancer Res., 1991, 51, 1426–1433 CAS.
- R. L. Nelson, F. G. Davis, E. Sutter, L. H. Sobin, J. W. Kikendall and P. Bowen, Body iron stores and risk of colonic neoplasia, J. Natl. Cancer Inst., 1994, 86, 455–460 CrossRef CAS PubMed.
- Z. Kırkalı, M. Güzelsoy, M. U. Mungan, G. Kırkalı and K. Yörükoğlu, Serum ferritin as a clinical marker for renal cell carcinoma: influence of tumor size and volume, Urol. Int., 1999, 62, 21–25 CrossRef PubMed.
- S. Hercberg, C. Estaquio, S. Czernichow, L. Mennen, N. Noisette, S. Bertrais, J. C. Renversez, S. Briancon, A. Favier and P. Galan, Iron status and risk of cancers in the SU.VI.MAX cohort, J. Nutr., 2005, 135, 2664–2668 CAS.
- L. R. Zacharski, B. K. Chow, P. S. Howes, G. Shamayeva, J. A. Baron, R. L. Dalman, D. J. Malenka, C. K. Ozaki and P. W. Lavori, Decreased cancer risk after iron reduction in patients with peripheral arterial disease: results from a randomized trial, J. Natl. Cancer Inst., 2008, 100, 996–1002 CrossRef CAS PubMed.
- K. H. Zhang, H. Y. Tian, X. Gao, W. W. Lei, Y. Hu, D. M. Wang, X. C. Pan, M. L. Yu, G. J. Xu, F. K. Zhao and J. G. Song, Ferritin heavy chain-mediated iron homeostasis and subsequent increased reactive oxygen species production are essential for epithelial-mesenchymal transition, Cancer Res., 2009, 69, 5340–5348 CrossRef CAS PubMed.
- A. A. Alkhateeb, K. Leitzel, S. M. Ali, C. Campbell-Baird, M. Evans, E. M. Fuchs, W. J. Köstler, A. Lipton and J. Connor, Elevation in inflammatory serum biomarkers predicts response to trastuzumab-containing therapy, PLoS One, 2012, 7, e51379 CAS.
- A. A. Alkhateeb and J. R. Connor, The significance of ferritin in cancer: Anti-oxidation, inflammation and tumorigenesis, Biochim. Biophys. Acta, 2013, 1836, 245–254 CAS.
- A. Amid, N. Barrowman, A. Vijenthira, P. Lesser, K. Mandel and R. Ramphal, Risk factors for hyperferritinemia secondary to red blood cell transfusions in pediatric cancer patients, Pediatr. Blood Cancer, 2013, 60, 1671–1675 Search PubMed.
- A. Alkhateeb, L. Zubritsky, B. Kinsman, K. Leitzel, C. Campbell-Baird, S. M. Ali, J. Connor and A. Lipton, Elevation in Multiple Serum Inflammatory Biomarkers Predicts Survival of Pancreatic Cancer Patients with Inoperable Disease, J. Gastrointest. Cancer, 2014 DOI:10.1007/12029-013-9564-9.
- R. Orlandi, M. De Bortoli, C. M. Ciniselli, E. Vaghi, D. Caccia, V. Garrisi, S. Pizzamiglio, S. Veneroni, C. Bonini, R. Agresti, M. G. Daidone, D. Morelli, C. Camaschella, P. Verderio and I. Bongarzone, Hepcidin and ferritin blood level as noninvasive tools for predicting breast cancer, Ann. Oncol., 2014 DOI:10.1093/annonc/mdt490.
- K. Jurczyk, M. Wawrzynowicz-Syczewska, A. Boroń-Kaczmarska and Z. Sych, Serum iron parameters in patients with alcoholic and chronic cirrhosis and hepatitis, Med. Sci. Monit., 2001, 7, 962–965 CAS.
- D. H. G. Crawford, T. L. Murphy, L. E. Ramm, L. M. Fletcher, A. D. Clouston, G. J. Anderson, V. N. Subramaniam, L. W. Powell and G. A. Ramm, Serum Hyaluronic Acid with Serum Ferritin Accurately Predicts Cirrhosis and Reduces the Need for Liver Biopsy in C282Y Hemochromatosis, Hepatology, 2009, 49, 418–425 CrossRef CAS PubMed.
- T. C. H. Tan, D. H. Crawford, M. E. Franklin, L. A. Jaskowski, G. A. Macdonald, J. R. Jonsson, M. J. Watson, P. J. Taylor and L. M. Fletcher, The serum hepcidin:ferritin ratio is a potential biomarker for cirrhosis, Liver Int., 2012, 32, 1391–1399 CrossRef CAS PubMed.
- M. E. Olesnevich, M. Fanelli Kuczmarski, M. Mason, C. Fang, A. B. Zonderman and M. K. Evans, Serum ferritin levels associated with increased risk for developing CHD in a low-income urban population, Public Health Nutr., 2012, 15, 1291–1298 CrossRef PubMed.
- K. C. Sung, S. M. Kang, E. J. Cho, J. B. Park, S. H. Wild and C. D. Byrne, Ferritin is independently associated with the presence of coronary artery calcium in 12,033 men, Arterioscler., Thromb., Vasc. Biol., 2012, 32, 2525–2530 CrossRef CAS PubMed.
- Y. Zhou, T. Liu, C. Tian, P. Kang and C. Jia, Association of serum ferritin with coronary artery disease, Clin. Biochem., 2012, 45, 1336–1341 CrossRef CAS PubMed.
- B. Ponikowska, T. Suchocki, B. Paleczny, M. Olesinska, S. Powierza, L. Borodulin-Nadzieja, K. Reczuch, S. von Haehling, W. Doehner, S. D. Anker, J. G. Cleland and E. A. Jankowska, Iron Status and Survival in Diabetic Patients With Coronary Artery Disease, Diabetes Care, 2013, 36, 4147–4156 CrossRef CAS PubMed.
- J. T. Salonen, T. P. Tuomainen, K. Nyyssonen, H. M. Lakka and K. Punnonen, Relation between iron stores and non-insulin dependent diabetes in men: case-control study, BMJ, 1998, 317, 727 CrossRef CAS.
- E. S. Ford and M. E. Cogswell, Diabetes and serum ferritin concentration among U.S. adults, Diabetes Care, 1999, 22, 1978–1983 CrossRef CAS.
- J. G. Wilson, J. H. Lindquist, S. C. Grambow, E. D. Crook and J. F. Maher, Potential role of increased iron stores in diabetes, Am. J. Med. Sci., 2003, 325, 332–339 CrossRef PubMed.
- R. Jiang, J. E. Manson, J. B. Meigs, J. Ma, N. Rifai and F. B. Hu, Body iron stores in relation to risk of type 2 diabetes in apparently healthy women, JAMA, 2004, 291, 711–717 CrossRef CAS PubMed.
- M. Mert, M. Korkmaz, M. Temizel and M. Acar, The Level of Ferritin in Diabetic and Nondiabetic Patients with Acute Myocardial Infarction, Turk. J. Med. Sci., 2005, 35, 25–34 CAS.
- R. T. Acton, J. C. Barton, L. V. Passmore, P. C. Adams, M. R. Speechley, F. W. Dawkins, P. Sholinsky, D. M. Reboussin, G. D. McLaren, E. L. Harris, T. C. Bent, T. M. Vogt and O. Castro, Relationships of serum ferritin, transferrin saturation, and HFE mutations and self-reported diabetes in the Hemochromatosis and Iron Overload Screening (HEIRS) study, Diabetes Care, 2006, 29, 2084–2089 CrossRef CAS PubMed.
- E. M. Alissa, W. H. Ahmed, N. Al-Ama and G. A. Ferns, Relationship between indices of iron status and coronary risk factors including diabetes and the metabolic syndrome in Saudi subjects without overt coronary disease, J. Trace Elem. Med. Biol., 2007, 21, 242–254 CAS.
- N. G. Forouhi, A. H. Harding, M. Allison, M. S. Sandhu, A. Welch, R. Luben, S. Bingham, K. T. Khaw and N. J. Wareham, Elevated serum ferritin levels predict new-onset type 2 diabetes: results from the EPIC-Norfolk prospective study, Diabetologia, 2007, 50, 949–956 CrossRef CAS PubMed.
- M. L. Jehn, E. Guallar, J. M. Clark, D. Couper, B. B. Duncan, C. M. Ballantyne, R. C. Hoogeveen, Z. L. Harris and J. S. Pankow, A prospective study of plasma ferritin level and incident diabetes: the Atherosclerosis Risk in Communities (ARIC) Study, Am. J. Epidemiol., 2007, 165, 1047–1054 CrossRef PubMed.
- L. Sun, O. H. Franco, F. B. Hu, L. Cai, Z. Yu, H. Li, X. Ye, Q. Qi, J. Wang, A. Pan, Y. Liu and X. Lin, Ferritin concentrations, metabolic syndrome, and type 2 diabetes in middle-aged and elderly chinese, J. Clin. Endocrinol. Metab., 2008, 93, 4690–4696 CrossRef CAS PubMed.
- J. A. Kolberg, T. Jørgensen, R. W. Gerwien, S. Hamren, M. P. McKenna, E. Moler, M. W. Rowe, M. S. Urdea, X. M. Xu, T. Hansen, O. Pedersen and K. Borch-Johnsen, Development of a type 2 diabetes risk model from a panel of serum biomarkers from the Inter99 cohort, Diabetes Care, 2009, 32, 1207–1212 CrossRef PubMed.
- S. N. Rajpathak, J. P. Crandall, J. Wylie-Rosett, G. C. Kabat, T. E. Rohan and F. B. Hu, The role of iron in type 2 diabetes in humans, Biochim. Biophys. Acta, 2009, 1790, 671–681 CrossRef CAS PubMed.
- C. H. Kim, H. K. Kim, S. J. Bae, J. Y. Park and K. U. Lee, Association of elevated serum ferritin concentration with insulin resistance and impaired glucose metabolism in Korean men and women, Metabolism, 2011, 60, 414–420 CrossRef CAS PubMed.
- B. K. Lee, Y. Kim and Y. I. Kim, Association of serum ferritin with metabolic syndrome and diabetes mellitus in the South Korean general population according to the Korean National Health and Nutrition Examination Survey 2008, Metabolism, 2011, 60, 1416–1424 CrossRef CAS PubMed.
- J. H. Ryoo, M. G. Kim, D. W. Lee and J. Y. Shin, The relationship between serum ferritin and metabolic syndrome in healthy Korean men, Diabetes/Metab. Res. Rev., 2011, 27, 597–603 CrossRef CAS PubMed.
- W. Bao, Y. Rong, S. Rong and L. Liu, Dietary iron intake, body iron stores, and the risk of type 2 diabetes: a systematic review and meta-analysis, BMC Med., 2012, 10, 119 CrossRef CAS PubMed.
- V. Lyssenko, T. Jørgensen, R. W. Gerwien, T. Hansen, M. W. Rowe, M. P. McKenna, J. Kolberg, O. Pedersen, K. Borch-Johnsen and L. Groop, Validation of a multi-marker model for the prediction of incident type 2 diabetes mellitus: combined results of the Inter99 and Botnia studies, Diabet. Vasc. Dis. Res., 2012, 9, 59–67 Search PubMed.
- J. Montonen, H. Boeing, A. Steffen, R. Lehmann, A. Fritsche, H. G. Joost, M. B. Schulze and T. Pischon, Body iron stores and risk of type 2 diabetes: results from the European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam study, Diabetologia, 2012, 55, 2613–2621 CrossRef CAS PubMed.
- Z. Zhao, S. Li, G. Liu, F. Yan, X. Ma, Z. Huang and H. Tian, Body iron stores and heme-iron intake in relation to risk of type 2 diabetes: a systematic review and meta-analysis, PLoS One, 2012, 7, e41641 CAS.
- A. O. Aregbesola, S. Voutilainen, J. K. Virtanen, J. Mursu and T. P. Tuomainen, Body Iron Stores and the Risk of Type 2 Diabetes in Middle-Aged Men, Eur. J. Endocrinol., 2013, 169, 247–253 CrossRef CAS PubMed.
- B. Batchuluun, T. Matsumata, N. Erdenebileg, G. Tsagaantsooj, K. Boldbaatar and A. Khasag, Serum ferritin level is higher in poorly controlled patients with Type 2 diabetes and people without diabetes, aged over 55 years, Diabetic Med., 2013 DOI:10.1111/dme.12343.
- X. Guo, D. Zhou, P. An, Q. Wu, H. Wang, A. Wu, M. Mu, D. Zhang, Z. Zhang, L. He, Y. Liu and F. Wang, Associations between serum hepcidin, ferritin and Hb concentrations and type 2 diabetes risks in a Han Chinese population, Br. J. Nutr., 2013, 1–6 CrossRef PubMed.
- C. H. Jung, M. J. Lee, J. Y. Hwang, J. E. Jang, J. Leem, J. Y. Park, J. Lee, H. K. Kim and W. J. Lee, Elevated serum ferritin level is associated with the incident type 2 diabetes in healthy korean men: a 4 year longitudinal study, PLoS One, 2013, 8, e75250 CAS.
- D. Kundu, A. Roy, T. Mandal, U. Bandyopadhyay, E. Ghosh and D. Ray, Relation of iron stores to oxidative stress in type 2 diabetes, Niger. J. Clin. Pract., 2013, 16, 100–103 CrossRef CAS PubMed.
- S. K. Kunutsor, T. A. Apekey, J. Walley and K. Kain, Ferritin levels and risk of type 2 diabetes mellitus: an updated systematic review and meta-analysis of prospective evidence, Diabetes/Metab. Res. Rev., 2013, 29, 308–318 CrossRef CAS PubMed.
- L. Sun, G. Zong, A. Pan, X. W. Ye, H. X. Li, Z. J. Yu, Y. Zhao, S. R. Zou, D. X. Yu, Q. L. Jin, F. B. Hu and X. Lin, Elevated Plasma Ferritin Is Associated with Increased Incidence of Type 2 Diabetes in Middle-Aged and Elderly Chinese Adults, J. Nutr., 2013, 143, 1459–1465 CrossRef CAS PubMed.
- D. L. White and A. Collinson, Red meat, dietary heme iron, and risk of type 2 diabetes: the involvement of advanced lipoxidation endproducts, Adv. Nutr., 2013, 4, 403–411 CrossRef CAS PubMed.
- N. Wlazlo, M. M. J. van Greevenbroek, I. Ferreira, E. H. J. M. Jansen, E. J. M. Feskens, C. J. H. van der Kallen, C. G. Schalkwijk, B. Bravenboer and C. D. A. Stehouwer, Iron metabolism is associated with adipocyte insulin resistance and plasma adiponectin: the Cohort on Diabetes and Atherosclerosis Maastricht (CODAM) study, Diabetes Care, 2013, 36, 309–315 CrossRef CAS PubMed.
- A. Piperno, P. Trombini, M. Gelosa, V. Mauri, V. Pecci, A. Vergani, A. Salvioni, R. Mariani and G. Mancia, Increased serum ferritin is common in men with essential hypertension, J. Hypertens., 2002, 20, 1513–1518 CrossRef CAS PubMed.
- E. Coban, E. Alkan, S. Altuntas and Y. Akar, Serum ferritin levels correlate with hypertensive retinopathy, Med. Sci. Monit., 2010, 16, CR92–CR95 CAS.
- K. S. Houschyar, R. Lüdtke, G. J. Dobos, U. Kalus, M. Broecker-Preuss, T. Rampp, B. Brinkhaus and A. Michalsen, Effects of phlebotomy-induced reduction of body iron stores on metabolic syndrome: results from a randomized clinical trial, BMC Med., 2012, 10 CrossRef CAS PubMed.
- M. K. Kim, K. H. Baek, K. H. Song, M. I. Kang, J. H. Choi, J. C. Bae, C. Y. Park, W. Y. Lee and K. W. Oh, Increased Serum Ferritin Predicts the Development of Hypertension Among Middle-Aged Men, Am. J. Hypertens., 2012, 25, 492–497 CrossRef CAS PubMed.
- B. Choi, K. J. Yeum, S. J. Park, K. N. Kim and N. S. Joo, Elevated serum ferritin and mercury concentrations are associated with hypertension; analysis of the fourth and fifth Korea national health and nutrition examination survey (KNHANES IV-2, 3, 2008-2009 and V-1, 2010), Environ. Toxicol., 2013 DOI:10.1002/tox.21899.
- M. Jehn, J. M. Clark and E. Guallar, Serum ferritin and risk of the metabolic syndrome in U.S. adults, Diabetes Care, 2004, 27, 2422–2428 CrossRef.
- C. Bozzini, D. Girelli, O. Olivieri, N. Martinelli, A. Bassi, G. De Matteis, I. Tenuti, V. Lotto, S. Friso, F. Pizzolo and R. Corrocher, Prevalence of body iron excess in the metabolic syndrome, Diabetes Care, 2005, 28, 2061–2063 CrossRef.
- K. M. Choi, K. W. Lee, H. Y. Kim, J. A. Seo, S. G. Kim, N. H. Kim, D. S. Choi and S. H. Baik, Association among serum ferritin, alanine aminotransferase levels, and metabolic syndrome in Korean postmenopausal women, Metabolism, 2005, 54, 1510–1514 CrossRef CAS PubMed.
- A. S. González, D. B. Guerrero, M. B. Soto, S. P. Diáz, M. Martinez-Olmos and O. Vidal, Metabolic syndrome, insulin resistance and the inflammation markers C-reactive protein and ferritin, Eur. J. Clin. Nutr., 2006, 60, 802–809 CrossRef PubMed.
- V. Tsimihodimos, I. Gazi, R. Kalaitzidis, M. Elisaf and K. C. Siamopoulos, Increased serum ferritin concentrations and liver enzyme activities in patients with metabolic syndrome, Metab. Syndr. Relat. Disord., 2006, 4, 196–203 CrossRef CAS PubMed.
- P. Trombini and A. Piperno, Ferritin, metabolic syndrome and NAFLD: elective attractions and dangerous liaisons, J. Hepatol., 2007, 46, 549–552 CrossRef CAS PubMed.
- I. S. Vari, B. Balkau, A. Kettaneh, P. André, J. Tichet, F. Fumeron, E. Caces, M. Marre, B. Grandchamp and P. Ducimetière, Ferritin and transferrin are associated with metabolic syndrome abnormalities and their change over time in a general population: Data from an Epidemiological Study on the Insulin Resistance Syndrome (DESIR), Diabetes Care, 2007, 30, 1795–1801 CrossRef CAS PubMed.
- S. Zelber-Sagi, D. Nitzan-Kaluski, Z. Halpern and R. Oren, NAFLD and hyperinsulinemia are major determinants of serum ferritin levels, J. Hepatol., 2007, 46, 700–707 CrossRef CAS PubMed.
- S. Tsimikas, J. Willeit, M. Knoflach, M. Mayr, G. Egger, M. Notdurfter, J. L. Witztum, C. J. Wiedermann, Q. Xu and S. Kiechl, Lipoprotein-associated phospholipase A2 activity, ferritin levels, metabolic syndrome, and 10-year cardiovascular and non-cardiovascular mortality: results from the Bruneck study, Eur. Heart J., 2009, 30, 107–115 CrossRef CAS PubMed.
- L. Valenti, P. Dongiovanni, B. M. Motta, D. W. Swinkels, P. Bonara, R. Rametta, L. Burdick, C. Frugoni, A. L. Fracanzani and S. Fargion, Serum hepcidin and macrophage iron correlate with MCP-1 release and vascular damage in patients with metabolic syndrome alterations, Arterioscler., Thromb., Vasc. Biol., 2011, 31, 683–690 CrossRef CAS PubMed.
- P. Hämäläinen, J. Saltevo, H. Kautiainen, P. Mäntyselkä and M. Vanhala, Erythropoietin, ferritin, haptoglobin, hemoglobin and transferrin receptor in metabolic syndrome: a case control study, Cardiovasc. Diabetol., 2012, 11, 116 CrossRef PubMed.
- S. K. Park, J. H. Ryoo, M. G. Kim and J. Y. Shin, Association of serum ferritin and the development of metabolic syndrome in middle-aged Korean men: a 5-year follow-up study, Diabetes Care, 2012, 35, 2521–2526 CrossRef CAS PubMed.
- H. T. Kang, J. A. Linton and J. Y. Shim, Serum ferritin level is associated with the prevalence of metabolic syndrome in Korean adults: the 2007-2008 Korean National Health and Nutrition Examination Survey, Clin. Chim. Acta, 2012, 413, 636–641 CrossRef CAS PubMed.
- J. H. Yoon, J. A. Linton, S. B. Koh and H. T. Kang, Serum ferritin concentrations predict incidence of metabolic syndrome in rural Korean adults, Clin. Chem. Lab. Med., 2012, 50, 2057–2059 CrossRef CAS PubMed.
- J. S. Chang, S. M. Lin, T. C. Huang, J. C. Chao, Y. C. Chen, W. H. Pan and C. H. Bai, Serum ferritin and risk of the metabolic syndrome: a population-based study, Asia Pac. J. Clin. Nutr., 2013, 22, 400–407 CAS.
- C. Datz, T. K. Felder, D. Niederseer and E. Aigner, Iron homeostasis in the Metabolic Syndrome, Eur. J. Clin. Invest., 2013, 43, 215–224 CrossRef CAS PubMed.
- L. Guo, F. Jiang, Y. T. Tang, M. Y. Si and X. Y. Jiao, The Association of Serum Vascular Endothelial Growth Factor and Ferritin in Diabetic Microvascular Disease, Diabetes Technol. Ther., 2013 DOI:10.1089/dia.2013.0181.
- J. Li, R. Wang, D. Luo, S. Li and C. Xiao, Association between Serum Ferritin Levels and Risk of the Metabolic Syndrome in Chinese Adults: A Population Study, PLoS One, 2013, 8, e74168 CAS.
- C. Sfagos, A. C. Makis, A. Chaidos, E. C. Hatzimichael, A. Dalamaga, K. Kosma and K. L. Bourantas, Serum ferritin, transferrin and soluble transferrin receptor levels in multiple sclerosis patients, Mult. Scler., 2005, 11, 272–275 CrossRef CAS.
- H. Orbach, G. Zandman-Goddard, H. Amital, V. Barak, Z. Szekanecz, G. Szucs, K. Danko, E. Nagy, T. Csepany, J. F. Carvalho, A. Doria and Y. Shoenfeld, Novel biomarkers in autoimmune diseases: prolactin, ferritin, vitamin D, and TPA levels in autoimmune diseases, Ann. N. Y. Acad. Sci., 2007, 1109, 385–400 CrossRef CAS PubMed.
- G. Zandman-Goddard and Y. Shoenfeld, Hyperferritinemia in autoimmunity, Isr. Med. Assoc. J., 2008, 10, 83–84 Search PubMed.
- R. Da Costa, M. Szyper-Kravitz, Z. Szekanecz, T. Csépány, K. Dankó, Y. Shapira, G. Zandman-Goddard, H. Orbach, N. Agmon-Levin and Y. Shoenfeld, Ferritin and Prolactin levels in multiple sclerosis, Isr. Med. Assoc. J., 2011, 13, 91–95 Search PubMed.
- J. T. Salonen, K. Nyyssonen, H. Korpela, J. Tuomilehto, R. Seppanen and R. Salonen, High stored iron levels are associated with excess risk of myocardial infarction in eastern Finnish men, Circulation, 1992, 86, 803–811 CrossRef CAS.
- C. Moroz, H. Bessler, M. Katz, I. Zahavi, H. Salman and M. Djaldetti, Elevated serum ferritin level in acute myocardial infarction, Biomed. Pharmacother., 1997, 51, 126–130 CrossRef CAS.
- T. P. Tuomainen, K. Punnonen, K. Nyyssonen and J. T. Salonen, Association between body iron stores and the risk of acute myocardial infarction in men, Circulation, 1998, 97, 1461–1466 CrossRef CAS.
- K. Klipstein-Grobusch, J. F. Koster, D. E. Grobbee, J. Lindemans, H. Boeing, A. Hofman and J. C. M. Witteman, Serum ferritin and risk of myocardial infarction in the elderly: the Rotterdam Study, Am. J. Clin. Nutr., 1999, 69, 1231–1236 CAS.
- D. Claeys, M. Walting, F. Julmy, W. A. Wuillemin and B. J. Meyer, Haemochromatosis mutations and ferritin in myocardial infarction: a case-control study, Eur. J. Clin. Invest., 2002, 32, 3–8 CrossRef CAS.
- W. D. Silvia, S. Biswas, S. Uthappa and P. Shetty, Ferritin, a potent threat for acute myocardial infarction?, J. Assoc. Physicians India, 2003, 51, 947–950 Search PubMed.
- X. M. Yuan and W. Li, The iron hypothesis of atherosclerosis and its clinical impact, Ann. Med., 2003, 35, 578–591 CrossRef CAS.
- M. P. Holay, A. A. Choudhary and S. D. Suryawanshi, Serum ferritin-a novel risk factor in acute myocardial infarction, Indian Heart J., 2012, 64, 173–177 CrossRef CAS.
- M. P. Iqbal, N. Mehboobali, A. K. Tareen, M. Yakub, S. P. Iqbal, K. Iqbal and G. Haider, Association of body iron status with the risk of premature acute myocardial infarction in a Pakistani population, PLoS One, 2013, 8, e67981 CAS.
- S. Fargion, M. Mattioli, A. L. Fracanzani, M. Sampietro, D. Tavazzi, P. Fociani, E. Taioli, L. Valenti and G. Fiorelli, Hyperferritinemia, iron overload, and multiple metabolic alterations identify patients at risk for nonalcoholic steatohepatitis, Am. J. Gastroenterol., 2001, 96, 2448–2455 CrossRef CAS PubMed.
- M. Koruk, S. Tayşi, M. C. Savaş, O. Yilmaz, F. Akçay and M. Karakök, Serum levels of acute phase proteins in patients with nonalcoholic steatohepatitis, Turk. J. Gastroenterol., 2003, 14, 12–17 Search PubMed.
- E. Bugianesi, P. Manzini, S. D’Antico, E. Vanni, F. Longo, N. Leone, P. Massarenti, A. Piga, G. Marchesini and M. Rizzetto, Relative contribution of iron burden, HFE mutations, and insulin resistance to fibrosis in nonalcoholic fatty liver, Hepatology, 2004, 39, 179–187 CrossRef CAS PubMed.
- T. J. Hsiao, J. C. Chen and J. D. Wang, Insulin resistance and ferritin as major determinants of nonalcoholic fatty liver disease in apparently healthy obese patients, Int. J. Obes. Relat. Metab. Disord., 2004, 28, 167–172 CrossRef CAS PubMed.
- C. Loguercio, T. De Simone, M. V. D’Auria, I. de Sio, A. Federico, C. Tuccillo, A. M. Abbatecola, C. Del Vecchio Blanco and Italian AISF Clinical Group, Non-alcoholic fatty liver disease: a multicentre clinical study by the Italian Association for the Study of the Liver, Dig. Liver Dis., 2004, 36, 398–405 CrossRef CAS PubMed.
- G. C. Farrell and C. Z. Larter, Nonalcoholic fatty liver disease: from steatosis to cirrhosis, Hepatology, 2006, 43, S99–S112 CrossRef CAS PubMed.
- L. Valenti, A. L. Fracanzani, P. Dongiovanni, E. Bugianesi, G. Marchesini, P. Manzini, E. Vanni and S. Fargion, Iron depletion by phlebotomy improves insulin resistance in patients with nonalcoholic fatty liver disease and hyperferritinemia: evidence from a case-control study, Am. J. Gastroenterol., 2007, 102, 1251–1258 CrossRef CAS PubMed.
- E. Aigner and C. Datz, Iron perturbations in human non-alcoholic fatty liver disease (NAFLD): Clinical relevance and molecular mechanisms, Hepatitis Monthly, 2008, 8, 213–220 Search PubMed.
- L. Valenti, D. W. Swinkels, L. Burdick, P. Dongiovanni, H. Tjalsma, B. M. Motta, C. Bertelli, E. Fatta, D. Bignamini, R. Rametta, S. Fargion and A. L. Fracanzani, Serum ferritin levels are associated with vascular damage in patients with nonalcoholic fatty liver disease, Nutr., Metab. Cardiovasc. Dis., 2010, 21, 568–575 CrossRef PubMed.
- M. Yoneda, Y. Nozaki, H. Endo, H. Mawatari, H. Iida, K. Fujita, K. Yoneda, H. Takahashi, H. Kirikoshi, M. Inamori, N. Kobayashi, K. Kubota, S. Saito, S. Maeyama, K. Hotta and A. Nakajima, Serum ferritin is a clinical biomarker in Japanese patients with nonalcoholic steatohepatitis (NASH) independent of HFE gene mutation, Dig. Dis. Sci., 2010, 55, 808–814 CrossRef CAS PubMed.
- K. V. Kowdley, The role of iron in nonalcoholic fatty liver disease: the story continues, Gastroenterology, 2010, 138, 817–819 CrossRef PubMed.
- P. Manousou, G. Kalambokis, F. Grillo, J. Watkins, E. Xirouchakis, M. Pleguezuelo, G. Leandro, V. Arvaniti, G. Germani, D. Patch, V. Calvaruso, D. P. Mikhailidis, A. P. Dhillon and A. K. Burroughs, Serum ferritin is a discriminant marker for both fibrosis and inflammation in histologically proven non-alcoholic fatty liver disease patients, Liver Int., 2011, 31, 730–739 CrossRef CAS PubMed.
- Y. Sumida, M. Yoneda, H. Hyogo, K. Yamaguchi, M. Ono, H. Fujii, Y. Eguchi, Y. Suzuki, S. Imai, K. Kanemasa, K. Fujita, K. Chayama, K. Yasui, T. Saibara, N. Kawada, K. Fujimoto, Y. Kohgo and T. Okanoue, A simple clinical scoring system using ferritin, fasting insulin, and type IV collagen 7S for predicting steatohepatitis in nonalcoholic fatty liver disease, J. Gastroenterol., 2011, 46, 257–268 CrossRef CAS PubMed.
- K. V. Kowdley, P. Belt, L. A. Wilson, M. M. Yeh, B. A. Neuschwander-Tetri, N. Chalasani, A. J. Sanyal and J. E. Nelson, Serum ferritin is an independent predictor of histologic severity and advanced fibrosis in patients with nonalcoholic fatty liver disease, Hepatology, 2012, 55, 77–85 CrossRef CAS PubMed.
- C. W. Kim, Y. Chang, E. Sung, H. Shin and S. Ryu, Serum ferritin levels predict incident non-alcoholic fatty liver disease in healthy Korean men, Metabolism, 2012, 61, 1182–1188 CrossRef CAS PubMed.
- K. M. Utzschneider, A. Largajolli, A. Bertoldo, S. Marcovina, J. E. Nelson, M. M. Yeh, K. V. Kowdley and S. E. Kahn, Serum ferritin is associated with non-alcoholic fatty liver disease and decreased Beta-cell function in non-diabetic men and women, J. Diabetes Complications, 2013 DOI:10.1016/j.jdiacomp.2013.11.007.
- S. S. Entman, L. D. Richardson and A. P. Killam, Elevated serum ferritin in the altered ferrokinetics of toxemia of pregnancy, Am. J. Obstet. Gynecol., 1982, 144, 418–422 CAS.
- S. S. Entman, L. D. Richardson and A. P. Killam, Altered ferrokinetics in toxemia of pregnancy – a possible indicator of decreased red cell survival, Clin. Exp. Hypertens., Part B, 1983, 2, 171–178 CAS.
- M. P. Rayman, J. Barlis, R. W. Evans, C. W. Redman and L. J. King, Abnormal iron parameters in the pregnancy syndrome preeclampsia, Am. J. Obstet. Gynecol., 2002, 187, 412–418 CrossRef CAS.
- C. A. Hubel, L. M. Bodnar, A. Many, G. Harger, R. B. Ness and J. M. Roberts, Nonglycosylated ferritin predominates in the circulation of women with preeclampsia but not intrauterine growth restriction, Clin. Chem., 2004, 50, 948–951 CAS.
- I. A. Siddiqui, A. Jaleel, H. M. Kadri, W. A. Saeed and W. Tamimi, Iron status parameters in preeclamptic women, Arch. Gynecol. Obstet., 2011, 284, 587–591 CrossRef CAS PubMed.
- D. R. Blake, P. A. Bacon, E. J. Eastham and K. Brigham, Synovial fluid ferritin in rheumatoid arthritis, BMJ, 1980, 281, 715–716 CrossRef CAS.
- R. S. Rothwell and P. Davis, Relationship between serum ferritin, anemia, and disease activity in acute and chronic rheumatoid arthritis, Rheumatol. Int., 1981, 1, 65–67 CrossRef CAS.
- P. Biemond, A. J. G. Swaak, H. G. Vaneijk and J. F. Koster, Intraarticular ferritin-bound iron in rheumatoid arthritis – a factor that increases oxygen free radical-induced tissue destruction, Arthritis Rheum., 1986, 29, 1187–1193 CrossRef CAS.
- C. Palermo, S. Maddali Bongi and G. Bianucci, Relationship between serum ferritin, iron stores and disease activity in rheumatoid arthritis, Ric. Clin. Lab., 1986, 16, 463–469 CAS.
- P. Biemond, A. J. G. Swaak, H. G. Vaneijk and J. F. Koster, Superoxide Dependent Iron Release from Ferritin in Inflammatory Diseases, Free Radical Biol. Med., 1988, 4, 185–198 CrossRef CAS.
- E. Abe and M. Arai, Synovial fluid ferritin in traumatic hemarthrosis, rheumatoid arthritis and osteoarthritis, Tohoku J. Exp. Med., 1992, 168, 499–505 CrossRef CAS.
- K. Yildirim, S. Karatay, M. A. Melikoglu, G. Gureser, M. Ugur and K. Senel, Associations between acute phase reactant levels and disease activity score (DAS28) in patients with rheumatoid arthritis, Ann. Clin. Lab. Sci., 2004, 34, 423–426 Search PubMed.
- F. Lv, L. J. Song and X. F. Li, Combined measurement of multiple acute phase reactants to predict relapse of rheumatoid arthritis, Int. J. Rheum. Dis., 2013 DOI:10.1111/1756185X.12186.
- R. L. Goldenberg, B. M. Mercer, M. Miodovnik, G. R. Thurnau, P. J. Meis, A. Moawad, R. H. Paul, S. F. Bottoms, A. Das, J. M. Roberts, D. McNellis and T. Tamura, Plasma ferritin, premature rupture of membranes, and pregnancy outcome, Am. J. Obstet. Gynecol., 1998, 179, 1599–1604 CrossRef CAS.
- P. C. R. Garcia, F. Longhi, R. G. Branco, J. P. Piva, D. Lacks and R. C. Tasker, Ferritin levels in children with severe sepsis and septic shock, Acta Paediatr., 2007, 96, 1829–1831 CrossRef PubMed.
- T. D. Bennett, K. N. Hayward, R. W. Farris, S. Ringold, C. A. Wallace and T. V. Brogan, Very high serum ferritin levels are associated with increased mortality and critical care in pediatric patients, Pediatr. Crit. Care Med., 2011, 12, e233–e236 CrossRef PubMed.
- M. Suárez-Santamaría, F. Santolaria, A. Pérez-Ramírez, M. R. Aléman-Valls, A. Martínez-Riera, E. González-Reimers, M. J. de la Vega and A. Milena, Prognostic value of inflammatory markers (notably cytokines and procalcitonin), nutritional assessment, and organ function in patients with sepsis, Eur. Cytokine Network, 2010, 21, 19–26 Search PubMed.
- A. Dávalos, J. M. Fernandezreal, W. Ricart, S. Soler, A. Molins, E. Planas and D. Genis, Iron-related damage in acute ischemic stroke, Stroke, 1994, 25, 1543–1546 CrossRef.
- A. K. Erdemoglu and S. Ozbakir, Serum ferritin levels and early prognosis of stroke, Eur. J. Neurol., 2002, 9, 633–637 CrossRef CAS.
- G. M. Bishop and S. R. Robinson, Quantitative analysis of cell death and ferritin expression in response to cortical iron: implications for hypoxia-ischemia and stroke, Brain Res., 2001, 907, 175–187 CrossRef CAS.
- A. Armengou and A. Davalos, A review of the state of research into the role of iron in stroke, J. Nutr., Health Aging, 2002, 6, 207–208 CAS.
- E. Millerot, A. S. Prigent-Tessier, N. M. Bertrand, P. J. Faure, C. M. Mossiat, M. E. Giroud, A. G. Beley and C. Marie, Serum ferritin in stroke: a marker of increased body iron stores or stroke severity?, J. Cereb. Blood Flow Metab., 2005, 25, 1386–1393 CrossRef CAS PubMed.
- D. L. van der A, D. E. Grobbee, M. Roest, J. J. M. Marx, H. A. Voorbij and Y. T. van der Schouw, Serum ferritin is a risk factor for stroke in postmenopausal women, Stroke, 2005, 36, 1637–1641 CrossRef CAS PubMed.
- M. Millan, T. Sobrino, M. Castellanos, F. Nombela, J. F. Arenillas, E. Riva, I. Cristobo, M. M. Garcia, J. Vivancos, J. Serena, M. A. Moro, J. Castillo and A. Dávalos, Increased body iron stores are associated with poor outcome after thrombolytic treatment in acute stroke, Stroke, 2007, 38, 90–95 CrossRef PubMed.
- M. Mehdiratta, S. Kumar, D. Hackney, G. Schlaug and M. Selim, Association between serum ferritin level and perihematoma edema volume in patients with spontaneous intracerebral hemorrhage, Stroke, 2008, 39, 1165–1170 CrossRef CAS PubMed.
- M. Millán, T. Sobrino, J. F. Arenillas, M. Rodriguez-Yáñez, M. Garcia, F. Nombela, M. Castellanos, N. Pérez de la Ossa, P. Cuadras, J. Serena, J. Castillo and A. Dávalos, Biological signatures of brain damage associated with high serum ferritin levels in patients with acute ischemic stroke and thrombolytic treatment, Dis. Markers, 2008, 25, 181–188 CrossRef.
- N. P. Pérez de la Ossa, T. Sobrino, Y. Silva, M. Blanco, M. Millán, M. Gomis, J. Agulla, P. Araya, S. Reverté, J. Serena and A. Dávalos, Iron-related brain damage in patients with intracerebral hemorrhage, Stroke, 2010, 41, 810–813 CrossRef PubMed.
- K. H. Choi, M. S. Park, J. T. Kim, T. S. Nam, S. M. Choi, B. C. Kim, M. K. Kim and K. H. Cho, The serum ferritin level is an important predictor of hemorrhagic transformation in acute ischaemic stroke, Eur. J. Neurol., 2012, 19, 570–577 CrossRef PubMed.
- I. García-Yébenes, M. Sobrado, A. Moraga, J. G. Zarruk, V. G. Romera, J. M. Pradillo, N. Perez de la Ossa, M. A. Moro, A. Dávalos and I. Lizasoain, Iron overload, measured as serum ferritin, increases brain damage induced by focal ischemia and early reperfusion, Neurochem. Int., 2012, 61, 1364–1369 CrossRef PubMed.
- K. Nishiya and K. Hashimoto, Elevation of serum ferritin levels as a marker for active systemic lupus erythematosus, Clin. Exp. Rheumatol., 1997, 15, 39–44 CAS.
- M. K. Lim, C. K. Lee, Y. S. Ju, Y. S. Cho, M. S. Lee, B. Yoo and H. B. Moon, Serum ferritin as a serologic marker of activity in systemic lupus erythematosus, Rheumatol. Int., 2001, 20, 89–93 CrossRef CAS.
- L. G. Xu, M. Wu, J. C. Hu, Z. H. Zhai and H. B. Shu, Identification of downstream genes up-regulated by the tumor necrosis factor family member TALL-1, J. Leukocyte Biol., 2002, 72, 410–416 CAS.
- E. Beyan, C. Beyan, A. Demirezer, E. Ertugrul and A. Uzuner, The relationship between serum ferritin levels and disease activity in systemic lupus erythematosus, Scand. J. Rheumatol., 2003, 32, 225–228 CrossRef CAS.
- G. Zandman-Goddard, H. Orbach, H. Amital, Z. Szekanecz, G. Szucs, K. Danko, E. Nagy, T. Csepany and Y. Shoenfeld, Elevated levels of ferritin in systemic lupus erythematosus and other autoimmune diseases, Ann. Rheum. Dis., 2007, 66, 488 Search PubMed.
- A. Parodi, S. Davi, A. B. Pringe, A. Pistorio, N. Ruperto, S. Magni-Manzoni, P. Miettunen, B. Bader-Meunier, G. Espada, G. Sterba, S. Ozen, D. Wright, C. S. Magalhaes, R. Khubchandani, H. Michels, P. Woo, A. Iglesias, D. Guseinova, C. Bracaglia, K. Hayward, C. Wouters, A. Grom, M. Vivarelli, A. Fischer, L. Breda, A. Martini, A. Ravelli and P. R. E. Soc, Macrophage Activation Syndrome in Juvenile Systemic Lupus Erythematosus A Multinational Multicenter Study of Thirty-Eight Patients, Arthritis Rheum., 2009, 60, 3388–3399 CrossRef CAS PubMed.
- K. Vanarsa, Y. Ye, J. Han, C. Xie, C. Mohan and T. Wu, Inflammation associated anemia and ferritin as disease markers in SLE, Arthritis Res. Ther., 2012, 14, R182 CAS.
- M. Abbasi, M. Sahebari, A. Amini and M. Saghafi, Hyperferritinemia: A possible marker for diagnosis of systemic lupus erythematosus?, Life Sci. J., 2013, 10, 335–337 Search PubMed.
- S. Y. Lee, S. W. Lee and W. T. Chung, Severe inflammation may be caused by hyperferritinemia of pseudo-pseudo Meigs’ syndrome in lupus patients: two cases reports and a literature review, Clin. Rheumatol., 2013, 32, 1823–1826 CrossRef PubMed.
- M. A. B. Lozovoy, A. N. C. Simão, S. R. Oliveira, T. M. V. Iryioda, C. Panis, R. Cecchini and I. Dichi, Relationship between iron metabolism, oxidative stress, and insulin resistance in patients with systemic lupus erythematosus, Scand. J. Rheumatol., 2013, 42, 303–310 CrossRef CAS PubMed.
- S. Vilaiyuk, N. Sirachainan, S. Wanitkun, K. Pirojsakul and J. Vaewpanich, Recurrent macrophage activation syndrome as the primary manifestation in systemic lupus erythematosus and the benefit of serial ferritin measurements: a case-based review, Clin. Rheumatol., 2013, 32, 899–904 CrossRef PubMed.
- G. Zandman-Goddard, H. Orbach, N. Agmon-Levin, M. Boaz, H. Amital, Z. Szekanecz, G. Szucs, J. Rovensky, E. Kiss, N. Corocher, A. Doria, L. Stojanovich, F. Ingegnoli, P. L. Meroni, B. Rozman, J. Gomez-Arbesu, M. Blank and Y. Shoenfeld, Hyperferritinemia is associated with serologic antiphospholipid syndrome in SLE patients, Clin. Rev. Allergy Immunol., 2013, 44, 23–30 CrossRef CAS PubMed.
- A. Undas, P. Podolec, K. Zawilska, M. Pieculewicz, I. Jedliński, E. Stępień, E. Konarska-Kuszewska, P. Weglarz, M. Duszynska, E. Hanschke, T. Przewlocki and W. Tracz, Altered fibrin clot structure/function in patients with cryptogenic ischemic stroke, Stroke, 2009, 40, 1499–1501 CrossRef CAS PubMed.
- I. Palka, J. Nessler, B. Nessler, W. Piwowarska, W. Tracz and A. Undas, Altered fibrin clot properties in patients with chronic heart failure and sinus rhythm: a novel prothrombotic mechanism, Heart, 2010, 96, 1114–1118 CrossRef CAS PubMed.
- A. Undas and R. A. S. Ariëns, Fibrin clot structure and function: a role in the pathophysiology of arterial and venous thromboembolic diseases, Arterioscler., Thromb., Vasc. Biol., 2011, 31, e88–e99 CrossRef CAS PubMed.
- A. Undas, M. Cieśla-Dulb, T. Drżkiewiczb and J. Sadowski, Altered fibrin clot properties are associated with residual vein obstruction: effects of lipoprotein(a) and apolipoprotein(a) isoform, Thromb. Res., 2012, 130, e184–e187 CrossRef CAS PubMed.
- J. Bester, A. V. Buys, B. Lipinski, D. B. Kell and E. Pretorius, High ferritin levels have major effects on the morphology of erythrocytes in Alzheimer’s disease, Front. Aging Neurosci., 2013, 5, 00088 Search PubMed.
- E. Pretorius and B. Lipinski, Thromboembolic ischemic stroke changes red blood cell morphology, Cardiovasc. Pathol., 2013, 22, 241–242 CrossRef PubMed.
- E. Pretorius and B. Lipinski, Iron alters red blood cell morphology, Blood, 2013, 121, 9 CrossRef.
- E. Pretorius, J. Bester, N. Vermeulen, B. Lipinski, G. S. Gericke and D. B. Kell, Profound morphological changes in the erythrocytes and fibrin networks of patients with hemochromatosis or with hyperferritinemia, and their normalization by iron chelators and other agents, PLoS One, 2014, 9, e85271 Search PubMed.
- E. Pretorius, N. Vermeulen and J. Bester, Atypical erythrocytes and platelets in a patient with a pro-thrombin mutation, Platelets, 2013 DOI:10.3109/09537104.2013.830709.
- E. Pretorius, N. Vermeulen, J. Bester, B. Lipinski and D. B. Kell, A novel method for assessing the role of iron and its functional chelation in fibrin fibril formation: the use of scanning electron microscopy, Toxicol. Mech. Methods, 2013, 23, 352–359 CrossRef CAS PubMed.
- B. Lipinski, E. Pretorius, H. M. Oberholzer and W. J. van der Spuy, Interaction of fibrin with red blood cells: the role of iron, Ultrastruct. Pathol., 2012, 36, 79–84 CrossRef PubMed.
- B. Lipinski and E. Pretorius, Novel pathway of iron-induced blood coagulation: implications for diabetes mellitus and its complications, Pol. Arch. Med. Wewn., 2012, 122, 115–122 CAS.
- E. Pretorius, J. du Plooy, P. Soma and A. Y. Gasparyan, An ultrastructural analysis of platelets, erythrocytes, white blood cells, and fibrin network in systemic lupus erythematosus, Rheumatol. Int., 2013 DOI:10.1007/s00296-013-2817-x.
- A. V. Buys, M. J. Van Rooy, P. Soma, D. Van Papendorp, B. Lipinski and E. Pretorius, Changes in red blood cell membrane structure in type 2 diabetes: a scanning electron and atomic force microscopy study, Cardiovasc. Diabetol., 2013, 12, 25 CrossRef PubMed.
- S. Gangopadhyay, V. K. Vijayan and S. K. Bansal, Lipids of erythrocyte membranes of COPD patients: a quantitative and qualitative study, COPD, 2012, 9, 322–331 Search PubMed.
- E. Pretorius, J. N. du Plooy, P. Soma, I. Keyser and A. V. Buys, Smoking and fluidity of erythrocyte membranes: A high resolution scanning electron and atomic force microscopy investigation, Nitric Oxide, 2013, 35C, 42–46 CrossRef PubMed.
- C. J. Smith and T. H. Fischer, Particulate and vapor phase constituents of cigarette mainstream smoke and risk of myocardial infarction, Atherosclerosis, 2001, 158, 257–267 CrossRef CAS.
- G. J. Kontoghiorghes, Future chelation monotherapy and combination therapy strategies in thalassemia and other conditions. Comparison of deferiprone, deferoxamine, ICL670, GT56-252, L1NAll and starch deferoxamine polymers, Hemoglobin, 2006, 30, 329–347 CrossRef CAS PubMed.
- H. Nick, Iron chelation, quo vadis?, Curr. Opin. Chem. Biol., 2007, 11, 419–423 CrossRef CAS PubMed.
- A. Maggio, A. Filosa, A. Vitrano, G. Aloj, A. Kattamis, A. Ceci, S. Fucharoen, P. Cianciulli, R. W. Grady, L. Prossomariti, J. B. Porter, A. Iacono, M. D. Cappellini, F. Bonifazi, F. Cassara, P. Harmatz, J. Wood and C. Gluud, Iron chelation therapy in thalassemia major: a systematic review with meta-analyses of 1520 patients included on randomized clinical trials, Blood Cells, Mol., Dis., 2011, 47, 166–175 CrossRef CAS PubMed.
- E. A. Rachmilewitz and P. J. Giardina, How I treat thalassemia, Blood, 2011, 118, 3479–3488 CrossRef CAS PubMed.
- Y. Ma, T. Zhou, X. Kong and R. C. Hider, Chelating agents for the treatment of systemic iron overload, Curr. Med. Chem., 2012, 19, 2816–2827 CrossRef CAS.
- T. P. Chang and C. Rangan, Iron poisoning: a literature-based review of epidemiology, diagnosis, and management, Pediatr. Emerg. Care, 2011, 27, 978–985 CrossRef PubMed.
- A. J. Matthews, G. M. Vercellotti, H. J. Menchaca, P. H. Bloch, V. N. Michalek, P. H. Marker, J. Murar and H. Buchwald, Iron and atherosclerosis: inhibition by the iron chelator deferiprone (L1), J. Surg. Res., 1997, 73, 35–40 CrossRef CAS PubMed.
- K. M. Mitchell, A. L. Dotson, K. M. Cool, A. Chakrabarty, S. H. Benedict and S. M. LeVine, Deferiprone, an orally deliverable iron chelator, ameliorates experimental autoimmune encephalomyelitis, Mult. Scler., 2007, 13, 1118–1126 CrossRef CAS PubMed.
- R. Galanello, Deferiprone in the treatment of transfusion-dependent thalassemia: a review and perspective, Ther. Clin. Risk Manage., 2007, 3, 795–805 CAS.
- W. T. Lindsey and B. R. Olin, Deferasirox for transfusion-related iron overload: a clinical review, Clin. Ther., 2007, 29, 2154–2166 CrossRef CAS PubMed.
- M. D. Cappellini and A. Taher, Long-term experience with deferasirox (ICL670), a once-daily oral iron chelator, in the treatment of transfusional iron overload, Expert Opin. Pharmacother., 2008, 9, 2391–2402 CrossRef CAS PubMed.
- C. McLeod, N. Fleeman, J. Kirkham, A. Bagust, A. Boland, P. Chu, R. Dickson, Y. Dundar, J. Greenhalgh, B. Modell, A. Olujohungbe, P. Telfer and T. Walley, Deferasirox for the treatment of iron overload associated with regular blood transfusions (transfusional haemosiderosis) in patients suffering with chronic anaemia: a systematic review and economic evaluation, Health Technol. Assess., 2009, 13 CAS , iii–iv, ix–xi, 1–121.
- M. D. Cappellini, J. Porter, A. El-Beshlawy, C. K. Li, J. F. Seymour, M. Elalfy, N. Gattermann, S. Giraudier, J. W. Lee, L. L. Chan, K. H. Lin, C. Rose, A. Taher, S. L. Thein, V. Viprakasit, D. Habr, G. Domokos, B. Roubert and A. Kattamis, Tailoring iron chelation by iron intake and serum ferritin: the prospective EPIC study of deferasirox in 1744 patients with transfusion-dependent anemias, Haematologica, 2010, 95, 557–566 CrossRef CAS PubMed.
- J. A. Joseph, N. A. Denisova, D. Bielinski, D. R. Fisher and B. Shukitt-Hale, Oxidative stress protection and vulnerability in aging: putative nutritional implications for intervention, Mech. Ageing Dev., 2000, 116, 141–153 CrossRef CAS.
- K. E. Heim, A. R. Tagliaferro and D. J. Bobilya, Flavonoid antioxidants: chemistry, metabolism and structure-activity relationships, J. Nutr. Biochem., 2002, 13, 572–584 CrossRef CAS.
- S. R. McAnulty, L. S. McAnulty, D. C. Nieman, C. L. Dumke, J. D. Morrow, A. C. Utter, D. A. Henson, W. R. Proulx and G. L. George, Consumption of blueberry polyphenols reduces exercise-induced oxidative stress compared to vitamin C, Nutr. Res., 2004, 24, 209–221 CrossRef CAS PubMed.
- P. Velayutham, A. Babu and D. M. Liu, Green tea catechins and cardiovascular health: an update, Curr. Med. Chem., 2008, 15, 1840–1850 CrossRef.
- M. Akhlaghi and B. Bandy, Mechanisms of flavonoid protection against myocardial ischemia-reperfusion injury, J. Mol. Cell. Cardiol., 2009, 46, 309–317 CrossRef CAS PubMed.
- N. R. Perron and J. L. Brumaghim, A review of the antioxidant mechanisms of polyphenol compounds related to iron binding, Cell Biochem. Biophys., 2009, 53, 75–100 CrossRef CAS PubMed.
- N. R. Perron, H. C. Wang, S. N. Deguire, M. Jenkins, M. Lawson and J. L. Brumaghim, Kinetics of iron oxidation upon polyphenol binding, Dalton Trans., 2010, 39, 9982–9987 RSC.
- N. R. Perron, C. R. Garcia, J. R. Pinzon, M. N. Chaur and J. L. Brumaghim, Antioxidant and prooxidant effects of polyphenol compounds on copper-mediated DNA damage, J. Inorg. Biochem., 2011, 105, 745–753 CrossRef CAS PubMed.
- R. A. Jacob, H. H. Sandstead, L. M. Klevay and L. K. Johnson, Utility of serum ferritin as a measure of iron deficiency in normal males undergoing repetitive phlebotomy, Blood, 1980, 56, 786–791 CAS.
- C. A. Finch, V. Bellotti, S. Stray, D. A. Lipschitz, J. D. Cook, M. J. Pippard and H. A. Huebers, Plasma ferritin determination as a diagnostic tool, West. J. Med., 1986, 145, 657–663 CAS.
- J. D. Cook, C. H. Flowers and B. S. Skikne, The quantitative assessment of body iron, Blood, 2003, 101, 3359–3364 CrossRef CAS PubMed.
- A. Kolnagou, D. Yazman, C. Economides, E. Eracleous and G. J. Kontoghiorghes, Uses and limitations of serum ferritin, magnetic resonance imaging T2 and T2* in the diagnosis of iron overload and in the ferrikinetics of normalization of the iron stores in thalassemia using the International Committee on Chelation deferiprone/deferoxamine combination protocol, Hemoglobin, 2009, 33, 312–322 CrossRef CAS PubMed.
- B. D. Maliken, W. F. Avrin, J. E. Nelson, J. Mooney, S. Kumar and K. V. Kowdley, Room-temperature susceptometry predicts biopsy-determined hepatic iron in patients with elevated serum ferritin, Ann. Hepatol., 2012, 11, 77–84 CAS.
- M. Worwood, S. J. Cragg, M. Wagstaff and A. Jacobs, Binding of human serum ferritin to concanavalin A, Clin. Sci., 1979, 56, 83–87 CAS.
- N. C. Andrews, Forging a field: the golden age of iron biology, Blood, 2008, 112, 219–230 CrossRef CAS PubMed.
- A. E. Hamburger, A. P. West, Z. A. Hamburger, P. Hamburger and P. J. Bjorkman, Crystal structure of a secreted insect ferritin reveals a symmetrical arrangement of heavy and light chains, J. Mol. Biol., 2005, 349, 558–569 CrossRef CAS PubMed.
- S. Ghosh, S. Hevi and S. L. Chuck, Regulated secretion of glycosylated human ferritin from hepatocytes, Blood, 2004, 103, 2369–2376 CrossRef CAS PubMed.
- T. N. Tran, S. K. Eubanks, K. J. Schaffer, C. Y. J. Zhou and M. C. Linder, Secretion of ferritin by rat hepatoma cells and its regulation by inflammatory cytokines and iron, Blood, 1997, 90, 4979–4986 CAS.
- J. Y. Li, N. Paragas, R. M. Ned, A. Qiu, M. Viltard, T. Leete, I. R. Drexler, X. Chen, S. Sanna-Cherchi, F. Mohammed, D. Williams, C. S. Lin, K. M. Schmidt-Ott, N. C. Andrews and J. Barasch, Scara5 is a ferritin receptor mediating non-transferrin iron delivery, Dev. Cell, 2009, 16, 35–46 CrossRef CAS PubMed.
- L. Li, C. J. Fang, J. C. Ryan, E. C. Niemi, J. A. Lebrón, P. J. Björkman, H. Arase, F. M. Torti, S. V. Torti, M. C. Nakamura and W. E. Seaman, Binding and uptake of H-ferritin are mediated by human transferrin receptor-1, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 3505–3510 CrossRef CAS PubMed.
- J. Han, W. E. Seaman, X. M. Di, W. Wang, M. Willingham, F. M. Torti and S. V. Torti, Iron Uptake Mediated by Binding of H-Ferritin to the TIM-2 Receptor in Mouse Cells, PLoS One, 2011, 6 Search PubMed.
- J. C. Sibille, H. Kondo and P. Aisen, Interactions between Isolated Hepatocytes and Kupffer Cells in Iron Metabolism – a Possible Role for Ferritin as an Iron Carrier Protein, Hepatology, 1988, 8, 296–301 CrossRef CAS.
- L. A. Cohen, L. Gutierrez, A. Weiss, Y. Leichtmann-Bardoogo, D. L. Zhang, D. R. Crooks, R. Sougrat, A. Morgenstern, B. Galy, M. W. Hentze, F. J. Lazaro, T. A. Rouault and E. G. Meyron-Holtz, Serum ferritin is derived primarily from macrophages through a nonclassical secretory pathway, Blood, 2010, 116, 1574–1584 CrossRef CAS PubMed.
- R. Wilkinson and K. Pickett, The spirit level: why equality is better for everyone, Penguin Books, London, 2009 Search PubMed.
- R. W. G. Chapman, A. Gorman, M. Laulicht, M. A. Hussain, S. Sherlock and A. V. Hoffbrand, Binding of Serum Ferritin to Concanavalin-a in Patients with Iron Overload and with Chronic Liver-Disease, J. Clin. Pathol., 1982, 35, 481–486 CrossRef CAS.
- T. V. Adamkiewicz, M. R. Abboud, C. Paley, N. Olivieri, M. Kirby-Allen, E. Vichinsky, J. F. Casella, O. A. Alvarez, J. C. Barredo, M. T. Lee, R. V. Iyer, A. Kutlar, K. M. McKie, V. McKie, N. Odo, B. Gee, J. L. Kwiatkowski, G. M. Woods, T. Coates, W. Wang and R. J. Adams, Serum ferritin level changes in children with sickle cell disease on chronic blood transfusion are nonlinear and are associated with iron load and liver injury, Blood, 2009, 114, 4632–4638 CrossRef CAS PubMed.
- T. Takikawa, H. Hayashi, N. Nishimura, M. Yano, T. Isomura and N. Sakamoto, Correlation between serum levels of alanine aminotransferase and ferritin in male blood donors with antibody to hepatitis C virus, J. Gastroenterol., 1994, 29, 593–597 CrossRef CAS.
- C. Caramelo, M. Albalate, T. Bermejillo, S. Navas, A. Ortiz, P. de Sequera, S. Casado and V. Carreño, Relationships between plasma ferritin and aminotransferase profile in haemodialysis patients with hepatitis C virus, Nephrol., Dial., Transplant., 1996, 11, 1792–1796 CrossRef CAS.
- B. Dubern, J. P. Girardet and P. Tounian, Insulin resistance and ferritin as major determinants of abnormal serum aminotransferase in severely obese children, Int. J. Pediatr. Obes., 2006, 1, 77–82 CrossRef.
- T. Nakagawa, Y. Muramoto, M. Hori, S. Mihara, T. Marubayashi and K. Nakagawa, A preliminary investigation of the association between haptoglobin polymorphism, serum ferritin concentration and fatty liver disease, Clin. Chim. Acta, 2008, 398, 34–38 CrossRef CAS PubMed.
- M. Iwasa, N. Hara, K. Iwata, M. Ishidome, R. Sugimoto, H. Tanaka, N. Fujita, Y. Kobayashi and Y. Takei, Restriction of calorie and iron intake results in reduction of visceral fat and serum alanine aminotransferase and ferritin levels in patients with chronic liver disease, Hepatol. Res., 2010, 40, 1188–1194 CrossRef CAS PubMed.
- E. Ozawa, S. Abiru, S. Nagaoka, K. Yano, A. Komori, K. Migita, H. Yatsuhashi, N. Taura, T. Ichikawa, H. Ishibashi and K. Nakao, Ferritin/alanine aminotransferase ratio as a possible marker for predicting the prognosis of acute liver injury, J. Gastroenterol. Hepatol., 2011, 26, 1326–1332 CrossRef CAS PubMed.
- P. C. Adams and J. C. Barton, A diagnostic approach to hyperferritinemia with a non-elevated transferrin saturation, J. Hepatol., 2011, 55, 453–458 CrossRef CAS PubMed.
- S. Uysal, F. Armutcu, T. Aydogan, K. Akin, M. Ikizek and M. R. Yigitoglu, Some inflammatory cytokine levels, iron metabolism and oxidan stress markers in subjects with nonalcoholic steatohepatitis, Clin. Biochem., 2011, 44, 1375–1379 CrossRef CAS PubMed.
- A. Oguz, A. E. Atay, A. Tas, G. Seven and M. Koruk, Predictive role of acute phase reactants in the response to therapy in patients with chronic hepatitis C virus infection, Gut Liver, 2013, 7, 82–88 CrossRef CAS PubMed.
- J. C. Waterlow, P. J. Garlick and D. J. Millward, Protein Turnover in Mammalian Tissues and in the Whole Body, Elsevier/North-Holland, Amsterdam, 1978 Search PubMed.
- H. A. Johnson, R. L. Baldwin, J. France and C. C. Calvert, A model of whole-body protein turnover based on leucine kinetics in rodents, J. Nutr., 1999, 129, 728–739 CAS.
- J. Pellettieri and A. Sanchez Alvarado, Cell turnover and adult tissue homeostasis: from humans to planarians, Annu. Rev. Genet., 2007, 41, 83–105 CrossRef CAS PubMed.
- A. J. Claydon and R. J. Beynon, Proteome dynamics: revisiting turnover with a global perspective, Mol. Cell. Proteomics, 2012, 11, 1551–1565 Search PubMed.
- G. Balla, H. S. Jacob, J. Balla, M. Rosenberg, K. Nath, F. Apple, J. W. Eaton and G. M. Vercellotti, Ferritin: a cytoprotective antioxidant strategem of endothelium, J. Biol. Chem., 1992, 267, 18148–18153 CAS.
- K. Orino, L. Lehman, Y. Tsuji, H. Ayaki, S. V. Torti and F. M. Torti, Ferritin and the response to oxidative stress, Biochem. J., 2001, 357, 241–247 CrossRef CAS.
- Y. Tsuji, H. Ayaki, S. P. Whitman, C. S. Morrow, S. V. Torti and F. M. Torti, Coordinate transcriptional and translational regulation of ferritin in response to oxidative stress, Mol. Cell. Biol., 2000, 20, 5818–5827 CrossRef CAS.
- K. Hailemariam, K. Iwasaki, B. W. Huang, K. Sakamoto and Y. Tsuji, Transcriptional regulation of ferritin and antioxidant genes by HIPK2 under genotoxic stress, J. Cell Sci., 2010, 123, 3863–3871 CrossRef CAS PubMed.
- B. W. Huang, P. D. Ray, K. Iwasaki and Y. Tsuji, Transcriptional regulation of the human ferritin gene by coordinated regulation of Nrf2 and protein arginine methyltransferases PRMT1 and PRMT4, FASEB J., 2013, 27, 3763–3774 CrossRef CAS PubMed.
- M. Nakano, Y. Kawanishi, S. Kamohara, Y. Uchida, M. Shiota, Y. Inatomi, T. Komori, K. Miyazawa, K. Gondo and I. Yamasawa, Oxidative DNA damage (8-hydroxydeoxyguanosine) and body iron status: a study on 2507 healthy people, Free Radical Biol. Med., 2003, 35, 826–832 CrossRef CAS.
- Y. Maruyama, M. Nakayama, K. Yoshimura, H. Nakano, H. Yamamoto, K. Yokoyama and B. Lindholm, Effect of repeated intravenous iron administration in haemodialysis patients on serum 8-hydroxy-2′-deoxyguanosine levels, Nephrol., Dial., Transplant., 2007, 22, 1407–1412 CrossRef CAS PubMed.
- T. P. Tuomainen, S. Loft, K. Nyyssonen, K. Punnonen, J. T. Salonen and H. E. Poulsen, Body iron is a contributor to oxidative damage of DNA, Free Radical Res., 2007, 41, 324–328 CrossRef CAS PubMed.
- N. Fujita, R. Sugimoto, N. Ma, H. Tanaka, M. Iwasa, Y. Kobayashi, S. Kawanishi, S. Watanabe, M. Kaito and Y. Takei, Comparison of hepatic oxidative DNA damage in patients with chronic hepatitis B and C, J. Viral Hepat., 2008, 15, 498–507 CrossRef CAS PubMed.
- K. L. Kuo, S. C. Hung, Y. H. Wei and D. C. Tarng, Intravenous iron exacerbates oxidative DNA damage in peripheral blood lymphocytes in chronic hemodialysis patients, J. Am. Soc. Nephrol., 2008, 19, 1817–1826 CrossRef CAS PubMed.
- K. Broedbaek, H. E. Poulsen, A. Weimann, G. D. Kom, E. Schwedhelm, P. Nielsen and R. H. Boger, Urinary excretion of biomarkers of oxidatively damaged DNA and RNA in hereditary hemochromatosis, Free Radical Biol. Med., 2009, 47, 1230–1233 CrossRef CAS PubMed.
- A. Hori, T. Mizoue, H. Kasai, K. Kawai, Y. Matsushita, A. Nanri, M. Sato and M. Ohta, Body iron store as a predictor of oxidative DNA damage in healthy men and women, Cancer Sci., 2010, 101, 517–522 CrossRef CAS PubMed.
- K. Broedbaek, V. Siersma, J. T. Andersen, M. Petersen, S. Afzal, B. Hjelvang, A. Weimann, R. D. Semba, L. Ferrucci and H. E. Poulsen, The association between low-grade inflammation, iron status and nucleic acid oxidation in the elderly, Free Radical Res., 2011, 45, 409–416 CrossRef CAS PubMed.
- T. P. Tuomainen, U. Diczfalusy, J. Kaikkonen, K. Nyyssonen and J. T. Salonen, Serum ferritin concentration is associated with plasma levels of cholesterol oxidation products in man, Free Radical Biol. Med., 2003, 35, 922–928 CrossRef CAS.
- S. Yeoh-Ellerton and M. C. Stacey, Iron and 8-isoprostane levels in acute and chronic wounds, J. Invest. Dermatol., 2003, 121, 918–925 CrossRef PubMed.
- C. Matayatsuk, C. Y. Lee, R. W. Kalpravidh, P. Sirankapracha, P. Wilairat, S. Fucharoen and B. Halliwell, Elevated F2-isoprostanes in thalassemic patients, Free Radical Biol. Med., 2007, 43, 1649–1655 CrossRef CAS PubMed.
- A. P. Jewell and R. E. Marcus, Platelet Derived Malonyldialdehyde Production in Patients with Thalassemia Major, J. Clin. Pathol., 1984, 37, 1043–1045 CrossRef CAS.
- F. Farinati, R. Cardin, N. Demaria, G. Dellalibera, C. Marafin, E. Lecis, P. Burra, A. Floreani, A. Cecchetto and R. Naccarato, Iron storage, lipid peroxidation and glutathione turnover in chronic anti-HCV positive hepatitis, J. Hepatol., 1995, 22, 449–456 CrossRef CAS.
- J. Mimić-Oka, A. Savić-Radojević, M. Plješa-Ercegovac, M. Opačić, T. Simić, N. Dimković and D. V. Simić, Evaluation of oxidative stress after repeated intravenous iron supplementation, Renal Failure, 2005, 27, 345–351 CrossRef.
- A. S. De Vriese, D. Borrey, E. Mahieu, I. Claeys, L. Stevens, A. Vanhaeverbeke, M. Roelens and M. R. Langlois, Oral vitamin C administration increases lipid peroxidation in hemodialysis patients, Nephron Clin. Pract., 2008, 108, c28–c34 CrossRef CAS PubMed.
- S. M. King, C. M. Donangelo, M. D. Knutson, P. B. Walter, B. N. Ames, F. E. Viteri and J. C. King, Daily supplementation with iron increases lipid peroxidation in young women with low iron stores, Exp. Biol. Med., 2008, 233, 701–707 CrossRef CAS PubMed.
- J. F. R. Mendes, S. F. Arruda, E. M. de Almeida Siqueira, M. K. Ito and E. F. da Silva, Iron status and oxidative stress biomarkers in adults: a preliminary study, Nutrition, 2009, 25, 379–384 CrossRef CAS PubMed.
- A. I. Alsultan, M. A. Seif, T. T. Amin, M. Naboli and A. M. Alsuliman, Relationship between oxidative stress, ferritin and insulin resistance in sickle cell disease, Eur. Rev. Med. Pharmacol. Sci., 2010, 14, 527–538 CAS.
- T. E. de Jesus dos Santos, G. F. de Sousa, M. C. Barbosa and R. P. Goncalves, The role of iron overload on oxidative stress in sickle cell anemia, Biomarkers Med., 2012, 6, 813–819 CrossRef PubMed.
- M. S. Elalfy, A. A. Adly, A. A. Attia, F. A. Ibrahim, A. S. Mohammed and A. M. Sayed, Effect of antioxidant therapy on hepatic fibrosis and liver iron concentrations in beta-thalassemia major patients, Hemoglobin, 2013, 37, 257–276 CrossRef CAS PubMed.
- E. A. Decker and B. Welch, Role of ferritin as a lipid oxidation catalyst in muscle food, J. Agric. Food Chem., 1990, 38, 674–677 CrossRef CAS.
- J. I. Gray, E. A. Gomaa and D. J. Buckley, Oxidative quality and shelf life of meats, Meat Sci., 1996, 43, S111–S123 CrossRef CAS.
- D. U. Ahn and S. M. Kim, Prooxidant effects of ferrous iron, hemoglobin, and ferritin in oil emulsion and cooked-meat homogenates are different from those in raw-meat homogenates, Poult. Sci., 1998, 77, 348–355 CrossRef CAS PubMed.
- D. U. Ahn, F. H. Wolfe and J. S. Sim, The Effect of Metal Chelators, Hydroxyl Radical Scavengers, and Enzyme-Systems on the Lipid-Peroxidation of Raw Turkey Meat, Poult. Sci., 1993, 72, 1972–1980 CrossRef CAS.
- T. Wang, R. Jónsdóttir and G. Ólafsdóttir, Total phenolic compounds, radical scavenging and metal chelation of extracts from Icelandic seaweeds, Food Chem., 2009, 116, 240–248 CrossRef CAS PubMed.
- F. Lang, E. Lang and M. Foller, Physiology and pathophysiology of eryptosis, Transfus. Med. Hemother., 2012, 39, 308–314 CrossRef PubMed.
- M. M. Aleman, C. Gardiner, P. Harrison and A. S. Wolberg, Differential contributions of monocyte- and platelet-derived microparticles towards thrombin generation and fibrin formation and stability, J. Thromb. Haemostasis, 2011, 9, 2251–2261 CrossRef CAS PubMed.
- C. T. Nielsen, O. Østergaard, C. Johnsen, S. Jacobsen and N. H. H. Heegaard, Distinct features of circulating microparticles and their relationship to clinical manifestations in Systemic Lupus Erythematosus, Arthritis Rheum., 2011, 63, 3067–3077 CrossRef PubMed.
- C. T. Nielsen, Circulating microparticles in systemic Lupus Erythematosus, Dan. Med. J., 2012, 59, B4548 Search PubMed.
- C. T. Nielsen, O. Østergaard, L. Stener, L. V. Iversen, L. Truedsson, B. Gullstrand, S. Jacobsen and N. H. H. Heegaard, Increased IgG on cell-derived plasma microparticles in systemic lupus erythematosus is associated with autoantibodies and complement activation, Arthritis Rheum., 2012, 64, 1227–1236 CrossRef CAS PubMed.
- L. V. Iversen, O. Østergaard, C. T. Nielsen, S. Jacobsen and N. H. H. Heegaard, A heparin-based method for flow cytometric analysis of microparticles directly from platelet-poor plasma in calcium containing buffer, J. Immunol. Methods, 2013, 388, 49–59 CrossRef CAS PubMed.
- L. Iversen, O. Østergaard, S. Ullman, C. T. Nielsen, P. Halberg, T. Karlsmark, N. H. H. Heegaard and S. Jacobsen, Circulating microparticles and plasma levels of soluble E- and P-selectins in patients with systemic sclerosis, Scand. J. Rheumatol., 2013, 42, 473–482 CrossRef CAS PubMed.
- O. Østergaard, C. T. Nielsen, L. V. Iversen, J. T. Tanassi, S. Knudsen, S. Jacobsen and N. H. H. Heegaard, Unique protein signature of circulating microparticles in systemic lupus erythematosus, Arthritis Rheum., 2013, 65, 2680–2690 Search PubMed.
- B. Parker, A. Al-Husain, P. Pemberton, A. P. Yates, P. Ho, R. Gorodkin, L. S. Teh, M. Y. Alexander and I. N. Bruce, Suppression of inflammation reduces endothelial microparticles in active systemic lupus erythematosus, Ann. Rheum. Dis., 2013 DOI:10.1136/annrheumdis-2012-203028.
- J. Pereira, G. Alfaro, M. Goycoolea, T. Quiroga, M. Ocqueteau, L. Massardo, C. Pérez, C. Sáez, O. Panes, V. Matus and D. Mezzano, Circulating platelet-derived microparticles in systemic lupus erythematosus. Association with increased thrombin generation and procoagulant state, Thromb. Haemostasis, 2006, 95, 94–99 CAS.
- A. A. G. Tantawy, A. A. M. Adly, E. A. R. Ismail and N. M. Habeeb, Flow cytometric assessment of circulating platelet and erythrocytes microparticles in young thalassemia major patients: relation to pulmonary hypertension and aortic wall stiffness, Eur. J. Haematol., 2013, 90, 508–518 CrossRef CAS PubMed.
- A. A. G. Tantawy, A. A. M. Adly, E. A. R. Ismail, N. M. Habeeb and A. Farouk, Circulating platelet and erythrocyte microparticles in young children and adolescents with sickle cell disease: Relation to cardiovascular complications, Platelets, 2013, 24, 605–614 CrossRef CAS PubMed.
- I. Porto, G. L. De Maria, L. Di Vito, C. Camaioni, M. Gustapane and L. M. Biasucci, Microparticles in health and disease: small mediators, large role?, Curr. Vasc. Pharmacol., 2011, 9, 490–500 CrossRef CAS.
- P. E. Spronk, H. Bootsma and C. G. M. Kallenberg, Anti-DNA antibodies as early predictor for disease exacerbations in SLE – Guideline for treatment?, Clin. Rev. Allergy Immunol., 1998, 16, 211–218 CrossRef CAS PubMed.
- N. Agmon-Levin, C. Rosário, B. S. Katz, G. Zandman-Goddard, P. Meroni, R. Cervera, L. Stojanovich, M. Blank, S. Pierangeli, S. Praprotnik, E. Meis, L. P. Seguro, A. Ruffatti, V. Pengo, A. Tincani, A. Doria and Y. Shoenfeld, Ferritin in the antiphospholipid syndrome and its catastrophic variant (cAPS), Lupus, 2013, 22, 1327–1335 CrossRef CAS PubMed.
- C. Rosário, G. Zandman-Goddard, E. G. Meyron-Holtz, D. P. D’Cruz and Y. Shoenfeld, The Hyperferritinemic Syndrome: macrophage activation syndrome, Still’s disease, septic shock and catastrophic antiphospholipid syndrome, BMC Med., 2013, 11, 185 CrossRef PubMed.
- D. B. Kell and R. Goodacre, Metabolomics and systems pharmacology: why and how to model the human metabolic network for drug discovery, Drug Discovery Today, 2014, 19, 171–182 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |