Is the action spectrum for the UV-induced production of previtamin D3 in human skin correct?
Mary Norval*a, Lars Olof Björnbc and Frank R. de Gruijld
aBiomedical Sciences, University of Edinburgh Medical School, Teviot Place, Edinburgh, EH8 9AG, UK. E-mail: M.Norval@ed.ac.uk; Tel: +44 131 650 3167
bDepartment of Cell and Organism Biology, Lund University, Lund, Sweden
cKey Laboratory of Ecology and Environmental Science in Guangdong Higher Education, School of Life Science, South China Normal University, Guangzhou, China
dDepartment of Dermatology, Leiden University Medical Centre, Leiden, The Netherlands
First published on 19th October 2009
Abstract
The universally recognised action spectrum for the UV-induced conversion of 7-dehydrocholesterol to previtamin D3 in human skin was published in 1982, and indicates a maximum at about 297 nm with essentially no production above 315 nm. This work represents a milestone in research on vitamin D, but limitations in the original data should be recognised. Various findings have arisen in recent years which cast doubts on the accuracy of the action spectrum and its application for spectral weighting in calculations of effective UV doses. In conclusion, the construction of an entirely new computational model to predict previtamin D levels is recommended.
 Mary Norval | Mary Norval, DSc, is Professor Emeritus at the University of Edinburgh in Scotland, with major research interests in the effects of ultraviolet radiation on human health, especially immunological aspects. |
 Lars Olof Björn | Lars Olof Björn is Professor Emeritus of Botany at Lund University in Sweden. His major research interest is photobiology, with an emphasis on plants and ultraviolet effects. |
 Frank R. de Gruijl | Frank de Gruijl, PhD, is Associate Professor in the Department of Dermatology at the Leiden University Medical Centre in the Netherlands. He leads experimental research on ultraviolet effects in the skin, specifically in relation to the formation of skin cancer. More recent research concerns the induction of vitamin D3 by ultraviolet radiation and possible anti-cancer effects of vitamin D. |
Introduction
Interest in vitamin D has increased greatly in recent years as evidence is accumulating to indicate that it may confer multiple health benefits.1–5 The classical role of vitamin D has been recognised for almost a century as preventing rickets in children, and osteomalacia, osteoporosis and bone fractures in adults. Now protection against several autoimmune diseases, internal cancers, respiratory diseases, hypertension and death (from all causes) has been proposed.1–5 As more than 90% of the vitamin D in the majority of human subjects is derived from sun exposure,6,7 it is of obvious importance to determine the waveband in the solar spectrum that is effective at inducing the production of this compound.As outlined in Fig. 1, following ultraviolet radiation (UVR), 7-dehydrocholesterol (7-DHC, also called provitamin D3) in the skin is converted to previtamin D3 as the first step in the metabolic pathway leading to the active form of vitamin D, 1,25-dihydroxyvitamin D3 (1,25(OH)2D). Previtamin D3 undergoes a thermochemical reaction with formation of vitamin D3 (cholecalciferol). Vitamin D3 is then released from the cell membrane into the extracellular space where it combines with a carrier protein and is further metabolised to 25-hydroxyvitamin D3 and finally 1,25(OH)2D. These last two steps occur commonly in the liver and kidney respectively, although the whole pathway can take place in the skin8 and other organs.9 It can be seen from Fig. 1 that other metabolites can be formed at various stages such as lumisterol, toxisterols and tachysterol from previtamin D3,10 and 5,6-trans-vitamin D3, suprasterol I and suprasterol II from vitamin D3.11 Thus the cascade is not a simple one, especially as many of the reactions are photoreversible.
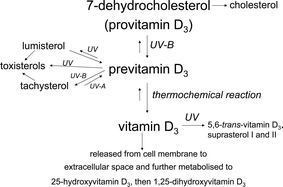 | ||
| Fig. 1 The metabolic pathway leading from 7-dehydrocholesterol to 1,25-dihydroxyvitamin D3. | ||
An action spectrum for the conversion of 7-DHC to previtamin D3 in human skin was determined by MacLaughlin et al. in 1982.12 The same data were further defined and extended by the CIE in 2006.13 The “official” CIE or the MacLaughlin et al.12 action spectrum is now being employed in various computations for weighting of solar spectra to ascertain effective doses with far reaching consequences (see final section), although probably never intended to be used for this purpose by the original authors. Therefore an investigation into its accuracy is urgently required. In this article we aim to raise concerns regarding the validity of some aspects of the action spectrum and, as a result of these comments, to outline several uncertainties about the adequacy of the action spectrum for calculating the production of vitamin D3 at different latitudes throughout the year.
Construction of the action spectrum for the conversion of 7-dehydrocholesterol to previtamin D3
Most of the 7-DHC in the skin is found in the cell membrane of the keratinocytes and, on irradiation, 7-DHC absorbs a quantum of energy that transforms it to an excited singlet state. Ring opening then yields previtamin D3. In vivo, the active cis (Z) cis (Z) conformer of previtamin D3 is trapped in the triglycerides of the keratinocyte membrane and converts to vitamin D3 within a few hours. The absorption spectrum for 7-DHC has been determined in vitro,12,14,15 although not in vivo. As shown in Fig. 2, it exhibits 3 peaks, the main ones at approximately 272 nm and 282 nm and a minor one at about 292 nm. While this last peak corresponds roughly to the action spectrum for the production of previtamin D3 from 7-DHC (see next section), the shorter-wavelength absorption bands do not appear to be relevant in vivo. The reasons for this are not entirely clear although it is possible that the stratum corneum may filter out such wavelengths,17 thus preventing them from reaching the 7-DHC which is located predominantly in the stratum spinosum and basale layers of the epidermis.18 Such filtering activity will vary between different subjects depending on the thickness of the epidermis, on cutaneous pigmentation and on the individual's history of UV exposure.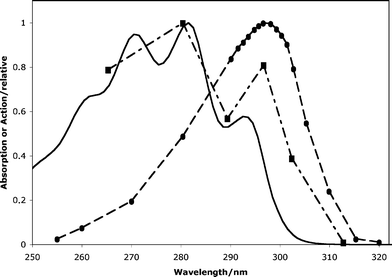 | ||
Fig. 2 The MacLaughlin et al.12 action spectrum for the conversion of 7-dehydrocholesterol to previtamin D3 in human skin (●), the absorption spectrum of 7-dehydrocholesterol in ethanol (![[thick line, graph caption]](https://www.rsc.org/images/entities/char_e117.gif) ; L. O. Björn, unpublished) and the anti-rachitic action spectrum in rats fed a diet deficient in vitamin D (■).16 ; L. O. Björn, unpublished) and the anti-rachitic action spectrum in rats fed a diet deficient in vitamin D (■).16 | ||
In constructing the action spectrum for previtamin D3 production, it should be remembered that this compound, like 7-DHC, can absorb UV photons, resulting in the back reaction to 7-DHC or the formation of tachysterol or lumisterol. The absorption coefficients of previtamin D3, tachysterol and lumisterol in organic solvents are known,12 and these, together with the quantum yields and the irradiation spectrum, determine the reaction rates and eventual photoequilibrium conditions. The quantum yields for these reactions at wavelengths ranging from 254 to 325 nm are shown in Fig. 3.19–22 The values vary to some extent between one publication and another but, as they were obtained in a variety of solvents at temperatures varying from 0 to 37 °C and were sometimes based on ergosterol and previtamin D2 rather than on 7-DHC and previtamin D3, perhaps this is not too surprising. The reaction 7-DHC → previtamin D3 has the same quantum yield at 254 and 302.5 nm implying that the action spectrum, at least for low UV exposures, should parallel the absorption spectrum of 7-DHC, which is not the case (see Fig. 2). While the reaction previtamin D3→ tachysterol is favoured over previtamin D3→ lumisterol and previtamin D3→ 7-DHC by about 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 over the UV-C waveband, the quantum yield for the back reaction tachysterol → previtamin D3 is high at longer wavelengths where the absorption is also high, thus accounting for the low accumulation of tachysterol at these longer wavelengths. To illustrate this effect, we present in Table 1 the calculated percentages of reaction products reached in the photostationary state at different wavelengths, using the quantum yields from Fig. 3 (interpolated where necessary) and the extinction coefficients from graphs (both the formation of toxisterols as products from over-irradiation and the slow thermochemical isomerisation to vitamin D3 were not considered).
1 over the UV-C waveband, the quantum yield for the back reaction tachysterol → previtamin D3 is high at longer wavelengths where the absorption is also high, thus accounting for the low accumulation of tachysterol at these longer wavelengths. To illustrate this effect, we present in Table 1 the calculated percentages of reaction products reached in the photostationary state at different wavelengths, using the quantum yields from Fig. 3 (interpolated where necessary) and the extinction coefficients from graphs (both the formation of toxisterols as products from over-irradiation and the slow thermochemical isomerisation to vitamin D3 were not considered).
| Wavelength/nm | εPre | εPro | εLumi | εTachy | %Prea | %Lumib | %Tachyc |
|---|---|---|---|---|---|---|---|
a %Pre =100(1 + kPre→Pro/kPro→Pre + kPre→Lumi/kLumi→Pre + kPre→Tachy/kTachy→Pre)−1, where rate constant kPre→Pro = σPreϕPre→Pro, with quantum yield ϕPre→Pro and the absorption cross section σPre = εPre× 103× ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (10)/NAv, with NAv as Avogradro's number; other k values are defined analogously.b %Lumi =%Pre ×kPre→Lumi/kLumi→Pre.c %Tachy =%Pre ×kPre→Tachy/kTachy→Pre. (10)/NAv, with NAv as Avogradro's number; other k values are defined analogously.b %Lumi =%Pre ×kPre→Lumi/kLumi→Pre.c %Tachy =%Pre ×kPre→Tachy/kTachy→Pre. | |||||||
| 254 | 8813 | 4795 | 4816 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 434 434 | 21 | 3 | 75 |
| 280 | 6034 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 497 497 | 8886 | 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 577 577 | 51 | 3 | 45 |
| 295 | 2322 | 5861 | 4018 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 686 686 | 64 | 4 | 31 |
| 300 | 1489 | 1575 | 1842 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 188 188 | 64 | 6 | 26 |
| 305 | 873 | 214 | 476 | 7027 | 43 | 26 | 12 |
| 310 | 496 | 95 | 163 | 4685 | 34 | 33 | 7 |
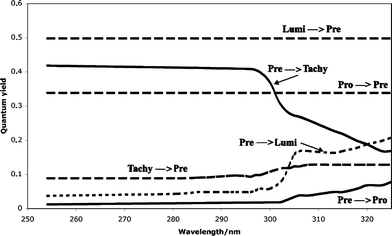 | ||
| Fig. 3 Quantum yields for the photoconversions of provitamin D3 and its photoderivatives. Although the values vary depending on factors such as solvent and temperature, the figure is intended to illustrate the main features of wavelength dependency. The quantum yields are wavelength-independent for lumisterol (Lumi) to previtamin D3 (Pre), provitamin D3 (7-dehydrocholesterol, Pro) to Pre, and tachysterol (Tachy) to Pre. For Pre to Pro and for Pre to Lumi, the quantum yield rises at long wavelengths, and for Pre to Tachy it falls at long wavelengths. Values are compiled from references 19–22. | ||
Such wavelength effects are borne out experimentally as it has been shown, for example, that, on monochromatic irradiation of a solution of 7-DHC, the production of previtamin D3 is favoured at 295 nm, tachysterol at 260 nm and lumisterol at 310 nm.25 This complex situation with different dose–response relationships for different wavelengths adds a degree of uncertainty or variability to experiments trying to establish a single action spectrum for the production of previtamin D3 from 7-DHC. It is illustrated by the demonstration that, when human skin is irradiated at 295 nm, a maximum of 65% 7-DHC can be converted to previtamin D3, while less than a maximum of 20% is converted if solar simulated radiation (SSR) is used.26 Over the wavelength range shown in Table 1, such a low maximum yield of previtamin D3 is not attained, suggesting that the longer wavelengths, over 305 nm, are very important for SSR. Furthermore it should be noted that the in vivo reactions differ somewhat from those in solvents because of molecular restraints in the bilipid membranes (e.g. lowering the yield of tachysterol). The proportion of the photoproducts can change as the UV dose increases so that the formation of lumisterol, in particular, is favoured on prolonged SSR exposure.12 Therefore assuming simple additivity from different wavelengths is questionable when calculating an effective dose with a single action spectrum for polychromatic irradiation, such as SSR.
The MacLaughlin et al. action spectrum
The action spectrum for the UV-induced synthesis of previtamin D3 in human skin was determined by MacLaughlin et al.12 more than 25 years ago and is shown in Fig. 2. Despite the importance of this result, only an outline of the experimental procedure was given. Human skin (site not specified) was obtained from an unspecified number of individuals of unknown age with phototype III. Samples of 6.25 cm2 were prepared and heated to 60 °C for 30 s in a water bath, then blotted dry. The stratum corneum side was irradiated in either 3 or 5 nm half-band widths, ranging from 255 to 320 nm at an unknown temperature. The dose or doses at each wavelength were not given. Immediately following the exposure, and leaving insufficient time for vitamin D3 to be formed from previtamin D3, the skin was presumably separated into the top layer containing the stratum corneum, granulosum and spinosum. The stratum basale cells were scraped off the remaining dermal layer and added to the other epidermal strata.18 Lipids were extracted from the epidermal cells with ethyl acetate in n-hexane for 24 h at −20 °C. The extracts were centrifuged, the supernatant taken to dryness, weighed and portions chromatographed by HPLC to determine the amounts of 7-DHC and its photoproducts. It was concluded that the optimum wavelengths for the production of previtamin D3 were between 295 and 300 nm with a maximum at 297 nm. There was no production detected below 260 nm or above 315 nm.Possibly the most crucial omission in the account of this experiment was to not detail the doses used at each wavelength. A 5 nm half-band width leads to noticeable smearing and broadening of the spectrum, and, to assess the extent of this, it would have been useful to know the precise band-shape of the radiation. To construct an action spectrum, it is normal practice to use a range of doses throughout the spectrum of interest and to assay the quantity of product. The dose required to ensure a chosen reference level of product can then be deduced and the action spectrum constructed as the inverse of these doses vs. wavelength. This complies with a very minimal definition of an action spectrum, limited to a certain response level. It would seem particularly important for two main reasons to assess UV dosages in the case of previtamin D3 production to ensure that they were not excessive.
First, more than 70 years ago, Knudson and Benford16 constructed an action spectrum for the anti-rachitic effect of monochromatic UVR in rats fed a diet deficient in vitamin D (see Fig. 2). It showed a clear maximum around 280 nm and a secondary peak about 297 nm, a finding that is roughly in accordance with the broad UV-B/UV-C absorption spectrum of 7-DHC. Remarkably at exposures higher than 125 J m−2 (750![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 erg over 6 cm2; one J = 107 erg) at wavelengths of 280 nm and shorter, the anti-rachitic effect increased only slightly, while at 297 and 302 nm it continued to increase substantially. At these bigger dosages, the maximum effect consequently shifted to 297 nm, similar to the action spectra reported in earlier publications, using the same rat model27 and a chicken model.28 Thus it appeared that the action spectrum for the anti-rachitic effect of UVR in vitamin D-deficient rats was dependent on the dose level used.
000 erg over 6 cm2; one J = 107 erg) at wavelengths of 280 nm and shorter, the anti-rachitic effect increased only slightly, while at 297 and 302 nm it continued to increase substantially. At these bigger dosages, the maximum effect consequently shifted to 297 nm, similar to the action spectra reported in earlier publications, using the same rat model27 and a chicken model.28 Thus it appeared that the action spectrum for the anti-rachitic effect of UVR in vitamin D-deficient rats was dependent on the dose level used.
Secondly, it is known that interference from the back reaction to 7-DHC and to the other photoproducts, tachysterol and lumisterol, can occur (see Fig. 1 and 3). The maximum (at 295 ± 5 nm) on the action spectrum curve of MacLaughlin et al.12 was 10 cm2 J−1 which corresponds to 0.1 J cm−2. As further shown by MacLaughlin et al.,12 this dose at 295 ± 5 nm induced about 8% previtamin D3 in human epidermis. A dose of approximately 0.3 J cm−2 at this wavelength gave about 28% previtamin D3, and higher doses at the same wavelength further increased the production of vitamin D3 but not in a linear fashion. Thus the portion of the curve of relevance for the action spectrum may be limited to doses up to 0.3 J cm−2 for the wavelength of maximum effectiveness, and doses less than this should ensure linearity, giving the dose of 0.1 J cm−2 some validity. However it should be noted that, even at 0.1 J cm−2 of wavelength 295 ± 5 nm, there was significant production of lumisterol (about 6%). This implies that at least 14% (6% plus 8%), rather than only 8% 7-DHC had been converted to previtamin D3. Thus the action spectrum is not a simple representation of the single forward reaction of 7-DHC to previtamin D3, and it could be affected by non-linear effects from additional UV-driven reactions. Using an estimate of the absorption cross section of 7-DHC in ethanol at 295 nm and the quantum yield for 7-DHC → previtamin D3 (see Fig. 3), we have calculated that about 65% 7-DHC should be converted to previtamin D3 following an irradiation dose of 0.1 J cm−2 (about 1.5 × 1017 photons cm−2), a much higher yield than the 8–14% found in vivo (corresponding to a transmission of 8 to 14%).
Equivalent information for wavelengths other than 295 nm was not given, but, as pointed out above, it is unlikely that the action spectrum could be used as a weighting function to assess previtamin D3 production as the basic assumption of additivity is not credible. Furthermore it was stated by MacLaughlin et al.12 that no more than 5% previtamin D3 was made at any wavelength. Thus the reliable dynamic range of previtamin D3 measurement was very limited, and the spectral analysis of effectiveness declined by at least 30-fold at wavelengths above 310 nm. This latter criticism could have been addressed by adequately high dosages at higher wavelengths, an approach that is technically difficult and was probably unfeasible in 1982. From a consideration of the action spectrum for erythema, we know that the relatively low values in the UV-A waveband can be very important for the assessment of the effectiveness of sun exposure, especially at low sun angles.
It is of interest to note that the relationship between conversion to previtamin D3 and radiation exposure in the MacLaughlin et al.12 action spectrum appears to be skewed towards the shorter wavelengths rather than having a curve similar to the absorption spectrum of 7-DHC. Under such conditions, the action spectrum may be ambiguous and dependent on the degree of conversion chosen as the endpoint. Indeed one explanation is that the dosages were already too high to measure the relatively strong contributions in the UV-C range, as earlier reported by Knudson and Benford in their experiments on the anti-rachitic effects of UVR in rats fed a vitamin D-deficient diet.16
It is also useful at this point to consider the ability of the epidermis and stratum corneum to transmit radiation of different wavelengths.29 These transmission values can be multiplied by the values from the 7-DHC absorption spectrum (Fig. 2). Fig. 4 shows the result, using transmission data of the stratum corneum from an unexposed skin site and from a skin site repeatedly exposed to UV-B. Comparison with the MacLaughlin et al.12 action spectrum, also depicted in Fig. 4, indicates that filtering by the stratum corneum cannot explain the high conversion efficiency by radiation of wavelengths longer than 300 nm compared with the shorter wavelengths.
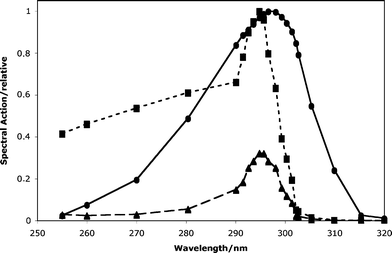 | ||
| Fig. 4 The MacLaughlin et al.12 action spectrum for the conversion of 7-dehydrocholesterol to previtamin D3 in human skin (●) and the absorption spectrum of 7-dehydrocholesterol multiplied by the transmission through stratum corneum from an unexposed skin site (■) and a skin site repeatedly exposed to UV-B (▲).27 | ||
Although there is some debate about the quantity of 7-DHC in human epidermis and it is likely to vary between one individual and another, most estimates for people in middle age are in the range of approximately 1 μg cm−2.17,30 The detection limit for previtamin D3 by conventional HPLC was stated as 0.3% of the epidermal 7-DHC concentration,31i.e. about 3 ng cm−2. Thus at wavelengths towards the upper end of the UV-B waveband (above 310 nm) and into the UV-A waveband (above 315 nm), the assessment of the amount of previtamin D3 formed as a result of UV irradiation may be inaccurate if it is at, or below, the limit of the sensitivity of the method. Such a situation is likely, judging from the absorption spectrum of 7-DHC and UV dosages which were inadequate due to technical difficulties in 1982. It is particularly important to obtain such data as several estimates of previtamin D3 production induced by solar exposure at different latitudes and at different times of the year have been made, based on the MacLaughlin et al.12 action spectrum (see final section below). It would be of interest to analyse epidermal samples by more sensitive techniques developed since 1982, such as LC-mass spectroscopy, that would allow accurate measurement of very small quantities of previtamin D3 and other photoproducts. It is possible by such methods to analyse concentrations as low as 200 pmol L−1.
The MacLaughlin et al.12 action spectrum was developed by the assessment of the response of an unspecified number of subjects of unknown age with phototype III. Age is an untested variable in the action spectrum but it is known that the amount of 7-DHC in the skin decreases with time so that an 80 year old has about half that of a 20 year old.30 As a result of intrinsic ageing, the epidermis atrophies, the 7-DHC content decreases and there are major changes in the dermis.
It is also not known if the skin samples came from a sun-exposed or unexposed site. The two important mechanisms involved in photoadaptation are melanogenesis and thickening of the stratum corneum, and chronic photodamage leads to a hyperproliferative cutaneous response with an increased stratum spinosum layer and mild dysplastic changes.32 The transmission properties of the epidermis and the stratum corneum differ between an unexposed site and one repeatedly exposed to UV-B (see Fig. 4). For example, at 305 nm, about 20% is transmitted through the stratum corneum from an exposed skin site, and 40% through the unexposed.29 Recently Meinhardt et al. have used optoacoustics to measure absorption spectra in human skin in vivo over the range 290–341 nm.33,34 The skin came from individuals of different phototypes and from sun-exposed and unexposed parts of the body. While phototype made little difference to the absorption spectra, they demonstrated that UV adaptation mechanisms for absorption were most effective in the UV-B waveband. This finding led to the conclusion that hyperkeratinisation was the major adaptive factor involved rather than increased production of melanin. The latter would show a relatively higher absorption in the short-wave UV-A part (UV-A-II) of the spectrum. Any impact of the change in absorption spectra due to photoadaptation on the action spectrum for the conversion of 7-DHC to previtamin D3 has not been investigated. Photoadaptation may account at least to some extent for the lack of a latitudinal gradient in vitamin D3 status at the overall population level,35 which might have been predicted, although a weak but statistically significant gradient has been reported for the Caucasian population (−0.7 ± 0.3 nmol L−1 25-hydroxyvitamin D3 per degree latitude, p = 0.02).35
The CIE “official” action spectrum
In 2006, the CIE formed a committee to produce an agreed standardised action spectrum for the production of previtamin D3 in human skin.13 The Figure of the action spectrum published by MacLaughlin et al.12 was taken as the starting point. A computer algorithm was employed to obtain values at 21 wavelengths between 255 and 315 nm. Intermediate points were calculated by spline interpolation. At wavelengths below 260 nm and above 315 up to 330 nm, the action spectrum was extrapolated by exponential decay functions. It is disappointing that the actual values used to construct the 1982 action spectrum and any error estimates were not available, and that the extrapolation, particularly at the higher wavelengths, was necessary. The authors of the report conclude that previtamin D3 formation occurs almost entirely in the UV-B band (280–315 nm) with only about 3–4% of the total production in the UV-A waveband.The Lehmann et al. action spectrum
Instead of using human skin, Lehmann et al. created a human skin equivalent model composed of fibroblasts in a collagen matrix representing the dermis and keratinocytes representing the epidermis which were cultured at the air/liquid interface, thus allowing differentiation.36 Different concentrations of 7-DHC, approximately equal to those found in human skin, were added to the cultures. Then the cultures were irradiated with monochromatic UV-B from 285–315 nm (band widths ±2.5 nm) followed by the identification of the products by normal phase HPLC. 7-DHC was converted via previtamin D3 to vitamin D3, this being complete after 15–20 h. Previtamin D3 was detectable within 10 min. The production of vitamin D3 was maximal at 302 nm and minimal at 293 nm. At wavelengths greater than or equal to 315 nm, no vitamin D3 was detected. Lumisterol and tachysterol were not found following the UV exposure. Their lack of production simplifies the reaction kinetics in comparison with the other systems described above, and would make the creation of a single action spectrum more realistic.Further comments follow. First, although there was some evidence for the peak efficiency of the conversion of 7-DHC to previtamin D3 being several nm higher than that found by MacLaughlin et al.,12 detailed data were not given as the report concentrated on the production of vitamin D3 rather than the intermediate previtamin D3. Secondly, in order to obtain substantial amounts of product following UV exposure, it was necessary to add 7-DHC to the cultures. It had been shown previously, using HaCaT cells, that such exogenous 7-DHC binds to the cell membrane and/or is transported into the cells37 and thus it may interact with the cellular constituents differently from when it is synthesised endogenously via the cholesterol pathway.
Why is it important to determine accurately the action spectra?
As sun exposure has both beneficial aspects, mainly the production of vitamin D, and harmful aspects, such as the induction of skin cancers, several eye diseases, sunburn and photoageing, helpful guidance for the public regarding personal sun behaviour is needed.38 To this end, several reports have estimated the influence of season and latitude on vitamin D synthesis, although it should be noted that personal behavioural choices are equally, if not more, important.39,40 Such calculations of potential vitamin D production generally use outdoor UV measurements weighted by the erythemal action spectrum41 together with the MacLaughlin et al.12 or derived CIE action spectrum13 as their starting basis.35,42–49On the basis that exposure of the body of young adults to one minimum erythema dose of solar UV-B radiation increases the circulating concentration of 25-hydroxyvitamin D3 to a peak of about 52 nmol L−1, Holick has suggested that, at mid-latitudes (around 40°) a fair skinned person should achieve maximum vitamin D3 synthesis during a sunny summer day by 5–10 min exposures, 2–3 times weekly, on the face and forearms around midday.50 The time should be increased to 30 min for dark skin or if the sky is cloudy. Other reports reach a similar conclusion.51,52 As a result, comparable figures have been adopted as position statements by various public health agencies in Australia and New Zealand53 and most recently by the International Agency for Research on Cancer.54 As explained above, it should be noted that a maximum synthesis of previtamin D3 occurs with a certain exposure to the sun depending on variable factors such as latitude, season and phototype, and further exposure will result in the conversion of previtamin D3 to the inactive isomers, tachysterol and lumisterol.
However few studies have attempted to actually measure sunlight-induced conversion of 7-DHC to previtamin D3 in human skin at different latitudes at different times of the year. Webb et al.55 analysed this reaction using, first, a solution of 7-DHC in methanol exposed to sunlight for one hour on a cloudless day over the noon period at locations from 18°N to 52°N. While conversion occurred throughout the year at latitudes of 34°N and below, no production of vitamin D3 was found in the winter months of November to February at 42°N (Boston) or October to March at 52°N (Edmonton), even if the exposure time was extended to 3 h. In the mid-summer months of June and July, the amount of vitamin D3 increased to a maximum of approximately 12% at lower latitudes, to 9% in one hour in Boston and 11% in 3 h in Edmonton. Analyses after exposure of the 7-DHC solution at several places in the southern hemisphere showed concordant results.56 Subsequent results using excised human skin placed in dishes were similar except a maximum of about 6% increase in previtamin D3 was obtained.55 This lower amount compared to the amount produced in the methanol solution was attributed to the attenuating properties of the stratum corneum. As exposure to a UV-B dominated source (295 nm) leads to a higher plateau level of previtamin D3 than exposure to SSR,25 there could be a shift in the irradiating spectrum towards longer wavelengths, leading to a further dominance of UV-A over UV-B and a consequent lowering of the plateau level of previtamin D3. On the basis of these experiments, it was concluded that no production of previtamin D3 by exposure to sunlight under cloudless conditions was possible at latitudes of 42° and above during the winter months.
McKenzie et al.48 have pointed out that, using the MacLaughlin et al.12 action spectrum, the vitamin D weighted UV in the winter months is roughly 5% of the summer value at mid-latitudes, around 40°. Thus sufficient vitamin D should be produced by whole-body exposure in a fair-skinned individual in winter and, even in a dark-skinned individual, some vitamin D should be produced. This calls into question either the accuracy of the single action spectrum or the sensitivity of the method for detecting small quantities of previtamin D3 in skin, or both. In addition calculations regarding vitamin D production have been based on the solar UV irradiating a horizontal surface while, in reality, the body is usually vertical. Around midday when the sun is overhead, there will be the largest difference in the amount of UV received compared with a horizontal surface.
In conclusion, we recognise the critical importance of vitamin D and its multiple health benefits in human subjects. We suggest that there are sufficient uncertainties in the officially recognised action spectrum for the conversion of 7-DHC to previtamin D3 in human skin to merit further experimentation, especially considering non-additivity and non-linearities in the cascade of UV-driven reaction and the incomplete data at wavelengths in the borderline between the UV-B and UV-A wavebands and into the UV-A. Indeed we recommend the construction of an entirely new computational model that would incorporate all the relevant action spectra including adequate data points in the UV-A, with, perhaps, a simplified version, especially for sunlight, based on only one or two action spectra. Such a model would provide the general public and health agencies with accurate scientific guidance regarding personal exposure to sunlight, in order to maximise vitamin D production whilst minimising any harmful effects.
References
- M. F. Holick, Vitamin D: a millennium perspective, J. Cell. Biochem., 2003, 88, 296–307 CrossRef CAS.
- M. F. Holick, Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease, Am. J. Clin. Nutr., 1994, 80(6 Suppl.), S1678–S1688.
- S. Kimball, Gel-H. Fuleihan and R. Veith, Vitamin D: a growing perspective, Crit. Rev. Clin. Lab. Sci., 2008, 45, 339–414 Search PubMed.
- J. R. Moro, M. Iwata and U. H. von Adriano, Vitamin effects on the immune system: vitamin A and D take centre stage, Nat. Rev. Immunol., 2008, 8, 685–698 CrossRef.
- W. B. Grant and S. B. Mohr, Ecological studies of ultraviolet B, vitamin D and cancer since 2000, Ann. Epidemiol., 2009, 19, 446–454 CrossRef.
- D. E. M. Lawson, A. A. Paul, A. E. Black, T. J. Cole, A. R. Mandal and M. David, Relative contributions of diet and sunlight to vitamin D state in the elderly, Br. Med. J., 1979, 2, 303–305 CrossRef CAS.
- E. M. E. Poskitt, T. J. Cole and D. E. M. Lawson, Diet, sunlight, and 25-hydroxyvitamin D in healthy children and adults, Br. Med. J., 1979, 1, 221–223 CrossRef CAS.
- B. Lehmann, The vitamin D3 pathway in human skin and its role for regulation of biological processes, Photochem. Photobiol., 2005, 81, 1246–1251 CrossRef CAS.
- P. H. Anderson and G. J. Atkins, The skeleton as an intracrine organ for vitamin D metabolism, Mol. Aspects Med., 2008, 29, 397–406 CrossRef CAS.
- F. Boomsma, H. J. C. Jacobs, E. Havinga and A. von der Gen, Studies of vitamin D and related compounds, part 24. New irradiation products of pre-vitamin D3, Tetrahedron Lett., 1975, 16, 427–430 CrossRef.
- A. R. Webb, B. R. DeCosta and M. F. Holick, Sunlight regulates the cutaneous production of vitamin D3 by causing its photodegradation, J. Clin. Endocrinol. Metab., 1989, 68, 882–887 CrossRef CAS.
- J. A. MacLaughlin, R. R. Anderson and M. F. Holick, Spectral character of sunlight modulates photosynthesis of previtamin D3 and its photoisomers in human skin, Science, 1982, 216, 1001–1003 CAS.
- R. Bouillon, J. Eisman, M. Garabedian, M. Holick, J. Kleinschmidt, T. Suda, I. Terenetskaya and A. Webb, Action spectrum for production of previtamin D3 in human skin, CIE Technical Report 174, Commission Internationale de l'Eclairage (CIE) Central Bureau, Vienna, Austria, 2006 Search PubMed.
- O. N. Galkin and I. P. Terenetskaya, Vitamin D’ biodosimeter: basic characteristics and potential application, J. Photochem. Photobiol., B, 1999, 53, 12–19 CrossRef CAS.
- M. Glover, J. Glover and R. A. Morton, Provitamin D3 in tissues and the conversion of cholesterol to 7-dehydrocholesterol in vivo, Biochem. J., 1952, 51, 1–9 CAS.
- A. Knudson and F. Benford, Quantitative studies of the effectiveness of ultraviolet radiation of various wave-lengths in rickets, J. Biol. Chem., 1938, 124, 287–299 CAS.
- A. R. Webb and M. F. Holick, The role of sunlight in the cutaneous production of vitamin D3, Annu. Rev. Nutr., 1988, 8, 375–399 CrossRef CAS.
- M. F. Holick, J. A. MacLaughlin, M. B. Clark, S. A. Holick, J. T. Potts, R. R. Anderson, I. H. Blank, J. A. Parrish and P. Elias, Photosynthesis of previtamin D3 in human skin and the physiological consequences, Science, 1980, 210, 203–205 CAS.
- E. Havinga, Vitamin D, example and challenge, Experientia, 1973, 29, 1181–1316 CAS.
- H. J. C. Jacobs, J. W. J. Gielen and E. Havinga, Effects of wavelength and conformation on the photochemistry of vitamin D and related conjugated trienes, Tetrahedron Lett., 1981, 22, 4013–4016 CrossRef CAS.
- W. G. Dauben, B. Disanayaka, J. H. Funhoff, B. Zhou, B. E. Kohler and D. E. Schilke, Polyene 21Ag and 11Bu states and the photochemistry of previtamin D3, J. Am. Chem. Soc., 1991, 113, 8367–8374 CrossRef CAS.
- S. Gliesing, M. Reinbacher, H.-D. Ilge and D. Faßler, Wellenlangeneffekte auf die quantenausbuten ten photoisomerisierung von pravitamin D3, Z. Chem., 1989, 29, 21–22 CAS.
- J. C. Sternberg, H. S. Stillo and R. H. Schwenderman, Spectrophotometric analysis of multicomponent systems using least squares method in matrix form. Ergosterol irradiation system, Anal. Chem., 1960, 32, 84–90 CrossRef.
- O. G. Dmitrenko, I. P. Terenetskaya and W. Reischl, Solvent effect on previtamin D conformational equilibrium and photoreactions, J. Photochem. Photobiol., A, 1997, 104, 113–117 CrossRef CAS.
- M. F. Holick, J. A. MacLaughlin, J. A. Parrish and R. R. Anderson, The photochemistry and photobiology of vitamin D3, in The Science of Photomedicine, ed. J. D. Regan and J. A. Parrish, Plenum, New York, 1982, pp. 195–218 Search PubMed.
- M. F. Holick, J. A. MacLaughlin and S. H. Doppelt, Regulation of cutaneous previtamin D3 photosynthesis in man: skin pigment is not an essential regulator, Science, 1981, 211, 590–593 CrossRef CAS.
- J. W. M. Bunker and R. S. Harris, Precise evaluation of ultraviolet therapy in experimental rickets, N. Engl. J. Med., 1937, 216, 165–169.
- G. H. Maughan, Ultra-violet wavelengths valuable in the cure of rickets in chickens, Am. J. Physiol., 1928, 87, 381–398 Search PubMed.
- W. A. Bruls, H. Slaper, J. C. van der Leun and L. Berrens, Transmission of human epidermis and stratum corneum as a function of thickness in the ultraviolet and visible wavelengths, Photochem. Photobiol., 1984, 40, 485–494 CrossRef CAS.
- J. MacLaughlin and M. F. Holick, Aging decreases the capacity of human skin to produce vitamin D3, J. Clin. Invest., 1985, 76, 1536–1538 CrossRef CAS.
- T. C. Chen, F. Chimeh, Z. Lu, J. Mattieu, K. S. Person, A. Zhang, N. Kohn, S. Martinello, R. Berkowitz and M. F. Holick, Factors that influence the cutaneous synthesis and dietary sources of vitamin D, Arch. Biochem. Biophys., 2007, 460, 213–217 CrossRef CAS.
- J. H. Rabe, A. J. Mamelak, P. J. McElgunn, W. L. Morison and D. N. Sauder, Photoaging: mechanisms and repair, J. Am. Acad. Dermatol., 2006, 55, 1–19 CrossRef.
- M. Meinhardt, R. Krebs, A. Anders, U. Heinrich and H. Tronnier, Effect of ultraviolet absorption spectra of human skin in vivo, Photodermatol., Photoimmunol. Photomed., 2008, 24, 76–82 CrossRef.
- M. Meinhardt, R. Krebs, A. Anders, U. Heinrich and H. Tronnier, Absorption spectra of human skin in vivo in the ultraviolet wavelength range measured by optoacoustics, Photochem. Photobiol., 2009, 85, 70–77 CrossRef CAS.
- T. Hagenau, R. Vest, T. N. Gissel, C. S. Poulsen, M. Erlandsen, L. Mosekilde and P. Vestergaard, Global vitamin D levels in relation to age, gender, skin pigmentation and latitude: an ecologic meta-regression analysis, Osteoporosis Int., 2009, 20, 133–140 Search PubMed.
- B. Lehmann, T. Genehr, P. Knuschke, J. Pietzsch and M. Meurer, UVB-induced conversion of 7-dehydrocholesterol to 1α,25-dihydroxyvitamin D3 in an in vitro human skin equivalent model, J. Invest. Dermatol., 2001, 117, 1179–1185 CrossRef CAS.
- B. Lehmann, P. Knuschke and M. Meurer, UVB-induced conversion of 7-dehydrocholesterol to 1alpha,25-dihydroxycholesterol D3 (calcitriol) in the human keratinocyte line HaCaT, Photochem. Photobiol., 2000, 72, 803–809 CrossRef CAS.
- R. M. Lucas, M. H. Repacholi and A. J. McMichael, Is the current public health message on UV exposure correct?, Bull. W. H. O., 2006, 84, 485–491 CrossRef.
- I. A. van der Mei, A. L. Ponsonby, O. Engelsen, J. A. Pasco, J. J. McGrath, D. W. Eyles, L. Blizzard, T. Dwyer, R. Lucas and G. Jones, The high prevalence of vitamin D insufficiency across Australian populations is only partly explained by season and latitude, Environ. Health Perspect., 2007, 115, 1132–1139 CAS.
- A. F. McKinlay and B. L. Diffey, CIE Research Note. A reference action spectrum for ultraviolet induced erythema in human skin, CIE J., 1987, 6, 17–22.
- R. M. Sayre and J. C. Dowdy, Darkness at noon: sunscreens and vitamin D3, Photochem. Photobiol., 2007, 83, 459–463 CAS.
- S. J. Pope, M. F. Holick, S. Mackin and D. E. Godar, Action spectrum conversion factors that change erythemally weighted to previtamin D3-weighted UV doses, Photochem. Photobiol., 2008, 84, 1277–1283 CrossRef CAS.
- M. G. Kimlin, The climatology of vitamin D producing ultraviolet radiation over the United States, J. Steroid Biochem. Mol. Biol., 2004, 89–90, 479–483 CrossRef CAS.
- M. G. Kimlin and K. A. Schallhorn, Estimations of the human ‘vitamin D’ exposure in the USA, Photochem. Photobiol. Sci., 2004, 3, 1067–1070 RSC.
- O. Engelsen, M. Brustad, L. Aksnes and E. Lund, Daily duration of vitamin D synthesis in human skin with relation to latitude, total ozone, altitude, ground cover, aerosols and cloud thickness, Photochem. Photobiol., 2005, 81, 1287–1290 CrossRef CAS.
- A. R. Webb and O. Engelsen, Calculated ultraviolet exposure levels for a healthy vitamin D status, Photochem. Photobiol., 2006, 82, 1697–1703 CAS.
- A. Kazantzidis, A. F. Bais, M. M. Zempila, S. Kazadzis, P. N. den Outer, T. Kosela and H. Slaper, Calculations of the human vitamin D exposure from UV spectral measurements at three European stations, Photochem. Photobiol. Sci., 2009, 8, 45–51 RSC.
- R. L. McKenzie, J. B. Liley and L. O. Bjorn, UV radiation: balancing risks and benefits, Photochem. Photobiol., 2009, 85, 88–98 CrossRef CAS.
- V. E. Fioletov, L. J. McArthur, T. W. Matthews and L. Marrett, On the relationship between erythemal and vitamin D action spectrum weighted ultraviolet radiation, J. Photochem. Photobiol., B, 2009, 95, 9–16 CrossRef CAS.
- M. F. Holick, Vitamin D deficiency, N. Engl. J. Med., 2007, 357, 266–281 CrossRef CAS.
- I. R. Reid, D. J. Gallagher and J. Bosworth, Prophylaxis against vitamin D deficiency in the elderly by regular sunlight exposure, Age Ageing, 1986, 15, 35–40 CrossRef CAS.
- G. Jones and T. Dwyer, Bone mass in prepubertal children: gender differences and the role of physical activity and sunlight exposure, J. Clin. Endocrinol. Metab., 1998, 83, 4274–4279 CrossRef CAS.
- Vitamin D, Cancer Council Australia, http://www.cancer.org.au/cancersmartlifestyle/SunSmart/VitaminD.htm, accessed October 2009 Search PubMed.
- Vitamin D and Cancer, Working Group Reports, International Agency for Research on Cancer, WHO Press, Geneva, vol. 5, 2008 Search PubMed.
- A. R. Webb, L. Kline and M. F. Holick, Influence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonston will not promote vitamin D3 synthesis in human skin, J. Clin. Endocrinol. Metab., 1988, 67, 373–378 CrossRef CAS.
- T. C. Chen, Photobiology of vitamin D, in Vitamin D Physiology, Molecular Biology, and Clinical Application, ed. M. F. Holick, Humana Press, New Jersey, 1998, pp. 17–37 Search PubMed.
| This journal is © The Royal Society of Chemistry and Owner Societies 2010 |
