Using acridinium ester as the sonochemiluminescent probe for labeling of protein
Yongquan
Lai
,
Yuanyuan
Qi
,
Jian
Wang
* and
Guonan
Chen
*
Ministry of Education Key Laboratory of Analysis and Detection Technology for Food Safety (Fuzhou University), and Department of Chemistry, Fuzhou University, Fuzhou, Fujian 350002, China. E-mail: gnchen@fzu.edu.cn; jwang@fzu.edu.cn; Fax: +86 591-83713866; Tel: +86 591-87893315 Tel: +86 591-87893229
First published on 20th October 2008
Abstract
A new sonogenerated chemiluminescence (SCL) system has been developed to investigate the SCL behavior of acidinium ester (AE). The optimum parameters for this SCL system have also been investigated in detail. Under the optimum conditions, a typical SCL signal could be obtained in a mixture of DMF and alkaline water solutions with volume ratio of 3 : 2. According to electrospray ionization mass spectrometry and sonochemiluminescence emission spectra, SCL emission could be ascribed to the formation of N-methylacridone with a different mechanism from that in both the chemiluminescence and electrochemiluminescence reaction of acridinium ester. The acridinium ester has been used as a SCL probe for labeling of anti-human insulin guinea pig polyclonal antibody, and it was found that the concentration of AE-conjugated antibody was linear with the SCL intensity in the range of 0.15–0.75 ppb with a linear correlation coefficient of 0.9980.
1 Introduction
Ultrasound science and technology has become a rapidly growing research field,1 and it has been widely used in many research areas, such as chemical synthesis, clinic therapeutics, environmental protection, electrochemistry and food processing.2 Ultrasonic irradiation of liquids provides a unique chemical environment originating from acoustic cavitations bubbles. The whole process of the generation, growth and collapse of cavities occurs in an extremely short period of time, about in the order of a few microseconds.3 Consequently, a liquid zone (hot spot) with extremely high temperature and pressure is produced,4,5 at which a high-energy chemical reaction can occur.6 Furthermore, the collapse of bubbles is accompanied by the simultaneous emission of light, called sonoluminescence (SL), which is manifested in two forms: multibubble SL and single-bubble SL.7 Alkaline luminol solution under the irradiation of ultrasound generates one kind of emission called sonochemiluminescence (SCL), which is distinguishable from SL.8 The SCL is identical to chemiluminescence (CL) in the bulk liquid region, which results from the chemical reaction of luminol molecules with OH radicals produced during the process of bubble collapse.9,10Acridinium ester has been usually used as the CL label in immunoassays due to its extremely high sensitivity and selectivity.11–15 Since CL was firstly used in immunoassays,12 a variety of configurations have been introduced in assays of clinical importance. Flow injection analysis (FIA) used in association with CL immunoassays has proved to offer advantages of faster speed and better reproducibility.16 Littigt and co-workers studied the ECL characteristics of acridinium ester by using FIA. They demonstrated that introduction of an electrode into the flow cell could eliminate the need for adding H2O2 in solution and afford greater spatial and temporal control over reaction initiation.17 Both of the above mentioned CL and ECL reactions are initiated by H2O2 in alkaline solution. The hydroperoxyl anion (HO2−) dissociated from H2O2 attacks the acridinium ring structure, which results in multiple bond cleavage to produce N-methylacridone in the excited state to emit CL and ECL.17,18
SCL as an analytical tool has some important advantages. The SCL signal can be produced by sonolysis avoiding the step of adding H2O2 or organic peroxides, which is absolutely necessarily in the liquid phase CL reactions. The detection limits of SCL may be lower than that of ECL, and an inconvenient renewal procedure for the electrode surface by using mechanical smoothing in ECL methods can be avoided.
In this paper, SCL behavior and reaction route of acridinium ester is reported for the first time. The investigation on the SCL mechanism of acridinium ester showed that the emission was due to the formation of N-methylacridone, which was identical to the CL or ECL. However, one of the most important reactants, H2O2, was obtained by sonolysis instead of adding H2O2. Therefore, based on our experiments, a new possible SCL reaction mechanism for acridinium ester was proposed. Meanwhile, the use of acridinium ester as the SCL probe for labeling of protein has also been investigated.
2 Experimental
2.1 Materials
Acridinium NHS ester was obtained from Cayman Co., Ltd and its structure is shown in Scheme 1. Acridinium NHS ester was diluted with DMF and stored at −20 °C before used. Anti-human insulin guinea pig polyclonal antibody was obtained from Beijing Saichi biology and technology Co., Ltd. The dialytic bag was obtained from Shanghai Green Bird Science & Technology Development Co., Ltd. The molecular weight cutoff (MWCO) of the dialytic bag is 2000. All other reagents used were analytical grade from China National Medicines Corporation Ltd, and used without further purification. Doubly distilled water was used throughout for the preparation of solutions. The following buffers were used in the study. The labeling buffer solution (pH 8.0) contained 0.1 mol/L phosphate buffer solution and 0.15 mol/L NaCl;11 dialysate buffer contained 0.1 mol/L NaH2PO3.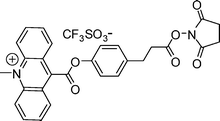 | ||
| Scheme 1 Molecular structure of acridinium NHS ester. | ||
2.2 Apparatus
The experimental apparatus is shown schematically in Fig. 1. SCL intensity was measured by using a BPCL Ultra-Weak Chemiluminescence Analyzer supplied by the Institute of Biophysics, Chinese Academy of Science, which was controlled by a personal computer with a BPCL program. The ultrasound source was supplied by a sonoreactor and piezoelectric transducer horn obtained from Shengxichaoshengyiqi Co. Ltd., Shanghai, China. The frequency of the sonoreactor is 20 kHz and its maximum out-power is 300 W. The transducer horn was used in conjunction with a 6 mm diameter cylindrical titanium sonoprobe tip. Mass spectrometry was performed on an Agilent USA (1100 LC-MSD Trap XCT).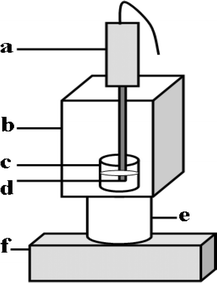 | ||
| Fig. 1 Schematic diagram of experimental apparatus (a) piezoelectric transducer horn; (b) light-proof box; (c) glass cell and sonicated solution; (d) titanium sonoprobe tip; (e) photomultiplier tube; (f) BPCL Ultra-Weak Chemiluminescence Analyzer. | ||
2.3 Procedures
3 Results and discussion
3.1 Phenomenon and reaction route of SCL for acridinium ester
It is well accepted that the CL reaction is typically initiated by the addition of alkaline H2O2, for example the CL reaction of luminol, lucigenin, acridinium ester derivatives and so on. H2O2 plays an important role in these CL reactions. In the case of ECL for acridinium ester, H2O2 was generated from reduction of oxygen by electrochemistry.17 The most probable CL reaction mechanism of acridinium ester is presented in Fig. 2a. H2O2 is dissociated to form the hydroperoxyl anion (HO2−), which attacks the 9-position of the acridinium ring. The attack would induce multiple bond cleavage to produce N-methylacridone in the excited state. Relaxation of the acridone to the ground state is via emission of light at 430 nm.12 All intermediates, except the dioxetanone, have been isolated and characterized.19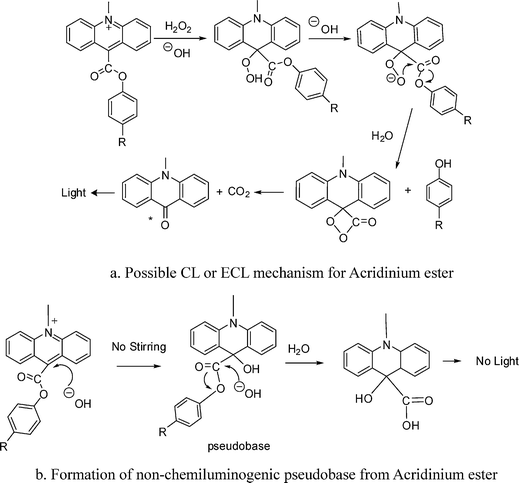 | ||
| Fig. 2 CL mechanism for acridinium ester. | ||
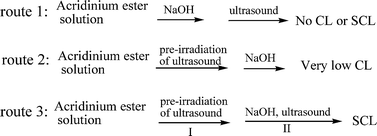
The SCL reaction of luminol has been well studied, which has been used as a tool to study the mechanism of cavitation.20–23 The SCL of luminol was initiated by sonochemical generation of radical species in alkaline solution.9 In order to confirm the possibility that sonochemical generation of highly active radicals initiated the CL reaction similar to SCL reaction of luminol, we examined the SCL reaction of acridinium ester through three different reaction routes:
The concentration of acridinium ester solution was 2 nmol/L and the solvent consisted of DMF and H2O at a volume ratio of 3 : 2.
In route 1, 16 µl of 0.8 mol/L sodium hydroxide solution was carefully transferred to acridinium ester solution, which was followed by ultrasound irradiation. Finally, there was no emission of CL or SCL observed. The reason may be due to the formation of pseudobase. In the alkaline solution, acridinium ester is in equilibrium with a pseudobase formed by hydroxyl substitution at the 9-position of the heterocycle, as shown in Fig. 2b. Although the acridinium ester is easily transferred into its pseudobase form in alkaline solution,13 it is impossible for the pseudobase to further react with hydroxyl (OH−) to produce SCL. This is because C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O is easier to be attacked by the nucleophilic OH− than the 9-position of the heterocycle. The product of the reaction between pseudobase and OH− is acridinium acid but not acridone, and the former is unable to produce emission (see Fig. 2b). Thus, SCL doesn't come from the reaction between pseudobase and hydroxyl (OH−). From the results, we conclude that one is unable to obtain SCL emission through reaction route 1 because of pre-exposure of acridinium ester to base.
O is easier to be attacked by the nucleophilic OH− than the 9-position of the heterocycle. The product of the reaction between pseudobase and OH− is acridinium acid but not acridone, and the former is unable to produce emission (see Fig. 2b). Thus, SCL doesn't come from the reaction between pseudobase and hydroxyl (OH−). From the results, we conclude that one is unable to obtain SCL emission through reaction route 1 because of pre-exposure of acridinium ester to base.
In the case of route 2, by contrast, when the acridinium ester solution was irradiated firstly by ultrasound for 10 seconds (this step is, in brief, called pre-irradiation of ultrasound in the following part), one kind of emission was generated simultaneously, which is called SL caused by active free radicals, shown in Fig. 3A(a). After 16 µl of 0.8 mol/L sodium hydroxide solution was subsequently added, another kind of low emission was observed, see Fig. 3A(b). Though the emission is much weaker relative to SL, this result indicates that some product, for example H2O2, was formed during the first step of pre-irradiation of ultrasound. This sonochemical generation ignited the light together with subsequently adding sodium hydroxide solution. We consider the reaction involving this emission as CL reaction, because it is identical to CL reaction generated by alkaline H2O2 solution. In addition, low CL emission illuminates the fact that there was too little amount of sonochemical generation produced in pre-irradiation of ultrasound to cause an efficient CL reaction.
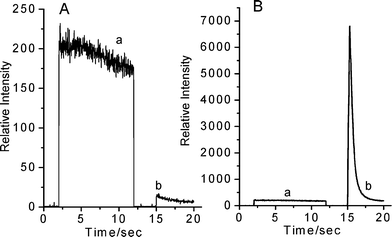 | ||
| Fig. 3 The phenomenon and reaction route of SCL for acridinium ester. Condition: The concentration of acridinium ester solution is 2 nmol/L; the solvent consisted of DMF and H2O at a volume ratio of 3 : 2 and 16 μl of 0.8 mol/L sodium hydroxide solution was added in the reaction of step II. | ||
The result obtained from step I of route 3 is equal to the result obtained from route 2 (see Fig. 3A(a) and 3B(a)). However, after 16 µl of 0.8 mol/L sodium hydroxide solution was carefully transferred to the solution, subsequent irradiation of ultrasound caused a highly intense emission of SCL, relative to SL, shown in Fig. 3B(b). From the figure, we can examine the kinetics of the SCL reaction of step II. The whole reaction was finished completely within 5 s, and the reaction speed increased sharply from the beginning. After reaching the peak at 0.3 s, the speed started to decline. This behavior and the results of the SCL reaction may be due to extraordinary physical and chemical conditions generated from cavitational collapse. This is because in clouds of cavitating bubbles, these hot-spots have equivalent temperatures of roughly 5000 K, pressures of about 1000 atmospheres, heating and cooling rates above 1010 K/s, and intensive shockwaves and microjet at high velocities estimated around 100 m s−1.
In a conclusion, we can get the efficient SCL through reaction route 3, which includes two steps, in brief, called pre-irradiation of ultrasound and ignition of SCL, respectively. During the period of pre-irradiation of ultrasound, some products like H2O2 were formed from free radicals caused by cavitational collapse and accumulated in the meantime; and then further irradiation of ultrasound ignites SCL emission together with sonochemical generation and adding sodium hydroxide solution.
3.2 Possible SCL mechanism for acridinium ester
It has been well known that the phenomenon of acoustic cavitation is the origin of sonochemical effects in liquids. The sonochemical reaction is thought to occur in the cavitation bubble or in its immediate vicinity.24 At the moment of the cavitation bubbles’ collapse, the local temperature at the hot spot reached a range of 2000–5000 K,4,25,26 which was sufficiently high to break any chemical bond.27 In air-saturated water, collapse of microscopic cavitation bubbles induced the homolytic cleavage of H2O and dissolved O2; the process could be described as below in reactions (1)–(2):28–30| H2O → H˙ + HO˙ | (1) |
| O2 → O + O | (2) |
| HO˙ + HO˙ → H2O2 | (3) |
| O + H2O → 2HO˙ | (4) |
| H˙ + O2 → HO2˙ | (5) |
| HO2˙ + HO2˙ → H2O2 + O2 | (6) |
The cleavage products, hydroxyl radicals (HO˙), hydrogen atoms (H˙) and oxygen atoms (O), react further and finally result in the formation of H2O2. The sonochemical generation of H2O2 has been well documented, and reactions (3)–(6) are reported to be significant routes of H2O2 formation.20,29–31
According to the above mechanism of sonochemical generation and discussion in section 3.1, the mostly probable sonochemical generation produced in pre-irradiation of ultrasound was H2O2, which plays a large role in the SCL reaction. This is because H2O2, unlike active radicals (OH˙) is stable enough to react with acridinium ester in step II of route 3. In order to confirm the importance of sonochemical generation H2O2, we examined the effect of ascorbic acid on SCL reaction. Aqueous solutions of ascorbic acid (H2A, vitamin C) have been widely used by inorganic chemists as a reducing agent.32 Its efficiency as an antioxidant is most likely due to a combination of the chemical properties of various components of the ascorbic acid system, namely, ascorbic acid (AH), ascorbate (AH−). AH2/AH− is a very efficient radical scavenger (OH, HO2/O2−, R˙, RO˙, RO2˙, etc.).33 Based on the reactions (3)–(6), ascorbic acid is able to prevent the formation of H2O2. We can see from the Fig. 4A, ascorbic acid can decrease efficiently the intensity of SCL. Furthermore, ascorbic acid almost quenches the emission SCL when its concentration reached 100 × 10−4 mg/ml. It is obvious that the result demonstrates the vital importance of H2O2 on the SCL reaction. In addition, we also studied the effect of pre-irradiation of ultrasound time on SCL reaction, shown in Fig. 4B. The intensity of SCL increases rapidly with pre-irradiation of ultrasound time from 2 to 11 s and reaches its peak at 20 s. However, ISCL decreases at 25 s, the reason may be ascribed to the fact that the amount of H2O2 declines as well.
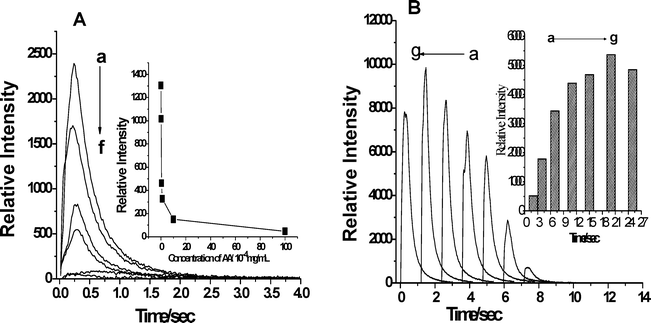 | ||
| Fig. 4 Relative SCL intensity as a function of concentration of ascorbic acid (A) and pre-irradiation of ultrasound time (B). (A) The concentration of ascorbic acid from a to f is 0, 0.2, 0.5, 1, 10, 100 × 10−4 mg/mL, respectively. The SCL was deducted from SL; in the case of the inset, the value of the y-axis is obtained by integrating SCL from time 0 to 4 s. Condition: The concentration of acridinium ester solution is 0.5 nmol/L; the solvent consisted of DMF and H2O at volume ratio of 3 : 2, the pre-irradiation of ultrasound time is 10 s and 16 μl of 0.8 mol/L sodium hydroxide solution was added in the reaction of step II. (B) The pre-irradiation of ultrasound time from a to g is 2 s, 4 s, 7 s, 11 s, 15 s, 20 s, and 25 s, respectively. The SCL was deducted from SL; in the case of the inset, the value of the y-axis is obtained by integrating SCL from time 0 to 4 s. Condition: The concentration of acridinium ester solution is 2 nmol/L; the solvent consisted of DMF and H2O at a volume ratio of 3 : 2 and 16 μl of 0.8 mol/L sodium hydroxide solution was added in the reaction of step II. | ||
Based on the above discussion, we proposed a probable SCL reaction mechanism for acridinium ester (see Fig. 5). In the first step, H2O and dissolved O2 was broken up by ultrasound and leads to the formation of H2O2; and then further irradiation of ultrasound ignites SCL emission together with H2O2 and subsequently adding sodium hydroxide solution. The key effect of ultrasound on the SCL reaction is that the ultrasound provides extraordinary physical and chemical conditions to generate SCL emission, which is impossible to occur under common conditions.
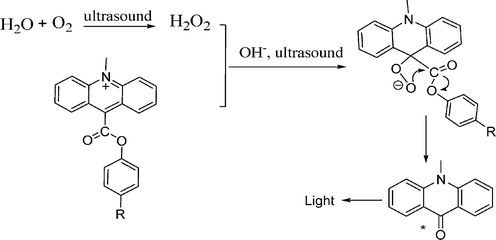 | ||
| Fig. 5 Possible SCL mechanism for acridinium ester. | ||
3.3 Sonolysis production of acridinium ester
In order to ascertain the SCL reaction mechanism, mass spectrometry measurements were performed. Fig. 6 shows the mass spectra of the sonolysis productions of acridinium NHS ester. The peak at 531.8 m/z is likely the result of the loss of the pyrrolidine-2, 5-dione when the N–O is cleaved, because the active ester is not very stabile and easy to hydrolyze. The strongest peak at 278.4 m/z belongs to N-methylacridone combined with trifluoromethyl group, which effectively supports the SCL reaction mechanism presented above. Similarly, the peak at 307.3 m/z suggests the formation of acridinium carboxylic acid adducted with the trifluoromethyl group. Although this peak at 307.3 m/z is much weaker than the peak at 278.4 m/z, the results indicate that the sonic energy caused attack by OH− at the phenyl ester carbonyl, the product of which is the acridinium carboxylic acid.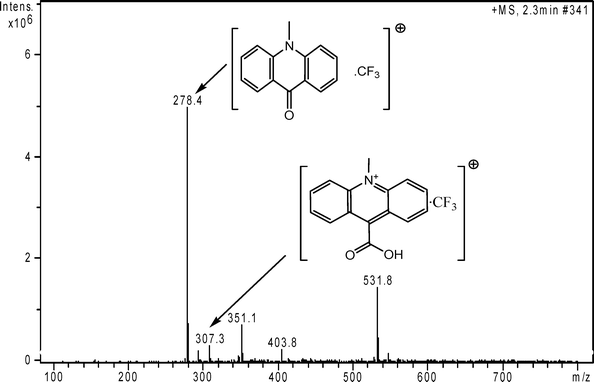 | ||
| Fig. 6 Mass spectra of sonolysis productions of acridinium NHS ester. Measurement conditions: Scanning range: 100–800 m/z; Flow rate of drying gas: 5 L/min; Evaporative gas pressure: 15 psi; Temperature: 325 °C; Ion source: positive electrospray ionizaion mode; Capillary voltage: 3.5 kV. | ||
Moreover, in order to further demonstrate the formation of N-methylacridone, we also examined the SCL spectra of acridinium ester, and the results showed that the maximum wavelength of the SCL spectrum was 440 nm, this peak is similar to that of the fluorescence emission of N-methylacridone, which has been reported at 430 nm with excitation at 395 nm.11,12 All these results suggested that when exposed to ultrasound and dilute NaOH solution in the present of sonochemical generation of H2O2, acridinium NHS ester was cleaved at the 9-position of the heterocycle and N-methylacridone in the excited state was finally formed, which was the oxidization product of acridinium ester and the primary emitter of the SCL reaction.
3.4 Using of acridinium ester as the SCL probe for protein
The appropriate reaction conditions for SCL reaction of acridinium ester were investigated. It was found that the volume percentage of DMF has a large effect on the SCL. It can be seen from Fig. 7 that the volume percentage of DMF has a large effect on the SCL, when the volume percentage of DMF was increased from 40% to 60%, the SCL intensity of c is about 50-times that of a. While the volume percentage was above 60%, the SCL decreased slightly.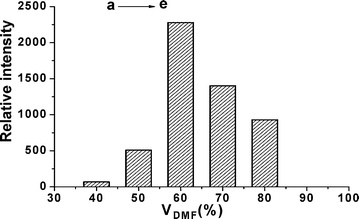 | ||
| Fig. 7 Relative SCL intensity as a function of volume percentage of DMF. The SCL was deducted from SL and the value of the y-axis is obtained by integrating SCL from time 0 to 4 s. The volume percentage from a to e were 40%, 50%, 60%, 70%, 80%, respectively. Conditions: The concentration of acridinium ester solution was 6.25 × 10−10 mol/L; the solvent consisted of DMF and H2O at volume ratio of 3 : 2; the pre-irradiation of ultrasound time was 15 s and 16 μl of 0.8 mol/L sodium hydroxide solution was added in the reaction of step II. | ||
The influence of NaOH concentration on SCL intensity was studied in the range of 0.08–1.6 mmol/L, and the results showed that ISCL increased with increasing NaOH concentration in the range of 0.08–0.4 mmol/L. However, the ISCL decreases slowly when the NaOH concentration is higher than 0.4 mmol/L of NaOH.
Acridinium ester was used as the SCL probe for labeling of the anti-human insulin guinea pig polyclonal antibody. The excess free acridiniuun ester was removed by dialysis, but the unlabeled antibody was not removed, which didn't influence the SCL reaction of AE-labeled antibody. Under the optimum conditions, the linear relationship between ISCL and concentration of AE-labeled antibody could be obtained in the range of 0.15–0.75 ppb with correlation coefficient of 0.9980 and detection limit of 0.09 ppb (see Fig. 8), and the RSD was less than 4%.
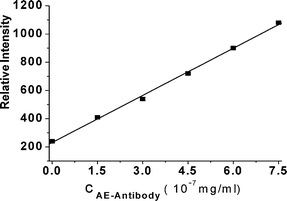 | ||
| Fig. 8 Relationship between ISCL and concentration of AE-conjugated antibody. The SCL was deducted from SL and the value of the y-axis is obtained by integrating SCL from time 0 to 4 s. Conditions: The solvent consisted of DMF and H2O at volume ratio of 3 : 2; the pre-irradiation of ultrasound time was 15 s and 16 μl of 0.8 mol/L sodium hydroxide solution was added in the reaction of step II. | ||
4 Conclusions
In this paper, a new SCL system for investigation of acidinium ester has been developed. The experiment showed that acidinium ester gave SCL in a mixture of DMF and alkaline water solutions with volume ratio of 3 : 2 under ultrasonic irradiation. The SCL reaction route and mechanism of acridinium ester have been investigated, and the results showed that the emission was due to the formation of N-methylacridone as the reactive intermediate, which was identical to the CL or ECL. However, one of the most important reactants, H2O2 was obtained by ultrasound instead of adding H2O2. Based on which, a new possible SCL reaction mechanism for acridinium ester was proposed. The acridinium ester was used as a SCL probe for labeling of anti-human insulin guinea pig polyclonal antibody, and it was found that the AE-labeled protein still gave a strong SCL.Acknowledgements
This project was supported by the National Nature Sciences Foundation of China (20735002, 20575011), Young Scientist Innovation Foundation of Fujian Province, china (2006F3071) and Science Foundation of Education Department of Fujian Province, China (JB06052).References
- T. J. Mason, Ultrason. Sonochem., 2003, 10, 175 CrossRef CAS.
- Bresançson, May. Fourth Conference in Applications of Power Ultrasound in Physical and Chemical Processing. 2003, France.
- V. S. Moholkar, P. S. Kumar and A. B. Pandit, Ultrason. Sonochem., 1999, 6, 53 CrossRef CAS.
- E. B. Flint and K. S. Suslick, Science, 1991, 253, 1397 CrossRef.
- W. B. McNamara III, Y. T. Didenko and K. S. Suslick, Nature, 1999, 401, 772 CrossRef.
- K. S. Suslick, Science, 1990, 247, 1439 CrossRef CAS.
- L. A. Crum, Physics Today, 1994, 47, 22 CrossRef CAS.
- E. N. Harvey, J. Am. Chem. Soc., 1939, 61(9), 2392 CrossRef CAS.
- H. N. McMurray and B. P. Wilson, J. Phys. Chem. A, 1999, 103(20), 3955 CrossRef CAS.
- S. Hatanaka, H. Mitome, K. Yasui and S. Hayashi, J. Am. Chem. Soc., 2002, 124, 10250 CrossRef CAS.
- I. Weeks, I. Beheshti, F. McCapra, A. K. Campbell and J. S. Woodhead, Clin. Chem., 1983, 29(8), 1474 CAS.
- I. Weeks, M. Sturgess, R. C. Brown and J. S. Woodhead, Methods Enzymol., 1986, 133, 366 Search PubMed.
- J. S. Littig and T. A. Nieman, J. Biolumin. Chemilumin., 1993, 8, 25 CrossRef CAS.
- J. M. Lin and M. Yamada, Microchem. J., 1998, 58, 105 CrossRef CAS.
- H. Sato, H. Mochizuki, Y. Tomita, T. Izako, N. Sato and T. Kanamori, J. Biolumin. Chemilumin., 1996, 11(1), 23 CrossRef CAS.
- H. J. Liu, J. C. Yu, D. S. Bindra, R. S. Givens and G. S. Wilson, Anal. Chem., 1991, 63, 666 CrossRef CAS.
- J. S. Littig and T. A. Nieman, Anal. Chem., 1992, 64, 1140 CrossRef CAS.
- F. McCapra, Acc. Chem. Res., 1976, 9, 201 CrossRef CAS.
- A. Townshend, Analyst, 1990, 115, 495 RSC.
- C. Petrier, M. F. Lamy, A. Francony, A. Benahcene, B. David, V. Renaudin and N. Gondrexon, J. Phys. Chem., 1994, 98, 10514 CrossRef CAS.
- A. Henglein, R. Ulrich and J. Lilie, J. Am. Chem. Soc., 1989, 111, 1974 CrossRef CAS.
- V. Renaudin, N. Gondrexon, P. Boldo, C. Petrier, A. Bernis and Y. Gonthier, Ultrason. Sonochem., 1994, 1, S81 CrossRef CAS.
- A. Henglein, D. Herburger and M. Gutierrez, J. Phys. Chem., 1992, 96, 1126 CrossRef CAS.
- A. Tuulmets, Ultrason. Sonochem., 1997, 4(2), 189 CrossRef CAS.
- E. J. Hart, C. H. Fischer and A. Henglein, Radiat. Phys. Chem., 1990, 36, 511 CrossRef CAS.
- V. Misík, N. Miyoshi and P. Riesz, J. Phys. Chem., 1995, 99, 3605 CrossRef CAS.
- L. S. Bernstein, M. R. Zakin, E. B. Flint and K. S. Suslick, J. Phys. Chem., 1996, 100, 6612 CrossRef CAS.
- K. Makino, M. M. Mossoba and P. Riesz, J. Am. Chem. Soc., 1982, 104, 3537 CrossRef CAS.
- C. H. Fischer, E. J. Hart and A. Henglein, J. Phys. Chem., 1986, 90, 1954 CrossRef.
- E. J. Hart and A. Henglein, J. Phys. Chem., 1987, 91, 3654 CrossRef CAS.
- A. Weissler, J. Am. Chem. Soc., 1959, 81, 1077 CrossRef CAS.
- A. V. Zelewsky, Inorg. Chem., 1981, 20, 4448 CrossRef.
- D. E. Cabelli and B. H. J. Bielski, J. Phys. Chem., 1983, 87, 1809 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2009 |