Surfactant-dispersed nanodiamond: biocompatibility evaluation and drug delivery applications†
Xiaoyong
Zhang
a,
ShiQi
Wang
a,
Meiying
Liu
b,
Junfeng
Hui
a,
Bin
Yang
a,
Lei
Tao
*a and
Yen
Wei
*a
aDepartment of Chemistry, Tsinghua University, Beijing, 100084, P. R. China
bBeijing National Laboratory for Molecular Sciences (BNLMS), Key Laboratory of Organic Solids, Laboratory of New Materials, Institute of Chemistry, Chinese Academy of Sciences, Beijing 100190, China. E-mail: leitao@mail.tsinghua.edu.cn; weiyen@tsinghua.edu.cn
First published on 24th June 2013
Abstract
Effective dispersion of nanodiamond (ND) in aqueous media especially in the physiological solution is of significant importance for its biomedical applications. Herein, the effect of surfactants on the water dispersibility of ND were investigated. On the basis of the dispersion results, biocompatibility as well as utilization of zwitterionic surfactant (lecithin) dispersed ND for intracellular delivery of doxorubicin hydrochloride were explored. We demonstrated that ND nanoparticles displayed enhanced dispersibility in both water and physiological solution in the present of lecithin. Given its facile, effective and low-cost features, the method for dispersion of ND nanoparticles described in this work will contribute significantly to the practical applications of ND.
1. Introduction
Since its first discovery in 1961, detonated nanodiamond (ND) has gained increasing research interest due to its excellent mechanical and physicochemical properties.1–5 Intensive research efforts have been devoted to developing such materials for various potential applications such as biosensing, cell imaging, intracellular drug delivery, etc.6–10 In particular, due to its small size, biocompatibility, specific photoluminiscent properties and inexpensive large scale production, the biomedical applications of ND have been extensively investigated over the past few years.11–17 It has been reported that surface-functionalized ND could adsorb or conjugate with various molecules such as proteins, drugs and nucleic acids for protein immobilization/separation and drug/gene delivery.18–23 Taking advantages of its bright fluorescence without photo-blinking and photo-bleaching, ND could also be used for the labeling and tracking of cancer cells and even small organisms.24–29 Furthermore, the biomedical applications of ND for tissue engineering have also been demonstrated in recent years.30,31 However, one of the main challenges for many of these biomedical applications is effective dispersion of ND in aqueous media, especially in physiological solution.32Over the last decade, considerable effort has been directed towards the dispersion of ND in aqueous and organic media.21,33–42 For example, Khabashesku et al. reported the synthesis of fluoro-nanodiamond via direct reaction of ND with mixed F2/H2 gas at temperature of 150–470 °C. Fluoro-nanodiamond can subsequently react with alkyllithium reagents, diamines and amino acids for preparation of a series of functionalized ND. These ND derivatives show improved solubility in polar organic solvents and reduced particle agglomeration.33 Surface functionalization of ND with polymers via “grafting to” and “grafting from” methods were also reported to improve the dispersibility of ND in organic and aqueous solutions.43–45 Furthermore, other surface functionalization methods based on ball milling, laser irradiation and click chemistry have also been developed.34,36–40,42,46 However, most of previous methods suffered from some significant limitations, such as time-consuming, rather cumbersome, and multi-step synthetic procedures. Therefore, the development of simple and effective methods for the dispersion of ND is still urgently needed for its practical applications.
Surfactants are amphiphilic molecules which can be obtained from commercial routes with rather low cost. Based on their charge, there are normally four types of surfactants: non-ionic, anionic, cationic and zwitterionic surfactants. Previous reports have confirmed that a number of nanomaterials including carbon nanotubes, gold nanoparticles, graphene, etc. could be dispersed in solution with the help of surfactants, which could modify the surface properties of the nanomaterials and prevent their aggregation over long time.47,48 However, to the best of our knowledge, dispersion of ND using surfactants and the biocompatibility of surfactant-modified ND have not been well studied.
In the current work, the effects of surfactants, i.e. non-ionic surfactants (polyoxyethylene lauryl ether (Brij35), Pluronic F127), anionic surfactants (sodium dodecyl benzene sulfonate (SDBS), dodecyl sulfate (SDS)), cationic surfactants (hexadecyl trimethyl ammonium bromide (CTAB)) and zwitterionic surfactants (lecithin (LEC)) on the stability of ND nanoparticles in aqueous media, especially in physiological solution (phosphate buffered saline (PBS)) were investigated (Scheme 1). On the basis of the dispersion results, the biocompatibility as well as the intracellular delivery of a model antitumor chemotherapeutic drug, doxorubicin hydrochloride (DOX), were explored. We expect that our method described here for the dispersion of ND nanoparticles will contribute significantly to the practical biomedical applications of ND.
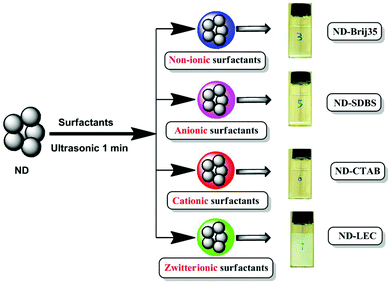 | ||
| Scheme 1 Schematic showing the dispersion of ND using surfactants with different surface charges (non-ionic, anionic, cationic and zwitterionic surfactants). The insets (no. 3, 5, 6, 7) are images of ND dispersed in PBS for 24 h using surfactants Brij35, SDBS, CTAB and LEC, respectively. | ||
2. Materials and methods
2.1. Materials and characterization
ND nanoparticles with diameters of 2–10 nm were purchased from Beijing Grish Hitech Co. Ltd. The as-received ND was used without further treatment. Surfactants Pluronic F127, Brij35, CTAB, SDS, SDBS and LEC and other chemical agents obtained from commercial routes were of at least analytical grade and used without further purification.Fourier transform infrared (FT-IR) spectra were obtained in transmission mode on a Perkin-Elmer Spectrum 100 spectrometer (Waltham, MA, USA).49 Typically, 4 scans at a resolution of 1 cm−1 were accumulated to obtain one spectrum. Transmission electron microscopy (TEM) images were recorded on a Hitachi 7650B microscope operated at 80 kV; the TEM specimens were made by placing a drop of the nanoparticle ethanol suspension on a carbon-coated copper grid. Thermal gravimetric analysis (TGA) was conducted on a TA instrument Q50 with a heating rate of 20 °C min−1. Samples weighing between 10 and 20 mg were heated from 25 to 500 °C in air flow (60 mL min−1), with N2 as the balance gas (40 mL min−1). UV-visible absorption spectra were recorded on UV/Vis/NIR Perkin-Elmer LAMBDA 750 spectrometer (Waltham, MA, USA) using quartz cuvettes of 1 cm path length. The fluorescence measurements were performed on a PE LS-55 spectrometer equipped with quartz cuvettes of 1 cm path length. The size distribution of ND and surfactant-modified ND nanoparticles in water and PBS were determined using a particle size analyzer (ZetaPlus, Brookhaven Instruments, Holtsville, NY). Each sample was ultrasonicated for 30 min prior to analysis. The reported values are the mean values of three measurements. The Brunauer–Emmett–Teller (BET) surface area, SBET, of the samples was determined from N2 adsorption–desorption isotherms obtained at 77 K using an ASAP 2010 Surface Area Analyzer (Micromeritics Instrument, USA). Prior to measurement, all samples were outgassed for 2 h at 473 K and 0.1 Pa. Surface areas were calculated by the BET method.
2.2. Surfactants-assisted dispersion of ND
The dispersion of ND nanoparticles with the help of surfactants was evaluated, four types of surfactants with different surface charges were investigated to understand the effects of surface charge properties of surfactants on the dispersion of ND. , 4 mg of ND and an equal quantity of surfactant (F127, Brij35, SDS, SDBS, CTAB or LEC) were added to 8 mL of H2O or PBS. The mixture was sonicated for about 1 min and then left to stand at room temperature for different periods of time. Photos were taken at different time intervals to evaluate the colloidal stability of ND suspensions. A suspension without the addition of a surfactant was used as the control. Furthermore, the size distribution of these suspensions was also determined to investigate the stability of ND suspensions. The insets of Scheme 1 are photos of ND suspensions dispersed in PBS for 24 h in the presence of surfactants (Brij35, SDBS, CTAB and LEC).2.3. Interactions between surfactants and ND
To understand the effect of surfactants on the stability of ND suspensions, the adsorption of surfactants F127 and Brij35 on ND was further investigated. In a typical experimental procedure, 60 mg of ND and 60 mg of F127/Brij35 were dispersed in 30 mL of deionized water. The mixture was ultrasonicated for 30 min and then deposited for 24 h to achieve adsorption equilibrium. Then the surfactant-dispersed ND nanoparticles were centrifuged at 7500 rpm for 30 min to remove the excess surfactants; the sediments were collected and dried under vacuum at 80 °C overnight. F127 and Brij35 coated ND were denoted as ND-F127 and ND-Brij35, respectively. The as-received ND as well as the surfactant-modified ND were well characterized by TEM, FT-IR and TGA.2.4. ND-LEC for drug delivery
| DLE (w/w%) = (weight of loaded |
| drug |
| /weight of ND nanoparticles) × 100% |
The cellular effects of ND-F127, ND-Brij35, ND-LEC and ND-LEC-DOX were determined using the WST assay as described in our previous reports.51–54 Briefly, cells were seeded in 96-well microplates at a density of 5 × 104 cells per mL in 160 μL of the respective media containing 10% FBS. After 24 h of cell attachment, the cells were incubated with 10, 20, 40, 80, 160 μg mL−1 of ND-LEC-DOX (the concentration of ND nanoparticles) for 24 h. Then the nanoparticles were removed and the cells were washed with PBS for three times. 10 μL of CCK-8 dye and 100 μL of Dulbecco's Modified Eagle's Medium (DMEM) cell culture media was added to each well and incubated for 2 h at 37 °C. Plates were then analyzed with a microplate reader (VictorIII, Perkin-Elmer). Measurements of dye absorbance were carried out at 450 nm, with the reference wavelength at 620 nm. The values were proportional to the number of live cells. The percent reduction of WST was compared to the control (cells not exposed to nanoparticles), which represented 100% WST reduction. Three replicate wells were used for each control and test concentrations per microplate, and the experiment was repeated three times. Cell survival was expressed as absorbance relative to that of untreated controls. Results are presented as mean ± standard deviation (SD).
2.5. Cellular uptake of ND-LEC-DOX complexes
Confocal microscopy images were taken to evaluate the cell uptake of ND-LEC-DOX.57–59 Briefly, on the day prior to treatment, cells were seeded in a glass bottomed dish with a density of 1 × 105 cells per dish. On the day of treatment, the cells were incubated with ND-LEC-DOX at a final concentration of 100 μg mL−1 (the concentration of ND) for 3 h at 37 °C. Afterwards, the cells were washed three times with PBS to remove the ND-LEC-DOX complex and then fixed with 4% paraformaldehyde for 10 min at room temperature. The cell nuclei were stained with Hoechst 33342. Cell images were taken with a Laser Scanning Confocal Microscope (LCSM) Zeiss 710 3-channel (Zeiss, Germany) with an excitation wavelength of 488 nm.3. Results and discussion
3.1. Stability of ND nanoparticles
The dispersibility of ND nanoparticles in water and PBS in the presence of different types of surfactants was investigated. Both bare ND and surfactant-dispersed ND suspensions are stable in water for at least 24 h (Fig. S1†). However, almost all of the ND suspensions were precipitated on bottom of bottles in PBS solution within 1 h, except for the LEC dispersed ND suspension (Fig. 1B). The LEC dispersed ND suspension retained excellent dispersibility after 24 h (Fig. 1D). The significantly different stability of ND in water and PBS is likely due to the ions in PBS, which may shield the electrostatic repulsion between ND nanoparticles. | ||
| Fig. 1 Colloidal stability of ND nanoparticles in PBS with concentration of 0.5 mg mL−1. (A) Immediately upon mixing and after (B) 1 h, (C) 6 h and (D) 24 h. (1) bare ND, (2) ND-F127, (3) ND-Brij35, (4) ND-SDS, (5) ND-SDBS, (6) ND-CTAB, (7) ND-LEC. | ||
The enhancement of dispersibility of ND nanoparticles in PBS by LEC was further confirmed by size distribution measurement. As listed in Table 1, the size distribution of bare ND nanoparticles in water is 2835 ± 365 nm. Their size distribution was reduced to 655 ± 11 nm in the presence of LEC. However, other surfactants showed no significant effects on the size distribution of ND nanoparticles based on size distribution measurements (Table 1). We speculated that the stability enhancement of ND nanoparticles in the present of anionic surfactants (Table 1, ND-SDS & ND-SDBS) is likely due to adsorption of surfactants on the surface of ND nanoparticles, thus changing their surface properties and stability in water. At least two factors may be involved in the dispersibility enhancement. The first one is that ND nanoparticles possess many hydrophobic areas, which can readily interact with surfactants’ hydrophobic segments through hydrophobic interactions, exposing the hydrophilic segments of the surfactants to the outside, which makes the surface of ND clusters hydrophilic and enhances their dispersibility in aqueous solution. The other factors which influence the stability of ND suspensions are their surface charges. As reported previously, positive charge exists on the surface of ND nanoparticles (its zeta-potential value is 19.2 ± 4.4 mV), which could interact with the negative charge of surfactants. However, the hydrophobic alkyl chains exposure to the outside could further interact with the hydrophobic segments of other surfactants, leading to the possible formation of surfactants bi-layers on the ND nanoparticles.
| H2O (nm) (PDI) | PBS (nm) (PDI) | |
|---|---|---|
| ND | 345 ± 19 (0.19) | 2835 ± 356 (0.07) |
| ND-F127 | 299 ± 1 (0.26) | 1653 ± 233 (0.01) |
| ND-Brij35 | 263 ± 3 (0.32) | 2227 ± 395 (0.24) |
| ND-SDS | 88 ± 1 (0.23) | 1914 ± 260 (0.01) |
| ND-SDBS | 72 ± 1 (0.20) | 3219 ± 555 (0.25) |
| ND-CTAB | 907 ± 37 (0.37) | 2265 ± 190 (0.1) |
| ND-LEC | 267 ± 10 (0.16) | 655 ± 11 (0.27) |
To better understand the interaction between ND nanoparticles and surfactants, more information including TEM, FT-IR spectra and TGA was examined. Compared with the as-received ND, the images of surfactants modified ND clusters are blurred, which is evidence that the surfactants are indeed coated on ND clusters (Fig. 2C). Despite the dispersibility of ND being enhanced by surfactants, the ND clusters are still present and are not deaggregated by the addition of surfactants.
 | ||
| Fig. 2 Representative TEM images of (A) ND and (B) ND-LEC, (C) ND-F127, (D) ND-Brij35. Scale bar = 50 nm. | ||
FT-IR spectra were used to give further evidence of the interaction between surfactants and ND clusters. As shown in Fig. 3A, a new band at 2900 cm−1 represents the C–H strength band is observed in the samples of ND-F127 and ND-Brij35, which provided direct evidence that surfactants were coated on the surface of ND clusters. Furthermore, we could also find the significant enhancement of intensity at the wavenumber of 1100 cm−1, which reflected that the density of C–O bonds is enriched. These results also demonstrated that the surfactants have adsorbed on the surface of ND. TGA curves of ND, ND-F127 and ND-Brij35 were obtained and compared to quantify the mass of surfactants adsorbed on ND. As shown in Fig. 3B, the mass loss of ND-F127 and ND-Brij35 is obviously greater than that of ND-OH. The significant mass loss of surfactant-modified ND is at the temperature of 300 °C, which is consistent with the decomposition temperature of surfactants (Fig. S2†). The percentages of surfactants coated on the surface of ND were calculated to be 13.6% and 13.9% for ND-F127 and ND-Brij35, respectively.
 | ||
| Fig. 3 (A) FT-IR spectra of ND-1, ND-1-F127 and ND-1-Brij35, the characteristic bands of F127 and Brij35 at 2900 and 1100 cm−1 were observed in the spectra, indicating that the surfactants were adsorbed on the surface of ND. (B) TGA curves of ND, ND-F127 and ND-Brij35, the mass percentages of on F127 and Brij35 adsorbed on the surface of ND were calculated to be 13.6% and 13.9%, respectively. | ||
The biocompatibility of surfactant-modified ND nanoparticles on A549 cells were evaluated using microscopy observation and a WST assay. As shown in Fig. 4B, no significant cell morphology changes were observed when cells incubated with 40 μg mL−1 of ND-LEC. Even when its concentration was increased to 160 μg mL−1, cells still strongly adhered to the cell plate (Fig. 4C), confirming the good biocompatibility of ND-LEC. Cell viability obtained from the WST assay further demonstrated the biocompatibility of ND-LEC. As shown in Fig. 4D, ND-LEC had a minimal effect on the viability of A549 cells at the concentrations of 20–320 μg mL−1 during an exposure time of 24 h. Even when the concentration of ND-LEC reached 320 μg mL−1, its cell viability was still greater than 75%, suggesting the potential biomedical applications of ND-LEC. Furthermore, the cell viability in the presence of ND-F127 and ND-Brij35 was also examined. Based on WST results, the half maximal inhibitory concentration (IC50) values of ND-F127, ND-Brij 35 and ND-LEC to A549 cells are 894.8, 2182.9 and 565.7 μg mL−1, respectively. Because of the well-known cytotoxicity of ionic surfactants, the biocompatibility of CTAB, SDS and SDBS modified ND nanoparticles were not examined in this work.60 Given its biocompatibility and water dispersibility in physiological solution, we expect that ND-LEC could be promising for biomedical applications.
 | ||
| Fig. 4 Microscopy images of A549 cells incubated with ND-LEC for 24 h, (A) control cells, (B) 40 μg mL−1 of ND nanoparticles, (C) 160 μg mL−1 of ND nanoparticles, scale bar = 200 μm. (D) Cell viability of ND-F127, ND-Brij35 and ND-LEC, concentrations of ND are ranged from 20–320 μg mL−1. | ||
ROS is an important biochemical indicator of intracellular oxidative stress, which is associated with a variety of biochemical processes and widely used to explain the cytotoxicity mechanism of extracellular stimuli. Herein, ROS production induced by ND-F127, ND-Brij35 and ND-LEC was further examined. As shown in Fig. 5, all the ND samples showed limited ROS level increase after they were incubated with cells for 8 h. Even when the incubation time was extended to 24 h, no significant ROS production was observed. Significantly different from many other nanomaterials, the ROS level decrease was also observed when cells were exposed to high concentration of ND nanoparticles.61,62 We speculated that this phenomenon might be ascribed to free radical scavenging capabilities of ND nanoparticles.63
 | ||
| Fig. 5 Generation of ROS from A549 cells determined by the hydrolysis of DCFH-DA after incubation with ND-F127, ND-Brij35 and ND-LEC for 8 h (A) and 24 h (B). | ||
Apart from the excellent biocompatibility, ND nanoparticles also have many other remarkable merits for biomedical applications. For example, ND nanoparticles readily form ND clusters with size greater than one hundred nanometers (Fig. 2). The ND clusters are novel porous nanostructures with BET surface areas greater than 350 cm3 g−1 and pore size mainly distributed between 2–10 nm (Fig. S3†), making it a promising candidate as drug container. In an attempt to explore their biomedical applications, the intracellular delivery of chemotherapeutic drug (DOX) using ND-LEC for cancer therapy was subsequently investigated. The amount of DOX loaded onto the composite was described as a function versus time and displayed in Fig. 6A, the adsorption of DOX onto ND nanoparticles quickly reached equilibrium after 6 h incubation. The inset of Fig. 6A gives further evidence of the efficient loading of DOX into ND nanoparticles. It can be seen that the suspension of ND-LEC-DOX turned to a pale red color after 36 h incubation. At these concentrations of nanoparticles (200 μg mL−1) and DOX (100 μg mL−1), drug loading efficiency can reach as high as 48% (w/w), which is much higher than that of ND-polyPEGMA.64 The saturated-loading efficiency of ND nanoparticles toward DOX can be further improved if the drug-loading parameters are optimized.65 In sharp contrast, DOX release from the ND-DOX complex is relative slow (Fig. 6B), suggesting its potential applications for controlled drug delivery.65
 | ||
| Fig. 6 (A) Adsorption of DOX onto ND, inset images are the DOX solution before (right) and after (left) adsorption by ND, concentration of DOX is 100 μg mL−1. (B) Fluorescence spectra of DOX release from ND-LEC-DOX complex in PBS (pH, 7.2–7.4) as a function of time. (C) Cell viability of ND-LEC-DOX complex, the concentration of ND is 20–320 μg mL−1, the corresponding concentrations of DOX ranges from 9.6–153.6 μg mL−1. (D–F) CLSM images of ND-LEC-DOX complex, (D) cells excited with a laser at 405 nm, cell nuclei dyed with Hoechst 33342, (E) cells excited with a laser at 488 nm, (F) merged images of D and E, the concentration of ND is 100 μg mL−1. | ||
In the following section, the anti-cancer effectiveness of ND-LEC-DOX complex was further determined. As shown in Fig. 6C, remarkable cytotoxicity was observed when cells were incubated with 20 μg mL−1 of ND-LEC-DOX for 24 h. When the concentration of ND-LEC-DOX was increased to 80 μg mL−1, almost no cells survived. Based on the cell viability data, the half maximal effective concentration (EC50) values of ND-LEC-DOX to A549 cells is 19.7 μg mL−1, which is corresponding to 9.8 μg mL−1 of DOX. The EC50 results are very close to the value of free DOX.64 Furthermore, the intracellular delivery of DOX was investigated using fluorescence microscopy and CLSM.66 As shown in Fig. S4,† red fluorescence from ND-LEC-DOX can be easily observed using fluorescence microscopy. Even when the concentration of ND-LEC-DOX is only 20 μg mL−1, the cellular uptake of ND-LEC-DOX can also been clearly distinguished (Fig. S5†). The internalization of ND-LEC-DOX was further investigated by CLSM images (Fig. 6D–F). These results demonstrated that ND-LEC-DOX could be transported into the cytoplasm, and DOX can be releaseed from ND-DOX and then enter into the cell nucleus. Considered its good biocompatibility and high drug-loading capability, ND-LEC should be promising for drug delivery.
4. Conclusion
In summary, the influence of surfactants on the dispersion of ND and the utilization of ND-LEC for intracellular delivery of chemotherapeutic drug DOX were reported for the first time. Results showed that the zwitterionic surfactant (LEC) could effectively disperse ND both in water and PBS. Optical microscopy observation, WST assay and ROS generation results demonstrated that ND-LEC is biocompatible with A549 cells. As evidenced by fluorescence microscopy and CLSM images, the intracellular delivery of an anticancer drug DOX into cells by ND-LEC and its release from ND-LEC-DOX complex were further confirmed. Given its excellent biocompatibility and stability in physiological solution, ND-LEC should have various promising biomedical applications such as controlled drug delivery.Acknowledgements
This research was supported by the National Science Foundation of China (no. 21134004, 21104039, 21201108), and the National 973 Project (no. 2011CB935700), China Postdoctoral Science Foundation (2011M500280).Notes and references
- P. S. DeCarli and J. C. Jamieson, Science, 1961, 133, 1821–1822 CAS.
- V. Y. Dolmatov, Russ. Chem. Rev., 2007, 76, 339–360 CrossRef CAS.
- A. Krueger, Chem.–Eur. J., 2008, 14, 1382–1390 CrossRef CAS.
- L. Lai and A. S. Barnard, Nanoscale, 2012, 4, 1130–1137 RSC.
- X. Cai, J. Hao, X. Zhang, B. Yu, J. Ren, C. Luo, Q. Li, Q. Huang, X. Shi and W. Li, Toxicol. Appl. Pharmacol., 2010, 243, 27–34 CrossRef CAS.
- A. Krueger, J. Mater. Chem., 2011, 21, 12571–12578 RSC.
- Y. L. Hsin, H. Y. Chu, Y. R. Jeng, Y. H. Huang, M. H. Wang and C. K. Chang, J. Mater. Chem., 2011, 21, 13213–13222 RSC.
- J. Li, Y. Zhu, W. Li, X. Zhang, Y. Peng and Q. Huang, Biomaterials, 2010, 31, 8410–8418 CrossRef CAS.
- X. Zhang, J. Yin, C. Peng, W. Hu, Z. Zhu, W. Li, C. Fan and Q. Huang, Carbon, 2011, 49, 986–995 CrossRef CAS.
- X. Zhang, J. Yin, C. Kang, J. Li, Y. Zhu, W. Li, Q. Huang and Z. Zhu, Toxicol. Lett., 2010, 198, 237–243 CrossRef CAS.
- L. Zhan, G. Yanxia, Z. Xiaoyong, Q. Wei, F. Qiaohui, L. Yan, J. Zongxian, W. Jianjun, T. Yuqin and D. Xiaojiang, J. Nanopart. Res., 2010, 13, 2939–2947 CrossRef.
- D. Ho, ACS Nano, 2009, 3, 3825–3829 CrossRef CAS.
- V. S. Bondar’, I. O. Pozdnyakova and A. P. Puzyr’, Phys. Solid State, 2004, 46, 758–760 CrossRef.
- A. M. Schrand, H. Huang, C. Carlson, J. J. Schlager, E. Ōsawa, S. M. Hussain and L. Dai, J. Phys. Chem. B, 2007, 111, 2–7 CrossRef CAS.
- Y. Yuan, X. Wang, G. Jia, J. H. Liu, T. Wang, Y. Gu, S. T. Yang, S. Zhen, H. Wang and Y. Liu, Diamond Relat. Mater., 2010, 19, 291–299 CrossRef CAS.
- Y. R. Chang, H. Y. Lee, K. Chen, C. C. Chang, D. S. Tsai, C. C. Fu, T. S. Lim, Y. K. Tzeng, C. Y. Fang and C. C. Han, Nat. Nanotechnol., 2008, 3, 284–288 CrossRef CAS.
- Y. C. Chen, D. C. Lee, C. Y. Hsiao, Y. F. Chung, H. C. Chen, J. P. Thomas, W. F. Pong, N. H. Tai, I. N. Lin and I. M. Chiu, Biomaterials, 2009, 30, 3428–3435 CrossRef CAS.
- R. Martin, M. Álvaro, J. R. Herance and H. García, ACS Nano, 2010, 4, 65–74 CrossRef CAS.
- M. Chen, X. Q. Zhang, H. B. Man, R. Lam, E. K. Chow and D. Ho, J. Phys. Chem. Lett., 2010, 1, 3167–3171 CrossRef CAS.
- E. K. Chow, X. Q. Zhang, M. Chen, R. Lam, E. Robinson, H. Huang, D. Schaffer, E. Osawa, A. Goga and D. Ho, Sci. Transl. Med., 2011, 3, 73ra21–10 CrossRef.
- X. Q. Zhang, M. Chen, R. Lam, X. Xu, E. Osawa and D. Ho, ACS Nano, 2009, 3, 2609–2616 CrossRef CAS.
- R. A. Shimkunas, E. Robinson, R. Lam, S. Lu, X. Xu, X. Q. Zhang, H. Huang, E. Osawa and D. Ho, Biomaterials, 2009, 30, 5720–5728 CrossRef CAS.
- A. H. Smith, E. M. Robinson, X. Q. Zhang, E. K. Chow, Y. Lin, E. Osawa, J. Xi and D. Ho, Nanoscale, 2011, 3, 2844–2848 RSC.
- I. P. Chang, K. C. Hwang and C. S. Chiang, J. Am. Chem. Soc., 2008, 130, 15476–15481 CrossRef CAS.
- N. Mohan, C. S. Chen, H. H. Hsieh, Y. C. Wu and H. C. Chang, Nano Lett., 2010, 10, 3692–3699 CrossRef CAS.
- S. J. Yu, M. W. Kang, H. C. Chang, K. M. Chen and Y. C. Yu, J. Am. Chem. Soc., 2005, 127, 17604–17605 CrossRef CAS.
- J. Tisler, G. Balasubramanian, B. Naydenov, R. Kolesov, B. Grotz, R. Reuter, J. P. Boudou, P. A. Curmi, M. Sennour and A. Thorel, ACS Nano, 2009, 3, 1959–1965 CrossRef CAS.
- A. M. Schrand, J. B. Lin, S. C. Hens and S. M. Hussain, Nanoscale, 2011, 3, 435–445 RSC.
- X. Zhang, S. Wang, C. Zhu, M. Liu, Y. Ji, L. Feng, L. Tao and Y. Wei, J. Colloid Interface Sci., 2013, 397, 39–44 CrossRef CAS.
- Q. Zhang, V. N. Mochalin, I. Neitzel, I. Y. Knoke, J. Han, C. A. Klug, J. G. Zhou, P. I. Lelkes and Y. Gogotsi, Biomaterials, 2011, 32, 87–94 CrossRef CAS.
- Q. Zhang, V. N. Mochalin, I. Neitzel, K. Hazeli, J. Niu, A. Kontsos, J. G. Zhou, P. I. Lelkes and Y. Gogotsi, Biomaterials, 2012, 33, 5067–5075 CrossRef CAS.
- L. Y. Chang, E. Ōsawa and A. S. Barnard, Nanoscale, 2011, 3, 958–962 RSC.
- Y. Liu, Z. Gu, J. L. Margrave and V. N. Khabashesku, Chem. Mater., 2004, 16, 3924–3930 CrossRef CAS.
- M. Ozawa, M. Inaguma, M. Takahashi, F. Kataoka, A. Kruger and E. Osawa, Adv. Mater., 2007, 19, 1201–1206 CrossRef CAS.
- T. Takimoto, T. Chano, S. Shimizu, H. Okabe, M. Ito, M. Morita, T. Kimura, T. Inubushi and N. Komatsu, Chem. Mater., 2010, 22, 3462–3471 CrossRef CAS.
- J. Cheng, J. He, C. Li and Y. Yang, Chem. Mater., 2008, 20, 4224–4230 CrossRef CAS.
- T. Meinhardt, D. Lang, H. Dill and A. Krueger, Adv. Funct. Mater., 2011, 21, 494–500 CrossRef CAS.
- A. Barras, J. Lyskawa, S. Szunerits, P. Woisel and R. Boukherroub, Langmuir, 2011, 27, 12451–12457 CrossRef CAS.
- L. Marcon, Z. Kherrouche, J. Lyskawa, D. Fournier, D. Tulasne, P. Woisel and R. Boukherroub, Chem. Commun., 2011, 47, 5178–5180 RSC.
- A. Pentecost, S. Gour, V. Mochalin, I. Knoke and Y. Gogotsi, ACS Appl. Mater. Interfaces, 2010, 2, 3289–3294 CAS.
- O. Shenderova, A. Koscheev, N. Zaripov, I. Petrov, Y. Skryabin, P. Detkov, S. Turner and G. Van Tendeloo, J. Phys. Chem. C, 2011, 115, 9827–9837 CAS.
- H. D. Wang, Q. Yang and C. H. Niu, Diamond Relat. Mater., 2010, 19, 441–444 CrossRef CAS.
- V. N. Mochalin, I. Neitzel, B. Etzold, A. M. Peterson, G. Palmese and Y. Gogotsi, ACS Nano, 2011, 5, 7494–7502 CrossRef CAS.
- H. Huang, Y. Wang, J. Zang and L. Bian, Appl. Surf. Sci., 2012, 258, 4079–4084 CrossRef CAS.
- X. Zhang, C. Fu, L. Feng, Y. Ji, L. Tao, Q. Huang, S. Li and Y. Wei, Polymer, 2012, 53, 3178–3184 CrossRef CAS.
- C. C. Li and C. L. Huang, Colloids Surf., A, 2010, 353, 52–56 CrossRef CAS.
- H. Hiramatsu and F. E. Osterloh, Chem. Mater., 2004, 16, 2509–2511 CrossRef CAS.
- L. Guardia, M. Fernandez-Merino, J. Paredes, P. Solis-Fernandez, S. Villar-Rodil, A. Martinez-Alonso and J. Tascon, Carbon, 2011, 49, 1653–1662 CrossRef CAS.
- X. Zhang, M. Liu, Y. Zhang, B. Yang, Y. Ji, L. Feng, L. Tao, S. Li and Y. Wei, RSC Adv., 2012, 2, 12153–12155 RSC.
- L. Xu, X. Zhang, C. Zhu, Y. Zhang, C. Fu, B. Yang, L. Tao and Y. Wei, J. Biomater. Sci., Polym. Ed., 2013 DOI:10.1080/09205063.2013.781934.
- Y. Zhu, X. Zhang, J. Zhu, Q. Zhao, Y. Li, W. Li, C. Fan and Q. Huang, Int. J. Mol. Sci., 2012, 13, 12336–12348 CrossRef CAS.
- Y. Zhang, B. Yang, X. Zhang, L. Xu, L. Tao, S. Li and Y. Wei, Chem. Commun., 2012, 48, 9305–9307 RSC.
- B. Yang, Y. Zhang, X. Zhang, L. Tao, S. Li and Y. Wei, Polym. Chem., 2012, 3, 3235–3538 RSC.
- X. Zhang, X. Zhang, B. Yang, S. Wang, M. Liu, Y. Zhang, L. Tao and Y. Wei, RSC Adv., 2013, 3, 9633–9636 RSC.
- X. Zhang, Y. Zhu, J. Li, Z. Zhu, W. Li and Q. Huang, J. Nanopart. Res., 2011, 13, 6941–6952 CrossRef CAS.
- X. Zhang, H. Qi, S. Wang, L. Feng, Y. Ji, L. Tao, S. Li and Y. Wei, Toxicol. Res., 2012, 1, 201–205 RSC.
- X. Zhang, S. Wang, L. Xu, Y. Ji, L. Feng, L. Tao, S. Li and Y. Wei, Nanoscale, 2012, 4, 5581–5584 RSC.
- J. Hui, X. Y. Zhang, Z. C. Zhang, S. Q. Wang, L. Tao, Y. Wei and X. Wang, Nanoscale, 2012, 4, 6967–6970 RSC.
- X. Zhang, X. Zhang, S. Wang, M. Liu, L. Tao and Y. Wei, Nanoscale, 2013, 5, 147–150 RSC.
- X. Zhang, X. Zhang, S. Wang, M. Liu, Y. Zhang, L. Tao and Y. Wei, ACS Appl. Mater. Interfaces, 2013, 5, 1943–1947 CAS.
- X. Zhang, W. Hu, J. Li, L. Tao and Y. Wei, Toxicol. Res., 2012, 1, 62–68 RSC.
- A. M. Schrand, L. Dai, J. J. Schlager, S. M. Hussain and E. Osawa, Diamond Relat. Mater., 2007, 16, 2118–2123 CrossRef CAS.
- R. Martín, C. Menchón, N. Apostolova, V. M. Victor, M. Álvaro, J. R. Herance and H. García, ACS Nano, 2010, 4, 6957–6965 CrossRef.
- X. Zhang, S. Wang, C. Fu, L. Feng, Y. Ji, L. Tao, S. Li and Y. Wei, Polym. Chem., 2012, 3, 2716–2719 RSC.
- H. Huang, E. Pierstorff, E. Osawa and D. Ho, Nano Lett., 2007, 7, 3305–3314 CrossRef CAS.
- X. Zhang, J. Hui, B. Yang, Y. Yang, D. Fan, M. Liu, L. Tao and Y. Wei, Polym. Chem., 2013 10.1039/c3py00489a.
Footnote |
| † Electronic supplementary information (ESI) available: Optical images, TGA, BET etc. See DOI: 10.1039/c3tx50021g |
| This journal is © The Royal Society of Chemistry 2013 |
