Bisphenol A determination in baby bottles by chemiluminescence enzyme-linked immunosorbent assay, lateral flow immunoassay and liquid chromatography tandem mass spectrometry
Elisabetta
Maiolini
a,
Elida
Ferri
a,
Agata Laura
Pitasi
a,
Angel
Montoya
b,
Manuela
Di Giovanni
c,
Ermanno
Errani
c and
Stefano
Girotti
*a
aDepartment of Pharmacy and Biotechnology, Alma Mater Studiorum – University of Bologna, Via San Donato 15, 40127 Bologna, Italy. E-mail: stefano.girotti@unibo.it; Web: http://bomet.fci.unibo.it/girotti/ Fax: +39 0512095652; Tel: +39 0512095660
bInter-University Research Institute for Bioengineering and Human Centered Technology (I3BH), Universitat Politècnica de València, Camino de Vera, s/n, 46022 Valencia, Spain
cRegional Agency for Environmental Protection (ARPA), Via F. Rocchi, 19, Bologna, Italy
First published on 13th November 2013
Abstract
Two immunoassays, a Lateral Flow ImmunoAssay (LFIA) based on colloidal gold nanoparticle labels and an indirect competitive chemiluminescence enzyme-linked immunosorbent assay (CL-ELISA), were developed and a high performance liquid chromatography-tandem mass spectrometry (LC-MS/MS) method was optimized to assess the possible release of bisphenol A (BPA, 4,4′-isopropylidenediphenol) from different plastic baby bottles treated with simulating solutions. Coating conjugate concentration, anti-BPA antibody dilution, incubation time of the primary and secondary antibodies, and tolerance to different organic solvents were optimized to obtain the best performance of the ELISA with chemiluminescent end-point detection. The influence of different buffers on LFIA performance was also evaluated. Both methods showed good repeatability (mean CV value around 13%) and sensitivity. Reproducibility tests for CL-ELISA gave a mean CV value of about 25%. The IC50 and Limit of Detection (LOD) values of CL-ELISA were 0.2 and 0.02 ng mL−1, respectively. The LOD of LFIA was 0.1 μg mL−1. A LC-MS/MS method was also optimized. The separation was performed in a C18 column with a triple-quadrupole mass spectrometer with electrospray ionisation interface. The method showed a good linearity in the range 2 to 500 ng mL−1, with a regression coefficient of 0.998. In the simulating solutions the detection and quantification limits, calculated by the signal to noise level of 3 (S/N = 3), were 5.8 ng mL−1 and 17.4 ng mL−1, respectively. This limit of quantification was about 3 and 35 times lower than the permitted limits set by the official method CEN/TS 13130-13 (0.05 μg mL−1) and by the Directive 2004/19/EC (0.6 μg mL−1), respectively. The methods were applied to determine BPA release from baby bottles, performing repeated procedures according to EU and national regulations. The results demonstrated that no BPA migration from the tested plastic materials occurred with only one exception. The migrated amount, above the regulatory limits, was detected by all the mentioned assays.
1. Introduction
Bisphenol A (BPA, 4,4′-isopropylidenediphenol) is a monomer used primarily in the production of polycarbonate plastics and epoxy resins.1 Polycarbonate plastic is used in a wide variety of digital media (e.g., CDs and DVDs) products, electrical and electronic equipment, sport safety equipment, reusable food and drink containers, etc. Epoxy resins are used in engineering applications, in paints and adhesives, and in a variety of protective coatings in metal cans for foods,1 bottle tops, and water supply pipes. To a lesser extent BPA is used in the production of polyester resins, polysulfone resins, polyacrylate resins, and flame retardants, in the processing of polyvinyl chloride plastic, and in the recycling of thermal paper.The primary way of exposure to bisphenol A for most people is assumed to be the diet: BPA in food and beverages accounts for the majority of daily human exposure.1 The highest estimated daily intakes of BPA in the general population occur in infants and children and this creates particular concern since BPA has been identified as an important endocrine disrupting compound (EDC). It can interfere with hormonal activities by modification of biosynthesis, metabolism and elimination of natural blood-borne hormones.2 Moreover, BPA can originate environmental problems: recent research demonstrates that BPA causes inter-species mating.3
The critical period of BPA exposure is during perinatal development, when the effects tend to be permanent due to the low binding affinity of BPA to serum proteins4 and the capability of xenoestrogenic compounds to bypass the mechanisms that limit the exposure of the baby's organs and brain to circulating estrogens.5 It has been reported that exposure to low doses of BPA results in alterations in the ovary, uterus or mammary glands6 and produces a decline in reproductive capacity.7 These data confirm that during the perinatal period there is an increased sensitivity to BPA with probable consequences for babies' health.
In addition, young children have immature organ systems, high metabolic rates, and relatively low bodyweight, and go through rapid physical development, so repeated exposure to even low levels of BPA may lead to adverse health effects.7 Therefore, it is urgently necessary to have effective analytical tools available to carefully evaluate the real levels of exposure to this compound.
The U.S. Food and Drug Administration (FDA) has established a tolerable daily intake (TDI) of 0.05 mg BPA per kg body weight per day, which is also the reference dose settled by the U.S. Environmental Protection Agency (EPA). On the other hand, a specific migration limit (SML) for BPA of 0.6 mg kg−1 was set by the EU Commission in 2004.8
In September 2010, Canada became the first country in which BPA was banned.9 The European Union states outlawed the manufacture of polycarbonate feeding bottles containing the compound from March 2011, and banned their import and sale from June 2011.10 This recent BPA banning in baby bottles has forced manufacturers to employ BPA-free plastics.11 A recent study by the Joint Research Centre, European Commission,12 shows that, unfortunately, possible problems derived from the use of these alternative plastics have not been thoroughly evaluated.13
Many methods have been reported with the aim of determining BPA in water and in food: liquid chromatography tandem mass spectrometry, yeast bioassays, immunochemical assays, and HPLC with fluorescence or diode-array detectors.14–23 Immunoassays appear to comply with the sensitivity, rapidity, and low cost requirements for effective monitoring activities.24,25
Here we report on the development of a colloidal gold-based lateral flow (LFIA) assay, on the optimization of an ELISA with chemiluminescent detection (CL-ELISA), both based on an indirect competitive immunoreaction, and on the optimization of a High Performance Liquid Chromatography Tandem Mass Spectrometry (LC-MS/MS) method for BPA analysis. The three methods were applied to analyze the possible BPA migration from baby bottles treated to simulate the real, repeated conditions of use, taking into account the food characteristics and the sterilization and heating procedures usually involved. This study was performed according to the statement: “heating bottles or pouring hot liquids into bottles, the presence of acidic or basic foods and beverages have all been shown to increase the rate of BPA leaching from bottles”.26
2. Materials and methods
2.1 Reagents
Bisphenol A standard powder (purity > 97.7%) and bisphenol A d16 (98% purity), the second one employed in HPLC-MS/MS measurements, were purchased from Acros Organics (Geel, Belgium) and Sigma Aldrich (Milan, Italy), respectively. Reagents of analytical grade such as glacial acetic acid, ethanol, methanol, ammonia (32% aqueous solution) and sodium hypochlorite (aqueous solution, 7% active chlorine) were provided by Carlo Erba Reagents (Milan, Italy). Luminol, hydrogen peroxide and p-iodophenol were from Sigma Aldrich Srl (Milano, Italy). Ultrapure water was produced in a MilliQ plant (Millipore Corp., Billerica, MA, USA).The main buffering solutions employed were: 0.05 M carbonate–bicarbonate buffer , pH 9.6; 10 mM PBS , pH 7.4 (10 mM Na2HPO4, 2 mM KH2PO4, 137 mM NaCl, 2.7 mM KCl); PBST (PBS containing 0.05% Tween20); PBSG 1 (10 mM PBS, 0.5% Fish Gelatine); PBSG 2 (20 mM PBS, 1% Fish Gelatine), and 0.2 M BB (borate buffer: 50 mM Na2B4O7, 200 mM H3BO3, pH 8.5).
2.2 Immunoreagents
Mouse anti-bisphenol A monoclonal antibody (BPAB-11) and hapten conjugate, OVA-BPAB, were produced by the Immunotechnology Group at Polytechnic University of Valencia (I3BH, UPV, Spain). Rabbit anti-mouse, goat anti-rabbit immunoglobulins, and tetrachloroauric acid were from Sigma; horseradish peroxidase labelled rabbit anti-mouse immunoglobulin was from Dako (Glostrup, Denmark).2.3 Sample treatment
Twelve polycarbonate (PC) and 12 polypropylene (PP) baby bottles of 260 mL of capacity were analyzed, the first ones containing BPA and the second ones being BPA free. The analyzed samples were representative of various brands of baby bottles available in the Italian market.In order to simulate the migration of bisphenol A from baby bottles under different conditions several assays were carried out using liquids simulating different kinds of food, as indicated in the European Commission Directives 93/8/EEC and 97/48/EEC.27,28 Three different solutions were used:
- Simulating solution A: distilled water. It reproduced the extraction capability of aqueous food with pH > 4.5.
- Simulating solution B: water–acetic acid 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 (v/v). It reproduced the extraction capability of acidic food with pH < 4.5.
3 (v/v). It reproduced the extraction capability of acidic food with pH < 4.5.
- Simulating solution C: water–ethanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 (v/v). It reproduced the extraction capability of fatty food (particularly milk).
50 (v/v). It reproduced the extraction capability of fatty food (particularly milk).
The migration tests were conducted in order to reproduce the conditions of real home-use during the disinfection and meal preparation steps, taking into account the indications reported in the leaflets of the different bottles which recommend a 5 minutes boiling in water prior to the first use. To this end, the bottles of the same material (PP or PC) were divided into two groups of 6 bottles each: one group was subjected to the initial sterilization by 5 minutes boiling whereas the other one was not. Each group of 6 bottles was further divided into two groups of 3 bottles made by the same material and each bottle was filled with one of the simulating solutions A, B, or C. One group was treated by microwaves and the second one by water bath heating for 6 consecutive cycles, replacing the simulating solutions after every cycle (Fig. 1). Only the simulating liquids obtained after the third and sixth migration cycles were analyzed, as indicated by the Italian legislation.29 Further tests were carried out under conditions of stress simulated by the two cycles of sterilization, the first one by heat and the second one by cold sterilization. After each sterilization cycle 3 migration cycles in a water bath or microwave oven were applied. Time and temperature exposure conditions complied with the EU legislation27 and the instructions for use of each bottle.
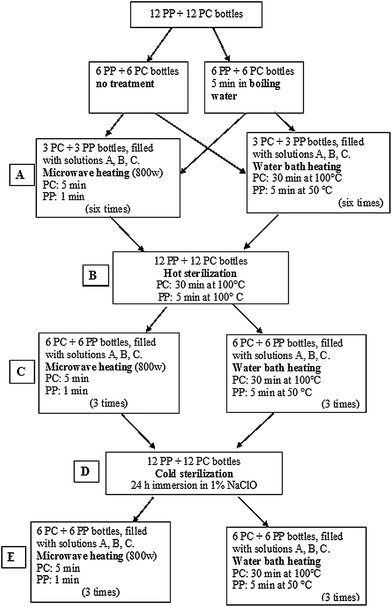 | ||
| Fig. 1 Scheme of the procedure to induce migration of BPA from baby bottle simulating conditions of repeated use. | ||
For polycarbonate bottles (containing BPA): 30 min exposure at 100 °C. For bottles made of PP: 5 min exposure at 50 °C (Fig. 1, step A, right).
2.4 Chemiluminescent ELISA
ELISAs were performed in 96-well microplates in the indirect competitive conjugate-coated format using the OVA-BPAB hapten conjugate and BPAB-11 monoclonal antibody previously employed to develop and validate a colorimetric ELISA.17,25 The optimal concentrations for the chemiluminescent assay were first established by checkerboard titration using the same concentration range, 0.05–0.2 μg mL−1, for both immunoreagents. In order to obtain the best competitive calibration curve other parameters were again examined such as tolerance to the organic solvents methanol and 1–4 dioxane at 1, 5, and 10% final proportions, time of the various immunoassay steps, and effect of shaking during the competitive step.The optimized calibration curves were performed in the 0.001–1000 ng mL−1 range. The OVA-BPAB conjugate (0.2 μg mL−1) in 0.05 M carbonate–bicarbonate buffer, pH 9.6 (100 μL per well) was added into the black polystyrene high-binding microplates (Costar, Cambridge, USA) and incubated overnight at 4 °C for coating. Plates were washed 3 times with 200 μL per well of PBST using a microplate washer (WellWash 4, Labsystem, Finland) and the uncoated sites were blocked by adding 200 μL per well of PBSG 1×. The plates were incubated at room temperature (RT) for 60 min with shaking and washed again 3 times as described above. For the competitive reaction 50 μL per well of 0.2 μg mL−1 of BPAB-11 antibody in PBSG 2× and 50 μL per well of standard solution or sample extract were added and incubated for 60 min at RT without shaking. In order to avoid the absorption of BPA on plastic surfaces only glass containers were used to manipulate the standard solutions (glass vials, Hamilton syringes, etc.). After another microplate washing, 100 μL per well of rabbit anti-mouse IgG-HRP diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2000 in PBSG 1× were added and incubated for 60 min at RT with shaking. Finally, after a new washing step, 100 μL of luminescent mixture (45 μL of 1 mM luminol, 10 μL of 0.5 mM p-iodophenol, 9.8 mL of 0.2 M borate buffer, pH 8.5 and 100 μL of 1 mM hydrogen peroxide) were added to each well and the luminescence emission at 425 nm was immediately recorded using a Victor 1420 luminometer (Wallac-Perkin Elmer, Waltham, MA, USA) and expressed as Relative Luminescence Units (RLU).
2000 in PBSG 1× were added and incubated for 60 min at RT with shaking. Finally, after a new washing step, 100 μL of luminescent mixture (45 μL of 1 mM luminol, 10 μL of 0.5 mM p-iodophenol, 9.8 mL of 0.2 M borate buffer, pH 8.5 and 100 μL of 1 mM hydrogen peroxide) were added to each well and the luminescence emission at 425 nm was immediately recorded using a Victor 1420 luminometer (Wallac-Perkin Elmer, Waltham, MA, USA) and expressed as Relative Luminescence Units (RLU).
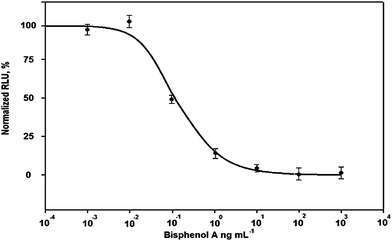
The chemiluminescent emission values (Ex) were normalized between 100% (the emission of a blank control, E0) and 0% (the emission of a sample with an excess of standard compound, Eexcess) according to the expression:
| %B/B0 = 100(Ex − Eexcess)/(E0 − Eexcess) |
The results, expressed as “%B/B0”, were the ratio between B, the emission of “X” ng mL−1 standard solution and B0, the emission of a 0 ng mL−1 standard solution.30 The normalized values were then mathematically fitted, using the Sigmaplot® software (SPSS) version 8.0, to the four parameter logistic equation:
| y = {(A − D)/[1 + (x/C)B]} + D |
2.5 Lateral flow immunoassay
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 × g for 30 min at 10 °C in a Sorvall RC2-B Superspeed centrifuge (Thermo Fisher Scientific Inc., USA).
000 × g for 30 min at 10 °C in a Sorvall RC2-B Superspeed centrifuge (Thermo Fisher Scientific Inc., USA).
2.6 Liquid chromatography tandem mass spectrometry assay
Alliance® 2695 High Performance Liquid Chromatography (HPLC) equipment and an Alliance® 2695 autosampler (Waters, Milliford, MA, USA), refrigerated at 5 °C, were used for chromatographic analyses, coupled to a triple-quadrupole mass spectrometer (MS/MS) “Quattro Micro” TM API (Waters, Milliford, MA, USA) with an electrospray ionization interface and a nitrogen Generator (Peak Scientific, Germany).The detection of BPA was carried out by reverse phase chromatography using a Supelco Discovery® C18 (150 × 2.1 mm, 5 μm) column from Supelco (Milan, Italy) thermostated at 30 °C, with phase eluent consisting of demineralized water MilliQ (mobile phase A) and a solution of 0.1% (v/v) of ammonia in methanol (mobile phase B) at a constant flow of 200 μL min−1. The elution conditions started from a mixture of 90% of phase A, followed by a linear gradient of 5 min up to 85% of phase B, a plateau of 10 min, return to initial conditions in 2 minutes and finally 5 min of column reconditioning. The injection volume was 50 μL. The total elution time was 22 min, with a retention time of 11 min for bisphenol A.
3. Results and discussion
3.1 Optimization of immunochemical methods
| Compound | Ion mode | Cone voltage (V) | Transition quantification | Coll. E (eV) | Transition confirmation | Coll. E (eV) | Dwell (s) |
|---|---|---|---|---|---|---|---|
| BPA | ESI | 31.0 | 227 > 212 | 20.0 | 227 > 133 | 50.0 | 0.3 |
| d16-BPA | ESI | 31.0 | 241 > 142 | 20.0 | — | — | 0.3 |
| Standard BPA conc. (ng mL−1) | Repeatability CV (%) (n = 6) | Reproducibility CV (%) (n = 6) | Recovery (%) (n = 3) |
|---|---|---|---|
| 0.02 | 22.3 | 35.5 | 84.3 |
| 0.2 | 15.7 | 28.9 | 86.1 |
| 2 | 12.5 | 25.6 | 85.4 |
| 7 | 10.6 | 19.8 | 95.6 |
| 50 | 9.7 | 22.1 | 91.7 |
| 100 | 8.2 | 18.2 | 104.2 |
The specificity of monoclonal antibody BPAB-11 had previously been evaluated by performing competitive assays with several compounds structurally related to bisphenol A.17 Significant cross reactivity values were observed only with the closely related compounds 4,4-ethylidenebisphenol (6–19%) and 4-cumylphenol (20–74%).
3.2 Optimization of the LC-MS/MS method
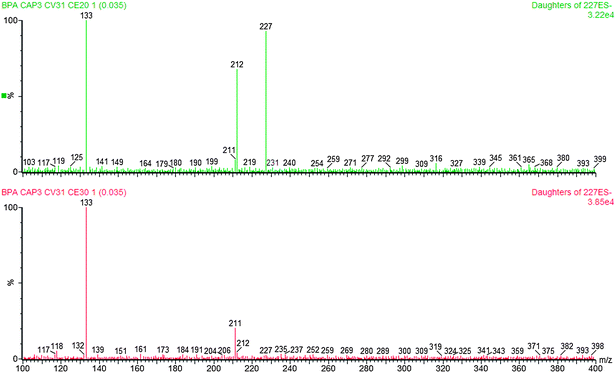
The optimization of the mass parameters was carried out by separate infusion of a solution of bisphenol A and d16-bisphenol A 1 μg mL−1 in methanol. The best conditions of ionization were obtained with a potential difference of 3.0 kV to the capillary (capillary voltage), ESI (electron spray ionization) negative mode, a source temperature of 120 °C, a desolvation temperature of 250 °C and a desolvation gas flow (nitrogen) of 250 L h−1.The quantification transitions chosen were 227 > 212 for the bisphenol A (due to the loss of a methyl group34) and 241 > 142 for the deuterated bisphenol A (due to the same molecular rupture), the confirmation transition detected of the bisphenol A was 227 > 133 (due to the loss of a phenol group). Table 1 summarizes the setting parameters of the spectrometer and Fig. 4 shows the spectra acquired during scanning in a range from 0 to 400 amu (atomic mass unit) for the identification of the molecular ion m/z 227 and in a range from 100 to 400 amu for the fragments of transition m/z 212 and 133 for the bisphenol A and m/z 142 for the d16 BPA.
The developed method showed a very good linearity in the 6–500 ng mL−1 range, with a correlation coefficient of 0.998. The detection limit in the simulating solutions calculated by the signal to noise level of 3 (S/N = 3) was 5.8 ng mL−1, 9 times lower than that stated in the official method UNI CEN/TS 13130-13 [Ref. 35] (0.05 μg mL−1) and about 100 times lower than the limit set by the 2011/10/EU Directive for the BPA migration in food (0.6 μg mL−1).8 The quantification limit, calculated according to the same criteria (S/N = 10), resulted in 17.4 ng mL−1, a value about 3 and 35 times lower, respectively, than the above mentioned permitted limits.
We determined some analytical parameters such as the repeatability, assessed at 450 ng mL−1 (n = 10) with a CV of 6%, and the reproducibility of a 121 ng mL−1 standard solution (n = 6), obtaining in this case a CV of 12.5%.
3.3 Real sample analysis
The analysis of simulating samples obtained from migration tests under the different temperature, timing and heating conditions revealed that only one sample resulted clearly positive both in the CL-ELISA and in the LC/MS/MS assay. That sample was the one obtained from the third migration cycle under microwave of the PC bottle filled with the simulant C and which was not boiled for 5 min before starting the migration tests. Also the LFIA was applied to the analysis of the simulating liquids and the positive sample gave a clearly positive result, though it was not possible to determine the exact content of BPA. In fact, this rapid method is semi-quantitative and in the case of a positive result the exact concentration should be determined by HPLC-MS/MS, ELISA, or CL-ELISA. The BPA concentrations obtained by the different analytical methods are summarized in Table 3.| Method | BPA content (μg mL−1) | Number of determinations (n) | SD (%) |
|---|---|---|---|
| CL-ELISA | 4.5 | 3 | 12.5 |
| LC-MS/MS | 3.3 | 3 | 9.8 |
| LFIA | + (up to 1000) | 3 | — |
All other samples were below the limit of quantification of the quantitative methods and then extensively below the admitted limits set by regulations, showing that the migration of BPA from baby bottles still containing this compound is a quite rare eventuality, even after repeated application of sterilization and heating treatments. Moreover, one sample is not enough to confirm that the 5 min boiling treatment can influence the migration process, at least in the case of subsequent treatment by microwave. These results are in accordance with recently reported studies12,13 that examined the BPA release from 277 baby bottles made of several types of plastic (polycarbonate, polyamide, polyethersulphone, polypropylene, silicone, and Tritan™) and purchased from 26 European Union countries, Canada, Switzerland and the USA.
It is surely important to underline that by comparing the data obtained from the LC-MS/MS analysis with those of the two immunoassays not one false positive result from the CL-ELISA or the LFIA was observed, supporting the reliability of these methods.
4. Conclusion
The application of the chemiluminescent detection resulted in the optimization of the ELISA, which led to a slight improvement of its sensitivity but also the expected, very useful reduction of the monoclonal antibody and conjugate consumption. The cost per analysis of this sensitive, fast, and high-throughput screening assay is in this way further reduced.The new, simple and fast LFIA has been revealed to be a very interesting method for the semi-quantitative determination of bisphenol A. The detection limit was very good for such a type of portable assay and the LFIA format was the only one suitable to solve the reproducibility problems of ELISA, connected with the property of BPA to absorb on plastic surfaces: the run can be entirely performed in a glass tube.
Bisphenol A is currently detected by chromatographic separation (LC) often coupled with mass spectrometry.14,34,36 To ascertain the reliability of the developed immunological methods we set up this simple LC-MS/MS method which was able to determine BPA at concentrations below the lower limits set by the present legal rules and with a LOQ lower than the LC-DAD official method. This LC-MS/MS method does not require any derivatization procedure or pre-concentration step. The significant reduction in the time per analysis has been further increased by employing the quicker separation on gradient instead of the isocratic one. The very high sensitivity achieved makes this assay a suitable control method to determine the absolute content of BPA and to reveal the migration of very low amounts of BPA from plastics.
Acknowledgements
This work was supported by a grant from the University of Bologna (RFO-Focused Fundamental Research Projects 2000-2011) grants.References
- The Bisphenol-A, http://www.bisphenol-a.org, accessed 04 July 2013.
- E. Diamanti-Kandarakis, J. P. Bourguignon, C. Giudice, R. Hauser, G. S. Prins, A. M. Soto, R. T. Zoeller and A. C. Gor, Endocr. Rev., 2009, 30, 293–342 CrossRef CAS PubMed.
- J. L. Ward and M. J. Blum, Evol. Appl., 2012, 5, 901–912 CrossRef CAS PubMed.
- S. C. Nagel, F. S. vom Saal and W. V. Welshons, Exp. Biol. Med., 1998, 217, 300–309 CrossRef CAS.
- N. J. MacLusky and F. Naftolin, Science, 1981, 211, 1294–1303 CAS.
- N. J. Cabaton, P. R. Wadia, B. S. Rubin, D. Zalko, C. M. Schaeberle, M. H. Askenase, J. L. Gadbois, A. P. Tharp, G. S. Whitt, C. Sonnenschein and A. M. Soto, Environ. Health Perspect., 2010, 119, 547–552 CrossRef PubMed.
- M. Munoz-de-Toro, C. Markey, P. R. Wadia, E. H. Luque, B. S. Rubin, C. Sonnenschein and A. M. Soto, Endocrinology, 2005, 146, 4138–4147 CrossRef CAS PubMed.
- Regulation of the European Commission (EU) No. 2011/10/EU of 14 January 2011 relating to materials and articles made of plastic material intended to come into contact with foodstuffs, http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2011:012:0001:0089:EN:PDF, accessed 04 July 2013.
- Canada Gazette Part II 144(21): 1806–18, 13 October 2010, http://www.gazette.gc.ca/rp-pr/p2/2010/2010-10-13/pdf/g2-14421.pdf, accessed 04 July 2013.
- Commission Directive 2011/8/EU of 28 January 2011 amending Directive 2002/72/EC, Off. J. Eur. Communities: Legis., 2011, 026, 11–14 Search PubMed.
- F. Baldi and A. Mantovani, Baby bottle: Struggling for safety, Food and Veterinary Toxicology Unit – ISS, http://www.iss.it/prvn/focu/cont.php?id=281%26lang=2%26tipo=42, accessed 04 July 2013.
- K. Aschberger, P. Castello, E. Hoekstra, S. Karakitsios, S. Munn, S. Pakalin and D. Sarigiannis, European Commission Joint Research Centre Institute for Health and Consumer Protection, http://publications.jrc.ec.europa.eu/repository/bitstream/111111111/14221/1/eur%2024389_bpa%20%20baby%20bottles_chall%20%20persp%20%282%29.pdf, accessed 04 July 2013.
- C. Simoneau, S. Valzacchi, V. Morkunas and L. Van den Eede, Food Addit. Contam., 2011, 28, 1763–1768 CAS.
- A. Ballesteros-Gómez, S. Rubio and D. Pérez-Bendito, J. Chromatogr., A, 2009, 1216, 449–469 CrossRef PubMed.
- Y. Feng, B. Ning, P. Su, H. Wang, C. Wang, F. Chen and Z. Gao, Talanta, 2009, 80, 803–808 CrossRef CAS PubMed.
- M. K. Mudiam, R. Jain, V. K. Dua, A. K. Singh, V. P. Sharma and R. C. Murthy, Anal. Bioanal. Chem., 2011, 401, 1695–1701 CrossRef PubMed.
- M. J. Moreno, P. d'Arienzo, J. J. Manclús and A. Montoya, J. Environ. Sci. Health, Part A, 2011, 46, 509–517 CrossRef CAS PubMed.
- G. O. Noonan, L. K. Ackerman and T. H. Begley, J. Agric. Food Chem., 2011, 59, 7178–7185 CrossRef CAS PubMed.
- A. Schecter, N. Malik, D. Haffner, S. Smith, T. R. Harris, O. Paepke and L. Birnbaum, Environ. Sci. Technol., 2010, 44, 9425–9430 CrossRef CAS PubMed.
- L. Viganò, E. Benfenati, A. van Cauwenberge, J. K. Eidem, C. Erratico, A. Goksøyr, W. Kloas, S. Maggioni, A. Mandich and R. Urbatzka, Chemosphere, 2008, 73, 1078–1089 CrossRef PubMed.
- H. Fukata, H. Miyagawa, N. Yamazaki and C. Mori, Toxicol. Mech. Methods, 2006, 16, 427–430 CrossRef CAS PubMed.
- M. Razee, Y. Yamini, S. Shariati, A. Esrafili and M. Shamsipur, J. Chromatogr., A, 2009, 1216, 1511–1514 CrossRef PubMed.
- X. Liu, Y. Yongsheng Ji, H. Zhang and M. Liu, Food Addit. Contam., Part A, 2008, 25, 772–778 CrossRef CAS PubMed.
- M. Farré, L. Kantiania and D. Barceló, Trends Anal. Chem., 2007, 26, 1100–1112 CrossRef PubMed.
- J. J. Manclùs, M. J. Moreno and A. Montoya, Anal. Methods, 2013, 5, 4244–4251 RSC.
- R. L. Gibson, Toxic Baby bottles: Scientific study finds leaching chemicals in clear plastic baby bottles, Environmental California Research & Policy Center, 2007, http://www.drldiego.com/docs/publications/Toxic_Baby_Bottles_sm.pdf, accessed 04 July 2013.
- Commission Directive 93/8/EEC of 15 March 1993, Off. J. Eur. Communities: Legis., 1993, 90, 22–25 Search PubMed.
- Directive 97/48/EC of 27 July 1997, Off. J. Eur. Communities: Legis., 1997, 222, 10–13 Search PubMed.
- Decreto Ministeriale 21/03/1973, Gazzetta Ufficiale, Supplemento Ordinario 104, 20/04/1973.
- S. Girotti, E. Maiolini, S. Ghini, E. Ferri, F. Fini, P. Nodet and S. Eremin, Anal. Lett., 2008, 41, 46–55 CrossRef CAS.
- A. E. Botchkareva, S. A. Eremin, A. Montoya, J. J. Manclùs, B. Mickova, P. Rauch, F. Fini and S. Girotti, J. Immunol. Methods, 2003, 283, 45–57 CrossRef CAS PubMed.
- G. Frens, Nature, Phys. Sci., 1973, 241, 20–22 CrossRef CAS.
- S. Girotti, A. Montoya, S. Eremin, M. J. Moreno, P. Caputo, M. D'Elia, L. Ripani, F. S. Romolo and E. Maiolini, Anal. Bioanal. Chem., 2010, 396, 687–695 CrossRef CAS PubMed.
- M. I. Santillana, E. Ruiz, M. T. Nieto, J. Bustos, J. Maia, R. Sendón and J. J. Sánchez, Food Addit. Contam., Part A, 2011, 28(11), 1610–1618 CrossRef CAS PubMed.
- Materials and articles in contact with foodstuffs – Plastics substances subject to limitation – Part 13: Determination of 2,2-bis(4-hydroxyphenyl)propane (bisphenol A) in food simulants UNI CEN/TS 13130-13:2005, http://www.cen.eu/CEN/Sectors/TechnicalCommitteesWorkshops/CENTechnicalCommittees/Pages/Standards.aspx?param=6175%26title=CEN/TC+194, accessed 04 July 2013.
- N. C. Maragou, A. Makri, E. N. Lampi, N. S. Thomaidis and M. A. Koupparis, Food Addit. Contam., Part A, 2008, 25, 373–383 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |