No time to lose—high throughput screening to assess nanomaterial safety
R.
Damoiseaux
*ac,
S.
George
bd,
M.
Li
d,
S.
Pokhrel
e,
Z.
Ji
d,
B.
France
d,
T.
Xia
d,
E.
Suarez
d,
R.
Rallo
df,
L.
Mädler
ce,
Y.
Cohen
d,
E. M. V.
Hoek
d and
A.
Nel
*bcd
aMolecular Screening Shared Resources, UC CEIN, NanoSystems Institute, University of California, 6522 CNSI, 570 Westwood, Los Angeles, California 90095-7277, USA. E-mail: Rdamoiseaux@mednet.ucla.edu
bDepartment of Medicine-Division of NanoMedicine, UC CEIN, NanoSystems Institute, University of California, 6522 CNSI, 570 Westwood Plaza, Los Angeles, California 90095-7277, USA. E-mail: ANel@mednet.ucla.edu
cCalifornia NanoSystems Institute at University of California, Los Angeles, CA, USA
dCenter for Environmental Implications of Nanotechnology, University of California, Los Angeles, USA
eIWT Foundation Institute of Materials Science, Department of Production Engineering, University of Bremen, Germany
fDepartament d'Enginyeria Informatica i Matematiques, Universitat Rovira i Virgili, Catalunya, Spain
First published on 7th February 2011
Abstract
Nanomaterials hold great promise for medical, technological and economical benefits. Knowledge concerning the toxicological properties of these novel materials is typically lacking. At the same time, it is becoming evident that some nanomaterials could have a toxic potential in humans and the environment. Animal based systems lack the needed capacity to cope with the abundance of novel nanomaterials being produced, and thus we have to employ in vitro methods with high throughput to manage the rush logistically and use high content readouts wherever needed in order to gain more depth of information. Towards this end, high throughput screening (HTS) and high content screening (HCS) approaches can be used to speed up the safety analysis on a scale that commensurate with the rate of expansion of new materials and new properties. The insights gained from HTS/HCS should aid in our understanding of the tenets of nanomaterial hazard at biological level as well as assist the development of safe-by-design approaches. This review aims to provide a comprehensive introduction to the HTS/HCS methodology employed for safety assessment of engineered nanomaterials (ENMs), including data analysis and prediction of potentially hazardous material properties. Given the current pace of nanomaterial development, HTS/HCS is a potentially effective means of keeping up with the rapid progress in this field—we have literally no time to lose.
 R. Damoiseaux | Dr Damoiseaux is the Scientific Director of the Molecular Shared Screening Resources (MSSR) at the California NanoSystems Institute of UCLA. He obtained his PhD degree while working on directed molecular evolution at the University of Lausanne (Switzerland) and joined the Novartis Institute for Functional Genomics (GNF) in La Jolla, CA, in 2001 and resides at the University of California, Los Angeles, since 2004. He oversees all drug discovery as well as the functional genomics projects at the MSSR and feels at home at the interface of chemistry, biology and engineering. His research diverse interests span biofilms, toxicological issues of nanomaterials and high throughput materials discovery and high throughput screening. |
 A. Nel | André Nel is a Professor of Medicine and Chief of the Division of NanoMedicine at UCLA. He directs the UC Center for the Environmental Impact of Nanotechnology. Dr Nel obtained his MD and Doctorate degrees in Cape Town, South Africa, and did Clinical Immunology and Allergy training at UCLA. He is the Director of the UCLA Asthma and Immunology Disease Center and a peer-selected member of the Best Doctors of America. His chief research interests are: (i) Nanomedicine, including drug and siRNA delivery systems; (ii) Nanobiology with interest in interfacial nanomaterial properties that can be used to improve biosafety; (iii) Nanotoxicology, with interest in predictive toxicological screening paradigms, nanomaterial safety testing, high throughput screening, and nanomaterial impact on the environment. |
Introduction
The production of engineered nanomaterials (ENMs) represents a scientific breakthrough in material design and the development of new consumer products that are expected to impact almost every industry.1–4 ENMs can be used for a host of applications: ranging from building materials, clothes, aircraft and motor vehicle parts and sports goods. ENMs are also anticipated to contribute significantly to the development of the next generation of electronics and will also play a key role in the development of new nanotherapeutics and diagnostics in the field of medicine.5–14Although nanotechnology is still considered a young science there is a rapid expansion of the number and identity of novel ENMs. This includes material compositions such as metals, metal oxides, metal chalcogenides (“quantum dots”), fullerenes, single walled nanotubes (SWNTs), multi-walled carbon nanotubes (MWCNTs), dendrimers, etc. Moreover, individual ENMs can be produced to exhibit different sizes, shapes, aspect ratios, crystallinity, surface modifications or hybridization with other nanomaterials. Thus, the number of commercial products that include ENMs has exceeded 1000 consumer products in 2009 and could grow to 104 materials within a decade.15
At the same time, it has to be acknowledged that nanomaterials are not new. The history of ultrafine materials can be traced to ∼2000 BC, including Chinese ink that contained ultrafine pigment particles or the colloidal gold suspensions that were used for the bright colors in painted church windows. Moreover, since the 50's, companies such as Cabot, Cristal (formerly SCM and Millennium Chemicals), DuPont, and Evonik (formerly Degussa) have been producing materials such as carbon black, SiO2, TiO2, and Al2O3 in millions of tons per year for use in paints, catalysts and surfactants. Currently, it is not only the quantity but also the diversity and novelty of nanomaterials that are prompting us to consider the toxicological properties of nanomaterials.
Soft-chemistry routes were developed to synthesize metal oxide ENMs with well defined shape, size and composition.16–18 Around the same time, flame spray pyrolysis (FSP) enabled the synthesis of complex nanomaterial systems using a scalable process. Due to the broad range of available precursor materials, FSP allows for a single step synthesis of a broad class of binary, mixed binary, ternary and mixed ternary metal oxide nanoparticles.19 Furthermore, control of the orientation of the particles and the capability to generate aligned and ordered nano- and microcrystallites onto a substrate were achieved by masking/patterning techniques such as template, lithography or by epitaxial electrodeposition.20–22
Almost all known ENM classes, including recently introduced materials, have been shown to pose some biological hazard under experimental conditions (Table 1). However, whether this is relevant from a health and environmental safety perspective is dependent on several additional factors such as the fate and transport of ENMs, exposure, bio-uptake and dose.
| ENM class | ENM property associated with toxicity | Toxicological phenomenon observed/mode of action |
|---|---|---|
| Metal | Shedding heavy metal (e.g.Ag, Cu, Pt) | DNA cleavage and damage leading to genotoxicity and mutation; heavy metal ions induced oxidative stress and inflammatory responses |
| Surface chemistries that affect the structure and function of proteins (e.g. Au) | Protein denaturation that may lead to inactivation of hormones or proteins, opening of cryptic epitopes that could lead to autoimmune diseases, protein fibrillation and accumulation of misfolded protein could lead to disease conditions such as Alzheimers | |
| Metal oxide | Dissolution and heavy metal release (e.g.ZnO) | Heavy metal ions induced oxidative stress and inflammatory responses |
| Electron–hole pair generation during photoactivation (e.g.TiO2) | Electron–hole pair generation during photoactivation leading to free radical generation | |
| Silica particles | Surface defects (e.g.SiO2) | Blood platelet, vascular endothelial and clotting abnormalities |
| Metal chalcogenide | Heavy metal release (e.g. CdSe quantum dots) | ROS generation, lipid peroxidation, DNA damage |
| Fullerenes and CNTs | Heavy metal contamination, aspect ratio > 5 | Fibrogenesis and tissue remodeling injury, oxygen radical production, GSH depletion, bio-catalytic mechanisms |
| Polymer | Cationic charge density | Membrane damage/leakage/thinning protein binding or unfolding responses/denaturation of proteins or fibrillation, lysosomal damage through proton pump inactivation or lysis of lysosomes |
Consequently, straightforward predictions about the toxicological behavior of the ENMs are not possible as one has to consider all the ENM properties that bring the materials in contact with the biophysical interface and all the biological properties that may contribute to hazard induction at the nano–bio interface.19 The inability to predict ENMs safety or hazard has raised considerable discussion about the safety of nanomaterials, including the best platform to use for material screening23–26 in academia, industry and regulatory agencies. It is clear from the experience dealing with >60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 industrial chemicals that it is not feasible to use animal testing as a primary screening platform because of the cost, duration and ethical concerns associated with such a venture.
000 industrial chemicals that it is not feasible to use animal testing as a primary screening platform because of the cost, duration and ethical concerns associated with such a venture.
Additional approaches are required to conduct initial screening. One possible solution to this bottleneck is the use of high throughput screening (HTS), one of the leading paradigms for drug discovery since the late 80's and early 90's. HTS has since also been adopted by the toxicological community as a screening tool in place of the labor-intensive and descriptive toxicological approaches from the past.27,28 As a result, an arsenal of toxicology assays and platforms that can be used in HTS mode has been adopted for chemicals and is now available for ENM toxicity screening. In fact, the National Academy of Sciences (NAS) has put forth a vision and strategy for using HTS approaches to assess multiple toxicants using a robust and mechanistic platform instead of using more descriptive and expensive animal studies.29 Similarly, the European Union has implemented the REACH program that requires all chemicals to undergo toxicology testing and has taken note of the EPA ToxCast program that utilizes HTS approaches similar to the NAS vision.30
Against this background, we will review the necessary characterization methods of nanomaterials, the current state of the art in high throughput toxicological approaches, its applications for ENM screening, as well as outlining the key steps for successful implementation of the HTS for ENMs. We will also review the possible pitfalls that are specific to ENM toxicity screening and will present examples of successful HTS screening approaches that have been developed for ENMs generating oxidative stress. We will review how HTS data can be used for ENMs hazard scoring and the important role of data mining approaches that use heat-map and self-organizing maps for data display and are useful for generating quantitative structure–activity relationships (QSARs).
HTS for ENM toxicity screening
The workflow that is required for ENM toxicity screening is outlined in Fig. 1. Typically, commercial ENMs are received as dry powders, which are subjected to a detailed class 1 characterization of the physicochemical properties in the “as-produced” state. This includes the assessment of properties such as the chemical composition, size, shape and phase. In the next step up to biological screening, the ENMs are suspended in deionized water and dispersed by sonification in the absence or presence of a dispersion agent. Class 2 characterization assesses the size, size distribution, state of dispersal/aggregation, stability and the ζ potential of the material in water as well as tissue culture media or other relevant assay buffers. This is followed by the actual HTS and data mining, processing and analysis. As evident from the workflow diagram, ENM characterization is a centrepiece of the approach without which it is impossible to relate to HTS outcome to individual material properties.31,32 It is important to understand that the reported ENM characteristics (e.g., size and surface charge) may vary depending on the specific experimental protocol that is used to initially prepare the ENMs. This has sparked efforts such as the Minimum Information of Nanoparticle Characterization (MINChar) to develop standards that ensure ENMs are characterized sufficiently to allow informed interpretation of data and cross-comparison of experimental results in a community of researchers. The MINChar set includes the following nine parameters: agglomeration/aggregation, chemical composition, crystal structure, particle size, purity, shape, surface area, surface charge and surface chemistry.33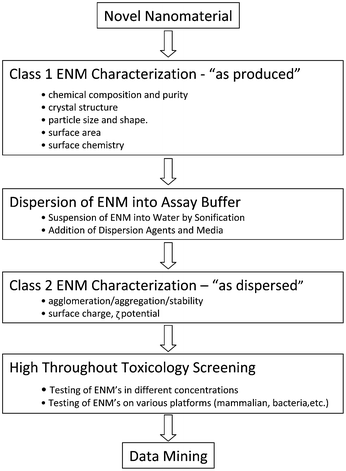 | ||
| Fig. 1 Assay development workflow for high throughput toxicity screening of nanomaterials. | ||
It is important to mention that the absent or incomplete nomenclature to classify ENM poses a considerable problem for correlating material properties with biological outcomes, including the generation of nano-QSARs. For example, carbon nanotubes (CNTs) at one point were assigned the same chemical abstract number (CAS number) as graphite, even though they differed in electrical conductivity, presence of metal impurities and different toxicological effects. Thus, while graphite tends to be biologically inert, CNTs are capable of eliciting granulomatous inflammation and fibrotic reactions in the lung. Not only is the ENM nomenclature made complex by differences in chemical composition, but a number of additional physicochemical properties could contribute to toxicological outcomes. It is therefore important to define a set of minimal class 1 parameters to understand the toxicological properties of ENMs in addition to defining more complicated class 2 parameters (e.g., dispersion, agglomeration and aggregation). The latter can be described as the acquirement of new properties due to the modification of the primary ENM properties in response to the composition of the suspending media, including the presence of mono- and divalent cations, pH, temperature and biological molecules, such as proteins. We will briefly review ENM characterization of class 1 and 2 properties (Table 2).
| Nanomaterial property | Available analytical techniques |
|---|---|
| Class 1: “as-produced”—morphology and structure | |
| Primary particle size, shape, and size distribution | TEM, SEM, AFM, ESEM, FIB-SEM, cryo-TEM |
| Fractal structure | TEM, SAXS, SANS |
| Pore size, porosity, and surface area | BET, SAXS, SANS |
| Crystallinity, framework structure, and crystal size | XRD, Raman, SAXS, NMR |
| Chemical composition | Raman, NMR, EDAX, FTIR, XPS, ICP |
| Elemental speciation and redox state | Raman, XAFS, XANES, NMR |
| Electronic, magnetic, and photonic properties | SAXS, Mossbauer, ESR, Raman, UV-Vis |
| “Dustiness” or tendency to aerosolize | EMPS, SMPS |
| Class 2: “as dispersed”—interfacial properties | |
| Dispersed size and size distribution | DLS, EMPS, SMPS, laser diffraction |
| Aggregate size | DLS, electro-acoustics |
| Charge density, pKa, PZC, ionization fraction | Direct titration in various suspending media |
| Surface (ζ) potential and IEP | EPM measurements in various suspending media |
| Surface tension components (LW, □ +, □ −) | Multiple probe liquid contact angle measurement |
| Roughness and chemical heterogeneity | AFM, CFM, FTIR, XPS, NMR, Raman |
Class 1 characterization of ENMs—“as produced”
A number of class 1 ENM properties have been reported to influence ENM hazard potential, including chemical composition, purity, size, shape, surface area, state of dispersion, crystallinity, and surface functionalization.25,26,34 The commonly used techniques and tools for assessing this class of physical–chemical properties include:Class 2 ENM characterization—“as dispersed”
While class 1 ENM properties are intrinsic material characteristics, class 2 properties (Table 2) arise from the interplay of the suspending medium (i.e. assay buffer) with the ENM surface. This could impact HTS analysis because ENMs may display altered or new properties, different from small molecules. While, for instance, most small molecules are easily dissoluble in the assay buffer, ENMs often form unstable suspensions and may therefore require dispersing agents to stabilize them in suspension.106 Even then, one has to be careful to ensure that the ENM suspension remains stable for the duration of the assay. This is not always possible and complicates data analysis as well as dosimetry calculations. Examples of dispersing agents that can be included in HTS assays include serum proteins, phospholipids, polyelectrolytes and a variety of surfactants.Serum albumin, including bovine serum albumin (BSA), is the most abundant protein in blood plasma. It is believed that nanoparticles are coated by BSA immediately upon the material addition to tissue culture medium. This non-specific binding occurs either through an electrostatic process or binding via a Ca2+ bridge.107 Fetal bovine serum (FBS) is another example of a biologically relevant dispersant that is often used as growth supplement for various mammalian cell culture media. Studies have shown that FBS at a typical concentration of 10% (v/v) in the culture medium can effectively improve nanoparticle dispersion. Another dispersing agent is dipalmitoylphosphatidylcholine (DPPC), which contains a hydrophilic acidic phosphate headgroup and two hydrophobic fatty acid tails. Since DPPC is the major component of lung surfactant, this phospholipid has often been used as a simple mimic of lung surfactant in nanotoxicity studies. Various other surfactants such as polyvinylpyrrolidone (PVP), Tween 80, and Pluronic F108 have been used for improving the dispersion of carbon allotropes (e.g., carbon nanotubes, carbon black, fullerenes). Special attention should be paid when using all of the aforementioned dispersing agents, as they modify the surface of the material and may therefore interfere in toxicity screening: for example, DPPC in relatively high doses has been shown to completely suppress the cytotoxicity and pro-apoptotic effects of quartz nanoparticles.108 This shielding can be avoided by using relatively low concentrations (e.g.160 µg mL−1). The commonly used techniques and tools for assessing class 2 properties include:
Liquid contact angle assessment elucidates additional information about surface chemistry, charge, and energy.47–51 Furthermore, the interfacial free energy at the site of cohesion (per unit area) is indicative of the inherent (thermodynamic) stability or aggregation propensity of a given material emerged in water:52,53 The higher the free energy at the site of adhesion, the higher the chance of an interaction taking place to lower the surface energy. Alternatively, the free energy of adhesion (per unit area) of two different materials (e.g., a nanoparticle and a sand grain) immersed in water gives an indication of inherent (thermodynamic) stability or deposition propensity. Thus, the higher the free energy, the more hydrophilic, stable, and deposition resistant is the material.53
ENM agglomeration can be measured by dynamic light scattering (DLS) techniques. For the purpose of a quick screening procedure to select a suitable assay buffer system in which to perform HTS, high throughput dynamic light scattering is an excellent choice. Dynamic light scattering measurement—as used in, e.g., the Dynapro™ plate reader (Wyatt Technology)—relies on measuring the Brownian motion that is proportional to the hydrodynamic diameter of the particles. DLS can be used as a convenient and effective technique for determining nanoparticle size distribution in the same format as being used for HTS, e.g., by using 384 well plates.
As DLS measures only the size of the particle in solution, it is important to keep in mind that precipitated materials at the bottom of the wells are not being measured. As an alternative method, a sedimentation approach using UV-vis spectroscopy can be used as a rough measure for evaluating nanoparticle sedimentation. Without aggregation and stability measurements, accurate dosing is impossible and will result in questionable HTS results. Unfortunately, few studies have addressed the stability of the dispersions towards assay development, including looking at the effect of proteins or other chemical surfactants on nanoparticle stability in tissue culture media.
Conceptual approach to high throughout toxicology screening
In the planning stage of selecting a HTS approach for ENM safety screening it is important to be cognizant of the workflow approaches that are used for toxicological screening of small molecules. The available approaches can be broken down into (i) classical plate reader-based assays using time-honored readouts such as luminescence, fluorescence, absorption, time-resolved fluorescence and fluorescence polarization or (ii) high content screening (HCS) approaches. Time-resolved fluorescence energy resonance transfer (TR-FRET) or fluorescence polarization (FP) platforms are less often used because of the high cost or sensitivity to interference from contaminants and temperature variations.Because traditionally plate reader assays have been used for small molecule safety assessment, they have yielded multiple platforms and readout modes that can be used for toxicological assessment. These platforms include the use of luminescence, fluorescence or absorption approaches to screen for mutagenicity or genotoxicity in bacteria, yeast or mammalian backgrounds. Examples include the bacterial-based VitoTox test54 that uses luciferase expression from the recN promoter in response to DNA damage. A similar-based approach in yeast is the GreenScreen assay55 in which the RAD54 promotor drives GFP expression that can be quantified by fluorescence. In the yeast-based RadarScreen56 the GFP is replaced by β-galactosidase, the activity of which is recorded by colorigenic or luminogenic substrates. Further up in the evolutionary chain, the GreenScreen HC assay makes use of a TK6 lymphoblastoma cell line that reports damage to a GADD45α promoter linked to a firefly luciferase reporter gene.
Cytotoxicity can be easily assessed by utilizing dyes such as propidium iodide (PI) and calcein-AM that can be assessed in a plate reader assay.57 While PI is impermeable to the intact surface membrane, calcein AM is a membrane permeable fluorescein-conjugated derivative that is cleaved by esterases and not capable of exiting the cell unless the membrane integrity is compromised. Cytotoxicity can also manifest itself as a depletion of cellular glutathione content that can be assessed by monochlobimane fluorescence58 or as increased production of radical oxygen species (ROS) that can be quantified by dichlorofluorescein acetate.59 The redox potential of the cell can also be followed using dyes such as MTT or alamarBlue.60 Luminescence-based alternatives are CytoLite (measures NADH),61ATP-Lite or CellTiter Glo (measure ATP).62 Another approach is to assess the generation of apoptosis, which is frequently induced by hazardous ENM, through a Z-DEVD-aminoluciferin63 assay that assesses caspase 3 and 7 activation or the use of LETD-aminoluciferin that assesses caspase 8 and 9 activation. Cleavage of these peptides by the caspases in question releases aminoluciferin that can be converted by firefly luciferase.
Another high throughput approach to toxicity assessment is to screen for endocrine disruptors using easy to perform reporter gene assays.64,65 This has allowed the assessment of androgen, estrogen, progesterone and glucocorticoid receptors in CHO cells through the use of luciferase and reporter gene assays.
HCS based assays are relatively new toxicological screening tools and provide significant advantages over the classical plate reader assays in terms of specificity and sensitivity. O'Brien et al. published a landmark study66 in 2006 during which they compared conventional single readouts to a five-parameter HCS assay that utilizes a combination of fluorescent compounds for toxicological assessment. The results were quite striking. While the single parameter readout had limited sensitivity (<25%) but high specificity (about 90%), the HCS approach showed a sensitivity of 93% and a specificity of 98%. Even more interestingly, although they used only a hepatocyte (HepG2) cell line, they were able to detect 92% of the toxic drugs, even those with non-hepatotoxic potential. Thus, HCS is potentially one of the best tools available for high throughput ENM screening. In fact, in 2008 Kotov recognized HCS as a universal tool for ENM cytotoxicity profiling and characterized cadmium telluride quantum dots and gold nanoparticles using this technique.115 However, while a variety of assays are available for toxicity screening of small organic molecules, it remains to be seen whether a wide range of ENM toxicology responses can be covered by HCS approaches. This will require discovery of ENM toxicological pathways in which the HCS approach can be used for hypothesis development about ENM properties capable of engaging those injury pathways. Last but not least novel assay platforms are also required for the assessment of the environmental impact of the ENM in organisms such as bacteria, yeast, zebrafish and oyster embryos. In order to elucidate the use of high throughput approaches for ENM screening, let us review the individual steps in such an effort.
Methodological approach to ENM toxicity testing
While the essential steps and workflow requirements are identical to small molecule screening (Fig. 2), it is important to elucidate special requirements for ENM toxicity screening, which overall poses more challenges than screening of small molecules: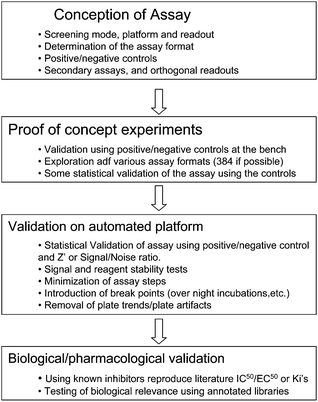 | ||
| Fig. 2 Assay development workflow. Adapted from: Molecular Screening, R. Damoiseaux, Handbook of Drug Discovery, Wiley and Sons, forthcoming. Reproduced with permission. | ||
| Toxicity type or paradigm | Analyte | Probes | Readout mode | Utility | Potential problem |
|---|---|---|---|---|---|
| Cytotoxicity | Cell number/proliferation | Hoechst 33342/DAPI | Fluorimetry/high content assay | Cell quantification, nuclear content | Background signal from NPs with blue fluorescence |
| Membrane leakage | Propidium iodide/Syto 9 | Fluorimetry/high content assay | Compromised cell membrane integrity | Background signal from NPs with red fluorescence (e.g. QDs) | |
| Membrane integrity | LDH assay | Absorbance at 490 nm | Cell viability | NPs may inhibit enzyme and/or absorb at 490 nm (CNTs, Ag) | |
| ATP | ATPlite™ | Luminescence | Mitochondrial activity and viability status of cells | Not appropriate for NPs that may inhibit enzyme and/or absorb light e.g. CNTs | |
| Mitochondrial membrane potential | JC1/TMRM/chloromethyl-X-rodhamine | Fluorimetry/high content assay | Loss of MMP | Background signal from NPs with red or green fluorescence | |
| Metabolic activity | MTT, WST-1, XTT | Absorbance | Mitochondrial activity and viability status of cells | NPs may inhibit enzyme and/or absorb light or substrates | |
| Intracellular calcium flux | Fluo-4/Fura 2-AM/Rho 2-AM | Fluorometry/high content assay | Increased intra-cellular calcium level | Background signal from NPs with green fluorescence | |
| Apoptosis | Calcein-AM | Fluorometry/high content assay | Mitochondrial membrane permeability transition (MPT) | Background signal from NPs with green fluorescence | |
| Genotoxicity | DNA cleavage | Micro-nuclei assay (HCS), BrDU incorporation | High content screening | Chromosome damage | Background signal from NPs with blue fluorescence |
| Inflammation | IL-1, IL-8, TNF-α | Antibody based ELISA or TR-FRET | Luminescence/TR-FRET | Expression level of inflammatory markers | Not appropriate for NPs that interfere with TR-FRET (e.g. absorb protein) or luminescence reactions |
| NF-κB and AP-1 activation | Reporter genes | Luminescence | Activation of inflammatory pathways | Not appropriate for NPs that may inhibit enzyme and/or absorb light | |
| Oxidative stress | GSH, ROS | Absorbance/HCS using fluorescence probes | Fluorometry/high content assay | Free radical generation, glutathione depletion | Not appropriate for NPs that may interfere with fluorescence output |
| Fibrogenesis | TGF-1β, collagen 1 and 3, MMPs | Fluorescence probe coupled antibody | Fluorometry/high content assay | Induction of fibrogenesis | Not appropriate for NPs that may interfere with fluorescence output |
In addition to screening of cells, whole-organism screening has been used successfully for toxicity studies.68 Whole organism screening using platforms such as the zebrafish allows for the screening of several developmental stages. While this type of screening can make the range of targets that can be covered even more complete, these methods have not yet been developed into high throughput approaches.
It is important for ENM toxicity screening to consider that cell-based and organism-based platforms differ in the temporal manifestation of their biological outcomes. Thus, while in vitro responses at cellular level often take place over the course of minutes to hours, the response in zebrafish embryos may require days and could therefore have an impact on the utility of the high throughput approach.
It is important to consider that the responsiveness of a particular cell type to potentially hazardous ENM properties could be target-specific and that cellular phenotype, lineage, transformed stage, and stage of differentiation are important. As an example, Weissleder showed that the predictive power of an in vitro ENM cytotoxicity assay is greatly improved by including multiple cell lines (4 different cell types) and using at least four different doses of the materials.69 Thus, the choice of cell type is essential to the success of HTS.
After the selection of the assay mode and toxicological paradigm for screening, one needs to decide on the appropriate assay readout. This choice has to take into consideration the type of material because several ENM compositions restrict the toxicological endpoints that can be assessed (Table 3). In contrast to small molecules, several unique ENM properties such as high adsorption capacity, optical properties, catalytic activity, etc. could interfere in the assay, leading to false positive or negative results.76 And old aphorism in HTS is “You get what you screen for” definitely holds true for toxicity screening. While designing this screening assay, care should be taken to counter the potential artifacts by inclusion of adequate controls and complementary assays that have an orthogonal readout. Thus, if the first assay relies on fluorescence, the second should use a luminescence readout. It is also important to consider that while some ENM cytotoxicity assays (e.g. MTT) use tetrazolium derivatives and optical density as readout, this can lead to invalid results with carbon based nanomaterials (carbon black, multiwalled carbon nanotubes, C60) or QDs due to dye/dye interactions and/or NM adsorption of the dye products.77 In a similar fashion the assessment of membrane integrity by measuring lactate dehydrogenase (LDH) activity in the culture supernatants through the use of the tetrazolium salts is prone to interference by ENMs. In this instance, the ENM could inhibit the enzymatic conversion and/or ability of the nanomaterial to absorb at the reading wavelength.78,79
While a HTS assay for each type of toxicity is theoretically possible, the cellular response that is being tested should be clinically or pathologically relevant. This approach could be supplemented with a larger panel of assays that cover additional injury mechanisms. General toxicity metrics such as cell number and redox potential are very useful to assess toxicity even at sub-lethal level. It is obvious that in addition to mammalian screening assays, HTS should also be implemented in bacteria, yeast and zebrafish in order to gain a comprehensive understanding of the ENM toxicity profiling in the environment.
Examples of successful implementation of HTS to assess ENM hazard potential
Although the use of HTS for ENM safety assessment is still in its infancy, some screening assays that have already been established have proven very useful and have allowed for prioritizing ENMS for in vivo testing based on the screening results. Shaw et al. devised a panel of assays in HTS format to evaluate the adverse biological effects of a panel of paramagnetic iron oxide nanoparticles with potential medical application in molecular imaging.69 They measured cytotoxic events such as ATP content, reducing equivalents, caspase-mediated apoptosis and mitochondrial membrane potential in different cell lines. Their analysis enabled them to identify nanomaterials with similar biologic activities elucidating the structure–activity relationships between particle properties and biological outcome.69 Moreover, they demonstrated that the predictive power of the in vitro assay increased as a function of the number of cell lines and cellular events tested.69Upon considering toxicological paradigms, one of the best HTS examples that have emerged is the screening of the ENM hazard potential according to the principle of cellular oxidative stress. In particular, we have demonstrated that ZnO nanoparticles are capable of engaging a toxicological pathway in a bronchial epithelial cell line that can be picked up by a multi-parametric oxidative stress assay that is also valid in the pro-inflammatory effects of the particles at cellular level.116 The in vitro screening, in turn, is analogous to the effect of welding fumes causing metal fume fever by exciting inflammation in human lung.82,83 The origin of this assay lies in prior demonstration that ambient particulate matter and a number of redox active ENMs are capable of inducing an incremental cellular oxidative stress according to ‘hierarchical oxidative stress paradigm’ (Fig. 3).19 Different levels of oxidant stress have been classified as antioxidant defense (Tier 1), pro-inflammatory (Tier 2), and cytotoxic (Tier 3) cellular responses, each of which are induced by specific sensing and signaling pathway. Thus, Tier 1 responses are initiated by the activation of the transcription factor, Nrf2, that transcriptionally activates the anti-oxidant response element (ARE) in the promoters of >200 phase II antioxidant defense, anti-inflammatory and cytoprotective genes. This pathway exerts anti-inflammatory effects and restores cellular redox equilibrium, including new GSH synthesis. This pathway may be engaged at relatively low levels of oxidative stress as well as electrophilic chemicals such as sulforaphane and resveratrol that are found in foods.84 Failure of this homeostatic response leads excessive ROS production to activate pro-inflammatory pathways such as the mitogen-activated protein kinase (MAPK) and nuclear factor-kappa B (NF-κB) cascades. In the context of oxidative stress, these pathways and the accompanying cytokines and chemokines represent Tier 2 of the hierarchical oxidative stress response and are relevant to particulate matter related disease processes such as asthma and atherosclerosis. Examples of pro-inflammatory responses in macrophages and epithelial cells include TNF-α and interleukin 8 (IL-8) production, respectively. These are also the characteristic cytokines that are found in the lung and bronchoalveolar lavage fluid of welders that develop metal fume fever.72 The increased redox in-equilibrium at the highest level of oxidative stress (Tier 3) triggers intracellular Ca2+ flux and perturbs mitochondria to the extent that could lead to triggering cellular apoptosis or apoptosis-necrosis. The cytotoxic response pathway can be triggered by a number of ENM types capable of ROS production as a result of the presence of transition metal impurities, ability to shed toxic metal ions, presence of electronically active surface (e.g., semiconductors), photoactivation or ability to catalyze biotic ROS production.70,71,116
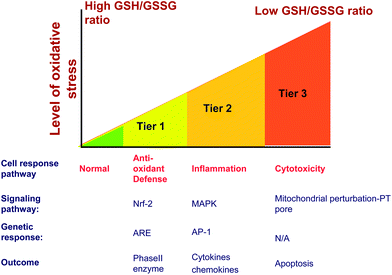 | ||
| Fig. 3 Hierarchical oxidative stress model explains cellular events occurring sequentially as the level of oxidative stress increases. A mild oxidative stress activates the protective antioxidant machinery in an attempt to restore the redox equilibrium (Tier 1). Higher levels of oxidative stress activate MAPK and Nf-κB cascade of molecular events leading to pro-inflammatory responses (Tier 2). Oxidative stress beyond the tolerable limit of cells triggers cell death pathways often manifested as increased cytosolic Ca2+ level, lowering of mitochondrial membrane potential, opening of mitochondrial permeability transition and cell membrane damage (Tier 3). | ||
A variety of biological assays and biomarkers have been developed for screening Tier 1, 2 and 3 cellular oxidative stress responses. Although, these endpoints were initially developed as individual assays that can be studied independently or in a batch, this is a labor intensive task that could theoretically be integrated in a multi-parametric assay that is useful for HTS (Table 3). After extensive experimentation we found compatible cellular dyes that could be used for contemporaneous screening of a number of Tier 3 responses by epifluorescence microscopy. This assay utilizes the DNA intercalating dye, Hoechst 33342, to assess cell number and nuclear size, 5,5′,6,6′-tetrachloro-1,1′,3,3′ tetraethylbenzimidazolylcarbocyanine iodide (JC 1) to assess perturbation of mitochondrial membrane potential (MMP), Fluo 4 to assess intracellular calcium flux and propidium iodide (PI) to report increased membrane permeability in dying cells.116 An example of the fluorescence changes that develop in cells being treated with ZnO nanoparticles is shown in Fig. 4. The data demonstrate that the drop in MMP (panel A) in a bronchial epithelial cell line (BEAS-2B) is accompanied by green cytoplasmic fluorescence distinct from the red fluorescence (and granular) staining of healthy mitochondria in untreated cells. Plasma membrane damage, leading to PI uptake and increased cellular [Ca2+] flux, results in increased red and green fluorescence, respectively (panel B). The functional link between cellular ROS production, [Ca2+]i flux, mitochondrial depolarization and plasma membrane damage is explained in Fig. 5 and explains the elements of a multiparametric assay that reflect sublethal and lethal oxidative stress responses.
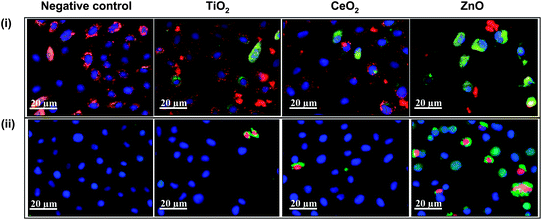 | ||
| Fig. 4 High content screening for assaying cytotoxic events triggered by nanoparticle interaction. (i) BEAS-2B cells subjected to nanoparticles, stained with nucleic acid staining Hoechst 33342 and mitochondrial dye JC1. Healthy cells show blue nuclei and red mitochondria (negative control, TiO2 and CeO2) while the mitochondrial depolarization causes the cytoplasm to fluoresce green (green cells in ZnO). (ii) BEAS-2B cells treated with nanoparticles and stained with a dye cocktail of Hoechst 33342, fluo-4 (for intracellular Ca2+) and propidium iodide (for accessing membrane damage). Healthy cells show blue nuclei, while damaged cells show green cytoplasm and red nuclei (cells in ZnO group). Depending upon the color profile, the percentage of cells affected can be assayed and nanoparticles can be ranked for their cytotoxic potential. Figure reproduced from ref. 116 with permission. | ||
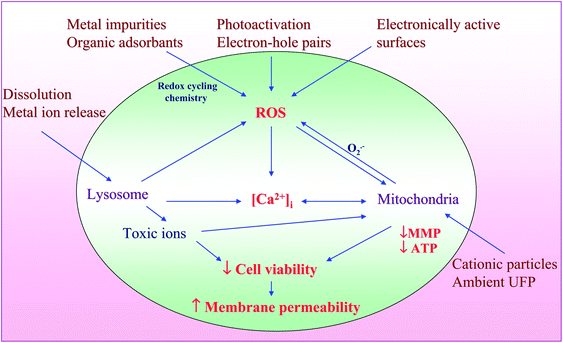 | ||
| Fig. 5 Hazardous nanomaterial properties leading to ROS generation and induction of integrated events in cytotoxicity pathway. Nanomaterials induce ROS production as a direct consequence of specific material properties or as a consequence of triggering cellular injury responses leading to oxidant radical generation. ROS production could trigger a range of oxidative stress effects as outlined in the hierarchical oxidative stress model. The induction of cellular toxicity at the highest level of oxidative stress involves a number of interrelated cellular responses that include intracellular Ca2+ release and mitochondrial perturbation leading to cell death with accompanying changes in cell membrane integrity and nuclear PI uptake. Figure reproduced from ref. 116 with permission. | ||
It is possible to assess Tier 2 oxidative stress responses in high throughput mode, either by measuring the cytokine release in the cell culture supernatant or by monitoring the activation of signaling cascades linked to transcriptional activation of cytokine promoters. This can be accomplished by developing appropriate reporter gene assays. The commonly tested human and murine inflammatory markers are interleukin-8 (IL-8), TNF-α and IL-6. The most common approach is to measure the production of cytokines or chemokines by classical ELISAs that can be combined with HTS, e.g., harvesting cellular supernatants through robotic processing. Because this requires a washing step that is problematic, the assay could be modified by using TR-FRET technology to measure cytokines in the supernatant with the cells. The detection of signaling pathways involved in inflammation through the use of reporter gene assays is feasible but not very specific. A number of key cellular signaling cascades are involved in oxidative stress, inflammation and cytotoxicity. These include the Nrf2, Jun kinase (JNK), NF-κB, AP-1 and P38 pathways.
While screening for oxidative stress is considered a pathway-specific approach, ENM toxicity screening in a bacterial or yeast platform can only be achieved by using phenotypic screening approaches. Among the phenotypic assays, growth assays utilizing optical density (OD) assessments offer the most straightforward approach to ENM toxicity screening. Frequently used OD metrics include doubling time and carrying capacity. However, these assays do not reflect the populations of bacteria that are dead or alive. A critical issue in bacterial ENM toxicity testing is the fact that frequently used growth media do not reflect the oligotrophic conditions that may be encountered in a real-life exposure environment. Assay buffers that reflect these oligotrophic conditions might be more helpful.
More information can be gained using assays that assess bacterial viability rather than measuring total biomass. These assessments can be performed by tailoring the assay buffer to reflect a real world exposure environment. One example is an assay that utilizes alamarBlue to assess the redox potential of the live bacteria.85 Another more universal assay is performance of ‘live/lead’ assays that distinguish the live from dead bacteria utilizing a pair of nucleic acid dyes, namely green-fluorescent SYTO® 9 and red-fluorescent propidium iodide (PI). While SYTO® 9 penetrates cell membranes and labels live and dead bacteria the PI stain only permeates the membrane of non-viable bacteria. Thus, the dead bacteria exhibit significantly less green fluorescence while showing a predominant red signal. The green to red fluorescence ratio represents a quantitative index of bacterial viability that is obtainable within minutes after the addition of the dyes.86–92 The live/dead assay has been applied to a variety of bacteria cultures including Acidianus brierleyi, Bacillus cereus, Bacillus subtilis, Clostridium perfringens, Escherichia coli, Klebsiella pneumoniae, Micrococcus luteus, Mycobacterium phlei, Pseudomonas aeruginosa, Pseudomonas syringae, Salmonella oranienburg, Serratia marcescens, Shigella sonnei, Staphylococcus aureus, Streptococcus pyogenes, and Sulfolobus acidocaldarius.89 The results obtained from the live/dead assay are normally in good agreement with standard plate count methods. This approach has been used by a number of research groups to evaluate the toxicological response to ENMs, including Ag, Au, ZnO, TiO2, QDs, etc.77,93–97 Since the assay is ratiometric it is more robust than standard single point assays. However, it needs to be mentioned that the assay requires establishment of a calibration curve which is different for each bacterial strain and the buffer in which the assay is being performed. Not surprisingly, the composition of the assay buffer exerts a big influence on the ENM toxicity assessment.
Data analysis and modeling of toxicological properties of ENMs
The lack of systematic toxicological assessment of well-characterized ENMs has hindered the development of predictive ENM toxicological models. HTS is one of the most powerful methods to acquire such data but has to be supplemented with equally powerful data analysis tools to establish property–activity relationships. HCS or HTS can be used to assess the influence of deliberate experimental variation of nanoparticle properties such as composition, size, charge, shape, aspect ratio, crystallinity, state of aggregation or dispersal. The same approach can be used to probe the effect of a variation in environmental parameters such as pH, temperature, salinity, solution chemistry and presence of natural dispersing agents. However, due to the lack of a priori quantitative models of NP toxicity, the challenge is to determine if the generated datasets span an adequate range (magnitude) of selected NP characteristics and toxicological parameters. Here, the development of models based on experimental data (i.e., data-driven models) has proven to be useful in situations where there is no prior knowledge of the complete set of equations governing the modeled phenomenon.The development of a computational framework to assess the toxicological effects of nanomaterials (NMs) requires a novel integrative approach that combines information technologies that allow data management through advanced algorithms and modeling approaches. The three main elements of the data analysis framework are illustrated in Fig. 6 and include: (a) an ENM database system, (b) modeling, and (c) hazard-ranking.
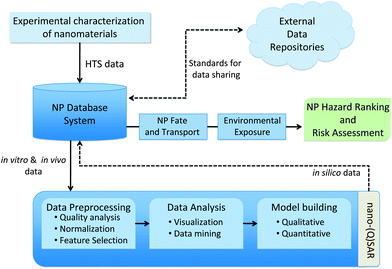 | ||
| Fig. 6 Components of a computational framework for predictive nanotoxicology. | ||
Experimental data related to ENM toxicity are derived from multiple assay platforms. The heterogeneous dataset is quite complex and highly dimensional as a result of ENM characteristics, screening approach, assay platform, etc. It has been recognized that, unlike single chemical molecules, chemical and structural descriptors that specify ENMs are a challenging endeavor. To this end, a number of initiatives have emerged that focus on standardizing the diverse aspects of nanotechnology datasets. Central to this effort is the development of a common nomenclature for nanoparticles to facilitate linkage of different ENM toxicity screening efforts. Towards this goal, a systematic approach towards codifying ENMs has been put forth by Gentleman and Chan18 while Tomalia98 suggested a system for ENM classification that is premised on atom mimicry and the periodic table of elements. Other efforts in this area are directed at the adoption of an approach similar to that used with Life Science Identifier99 in order to identify bioinformatics data objects. For example, the cancer nanotechnology laboratory portal is designed to facilitate data sharing activities by providing support for the annotation of ENM with physicochemical and in vitro characterizations, as well as the distribution of these characterizations and associated protocols in a secure fashion.
Although computational chemistry and predictive toxicology for nanoparticles are still in their infancy, these fields are fully developed for chemical compounds. Initiatives such as the EPA ToxCast™ program and the collaborative OpenTox initiative have already identified the main issues (e.g., ensuring unified access to distributed toxicological resources including data, computer models, validation and reporting) and provide the basis for the development of a predictive toxicology framework for chemicals. Lessons learned from these initiatives are key towards establishing an integrated framework for predictive toxicity of ENM.
HTS data pre-processing
The application of data-driven techniques for data mining and model development requires high data quality to ensure correctness and reliability of the results. Errors such as liquid handling errors contribute to cell growth variability or fluctuations in nanoparticle concentration and may introduce significant levels of noise in HTS data.100 Such errors can be partially compensated for by using replicate measurements and procedural quality controls such as positive and negative controls to reduce assay variability. Indeed, the use of control wells (i.e., cells not exposed to ENMs) helps to characterize plate-to-plate variability in multi-plate assays and to establish the proper assay background levels. The normalization of raw data between plates using control wells removes interplate variability and leads to experimental measurements that are comparable across plates. A detailed discussion of statistical methods for pre-processing HTS data can be found elsewhere.101Since the readouts from HTS plates are in the form of numbers, the categorization of the results requires distinguishing between significantly reactive and non-reactive ENMs. The common practice is to apply predefined activity thresholds to discriminate between active and non-active NPs. However, this approach tends to be biased towards the detection of highly active NPs whereas lower activities close to the threshold have a higher probability of being masked due to measurement errors. Different methods have been proposed in the literature to address this critical issue, including the use of local thresholds based on similarity clustering.102,103
HTS data analysis
Since HTS data are high dimensional, it is a common practice to use dimension reduction (or feature extraction) methods to project the data onto lower dimensional feature spaces more suitable for interpretation and visualization. Related to dimension reduction, cluster analysis can be used in the context of HTS data to extract similar activity patterns and to perform ENM grouping based on similar biological effects and physicochemical properties.117Agglomeration algorithms for clustering begin with each element as a separate cluster and merge them into successively larger clusters. Divisive algorithms begin with the whole set and proceed to divide it into successively smaller clusters. In contrast, partition clustering methods such as K-means determine a single cluster partition.117
Heat-map clustering has played an important role in HTS data analysis.104 Heat-maps provide, through row and/or column clustering, ordered representations of data that facilitate the identification of similarity patterns of large datasets. The same principles that have been used for heat-map based analysis of gene expression data can be adapted to the analysis of HTS nanoparticle toxicity datasets. Once properly pre-processed, the different plates containing data for specific HTS assays can be arranged in a form that is suitable for heat-map analysis. Fig. 7 illustrates the use of heat-maps to analyze a HTS dataset obtained from the experimental setup described in Fig. 7a. Fig. 7b depicts the cytotoxic activity patterns projected over the heat-map where rows and columns correspond to nanoparticles at a specific concentration and to the measured cytotoxic effects (after some exposure time), respectively. In this representation blue colors indicate no harmful activity while yellow/orange indicates adverse effects with respect to the oxidative stress paradigm presented above, e.g., reactive oxygen species (ROS) generation, membrane damage, loss of mitochondrial membrane potential and intracellular Ca flux. Specific patterns of activity can be extracted from this representation.
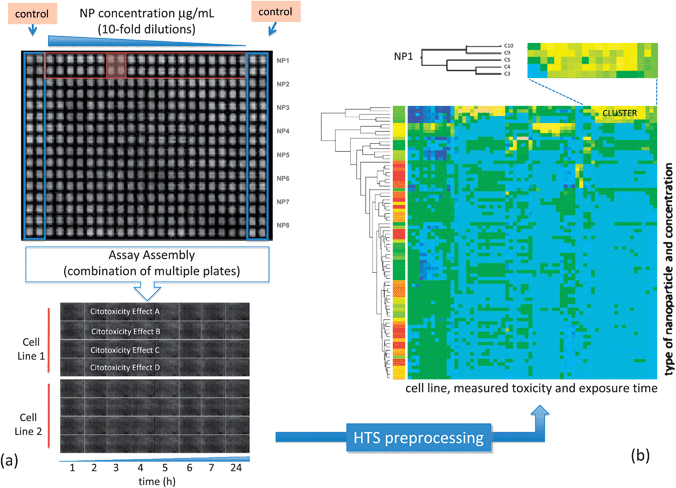 | ||
| Fig. 7 (a) Setup information for a HTS cytotoxicity assay showing plate arrangement. (b) Clustered heat-map corresponding to this data, blue colors indicate no activity, while yellow indicates toxic activity. Visual inspection identifies clusters of biologically active nanoparticles. | ||
A complementary approach to heat-maps is the use of topology preservation methods which are capable of maintaining, in the projected space, the distances measured in the original feature space. The self-organizing map (SOM) algorithm105,117 is the most important of these algorithms. The SOM approach provides a valuable computational tool that can facilitate knowledge extraction from high dimensional HTS data. Compared with heat-maps, SOM projections facilitate the discovery of hidden relationships between variables by providing topologically ordered representations of the input space. The SOM is based on an unsupervised competitive learning approach through which the SOM grid is generated. The initial generation of the SOM grid is entirely data-driven and once the initial generation of the SOM grid is finished, the grid can be used for clustering of the data. The clustering structure of the data can be visualized over the SOM grid by displaying the distances between cluster centers (distance matrices) or by projecting features onto the different component planes which allows for a more detailed evaluation of the parameters in these planes.116,117
Summary
There is the expectation that nanotechnology will help solve some of mankind's most pressing problems such as energy, environmental cleanup, preservation of water resources, improved diagnostics and therapeutics, etc. It is already clear that ENMs have the potential to revolutionize every aspect of daily life with applications ranging from electronics, building materials, airplane bodies, clothes, cosmetic products, etc. However, because of their unique properties, ENMs also have the potential to create hazardous interactions at the nano–bio interface. For example, some single walled CNTs have the capacity to induce granulomatous inflammation and fibrosis in rodent lung exposure models.14 The presence of CNTs in consumer products such as brake pads or tires may create inhalable wear particles and demonstrates the pressing need to establish screening procedures for ENM hazard assessment as well as for prioritizing animal experimentation. The use of animals as a primary screening platform does not have the capacity to keep up with the rate of expansion of nanotechnology. HTS has the necessary capacity as a primary screening tool and can be applied for ENM similar to its use in the pharmaceutical industry for flagging potentially toxic small organic molecules. HCS in particular appears to be a very promising platform for ENM screening. High throughput methods have helped the pharmaceutical industry to save resources through early recognition of the potential toxicological properties of drug candidates. A similar application in the nanotechnology industry should allow us to identify materials with potential hazardous properties and prioritizing animal testing. The idea would be to establish predictive toxicological paradigms where in vitro screening principles can be established that are representative of in vivo injury responses. Having established the relevance, observations can then be accomplished in vitro with only occasional in vivo relevance checks. How much of what we currently know about in vitro ENM toxicology is directly applicable to human disease is currently unknown. However, the screening for oxidative stress responses does represent a predictive platform for ambient air pollution particles where there is some correlation between in vitro oxidative stress responses and in vivo outcomes of pro-inflammatory disease processes such as asthma and atherosclerosis.109–113 It would appear that the in vitro injury responses that register in our multi-parametric assay also have some in vivo relevance, e.g., linkage of cellular oxidative stress by ZnO to metal fume fever in welders.112 There is also emerging data that suggest that pro-inflammatory responses in bronchial epithelial cells may be predictive of some classes of ENM to generate neutrophilic inflammation in rodent lungs.113,114 Thus, it would appear as if the shallow dose–response characteristics of nuisance dusts such as carbon black and TiO2in vitro as well as in animal lung differ from the steep dose–response characteristics of more toxic ENMs such as Co, Ni, Cu and crystalline silica.113,114 Moreover, these differences in pro-inflammatory effects were in some cases also reflected by higher levels of oxidative stress in response to the more potent lung toxicant.We can expect to uncover more ENM toxicity paradigms and injury pathways through the implementation of high throughput methods, which could be used together with compositional and combinatorial ENM libraries to also establish qualitative structure–activity relationships that will reveal more of the secrets at the nano–bio interface. In addition, this knowledge will also be able to promote safe-by-design methods to produce ENMs with improved safety profiles. However, the lack of a systematic ENM nomenclature has stymied our efforts at establishing robust structure–activity relationship to date. As a result, there are a lot of gaps in our understanding of the specific ENM properties that are responsible for hazard generation under biological conditions. It is also becoming clear that chemisorption or the presence of impurities such as transition metals contribute to ENM toxicity. This is particularly relevant from the perspective that there are potentially a large number of commercially available nanomaterials that are not adequately characterized and may contain impurities that lead to toxicity.
While HTS methodologies to date have focused largely on mammalian cellular screening systems it is necessary to develop similar screening efforts for prokaryotic systems and lower environmental life forms to also ENM safety screening for protection of the environment. For example, we have instigated high throughput screening assays in E. coli and S. cervisiae using genome-wide knockout libraries to study the applicability of these platforms to ENM eco-toxicological screening. Similar efforts are on the way in the use of zebrafish and oyster embryos. Last but not least efforts are on the way for using reporter cell lines selected to represent the most important cell types, lineages and cellular responses to perform HTS ENM screening.
Conclusion
HTS is an extremely useful methodology that has enhanced the ability for knowledge generation about ENM hazard in our multi-disciplinary University of California Center for the Environmental Impact of Nanotechnology. The data generated to date show that a number of toxicological paradigms can be effectively incorporated into high throughput or high content efforts and that the resulting data can be used for initial hazard ranking and prioritizing of ENM screening in the wider health and safety context.Acknowledgements
This work is supported by the National Science Foundation and the Environmental Protection Agency under Cooperative Agreement Number DBI 0830117. Any opinions, findings, conclusions or recommendations expressed herein are those of the author(s) and do not necessarily reflect the views of the National Science Foundation or the Environmental Protection Agency. This work has not been subjected to an EPA peer and policy review. Key support was provided by the US Public Health Service Grants U19 ES019528 (UCLA Center for NanoBiology and Predictive Toxicology), RO1 ES016746, and RC2 ES018766.References
- R. H. Baughman, A. A. Zakhidov and W. A. de Heer, Carbon nanotubes—the route toward applications, Science, 2002, 297(5582), 787–792 CrossRef CAS.
- N. A. Kotov, J. O. Winter, I. P. Clements, E. Jan, B. P. Timko, S. Campidelli, S. Pathak, A. Mazzatenta, C. M. Lieber, M. Prato, R. V. Bellamkonda, G. A. Silva, N. W. S. Kam, F. Patolsky and L. Ballerini, Nanomaterials for neural interfaces, Adv. Mater., 2009, 21(40), 3970–4004 CrossRef CAS.
- K. Ariga, X. L. Hu, S. Mandal and J. P. Hill, By what means should nanoscaled materials be constructed: molecule, medium, or human?, Nanoscale, 2010, 2(2), 198–214 RSC.
- C. R. Patra, R. Bhattacharya, D. Mukhopadhyay and P. Mukherjee, Fabrication of gold nanoparticles for targeted therapy in pancreatic cancer, Adv. Drug Delivery Rev., 2010, 62(3), 346–361 CrossRef CAS.
- J. Chen and F. Y. Cheng, Combination of lightweight elements and nanostructured materials for batteries, Acc. Chem. Res., 2009, 42(6), 713–723 CrossRef CAS.
- A. Dong, H. Yu, F. Wang and W. E. Buhro, Colloidal GaAs quantum wires: solution–liquid–solid synthesis and quantum-confinement studies, J. Am. Chem. Soc., 2008, 130(18), 5954–5961 CrossRef CAS.
- J. L. G. Fierro, Metal Oxides: Chemistry and Applications, CRC Press, Boca Raton, FL, 2006 Search PubMed.
- X. L. Gao, J. Chen, J. Y. Chen, B. X. Wu, H. Z. Chen and X. G. Jiang, Quantum dots bearing lectin-functionalized nanoparticles as a platform for in vivo brain imaging, Bioconjugate Chem., 2008, 19(11), 2189–2195 CrossRef CAS.
- J. Goldberger, R. Fan and P. D. Yang, Inorganic nanotubes: a novel platform for nanofluidics, Acc. Chem. Res., 2006, 39(4), 239–248 CrossRef CAS.
- R. C. Haddon, Carbon nanotubes, Acc. Chem. Res., 2002, 35(12), 997–997 CrossRef.
- A. I. Hochbaum and P. D. Yang, Semiconductor nanowires for energy conversion, Chem. Rev., 2010, 110(1), 527–546 CrossRef CAS.
- J. F. Liu and G. P. Miller, Field-assisted nanopatterning, J. Phys. Chem. C, 2007, 111(29), 10758–10760 CrossRef CAS.
- C. I. Richards, J. C. Hsiang, D. Senapati, S. Patel, J. H. Yu, T. Vosch and R. M. Dickson, Optically modulated fluorophores for selective fluorescence signal recovery, J. Am. Chem. Soc., 2009, 131(13), 4619–4621 CrossRef CAS.
- Y. L. Zhao and J. F. Stoddart, Noncovalent functionalization of single-walled carbon nanotubes, Acc. Chem. Res., 2009, 42(8), 1161–1171 CrossRef CAS.
- L. Muller-Kuhrt, Putting nature back into drug discovery, Nat. Biotechnol., 2003, 21(6), 602 CrossRef.
- M. Niederberger, Nonaqueous sol–gel routes to metal oxide nanoparticles, Acc. Chem. Res., 2007, 40(9), 793–800 CrossRef CAS.
- G. R. Patzke, F. Krumeich and R. Nesper, Oxidic nanotubes and nanorods—anisotropic modules for a future nanotechnology, Angew. Chem., Int. Ed., 2002, 41(14), 2446–2461 CrossRef.
- S. Pokhrel, C. E. Simion, V. S. Teodorescu, N. Barsan and U. Weimar, Synthesis, mechanism, and gas-sensing application of surfactant tailored tungsten oxide nanostructures, Adv. Funct. Mater., 2009, 19(11), 1767–1774 CrossRef CAS.
- A. Nel, T. Xia, L. Mädler and N. Li, Toxic potential of materials at the nanolevel, Science, 2006, 311(5761), 622–627 CrossRef CAS.
- L. L. Hench and J. K. West, The sol–gel process, Chem. Rev., 1990, 90(1), 33–72 CrossRef CAS.
- D. P. Sanders, Advances in patterning materials for 193 nm immersion lithography, Chem. Rev., 2010, 110(1), 321–360 CrossRef CAS.
- J. A. Switzer, R. V. Gudavarthy, E. A. Kulp, G. J. Mu, Z. He and A. J. Wessel, Resistance switching in electrodeposited magnetite superlattices, J. Am. Chem. Soc., 2010, 132(4), 1258–1260 CrossRef CAS.
- R. Service, Nanotechnology—can high-speed tests sort out which nanomaterials are safe?, Science, 2008, 321(5892), 1036–1037 CrossRef CAS.
- R. F. Service, American chemical society meeting: nanomaterials show signs of toxicity, Science, 2003, 300(5617), 243–243.
- V. L. Colvin, The potential environmental impact of engineered nanomaterials, Nat. Biotechnol., 2003, 21(10), 1166–1170 CrossRef CAS.
- G. Oberdorster, E. Oberdorster and J. Oberdorster, Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles, Environ. Health Perspect., 2005, 113(7), 823–839 CrossRef CAS.
- D. A. Pereira and J. A. Williams, Origin and evolution of high throughput screening, Br. J. Pharmacol., 2007, 152(1), 53–61 CrossRef.
- J. A. Dimasi, Risks in new drug development: approval success rates for investigational drugs, Clin. Pharmacol. Ther., 2001, 69(5), 297–307 CrossRef CAS.
- S. Gibb, Toxicity testing in the 21st century: a vision and a strategy, Reprod. Toxicol., 2008, 25(1), 136–138 CrossRef CAS.
- R. S. Judson, K. A. Houck, R. J. Kavlock, T. B. Knudsen, M. T. Martin, H. M. Mortensen, D. M. Reif, D. M. Rotroff, I. Shah, A. M. Richard and D. J. Dix, In vitro screening of environmental chemicals for targeted testing prioritization: the ToxCast project, Environ. Health Perspect., 2010, 118(4), 485–492 CAS.
- M. Auffan, J. Rose, J.-Y. Bottero, G. V. Lowry, J.-P. Jolivet and M. R. Wiesner, Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective, Nat. Nanotechnol., 2009, 4(10), 634–641 CrossRef CAS.
- D. B. Warheit, How meaningful are the results of nanotoxicity studies in the absence of adequate material characterization?, Toxicol. Sci., 2008, 101(2), 183–185 CrossRef CAS.
- J. W. Card and B. A. Magnuson, Proposed minimum characterization parameters for studies on food and food-related nanomaterials, J. Food Sci., 2009, 74(8) Search PubMed , vi–vii.
- K. W. Powers, S. C. Brown, V. B. Krishna, S. C. Wasdo, B. M. Moudgil and S. M. Roberts, Research strategies for safety evaluation of nanomaterials. Part VI. Characterization of nanoscale particles for toxicological evaluation, Toxicol. Sci., 2006, 90(2), 296–303 CAS.
- S. Lowell, Characterization of porous solids and powders: surface area, pore size and density, 2006, 15–18 Search PubMed.
- A. Bootz, V. Vogel, D. Schubert and J. Kreuter, Comparison of scanning electron microscopy, dynamic light scattering and analytical ultracentrifugation for the sizing of poly(butyl cyanoacrylate) nanoparticles, Eur. J. Pharm. Biopharm., 2004, 57(2), 369–375 CrossRef CAS.
- A. Jillavenkatesa and J. F. Kelly, Nanopowder characterization: challenges and future directions, J. Nanopart. Res., 2002, 4(5), 463–468 CrossRef CAS.
- A. Bootz, T. Russ, F. Gores, M. Karas and J. Kreuter, Molecular weights of poly(butyl cyanoacrylate) nanoparticles determined by mass spectrometry and size exclusion chromatography, Eur. J. Pharm. Biopharm., 2005, 60(3), 391–399 CrossRef CAS.
- H. Fritz, M. Maier and E. Bayer, Cationic polystyrene nanoparticles: preparation and characterization of a model drug carrier system for antisense oligonucleotides, J. Colloid Interface Sci., 1997, 195(2), 272–288 CrossRef CAS.
- M. Wiesner, Environmental Nanotechnology: applications and impacts of nanomaterials, McGraw-Hill Professional, 2007.
- S. Pokhrel, J. Birkenstock, M. Schowalter, A. Rosenauer and L. Mädler, Growth of ultrafine single crystalline WO3 nanoparticles using flame spray pyrolysis, Cryst. Growth Des., 2010, 10(2), 632–639 CrossRef CAS.
- S. J. L. Billinge, Strain, Nano-phase Separation, Multi-scale Structures and Function of Advanced Materials, in Intrinsic Multiscale Structure and Dynamics in Complex Electronic Oxides, Proceedings, ed. A. R. Bishop, S. R. Shenoy and S. Sridhar, World Scientific Publ. Co. Pte. Ltd., Singapore, 2003, pp. 25–40 Search PubMed.
- P. Deb, T. Biswas, D. Sen, A. Basumallick and S. Mazumder, Characteristics of Fe2O3 nanoparticles prepared by heat treatment of a nonaqueous powder precipitate, J. Nanopart. Res., 2002, 4(1–2), 91–97 CrossRef CAS.
- M. Ohnuma, K. Hono, H. Onodera, S. Ohnuma, H. Fujimori and J. S. Pedersen, Microstructures and magnetic properties of Co–Al–O granular thin films, J. Appl. Phys., 2000, 87(2), 817–823 CrossRef CAS.
- C. R. Hubbard, E. H. Evans and D. K. Smith, The reference intensity ratio, I/Ic, for computer simulated powder patterns, J. Appl. Crystallogr., 1976, 9(2), 169–174 CrossRef.
- Y.-L. Lo, T.-C. Yu, L.-S. Su and Y.-S. Huang, Modified total intensity ratio methods for measuring cell gap of twisted nematic liquid crystal cells, Opt. Commun., 2008, 281(18), 4560–4565 CrossRef CAS.
- A. Jawor, B. H. Jeong and E. M. V. Hoek, Synthesis, characterization, and ion-exchange properties of colloidal zeolite nanocrystals, J. Nanopart. Res., 2009, 11(7), 1795–1803 CrossRef CAS.
- C. J. van Oss, Acid–base interfacial interactions in aqueous-media, Colloids Surf., A, 1993, 78, 1–49 CrossRef CAS.
- C. J. van Oss, The Properties of Water and their Role in Colloidal and Biological Systems, Academic Press, Amsterdam, 2008, vol. 16 Search PubMed.
- C. J. van Oss and R. F. Giese, Influence of the pH of water on its electron-accepticity and donicity, J. Adhes., 2005, 81(3–4), 237–244 CAS.
- X. F. Huang, S. Bhattacharjee and E. M. V. Hoek, Is surface roughness a “Scapegoat” or a primary factor when defining particle–substrate interactions?, Langmuir, 2010, 26(4), 2528–2537 CrossRef CAS.
- F. M. Fowkes, Determination of interfacial tensions, contact angles, and dispersion forces in surfaces by assuming additivity of intermolecular interactions in surfaces, J. Phys. Chem., 1962, 66(2), 382 CAS.
- C. J. van Oss, Surface Tension Components and Parameters of Liquids and Solids, in Interfacial Forces in Aqueous Media, Marcel Dekker, Inc., New York, NY, 1994, ch. XIII, pp. 170–185 Search PubMed.
- L. Verschaeve, J. Van Gompel, L. Thilemans, L. Regniers, P. Vanparys and D. van der Lelie, VITOTOX bacterial genotoxicity and toxicity test for the rapid screening of chemicals, Environ. Mol. Mutagen., 1999, 33(3), 240–248 CrossRef CAS.
- J. Van Gompel, F. Woestenborghs, D. Beerens, C. Mackie, P. A. Cahill, A. W. Knight, N. Billinton, D. J. Tweats and R. M. Walmsley, An assessment of the utility of the yeast GreenScreen assay in pharmaceutical screening, Mutagenesis, 2005, 20(6), 449–454 CrossRef CAS.
- W. M. Westerink, J. C. Stevenson, A. Lauwers, G. Griffioen, G. J. Horbach and W. G. Schoonen, Evaluation of the Vitotox and RadarScreen assays for the rapid assessment of genotoxicity in the early research phase of drug development, Mutat. Res., 2009, 676(1–2), 113–130 CAS.
- C. Marbeuf-Gueye, M. Salerno, P. Quidu and A. Garnier-Suillerot, Inhibition of the P-glycoprotein- and multidrug resistance protein-mediated efflux of anthracyclines and calceinacetoxymethyl ester by PAK-104P, Eur. J. Pharmacol., 2000, 391(3), 207–216 CrossRef CAS.
- J. C. Fernandez-Checa and N. Kaplowitz, The use of monochlorobimane to determine hepatic GSH levels and synthesis, Anal. Biochem., 1990, 190(2), 212–219 CAS.
- C. Gabriel, A. Camins, F. X. Sureda, L. Aquirre, E. Escubedo, M. Pallas and J. Camarasa, Determination of nitric oxide generation in mammalian neurons using dichlorofluorescin diacetate and flow cytometry, J. Pharmacol. Toxicol. Methods, 1997, 38(2), 93–98 CrossRef CAS.
- G. R. Nakayama, M. C. Caton, M. P. Nova and Z. Parandoosh, Assessment of the alamarBlue assay for cellular growth and viability in vitro, J. Immunol. Methods, 1997, 204(2), 205–208 CrossRef CAS.
- J. H. Chan, H. S. Dua, A. Powell-Richards, D. R. Jones and I. M. Harris, Effect of ABO blood group mismatching on corneal epithelial cells: an in vitro study, Br. J. Ophthalmol., 2001, 85(9), 1104–1109 CrossRef CAS.
- R. Hannah, M. Beck, R. Moravec and T. Riss, CellTiter-Glo™ luminescent cell viability assay: a sensitive and rapid method for determining cell viability, Promega Cell Notes, 2001, 2, 11–13 Search PubMed.
- M. A. O'Brien, W. J. Daily, P. E. Hesselberth, R. A. Moravec, M. A. Scurria, D. H. Klaubert, R. F. Bulleit and K. V. Wood, Homogeneous, bioluminescent protease assays: caspase-3 as a model, J. Biomol. Screening, 2005, 10(2), 137–148 CrossRef CAS.
- E. Sonneveld, J. A. Riteco, H. J. Jansen, B. Pieterse, A. Brouwer, W. G. Schoonen and B. van der Burg, Comparison of in vitro and in vivo screening models for androgenic and estrogenic activities, Toxicol. Sci., 2006, 89(1), 173–187 CAS.
- W. G. Schoonen, R. J. de Ries, J. W. Joosten, G. J. Mathijssen-Mommers and H. J. Kloosterboer, Development of a high-throughput in vitro bioassay to assess potencies of progestagenic compounds using Chinese hamster ovary cells stably transfected with the human progesterone receptor and a luciferase reporter system, Anal. Biochem., 1998, 261(2), 222–224 CrossRef CAS.
- P. J. O'Brien, W. Irwin, D. Diaz, E. Howard-Cofield, C. M. Krejsa, M. R. Slaughter, B. Gao, N. Kaludercic, A. Angeline, P. Bernardi, P. Brain and C. Hougham, High concordance of drug-induced human hepatotoxicity with in vitro cytotoxicity measured in a novel cell-based model using high content screening, Arch. Toxicol., 2006, 80(9), 580–604 CrossRef CAS.
- S. Onoue, N. Igarashi, S. Yamada and Y. Tsuda, High-throughput reactive oxygen species (ROS) assay: an enabling technology for screening the phototoxic potential of pharmaceutical substances, J. Pharm. Biomed. Anal., 2008, 46(1), 187–193 CrossRef CAS.
- C. Y. Usenko, S. L. Harper and R. L. Tanguay, In vivo evaluation of carbon fullerene toxicity using embryonic zebrafish, Carbon, 2007, 45(9), 1891–1898 CrossRef CAS.
- S. Y. Shaw, E. C. Westly, M. J. Pittet, A. Subramanian, S. L. Schreiber and R. Weissleder, Perturbational profiling of nanomaterial biologic activity, Proc. Natl. Acad. Sci. U. S. A., 2008, 105(21), 7387–7392 CrossRef CAS.
- H. Meng, T. Xia, S. George and A. E. Nel, A predictive toxicological paradigm for the safety assessment of nanomaterials, ACS Nano, 2009, 3(7), 1620–1627 CrossRef CAS.
- T. Xia, M. Kovochich, J. Brant, M. Hotze, J. Sempf, T. Oberley, C. Sioutas, J. I. Yeh, M. R. Wiesner and A. E. Nel, Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm, Nano Lett., 2006, 6(8), 1794–1807 CrossRef CAS.
- T. Xia, M. Kovochich, M. Liong, L. Mädler, B. Gilbert, H. Shi, J. I. Yeh, J. I. Zink and A. E. Nel, Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties, ACS Nano, 2008, 2(10), 2121–2134 CrossRef CAS.
- T. Xia, M. Kovochich, M. Liong, J. I. Zink and A. E. Nel, Cationic polystyrene nanosphere toxicity depends on cell-specific endocytic and mitochondrial injury pathways, ACS Nano, 2008, 2(1), 85–96 CrossRef CAS.
- E. J. Jung, N. K. Avliyakulov, P. Boontheung, J. A. Loo and A. E. Nel, Pro-oxidative DEP chemicals induce heat shock proteins and an unfolding protein response in a bronchial epithelial cell line as determined by DIGE analysis, Proteomics, 2007, 7(21), 3906–3918 CrossRef CAS.
- G. G. Xiao, M. Wang, N. Li, J. A. Loo and A. E. Nel, Use of proteomics to demonstrate a hierarchical oxidative stress response to diesel exhaust particle chemicals in a macrophage cell line, J. Biol. Chem., 2003, 278(50), 50781–50790 CrossRef CAS.
- A. Kroll, M. H. Pillukat, D. Hahn and J. Schnekenburger, Current in vitro methods in nanoparticle risk assessment: limitations and challenges, Eur. J. Pharm. Biopharm., 2009, 72(2), 370–377 CrossRef.
- N. A. Monteiro-Riviere, A. O. Inman and L. W. Zhang, Limitations and relative utility of screening assays to assess engineered nanoparticle toxicity in a human cell line, Toxicol. Appl. Pharmacol., 2009, 234(2), 222–235 CrossRef CAS.
- A. Casey, E. Herzog, M. Davoren, F. M. Lyng, H. J. Byrne and G. Chambers, Spectroscopic analysis confirms the interactions between single walled carbon nanotubes and various dyes commonly used to assess cytotoxicity, Carbon, 2007, 45(7), 1425–1432 CrossRef CAS.
- J. M. Worle-Knirsch, K. Pulskamp and H. F. Krug, Oops they did it again! Carbon nanotubes hoax scientists in viability assays, Nano Lett., 2006, 6(6), 1261–1268 CrossRef CAS.
- J. H. Zhang, T. D. Chung and K. R. Oldenburg, A simple statistical parameter for use in evaluation and validation of high throughput screening assays, J. Biomol. Screening, 1999, 4(2), 67–73 CrossRef.
- J. Inglese, D. S. Auld, A. Jadhav, R. L. Johnson, A. Simeonov, A. Yasgar, W. Zheng and C. P. Austin, Quantitative high-throughput screening: a titration-based approach that efficiently identifies biological activities in large chemical libraries, Proc. Natl. Acad. Sci. U. S. A., 2006, 103(31), 11473–11478 CrossRef CAS.
- C. J. Martin, X. C. Le, T. L. Guidotti, S. Yalcin, E. Chum, R. J. Audette, C. K. Liang, B. S. Yuan, X. Zhang and J. Wu, Zinc exposure in Chinese foundry workers, Am. J. Ind. Med., 1999, 35(6), 574–580 CrossRef CAS.
- L. C. Rohrs, Metal-fume fever from inhaling zinc oxide, Arch. Intern. Med., 1957, 100(1), 44–49.
- J. A. Rubiolo, G. Mithieux and F. V. Vega, Resveratrol protects primary rat hepatocytes against oxidative stress damage: activation of the Nrf2 transcription factor and augmented activities of antioxidant enzymes, Eur. J. Pharmacol., 2008, 591(1–3), 66–72 CrossRef CAS.
- L. E. DeForge, K. L. Billeci and S. M. Kramer, Effect of IFN-γ on the killing of S. aureus in human whole blood. Assessment of bacterial viability by CFU determination and by a new method using alamarBlue, J. Immunol. Methods, 2000, 245(1–2), 79–89 CrossRef CAS.
- C. Laflamme, S. Lavigne, J. Ho and C. Duchaine, Assessment of bacterial endospore viability with fluorescent dyes, J. Appl. Microbiol., 2004, 96(4), 684–692 CrossRef CAS.
- Y. Shimomura, R. Ohno, F. Kawai and K. Kimbara, Method for assessment of viability and morphological changes of bacteria in the early stage of colony formation on a simulated natural environment, Appl. Environ. Microbiol., 2006, 72(7), 5037–5042 CrossRef CAS.
- L. Boulos, M. Prevost, B. Barbeau, J. Coallier and R. Desjardins, LIVE/DEAD® BacLight (™): application of a new rapid staining method for direct enumeration of viable and total bacteria in drinking water, J. Microbiol. Methods, 1999, 37(1), 77–86 CrossRef CAS.
- S. Leuko, A. Legat, S. Fendrihan and H. Stan-Lotter, Evaluation of the LIVE/DEAD BacLight kit for detection of extremophilic archaea and visualization of microorganisms in environmental hypersaline samples, Appl. Environ. Microbiol., 2004, 70(11), 6884–6886 CrossRef CAS.
- C. M. Sayes, A. M. Gobin, K. D. Ausman, J. Mendez, J. L. West and V. L. Colvin, Nano-C-60 cytotoxicity is due to lipid peroxidation, Biomaterials, 2005, 26(36), 7587–7595 CrossRef CAS.
- J. Porter, D. Deere, R. Pickup and C. Edwards, Fluorescent probes and flow cytometry: new insights into environmental bacteriology, Cytometry, 1996, 23(2), 91–96 CrossRef CAS.
- A. S. Kaprelyants and D. B. Kell, Rapid assessment of bacterial viability and vitality by rhodamine 123 and flow-cytometry, J. Appl. Bacteriol., 1992, 72(5), 410–422 CAS.
- O. Choi, K. K. Deng, N. J. Kim, L. Ross, R. Y. Surampalli and Z. Q. Hu, The inhibitory effects of silver nanoparticles, silver ions, and silver chloride colloids on microbial growth, Water Res., 2008, 42(12), 3066–3074 CrossRef CAS.
- J. S. Park, K. Park, H. T. Moon, D. G. Woo, H. N. Yang and K. H. Park, Electrical pulsed stimulation of surfaces homogeneously coated with gold nanoparticles to induce neurite outgrowth of PC12 cells, Langmuir, 2009, 25(1), 451–457 CrossRef CAS.
- J. Chen, J. M. Zhu, H. H. Cho, K. M. Cui, F. H. Li, X. B. Zhou, J. T. Rogers, S. T. C. Wong and X. D. Huang, Differential cytotoxicity of metal oxide nanoparticles, J. Exp. Nanosci., 2008, 3(4), 321–328 Search PubMed.
- X. Jin, M. Li, J. Wang, C. Marambio-Jones, X. Huang, R. Damoiseaux and E. M. V. Hoek, High-throughput screening of nano-silver stability and bacterial toxicity in simulated fresh water: influence of specific ions, Environ. Sci. Technol., 2010, 44(19), 7321–7328 CrossRef CAS.
- M. Li, S. Pokhrel, X. Jin, L. Mädler, R. Damoiseaux and E. M. V. Hoek, Stability, bioavailability and bacterial toxicity of ZnO and iron-doped ZnO nanoparticles in aquatic media, Environ. Sci. Technol., 2010, 45(2), 755–761.
- D. A. Tomalia, In quest of a systematic framework for unifying and defining nanoscale, J. Nanopart. Res., 2009, 11, 61.
- T. Clark, S. Martin and T. Liefeld, Globally distributed object identification for biological knowledgebases, Briefings Bioinf., 2004, 5(1), 59–70 Search PubMed.
- M. Wawer and J. Bajorath, Extraction of structure–activity relationship information from high-throughput screening data, Curr. Med. Chem., 2009, 16(31), 4049–4057 CAS.
- N. Malo, J. Hanley, S. Cerquozzi, J. Pelletier and R. Nadon, Statistical practice in high-throughput screening data analysis, Nat. Biotechnol., 2006, 24(2), 167–176 CrossRef CAS.
- B. A. Posner, H. Xi and J. E. J. Mills, Enhanced HTS hit selection via a local hit rate analysis, J. Chem. Inf. Model., 2009, 49(10), 2202–2210 CrossRef CAS.
- S. C. K. Sukuru, J. L. Jenkins, R. E. J. Beckwith, J. Scheiber, A. Bender, D. Mikhailov, J. W. Davies and M. Glick, Plate-based diversity selection based on empirical HTS data to enhance the number of hits and their chemical diversity, J. Biomol. Screening, 2009, 14(6), 690–699 CrossRef CAS.
- M. Eisen, P. Spellman, P. Brown and D. Botstein, Cluster analysis and display of genome-wide expression patterns, Proc. Natl. Acad. Sci. U. S. A., 1998, 95(25), 14863–14868 CrossRef CAS.
- T. Kohonen, The self-organizing map, Proc. IEEE, 1990, 78(9), 1464–1480 CrossRef.
- T. M. Sager, D. W. Porter, V. A. Robinson, W. G. Lindsley, D. E. Schwegler-Berry and V. Castranova, Improved method to disperse nanoparticles for in vitro and in vivo investigation of toxicity, Nanotoxicology, 2007, 1, 118–129 CrossRef CAS.
- Z. Ji, X. Jin, S. George, T. Xia, H. Meng, X. Wang, E. Suarez, H. Zhang, E. M. Hoek, H. Godwin, A. E. Nel and J. I. Zink, Dispersion and stability optimization of TiO2 nanoparticles in cell culture media, Environ. Sci. Technol., 2010, 44(19), 7309–7314 CrossRef CAS.
- N. Gao, M. J. Keane, T. Ong, J. Ye, W. E. Miller and W. E. Wallace, Effects of phospholipid surfactant on apoptosis induction by respirable quartz and kaolin in NR8383 rat pulmonary macrophages, Toxicol. Appl. Pharmacol., 2001, 175(3), 217–225 CrossRef CAS.
- N. Li, M. Wang, L. A. Bramble, D. A. Schmitz, J. J. Schauer, C. Sioutas, J. R. Harkema and A. E. Nel, The adjuvant effect of ambient particulate matter is closely reflected by the particulate oxidant potential, Environ. Health Perspect., 2009, 117(7), 1116–1123 CAS.
- N. Li, J. R. Harkema, R. P. Lewandowski, M. Wang, L. A. Bramble, G. R. Gookin, Z. Ning, M. T. Kleinman, C. Sioutas and A. E. Nel, Ambient ultrafine particles provide a strong adjuvant effect in the secondary immune response: implication for traffic-related asthma flares, Am. J. Physiol.: Lung Cell. Mol. Phys., 2010, 299(3), L374–383 Search PubMed.
- J. A. Araujo, B. Barajas, M. Kleinman, X. Wang, B. J. Bennett, K. W. Gong, M. Navab, J. Harkema, C. Sioutas, A. J. Lusis and A. E. Nel, Ambient particulate pollutants in the ultrafine range promote early atherosclerosis and systemic oxidative stress, Circ. Res., 2008, 102(5), 589–596 CrossRef CAS.
- T. Xia, N. Li and A. E. Nel, Potential health impact of nanoparticles, Annu. Rev. Public Health, 2009, 30, 137–150 CrossRef.
- R. Duffin, L. Tran, D. Brown and V. Stone, Proinflammogenic Effects of Low-Toxicity and Metal Nanoparticles in vivo and in vitro: Highlighting the Role of Particle Surface Area and Surface Reactivity, Inhalation Toxicology: International Forum for Respiratory Research, 2007, vol. 19, 10849–856 Search PubMed.
- E. K. Rushton, J. Jiang, S. S. Leonard, S. Eberly, V. Castranova, P. Biswas, A. Elder, X. Han, R. Gelein, J. Finkelstein and G. Oberdorster, Concept of assessing nanoparticle hazards considering nanoparticle dosemetric and chemical/biological response metrics, J. Toxicol. Environ. Health, 2010, 73(5), 445–461 CrossRef CAS.
- E. Jan, S. J. Byrne, M. Cuddihy, A. M. Davies, Y. Volkov, Y. K. Gun'ko and N. A. Kotov, High-content screening as a universal tool for fingerprinting of cytotoxicity of nanoparticles, ACS Nano, 2008, 2(5), 928–938 CrossRef CAS.
- S. George, S. Pokhrel, T Xia, G. Benjamin, Z. Gi, S. Marco, R. Andreas, R. Damoiseaux, K. . Bradley, L. Mädler and A. E. Nel, Use of a rapid cytotoxicity screening approach to engineer a safer zinc oxide nanoparticle through iron doping, ACS Nano, 2010, 4(1), 15–29 CrossRef CAS.
- R. Rallo, B. France, R. Liu, S. Nair, K. Bradley, R. Damoiseaux, S. George, A. Nel, F. Giralt and Y. Cohen, Analysis of Metal and Metal Oxide Nanoparticles Toxicity-Related Signaling Pathways via Self-Organizing Maps, Environ. Sci. Technol. Search PubMed (in press).
- D. J. Gentleman and W. C. Chan, A systematic nomenclature for codifying engineered nanostructures, Small, 2009, 5(4), 426–431 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2011 |
