Efficient exciton dissociation in isomeric BINOL-based porous polymers for sacrificial agent-free H2O2 photosynthesis and biomass valorization†
Flora
Banerjee
a,
Sougata
Saha‡
b,
Soumitra
Sau‡
 a,
Shubhangi
Majumdar
a,
Shubhangi
Majumdar
 c,
Shiladitya
Roy
a,
Pramit K.
Chowdhury
c,
Shiladitya
Roy
a,
Pramit K.
Chowdhury
 c,
Swapan K.
Pati
b and
Suman Kalyan
Samanta
c,
Swapan K.
Pati
b and
Suman Kalyan
Samanta
 *a
*a
aDepartment of Chemistry, Indian Institute of Technology Kharagpur, Kharagpur 721302, India. E-mail: sksamanta@chem.iitkgp.ac.in
bTheoretical Sciences Unit, School of Advanced Materials, Jawaharlal Nehru Centre for Advanced Scientific Research, Bangalore 560064, India
cDepartment of Chemistry, Indian Institute of Technology Delhi, Hauz Khas, New Delhi-110016, India
First published on 30th June 2025
Abstract
Achieving sustainable, sacrificial agent-free hydrogen peroxide (H2O2) production at the millimolar scale through molecular-level modulation of organic semiconductors is a crucial global challenge. In this study, novel hierarchical porous polymers incorporating triphenylamine and BINOL (1,1′-bi-2-naphthol) were synthesized using FeCl3-mediated homopolymerization, forming BINOL in situ, unlike conventional approaches that rely on pre-formed derivatives. These polymers, designed with varied linkage positions, exhibit remarkable optoelectronic properties, enabling efficient artificial photosynthesis of H2O2 up to 2.5 mmol·g−1·h−1 from natural water sources (river, tap, and seawater) without any additives. A direct 2e− oxygen reduction and water oxidation pathway facilitated stable H2O2 generation, achieving 6.47 mmol·g−1·h−1 in pure water under AM 1.5 G illumination, with a significantly high solar-to-chemical conversion efficiency of 1.6%. This rate was further increased to 27.5 mmol·g−1·h−1 in isopropanol/water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), ranking among the highest reported values thus far. Biomass-derived sacrificial agents such as 5-hydroxymethyl furfural and tetrahydrofuryl alcohol (THFA) further increased the generation rate (5.17 mmol g−1 h−1 in 1
1), ranking among the highest reported values thus far. Biomass-derived sacrificial agents such as 5-hydroxymethyl furfural and tetrahydrofuryl alcohol (THFA) further increased the generation rate (5.17 mmol g−1 h−1 in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 THFA/water), mitigating energy demands in both ways: H2O2 production and biomass valorization. Notably, the polymers were recycled up to ten consecutive runs without any loss in their catalytic efficiency. In addition, DFT calculations confirmed the BINOL served as the potential oxygen reduction site with thermodynamic feasibility for H2O2 formation, with a free energy release of 2.86 eV in IPA/water (1
10 THFA/water), mitigating energy demands in both ways: H2O2 production and biomass valorization. Notably, the polymers were recycled up to ten consecutive runs without any loss in their catalytic efficiency. In addition, DFT calculations confirmed the BINOL served as the potential oxygen reduction site with thermodynamic feasibility for H2O2 formation, with a free energy release of 2.86 eV in IPA/water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) and 0.38 eV in pure water.
10) and 0.38 eV in pure water.
1 Introduction
With the surging deficit of fossil fuels, photocatalytic production of hydrogen peroxide (H2O2) is receiving considerable interest in terms of green economy, water remediation, textile bleaching and forming rubber and other organic chemicals.1 In addition, a fuel cell can effortlessly transform the chemical energy present in H2O2 into electrical energy. H2O2 has the advantages of zero carbon emissions, a high energy density (3.0 MJ·L−1, 60 wt%) comparable to that of H2 fuel cells (2.8 MJ·L−1, 35 MPa H2), and an easy-to-assemble single-chamber cell design when utilized in fuel cells (oxidation and reduction of H2O2 as a single component).2,3 The production of H2O2 across the globe has reached 6.5 million tons in 2022, which itself expresses the present concern from the perspective of H2O2 as a versatile oxidant, easy to transport, and feasible to store, along with meeting the energy crisis as a single-component fuel cell.4 The most convenient pathway for H2O2 synthesis is estimated to be the 2e− oxygen reduction into peroxide.5 However, the reaction of molecular oxygen in its triplet state is spin-forbidden according to Wigner's spin selection rule; thus, oxygen activation is a necessary step in performing an oxygen reduction reaction. H2O2 synthesis on an industrial scale is majorly (95% of the overall H2O2) dependent on the anthraquinone oxidation method, which comes with compromised energy efficiency and the generation of toxic side products.6 Metal-based catalysts (Pd, Pd–Au) are also employed in direct H2O2 generation but are less preferable considering their explosive nature stemming from the use of an H2/O2 gas mixture.7 On this account, the artificial photosynthesis approach provides a suitable alternative to the conventional protocol involving oxygen and abundant water, using sunlight as the natural source of irradiation.8–10 Until now, several systems have been employed to achieve the photo production of H2O2, including TiO2 and metal–organic frameworks, which are associated with some drawbacks such as limited visible-light absorption and faster kinetics toward H2O2 decomposition, resulting in compromised efficiency.11–13 Presently, systems are designed rationally, keeping the criteria of environmental friendliness, potential recyclability and cost-effectiveness, and organic semiconductors could offer several advantages compared to metal alloys. Organic semiconductors are associated with superior visible light absorptivity and facile charge transfer property, furnishing solar-driven H2O2 production with high apparent quantum efficiency, a lower probability for any side reactions and high selectivity for H2O2 as the sole product.Sacrificial agent-mediated H2O2 production has garnered considerable interest owing to its potential to minimize electron–hole (e−–h+) pair recombination and improve electron consumption efficiency. To further improve the performance of semiconductors in photocatalytic H2O2 production by way of hole scavenging, several sacrificial agents have been used, including furyl alcohol and benzyl alcohol, which in turn produced value-added chemicals as oxidation products, simultaneously generating renewable energy sources and producing valuable synthons. In this context, lignocellulose and hemicellulose-derived natural feedstock chemicals and their further oxidation into carboxylic acids have garnered considerable attention among researchers owing to their large abundance and significant contribution in producing biofuel, electricity, and biogas; they could act as illuminating alternatives to overcome the current depletion in fossil fuels.14 At present, global energy consumption has increased by 37% within the last 15 years, of which 72% is coming from biomass valorization.15 Thus, designing a cascade chemical transformation pathway employing a suitable heterogeneous catalyst with tailorable redox motifs is pivotal. Fascinating catalytic systems such as metal–organic frameworks16 and Co nanoparticle-embedded carbon nanotubes with VO2/polyaniline17 have been reported for the oxidation of vanillyl and furyl alcohols.18 Among them, porous polymers are preferred owing to their superior thermochemical stability and long cycle life. Keeping that in mind, Thomas and co-workers synthesized covalent organic frameworks using the Povarov reaction, furnishing H2O2 with a production rate as high as 5.6 mmol·g−1·h−1 under solar radiation with benzyl alcohol as the sacrificial agent.19 However, the capability of benzyl alcohol alone to produce H2O2 under light irradiation makes it necessary to cultivate a sacrificial agent-free protocol to achieve a more sustainable approach.20–22 Furthermore, it is uneconomical to use an electron donor as a sacrificial agent since additional efforts are required to remove H2O2 from the solvent-containing sacrificial agent.23
Recently, triphenylamine was incorporated into a porous polymer by Tan and co-workers, and the polymer could facilitate suitable oxygen reduction to generate H2O2 even in the absence of any sacrificial agent.24 Here, the triphenylamine core offers suitable electron-donating properties under photoexcitation. Its photo- and electrochromic features and stable cation-radical generation tendency have made it an attractive candidate as a photocatalytic synthon and even a hole–transporting material. While the triphenylamine core is a well-studied photoredox active moiety, the BINOL (1,1′-bi-2-naphthol) entity is still underexplored in terms of photocatalytic applications, although it can potentially be used as a photoactive core.25 Due to their inherent axial chirality and Brønsted acid functionality, chiral BINOL phosphoric acid-based insoluble microporous polymers have performed well in catalyzing aza–ene reactions and Friedel–Crafts type reactions. Blechert and co-workers reported that a phosphoric acid-functionalized BINOL-based porous organic polymer (POP) showed intrinsic microporosity, with a Brunauer–Emmett–Teller (BET) surface area of 386 m2 g−1, and displayed chiral heterogeneous catalysis, such as asymmetric transfer hydrogenation of 2-aryl quinoline and benzoxazines (with 98% ee).26
This study presents an innovative strategy for enhancing photocatalytic performance by synthetically locking a triphenylamine (TPA) unit within a BINOL-based porous organic polymer (POP), which broadens the light absorption spectrum and effectively suppresses exciton recombination (Fig. 1). Noteworthily, we adopted a bottom-up synthetic route that enables the in situ formation of the BINOL entity during polymerization while integrating a propeller-shaped TPA moiety into the hierarchically porous TPA-BINOL polymers (Scheme 1). This synthetic tailoring of the resulting polymers finally resulted in the engineered intramolecular charge transfer characteristics that underpin their efficiency in converting lignocellulose-derived biomass into biofuel precursors, offering a sustainable and high-performing alternative to fossil fuels. Additionally, our in-depth structural investigation uncovered underexplored exciton dissociation behaviors, along with exceptional oxygen adsorption and reduction capabilities. These features enabled photosynthesis of hydrogen peroxide with a production rate of 6.47 mmol·g−1·h−1, exceeding the performance of current state-of-the-art organic and inorganic photocatalysts.
2 Results and discussion
2.1 Synthesis and characterization
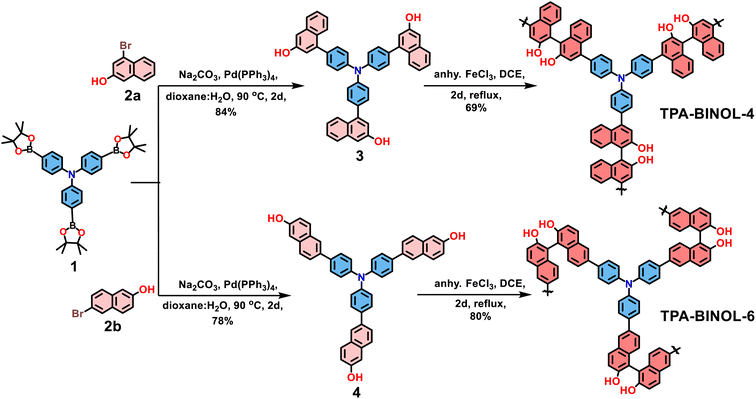
Symmetry engineering within porous organic polymers can tune their optoelectronic and physicochemical properties, which can pave the way to generating several organic polymer systems, boosting their catalytic efficiency, sensing, and light harvesting efficiency. In view of this, our objective is to generate two different hypercrosslinked polymers featuring isomeric BINOL moieties where the BINOL could be constructed during the FeCl3-mediated polymerization step. To realize this, compound 1 and precursors 2a/2b were reacted under Suzuki–Miyaura cross-coupling conditions to produce compounds 3 and 4 with 84% and 78% yields, respectively (Scheme 1). Next, our design strategy aimed to exploit oxidative homopolymerization using FeCl3 to generate symmetry-engineered TPA-BINOL-4 and TPA-BINOL-6, respectively, with 69% and 80% yields. Containing a similar TPA core, the regioisomeric TPA-BINOL-4 possesses BINOL moieties connected in a linear fashion compared to the twisted BINOL connectivity in TPA-BINOL-6, which could bring considerable changes in morphology and porosity. Such tailored connectivity within regioisomeric analogs can show pronounced effects on their optical and electrochemical redox properties.2.2 Structural features
The effectiveness of the oxidative homocoupling was evident from the structural features of the monomer that are retained within the polymers. Structural elucidation for TPA-BINOL-4 and TPA-BINOL-6 was accomplished with solid-state 13C CP/MAS NMR and FT-IR spectroscopy. The solid-state NMR spectrum of TPA-BINOL-6 exhibited peaks at 110 ppm, 117 ppm, 123–130 ppm and 155 ppm corresponding to aryl carbons, indicating the presence of the BINOL skeleton. The peaks between 123–130 and 142–146 ppm indicated the presence of the TPA moiety within the polymer network, indicating a successful homopolymerization of the corresponding monomers (Fig. 2a, S1 and S2, ESI†).27 The FT-IR spectra of TPA-BINOL-6 displayed peaks at 1252 cm−1 and 812 cm−1 arising from C–N symmetric stretching and aryl C–H bending, respectively; these were retained in the polymer with a 15 cm−1 blue shift from the corresponding monomer 4 (Fig. 2b). Alternatively, TPA-BINOL-4 displayed C–N and C–O stretching frequencies at 1265 cm−1 and 1076 cm−1, respectively, inherited from monomer 3. The O–H stretching peak appeared at 3450–3475 cm−1 in both the monomers and the polymers, which is in accordance with naphthol-based systems (Fig. S3, ESI†).28 The structure of monomer 3 was also verified with a mass spectrum (Fig. S4, ESI†). X-ray photoelectron spectroscopy provided crucial information about the elemental states of the carbon, oxygen, and nitrogen present within the POPs (Fig. S5, ESI†). For TPA-BINOL-4, the deconvoluted spectrum in specific regions displayed the C 1s binding energy between 284.5 and 287.2 eV for the C–C bond and C–N/–C–O bond, corresponding to the TPA core, along with an aromatic –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C peak positioned at 282.9 eV. Meanwhile, the O 1s spectrum exhibited a single broad peak at 532.5 eV, corresponding to the C–OH single bond originating from the BINOL motif (Fig. 2c and d).29,30 The effect of symmetry engineering was further evident in their morphological traits. Scanning electron microscope (SEM) images of TPA-BINOL-6 showed aggregated particle-like morphologies with elongated fibers (Fig. 2e). In contrast, TPA-BINOL-4 showed an aggregate of globules with a nearly spherical shape (Fig. S6, ESI†). Energy-dispersive X-ray (EDX) elemental mapping confirmed the homogeneous distribution of carbon, nitrogen, and oxygen within the polymer matrix (Fig. S7, ESI†). The bulk morphology was further elucidated with transmission electron microscopy (TEM), where spheres, rods and smaller aggregates were observed in the case of TPA-BINOL-6 and a spherical shape was predominant for TPA-BINOL-4 (Fig. S8, ESI†). The EDX profile confirmed the absence of Fe in these porous polymers, indicating that the FeCl3 residues were completely removed after washing. The powder X-ray diffraction (PXRD) patterns of both POPs did not show any sharp crystalline peaks; instead, a broad hump between 20–25° indicated the amorphous nature of these POPs (Fig. 2f).31,32 Thermogravimetric analysis (TGA) confirmed the thermal stability of TPA-BINOL-6 up to 210 °C with 5% weight loss (Fig. 2g). However, TPA-BINOL-4 showed 5% weight loss at ∼185 °C, which could be due to the trapped water and organic solvent molecules within the pores of the as-prepared porous polymers.33
C peak positioned at 282.9 eV. Meanwhile, the O 1s spectrum exhibited a single broad peak at 532.5 eV, corresponding to the C–OH single bond originating from the BINOL motif (Fig. 2c and d).29,30 The effect of symmetry engineering was further evident in their morphological traits. Scanning electron microscope (SEM) images of TPA-BINOL-6 showed aggregated particle-like morphologies with elongated fibers (Fig. 2e). In contrast, TPA-BINOL-4 showed an aggregate of globules with a nearly spherical shape (Fig. S6, ESI†). Energy-dispersive X-ray (EDX) elemental mapping confirmed the homogeneous distribution of carbon, nitrogen, and oxygen within the polymer matrix (Fig. S7, ESI†). The bulk morphology was further elucidated with transmission electron microscopy (TEM), where spheres, rods and smaller aggregates were observed in the case of TPA-BINOL-6 and a spherical shape was predominant for TPA-BINOL-4 (Fig. S8, ESI†). The EDX profile confirmed the absence of Fe in these porous polymers, indicating that the FeCl3 residues were completely removed after washing. The powder X-ray diffraction (PXRD) patterns of both POPs did not show any sharp crystalline peaks; instead, a broad hump between 20–25° indicated the amorphous nature of these POPs (Fig. 2f).31,32 Thermogravimetric analysis (TGA) confirmed the thermal stability of TPA-BINOL-6 up to 210 °C with 5% weight loss (Fig. 2g). However, TPA-BINOL-4 showed 5% weight loss at ∼185 °C, which could be due to the trapped water and organic solvent molecules within the pores of the as-prepared porous polymers.33
The porous properties of our as-prepared polymers were inspected using nitrogen adsorption–desorption isotherms at 77 K. Both polymers TPA-BINOL-4 and TPA-BINOL-6 showed an open-ended BET isotherm in the low-pressure region (Fig. 2h and S9a, ESI†). This could develop from either swelling of the polymer networks during adsorption or irreversible adsorption of N2 within the pores.3,34,35 The steep rise in N2 uptake at P/P0 ∼ 0.9 could be attributed to large populated mesopores, i.e. interparticle voids within the polymer which could be helpful for facile mass transfer during the catalytic cycle.36 A large hysteresis loop could result from the rigid and non-planar binaphthalene backbone of the network with capillary condensation within the mesopores, hampering the reversibility of the physisorption process.37,38 The pore size distribution derived from the Barrett–Joyner–Halenda (BJH) method showed narrow distributions located at 1.86 nm and 3.96 nm for TPA-BINOL-4 and 1.6 nm and 2.2 nm for TPA-BINOL-6, each exhibiting both micro- and mesopores within the network, i.e. both the POPs were hierarchically porous in nature (Fig. 2i and S9b, ESI†).39TPA-BINOL-4 showed a moderately high BET surface area of 479 m2 g−1. Interestingly, the microporous surface area was found to be 220 m2 g−1, which confirmed that nearly 40% of the total pores were micropores. The extent of mesopores was around 60%, which could promote diffusion of the substrate and product during catalytic cycles. Under similar conditions, TPA-BINOL-6 possessed a lower BET surface area of 143 m2 g−1, which presumably stems from the comparatively twisted backbone and its pore-blocking effect. The contorted structure in TPA-BINOL-4 and the resulting high BET surface area may confer the polymer with abundant oxygen adsorption sites to facilitate effective oxygen adsorption and reduction, along with suitable mesoporous channels for the diffusion of the product.
2.3 Photophysical properties
Photocatalytic usage demands that light absorption should promote charge carrier mobility within the polymer backbone. Therefore, to investigate the influence of the TPA moiety on the photophysical properties of the synthesized POPs, solid-state UV-vis spectroscopy and electrochemical measurements were performed. The UV-vis diffuse reflectance spectroscopy revealed that the absorption maximum of TPA-BINOL-4 was located near 485 nm, with the absorption edge reaching the near-infrared (NIR) region (Fig. 3a). Notably, compared to TPA-BINOL-4, a slight blue-shift of ∼34 nm in the absorption maximum was observed for TPA-BINOL-6. The optical band gaps (Eg) of the as-synthesized POPs were derived from the corresponding Tauc plots (Fig. 3b). The Eg of TPA-BINOL-4 was calculated to be 1.96 eV. In contrast, TPA-BINOL-6 exhibited a higher optical band gap value (Eg = 2.66 eV). The solid-state PL spectra (λex = 440 nm) highlighted slightly higher fluorescence responses for TPA-BINOL-4 than TPA-BINOL-6 which may have originated from the increased rigidity and restricted bond rotation in the former (Fig. 3c). The energy positions of the valence band were obtained from the valence band XPS (VB-XPS) profiles of both TPA-BINOL-4 and TPA-BINOL-6, with VB onset values at 1.40 and 1.92 eV, respectively (Fig. 3d). Detailed calculations, as given in eqn (S1),† afforded the valence band potentials of 1.31 and 1.83 V, respectively, for TPA-BINOL-4 and TPA-BINOL-6 (w.r.t. NHE). Corresponding conduction band energies were computed as −0.65 V for TPA-BINOL-4 and −0.83 V for TPA-BINOL-6 (eqn (S2), ESI†). The lowest unoccupied molecular orbitals (LUMO) of the as-synthesized TPA-BINOL-based POPs were obtained from cyclic voltammetry (CV) studies (Fig. S10, ESI†). The electrode potential values were calculated with respect to EAg/AgCl = 0.197 vs. NHE.40,41 Thus, the LUMO energy levels were determined for TPA-BINOL-4 (−0.62 V vs. NHE) and TPA-BINOL-6 (−0.67 V vs. NHE). Meanwhile, the reduction potentials were at −0.33 V for O2/O2−˙ and 0.68 V for O2/H2O2 with respect to NHE, which was indicative of greater accountability for the oxygen reduction by our POPs generating superoxide and H2O2 sufficiently (Fig. 3e).28Summarizing these results, TPA-BINOL-4 evidenced a greater propensity towards ROS generation from having a lower-lying LUMO (Fig. 3e). The interesting results obtained from the UV-vis and VB-XPS studies prompted us to explore their theoretical band positions and band gaps. In the case of TPA-BINOL-4, the highest occupied molecular orbital (HOMO) was confined within the triphenylamine moiety, whereas the LUMO was localized over BINOL, indicating its proficiency towards intramolecular charge transfer (Fig. 3f). A similar optimization protocol showed that the HOMO and LUMO of TPA-BINOL-6 were distributed over the TPA-naphthol arm, lowering the possibility of charge separation, which could facilitate a much lower charge transfer property. However, the theoretical HOMO–LUMO gaps were comparable for both polymers TPA-BINOL-6 (3.82 eV) and TPA-BINOL-4 (3.85 eV) (Fig. 3f and S11, ESI†). The photoexciton generation was further established through electron paramagnetic resonance (EPR) spectroscopy for the polymers. The observed single Lorentzian line could be ascribed to the radical generation property of triphenylamine under light irradiation which may have resulted in photoelectron generation properties in both polymers (Fig. S12, ESI†).42 The estimated conduction band edges (ECB) of the samples were well-aligned with the reduction potential of O2/H2O2 (0.68 eV vs. NHE at pH 0), indicating their capability to facilitate the photocatalytic reduction of O2 to H2O2. Compared to conventional diphase systems, the introduction of π-rich BINOL in the polymer significantly enhanced the photocatalytic performance. The established triphase interface could serve as an effective platform for facilitating photocatalytic reactions. Within this interface, gaseous O2, photogenerated charge carriers from the catalyst, and protons derived from water were brought into close proximity, enabling efficient interaction and promoting the desired redox processes.43 This innovative system not only optimized light absorption and accelerated charge transfer but is also expected to improve the diffusion and adsorption of O2 onto the photocatalysts, thereby increasing the interaction efficiency between the catalysts and reactants. Furthermore, the generated H2O2 could readily diffuse into water, minimizing self-decomposition and ensuring sustained production efficiency.44
2.4 Detection of hydrogen peroxide
H2O2 generation was observed in the cases of both TPA-BINOL-6 and TPA-BINOL-4 because the LUMOs of these polymers matched well with the O2/H2O2 redox potential of 0.68 V with respect to NHE.45 Visible-light-driven H2O2 generation was confirmed using the N,N-dimethyl-p-phenylenediamine (DPD) test. The addition of TPA-BINOL-4 to a colourless DPD solution resulted in an immediate change to an intense red solution, indicating the generation of DPD+˙, which confirmed the in situ H2O2 generation by the POPs as the active oxidizing species (Fig. S13a, ESI†) (for details, see eqn (S3), ESI†). A similar observation was noticed with ceric sulfate in 2 N H2SO4, where complete decolorization of the yellow Ce(SO4)2 solution took place upon adding the TPA-BINOL-4 photocatalyst. This was accompanied by a diminished characteristic signal of Ce(IV) at 416 nm, as it was converted to the colorless Ce(III), thus confirming the generation of H2O2 in the medium (Fig. S13b, ESI†) (eqn (S4), ESI†).2.5 Estimation of H2O2
The generated H2O2 was quantified via volumetric titration using ceric sulfate with a ferroin indicator (Experimental section, ESI†).46 The photocatalytic production of H2O2 from water and oxygen was initially examined under blue LED illumination (50 W, λ > 420 nm). Among the synthesized polymers, TPA-BINOL-6 demonstrated a notable H2O2 generation rate of 3.1 mmol g−1 h−1 without any additives (eqn (S5)–(S8), ESI†). Meanwhile, under similar conditions, TPA-BINOL-4 exhibited even greater efficiency, achieving an impressive rate of 4.33 mmol g−1 h−1, which is much higher than those of previously reported metal or metal-free photocatalysts working at high-intensity irradiation (Table S1, ESI†). This enhanced performance is well-correlated with its distinct absorption characteristics and electron paramagnetic resonance (EPR) profile. These results, in turn, were substantiated for naturally abundant water bodies (tap water, river water, and seawater), offering H2O2 up to 2.5 mmol·g−1·h−1 for tap water and a considerable generation rate of 1.9 mmol·g−1·h−1 for seawater without adding any sacrificial agent (Fig. 4a).47,48 These results show that our as-synthesized polymer is capable of producing bulk-scale H2O2 from natural water bodies without the prior addition of any hole scavengers.Plants convert sunlight into chemical energy with an efficiency of about 0.1% during the photosynthesis process.49 Since the as-synthesized polymers showed H2O2 photosynthesis in the absence of any sacrificial agents, artificial photosynthesis tests were conducted under AM 1.5 G simulated sunlight, and a H2O2 production rate of 6.47 mmol·g−1·h−1 was achieved with a solar to chemical conversion efficiency of 1.6% using TPA-BINOL-4 as the photocatalyst (eqn (S9), ESI†). This value clearly surpassed those of the reported organic polymers and inorganic photocatalysts to date in terms of sacrificial agent-free H2O2 generation (Fig. 4b and Table S1, ESI†).50 Utilizing solar radiation as the source of energy makes the entire protocol more practically relevant and less energy-invasive.51 Interestingly, taking natural sunlight as the irradiation source, H2O2 production reached nearly 2.05 and 0.98 mmol·g−1·h−1 for distilled water and seawater, respectively, without any additives, which resulted from the suitable overlap between the absorption spectrum of TPA-BINOL-4 and the solar radiation spectrum (Fig. 4c). The apparent quantum yield with TPA-BINOL-4 was 5.87% (at λ = 456 nm) without any sacrificial agent, which can be placed high among the literature-reported values (Fig. S14 and eqn (S10), ESI†).48,52,53 Moreover, a significant amount of H2O2 was produced, with a rate of 2.53 mmol·g−1·h−1 under a 50 W blue LED in the presence of aerial oxygen under sacrificial agent-free conditions (Fig. 4c). Interestingly, the H2O2 production was observed to have an enhanced rate in the presence of 0.2 N HCl, reaching 5.00 mmol·g−1·h−1, confirming the imperative role of additional proton donors to make the photoproduction of H2O2 more facile (Fig. 4c).
In this context, efficient exciton separation can be further promoted by hole scavenging, which prompted us to investigate the use of different sacrificial agents on the synthesis of H2O2 under a 50 W blue LED. At the same time, their oxidation can lead to value-added products that can find suitable applications in green energy production and biomass valorization. For this purpose, the commonly used sacrificial agents tetrahydrofurfuryl alcohol and ascorbic acid were tested as electron donors. As expected, the oxygen reductions were furnished with strikingly high rates of 5.17 and 8.23 mmol·g−1·h−1, respectively, recording the highest value achieved for H2O2 generation with TPA-BINOL-4 as the metal-free photocatalyst under these conditions (Fig. 4d). A satisfactory rate was obtained even with its regioisomer homolog TPA-BINOL-6, furnishing H2O2 at 4.72 and 8.01 mmol·g−1·h−1, respectively. This considerable increase in H2O2 production rate for the sacrificial agents could originate from the compatible oxidation potential for each of them and the superior electron donor property of the TPA moiety, which facilitates charge transfer at the liquid/vapor interface. H2O2 production was found to be further promoted in the presence of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 isopropanol (IPA)/water, with a production rate of 13.02 mmol·g−1·h−1 when using TPA-BINOL-4 as the photocatalyst (Fig. 4d). Furthermore, addressing naturally abundant water bodies, the H2O2 generation performance was elevated, with rates of formation of 6.62 and 6.01 mmol·g−1·h−1 with TPA-BINOL-4 for tap water and seawater, respectively, after adding isopropanol as the optimal sacrificial agent (10
10 isopropanol (IPA)/water, with a production rate of 13.02 mmol·g−1·h−1 when using TPA-BINOL-4 as the photocatalyst (Fig. 4d). Furthermore, addressing naturally abundant water bodies, the H2O2 generation performance was elevated, with rates of formation of 6.62 and 6.01 mmol·g−1·h−1 with TPA-BINOL-4 for tap water and seawater, respectively, after adding isopropanol as the optimal sacrificial agent (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v) (Fig. 4a). To get the best results, the effects of varying the sacrificial agent (IPA) concentration were studied for both the photocatalysts. With an increasing ratio of IPA, the H2O2 photosynthesis rate was enhanced significantly, and an equal ratio of water and IPA dramatically raised the H2O2 production to 27.5 mmol·g−1·h−1 for TPA-BINOL-4, which implied a considerably high value compared to those reported so far with organic semiconductor photocatalysts (Fig. 4e).
1 v/v) (Fig. 4a). To get the best results, the effects of varying the sacrificial agent (IPA) concentration were studied for both the photocatalysts. With an increasing ratio of IPA, the H2O2 photosynthesis rate was enhanced significantly, and an equal ratio of water and IPA dramatically raised the H2O2 production to 27.5 mmol·g−1·h−1 for TPA-BINOL-4, which implied a considerably high value compared to those reported so far with organic semiconductor photocatalysts (Fig. 4e).
Several energy alternatives to fossil fuels are emerging through sustainable production strategies, and biomass valorization of lignin and other hemicellulose components is a leading one. The H2O2 formation was not limited by the commonly used sacrificial agents, rather its evolution played an imperative role when organic catalytic transformation, i.e., biomass valorization, was chosen to test the generation of H2O2. As a proof of concept, we inspected 5-hydroxymethyl furfural (HMF) as a sacrificial agent while performing H2O2 production. The H2O2 production rate was observed to be 5.84 mmol·g−1·h−1 for TPA-BINOL-4 with 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 HMF in water under a 50 W blue LED, which indicates its possibility to act as a suitable sacrificial agent. The test results suggested a significant rate of formation of the selective oxidation product (Fig. 4f). This further evidenced the concept of killing two birds with one stone, i.e., biofuel production with simultaneous photosynthesis of H2O2 which itself is recognized as a value-added liquid fuel. The oxidation of HMF furnished 2,5-furandicarboxylic acid (m/z = 156) and maleic acid (m/z = 116), as reflected from the LC-MS mass spectra (Fig. S15, ESI†). A similar observation was found for tetrahydrofuryl alcohol when it was utilized as the sacrificial agent. The LC-MS profile of the reaction mixture after blue-light irradiation in the presence of TPA-BINOL-4 indicated the strong presence of tetrahydrofuryl carboxylic acid (m/z 116) and 2-formyl tetrahydrofuran (m/z 100) as the oxidation products (Fig. S16, ESI†), accompanied by a considerable rate for H2O2 production of up to 5.17 mmol·g−1·h−1 (Fig. 4d). Meanwhile, TPA-BINOL-4 was shown to produce α-hydroxyfuryl radicals (˙CH(OH)furan) by EPR in the reaction mixture. The analysis yielded six distinct peaks, with αN and αH measuring 15.4 G and 22.6 G, respectively. These values are indicative of the hyperfine splitting of nitrogen and hydrogen in nitrogen oxides.54 The introduction of the triphenylamine core improved the system's capacity to harvest light and could produce more carbon-radicals in the catalytic system, as evidenced by the greater intensity of DMPO-carbon-radical adduct on TPA-BINOL-4 (Fig. S17, ESI†).
1 HMF in water under a 50 W blue LED, which indicates its possibility to act as a suitable sacrificial agent. The test results suggested a significant rate of formation of the selective oxidation product (Fig. 4f). This further evidenced the concept of killing two birds with one stone, i.e., biofuel production with simultaneous photosynthesis of H2O2 which itself is recognized as a value-added liquid fuel. The oxidation of HMF furnished 2,5-furandicarboxylic acid (m/z = 156) and maleic acid (m/z = 116), as reflected from the LC-MS mass spectra (Fig. S15, ESI†). A similar observation was found for tetrahydrofuryl alcohol when it was utilized as the sacrificial agent. The LC-MS profile of the reaction mixture after blue-light irradiation in the presence of TPA-BINOL-4 indicated the strong presence of tetrahydrofuryl carboxylic acid (m/z 116) and 2-formyl tetrahydrofuran (m/z 100) as the oxidation products (Fig. S16, ESI†), accompanied by a considerable rate for H2O2 production of up to 5.17 mmol·g−1·h−1 (Fig. 4d). Meanwhile, TPA-BINOL-4 was shown to produce α-hydroxyfuryl radicals (˙CH(OH)furan) by EPR in the reaction mixture. The analysis yielded six distinct peaks, with αN and αH measuring 15.4 G and 22.6 G, respectively. These values are indicative of the hyperfine splitting of nitrogen and hydrogen in nitrogen oxides.54 The introduction of the triphenylamine core improved the system's capacity to harvest light and could produce more carbon-radicals in the catalytic system, as evidenced by the greater intensity of DMPO-carbon-radical adduct on TPA-BINOL-4 (Fig. S17, ESI†).
2.6 Mechanism of H2O2 photosynthesis
To elucidate the mechanistic path for the overall photosynthesis of H2O2, four different possibilities could be considered.(i) Direct 2e− oxygen reduction (0.68 V vs. NHE).
(ii) Indirect two-step 1e− reduction of O2via superoxide (0.94 V vs. NHE).
(iii) 4e− water oxidation into oxygen followed by O2 reduction (1.23 V vs. NHE).10
(iv) Direct 2e− water oxidation (1.78 V vs. NHE).
In this aspect, the mechanism for photocatalytic H2O2 generation was monitored using scavenger control tests where AgNO3 and p-benzoquinone (p-BQ) were chosen as the photoelectron and superoxide scavengers, respectively. In the case of TPA-BINOL-4, under dark or with AgNO3 resulted in no trace of H2O2 even after 1 h of irradiation, which suggests the possibility of light and photoelectrons playing pivotal roles in achieving the sustainable synthesis of H2O2 (Fig. 4c). The H2O2 synthesis with respect to time showed a production rate of 13.02 mmol·g−1·h−1 in the presence of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 isopropanol (IPA)/water, following nearly zero-order kinetics when TPA-BINOL-4 was taken as the photocatalyst (Fig. 4g). Meanwhile, in the presence of p-benzoquinone (p-BQ), the H2O2 generation rate displayed a small decrease, with a value of 10.43 mmol·g−1·h−1 (with TPA-BINOL-4 in 1
10 isopropanol (IPA)/water, following nearly zero-order kinetics when TPA-BINOL-4 was taken as the photocatalyst (Fig. 4g). Meanwhile, in the presence of p-benzoquinone (p-BQ), the H2O2 generation rate displayed a small decrease, with a value of 10.43 mmol·g−1·h−1 (with TPA-BINOL-4 in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 IPA/water), indicating a greater possibility for a direct 2e− oxygen reduction reaction (ORR), while the indirect path (O2 → O2˙− → H2O2) cannot be ruled out completely (Fig. 4g). However, the addition of sodium azide as the singlet oxygen scavenger had no effect on the catalytic efficiency of TPA-BINOL-4 (Fig. 4g).
10 IPA/water), indicating a greater possibility for a direct 2e− oxygen reduction reaction (ORR), while the indirect path (O2 → O2˙− → H2O2) cannot be ruled out completely (Fig. 4g). However, the addition of sodium azide as the singlet oxygen scavenger had no effect on the catalytic efficiency of TPA-BINOL-4 (Fig. 4g).
To get detailed mechanistic insight into H2O2 formation using TPA-BINOL-4, the electron transfer path needed to be considered. Rotating disc electrode (RDE) measurements at different angular velocities gave the Koutecky–Levich plot, which showed that the number of electrons for ORR with the polymers are 1.67, i.e., nearly 2, indicating the streamlined possibility of H2O2 formation other than 4e− ORR forming H2O as the product (Fig. S18, eqn (S11) and (S12), ESI†).55 The photocatalytic H2O2 production performances of TPA-BINOL-4 under various atmospheric circumstances (Ar or O2) are shown in Fig. 4g, displaying the time-dependent H2O2 production curves. However, no traces of H2O2 were found when Ar was continually bubbled into the water to extract O2. This observation suggests that the 2e− water oxidation reaction (WOR) pathway makes a minuscule contribution to the total quantity of H2O2 in our investigation. These results were further authenticated by the band positions of TPA-BINOL-4 guiding its performance, i.e., the HOMO energy position lies well above the oxidation potential of H2O/H2O2 at 1.78 V w.r.t. NHE, underpinning the minimal possibility of 2e− water oxidation (Fig. 1). Nevertheless, in Ar-presaturated water, the H2O2 level rose to 0.0056 mmol, indicating that the O2 generated by 4e− WOR under radiation might be utilized as an additional O2 source for 2e− ORR to make H2O2 (Fig. 4g).56 Furthermore, the amount of H2O2 generation was minimally affected upon the addition of tert-butyl alcohol, i.e. a scavenger of ˙OH. This finding suggests that the 2e− ORR (E = 0.68 V w.r.t. NHE) as the dominating H2O2 production pathway (Fig. 1) and the O2˙− to ˙OH or ˙OH + ˙OH = H2O2 pathway are not operative in our experimental conditions.
When H2O2 production studies were conducted under constant Ar purging, no trace of H2O2 was detected, indicating that the source of H2O2 synthesis entirely relied upon oxygen reduction and not the 2e− water oxidation for TPA-BINOL-4 (Fig. 4g). This experimental observation was in accordance with its valence band, which was aligned well above the potential required for 2e− WOR, making the process energetically uphill (EVB = 1.31 V w.r.t. NHE). In contrast, TPA-BINOL-6, although with lower H2O2 generation performance, could furnish H2O2 production even under constant Ar atmosphere with a production rate of 1.02 mmol·g−1·h−1, which corroborates well with its valence band alignment towards 2e− WOR (EVB = 1.83 V w.r.t. NHE) (Fig. 1 and 4h). The zero-order kinetics was reflected for the water oxidation path in the kinetic data recorded for up to three hours. This, in other words, depicts the promise of TPA-BINOL-based polymers in artificial photosynthesis towards the overall generation of H2O2via ORR and 2e− WOR even without the addition of a sacrificial agent.51 Since TPA-BINOL-6 could furnish H2O2 from direct 2e− water oxidation, its real-time operational sustainability was further tested with seawater, where results showed a compromised production rate of 253 μmol·g−1·h−1. This was attributed to the presence of interfering ions and sediments prohibiting the optical absorption and transfer of photoexcitons from the catalyst to water (Fig. 4h).
Monitoring the intermediates towards the photosynthesis of hydrogen peroxide is crucial to get a clear picture of the oxygen binding modes and possible reactive oxygen species involved. The in situ IR spectrum of TPA-BINOL-4 in the presence of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water/IPA and 50 W blue LED irradiation presented additional peaks at 1365 and 1310–1332 cm−1, along with existing characteristic peaks for the catalyst, which could be ascribed to surface-adhered H2O2 and hydroperoxide, respectively (Fig. 4i). Interestingly, a signal at 915 cm−1 could be assigned to the chemisorbed oxygen over the surface of the photocatalyst after oxygen exposure (Fig. S19, ESI†). Moreover, the O–H vibration from H2O2 possibly displayed a small rise around 2725 cm−1 due to intermolecular H-bonds (Fig. S19, ESI†). A similar trend was depicted in the in situ EPR spectra with DMPO as the radical trapper which reflected the effective generation of hydroperoxide signals upon continuous irradiation for up to 10 min. Although 2e− ORR is presented as the dominant path, the mechanism for H2O2 production can also proceed along the generation of superoxide, which was confirmed upon 10 min of irradiation in the presence of 10
1 water/IPA and 50 W blue LED irradiation presented additional peaks at 1365 and 1310–1332 cm−1, along with existing characteristic peaks for the catalyst, which could be ascribed to surface-adhered H2O2 and hydroperoxide, respectively (Fig. 4i). Interestingly, a signal at 915 cm−1 could be assigned to the chemisorbed oxygen over the surface of the photocatalyst after oxygen exposure (Fig. S19, ESI†). Moreover, the O–H vibration from H2O2 possibly displayed a small rise around 2725 cm−1 due to intermolecular H-bonds (Fig. S19, ESI†). A similar trend was depicted in the in situ EPR spectra with DMPO as the radical trapper which reflected the effective generation of hydroperoxide signals upon continuous irradiation for up to 10 min. Although 2e− ORR is presented as the dominant path, the mechanism for H2O2 production can also proceed along the generation of superoxide, which was confirmed upon 10 min of irradiation in the presence of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 IPA/water under a 50 W blue LED. The characteristic DMPO–O2˙− signal was observed initially and further split into additional peaks confirming the generation of DMPO–*OOH upon increasing time (Fig. S20, ESI†).
1 IPA/water under a 50 W blue LED. The characteristic DMPO–O2˙− signal was observed initially and further split into additional peaks confirming the generation of DMPO–*OOH upon increasing time (Fig. S20, ESI†).
2.7 Effect of pH and recycling
To understand the usefulness of proton donors in promoting H2O2 generation, H2O2 photosynthesis was effectively monitored while varying the pH of the water medium. Under acidic conditions (pH = 1), H2O2 production reached its maximum of 5.2 mmol·g−1·h−1; however, at pH = 12, no traces of H2O2 were detected, indicating sluggish proton transfer, i.e., lack of availability of proton donors under basic conditions (Fig. S21, ESI†).57 These results also supported the reduction of oxygen to peroxide, which plays a pivotal role in the photoproduction of H2O2. Besides the photoproduction of H2O2, its stability under irradiation in presence of our as-designed photocatalyst is a concern owing to the requirement for minimization of the photodecomposition reaction (low kd), which is desired for a practically reliable catalyst. The first order kinetic rate equation afforded a kd of 0.0015 min−1 with 8.8% photodecomposition for TPA-BINOL-4 and 11.2% for TPA-BINOL-6, respectively, within one hour of irradiation under argon atmosphere (Fig. 5a). The remarkable photostability of our as-synthesized TPA-BINOL-4 polymer was monitored for employment in long-cycling runs. For this, the polymer TPA-BINOL-4 was recovered via centrifugation and washed with methanol, chloroform, and acetone separately to test the consecutive cycles (Fig. 5b). In this fashion, it could be retrieved up to ten successive cycles without any considerable loss in photocatalytic activity (Fig. 5b). Notably, the polymer maintained its morphological and structural characteristics after recycling, without any significant loss (Fig. S22, ESI†).2.8 Photocurrent and electrochemical impedance spectra (EIS) comparison
To scrutinize the factors behind the superior photocatalytic activity of TPA-BINOL-4, transient photocurrent response and EIS data were necessitated as supporting evidence.58 As shown in Fig. 5c, TPA-BINOL-4 displayed a three times higher photocurrent response within 50 s intervals when compared with TPA-BINOL-6. This data was in line with the results obtained from the EPR spectra (Fig. S12, ESI†), i.e., the higher photoexciton generation property of TPA-BINOL-4. The origin of the photogenerated charge carrier migration was investigated by monitoring the EIS patterns under both dark and irradiated conditions. The charge transfer resistance observed under dark conditions dropped drastically upon exposure to AM 1.5 G light, showing good agreement with the transient photocurrent and EPR responses (Fig. S23, ESI†). The feasibility of plausible exciton separation was further tested with the Nyquist plots for both. The semi-circular arc in the case of TPA-BINOL-4 indicated its lower charge transfer resistance (Rct), i.e., lower resistance towards the migration of photogenerated charge carriers (Fig. 5d). These experimental observations agreed well with the picture from the theoretical molecular orbital distribution, i.e., charge separation (Fig. 3f and S11, ESI†). Moreover, the decay dynamics were examined with time-resolved photoluminescence (TRPL) spectra and, upon biexponential fitting, revealed the prolonged excited state lifetime for TPA-BINOL-4 (τ = 0.39 ns) as compared to that of TPA-BINOL-6 (τ = 0.35 ns), indicating less inclination towards radiative decay and charge carrier recombination for the former (Fig. S24, ESI†).2.9 Transient absorption spectra analysis
To further grasp the exciton dissociation dynamics, the femtosecond transient absorption spectra (TAS) of the polymers TPA-BINOL-4 and TPA-BINOL-6 were collected under oxygen and nitrogen atmospheres separately by pumping at 350 nm (see Experimental section, eqn (S13) and (S14), ESI†). The results depicted that both photocatalysts show broad positive absorption in the range of 450–535 nm, categorized as excited state absorption (ESA), due to the electronic transition from S1 → Sn (Fig. 5e and f).59,60 Under an oxygen atmosphere, TPA-BINOL-4 in the 2D mapping of TAS could be ascertained to have broad positive ESA up to the 535 nm range (Fig. S25, ESI†). It is interesting to note that both TPA-BINOL-6 and TPA-BINOL-4 exhibit faster TA kinetics in the oxygen atmosphere (τavg. O2 < τavg. N2), which may stem from electron transfer from the catalyst to oxygen, i.e., the electron transfer adds an additional decay channel compared to other relaxation pathways (i.e., fluorescence or internal conversion) under the O2 atmosphere (Fig. 5g and h).60,61 Notably, TPA-BINOL-4 exhibits faster decay (τavg. = 0.54 ps) and hence a lower charge carrier recombination compared to TPA-BINOL-6 (τavg. = 22.6 ps), indicating a faster electron transfer from TPA-BINOL-4 to O2 that may result in its superior photocatalytic generation of H2O2via ORR (Fig. S26, ESI†).62–64 Alternatively, under the nitrogen atmosphere, exceptionally longer decay times for both TPA-BINOL-4 (40.7 ± 4.4 ps) and TPA-BINOL-6 (199.6 ± 18 ps) was observed, which indicated reduced possibilities for exciton dissociation (Fig. S26 and S27, ESI†).2.10 Theoretical insight
For an insightful discussion on the evolution of TPA-BINOL-4 as the optimal photocatalyst for visible-light-driven H2O2 production, density functional theory (DFT) based calculations were employed. The optimized structure of TPA-BINOL-4 is depicted in Fig. S28,† where the two naphthol rings of BINOL moieties are positioned perpendicular to each other in each arm, further supporting the preferred orientation of BINOL moieties. To elucidate the photocatalytic reaction pathway, identifying the electronic states involved in the photoexcited transition of electrons and subsequent charge separation process plays a pivotal role. The time-dependent density functional theory (TDDFT) calculations unveiled that the electronic transitions between the HOMO → LUMO (S0 → S1) and HOMO → LUMO+1 (S0 → S2) had oscillator strengths (f) of 1.3787 and 1.3318, respectively. The electrons on the HOMO were mainly located on the three phenyl rings and the nitrogen atom of TPA in a symmetrical arrangement, as depicted in Fig. S29.† Conversely, the electronic distribution of the LUMO was concentrated on one of the arms of the TPA-BINOL-4 molecule and mainly focused on the (i.e., the major contributions are from) naphthol ring of the BINOL moiety adjacent to the TPA core (Fig. S29, ESI†). The electrons on the LUMO+1 were spread on the rest of the two arms of the TPA-BINOL-4 molecule in the same way. The charge distributions of the LUMO and LUMO+1 exhibited similar patterns on the first naphthol ring of the BINOL moiety (Fig. S29, ESI†). These HOMO, LUMO and LUMO+1 charge distributions suggested that the electrons (e−) are mainly situated on the first naphthol ring of the BINOL moiety, while the holes (h+) are concentrated on the phenyl rings and the nitrogen atom of the triphenylamine core. The different positions of h+ and e− ensured spatial charge separation after the photo excitation, which suppressed the rapid recombination of the opposite charges, thereby enhancing the catalytic activity.To screen the most favourable position for molecular oxygen adsorption, we considered a simplified model of TPA-BINOL-4. We substituted the two arms, each containing a phenyl and BINOL moiety, with hydrogen atoms, as illustrated in Fig. S29.† Analogous transition types to those seen in the TPA-BINOL-4 system were observed by TDDFT calculations on this simplified model system. The primary electronic transition, with an oscillator strength of 0.7963, was HOMO → LUMO+1 (S0 → S1). Much like the TPA-BINOL-4 system, the LUMO+1 charge density, which functions as an electron source following photoexcitation, is primarily localized on the naphthol ring of the BINOL moiety adjacent to TPA in the model unit (Fig. S30, ESI†), with just a small percentage of the charge density located on the naphthol ring away from the TPA core. Since the charge density along LUMO is positioned on the naphthol ring close to TPA in both the TPA-BINOL-4 and the truncated systems, we have methodically screened every available carbon atom for possible oxygen adsorption on the truncated system (Fig. S31a, ESI†). It was discovered that C2 is the most favourable site for oxygen adsorption (Fig. S31b, ESI†), offering the maximum adsorption energy among all other possible positions (Fig. S32, ESI†).
Upon molecular oxygen adsorption in the truncated TPA-BINOL-4 system, the bond distance increased to 1.46 Å. The NBO charge analysis shows charges of −0.79 on O1 and −0.41 on O2, indicating clear electron transfer from the catalytic surface to the adsorbed molecular oxygen (Fig. S30 and S31b, ESI†). This C2 position was also considered in the TPA-BINOL system, as both the TPA-BINOL-4 and truncated TPA-BINOL-4 systems show almost the same types of MO distributions (Fig. S30, ESI†). Further DFT calculations were conducted considering this O2 adsorption site to explore the catalytic pathway.
The photoexcited hole (h+) can oxidize the alcohol and produce the H+ and ketone, while the photo-excited electron (e−) can facilitate the two-electron reduction of O2, yielding H2O2.
| (CH3)2CHOH + 2h+ → CH3COCH3 + 2H+ | (1) |
| O2 + 2H+ + 2e− → H2O2 | (2) |
Proceeding along the reaction pathway, as shown in Fig. 6a, the next step involves the migration of the H+ (H2) from TPA-BINOL-4 to the O2 atom of the adsorbed O2 molecule (Ts-1) (Fig. S34b, ESI†). The transition barrier for this migration is 0.15 eV, which makes the intermediate *OOH (Int-3) stabilized by 1.76 eV, as depicted in Fig. 6b and S35a.† Interestingly, the hydroperoxide intermediate generation was confirmed with the generation of Int-3, which further proved the observation from the in situ EPR studies with DMPO as scavenger. Following the reaction path, the next hydrogen, H1, gets transferred from TPA-BINOL-4 to the O1 of the adsorbed O2 molecule in Ts-2 (Fig. 6b and S35b, ESI†) and makes the H2O2 molecule stabilized with 3.01 eV of energy liberation (Fig. 6a and b). The second transition state involves a lower energy barrier of 0.88 eV, as illustrated in the energy profile diagram (Fig. 6b and S34b, ESI†). The overall reaction is thermodynamically favorable, with a free energy of −2.86 eV (Fig. 6b). The solvent contains both water and isopropyl alcohol, so hydrogen transfer is possible from both the H2O and (CH3)2CHOH. Due to the higher acidic character of isopropyl alcohol, the proton transfer is more favourable from it than from water. We examined the proton transfer barrier from the different proton donors, and we found that the isopropyl alcohol is energetically more favorable than water (Fig. 6c and S36, ESI†).
We also examined the reaction pathway using H2O as the proton source (Fig. S36, ESI†). It was found that the thermodynamic barrier of proton transfer on TPA-BINOL-4 from the H2O molecule is 3.64 eV, significantly higher than the 1.15 eV barrier for IPA, as shown in Fig. 6c. This high energy barrier for proton transfer from water makes it a less efficient solvent compared to isopropyl alcohol, therefore facilitating the better efficiency of H2O2 production (Fig. 6c). The energy profile for sacrificial agent-free H2O2 production revealed the proton transfer from water leading to water getting oxidized into oxygen, which itself is a kinetically sluggish process, although overall stabilization is achieved with a free energy liberation of 0.38 V (Fig. S36, ESI†).
3 Conclusion
To summarize, a novel synthetic strategy was adopted to furnish TPA-BINOL-based porous organic polymers via in situ formation of the two different regioisomeric BINOLs during the homopolymerization of the 2-naphthol-based monomers. To our delight, these polymers could efficiently generate photo excitons to convert oxygen into several ROS (superoxide, H2O2), leading to unprecedented photosynthesis of H2O2 (a record activity of 6.47 mmol·g−1·h−1 in pure water) under AM 1.5 G solar radiation with an SCC efficiency of 1.6% and an apparent quantum yield of 5.87% without any additives. The H2O2 production for TPA-BINOL-4 reached 4.33 mmol·g−1·h−1 under a 50 W blue LED without any additives and could be elevated to 27.5 mmol·g−1·h−1 in the presence of isopropyl alcohol as the sacrificial agent. Surprisingly, under argon atmosphere, the H2O2 generation rate was 1.02 mmol·g−1·h−1 with TPA-BINOL-6 without employing any sacrificial proton donor, surpassing most of the POP materials reported to date. The H2O2 generation was further furnished from the separate oxidation of hydroxymethyl furfural and tetrahydrofurfuryl alcohol. Theoretical insights helped identify the reaction pathway and the best site for oxygen adsorption within the prepared photocatalyst TPA-BINOL-4. Thus, engineering the connectivity of BINOL-based porous polymers resulted in improved charge carrier migration for sustainable H2O2 production via the oxygen reduction reaction.Data availability
The data supporting this article have been included as part of the ESI.†Author contributions
F. B. performed the synthesis and characterizing of the polymers, conducted photocatalytic H2O2 production studies, and wrote the manuscript. S. S. performed the DFT calculations and wrote the manuscript. S. S. and S. R. performed some of the synthesis and electrochemical measurements. S. M. performed the TAS measurements for both polymers. P. K. C. supervised the measurements and contributed to review-editing. S. K. P. supervised the DFT studies and contributed to review-editing. S. K. S. designed the project, supervised all the experiments and revised the manuscript. The manuscript was written with permission from all authors.Conflicts of interest
There are no conflicts to declare.Acknowledgements
F. B. acknowledges the MoE, Govt. of India, for the PMRF fellowship. S. S. is thankful to the Indian Institute of Technology Kharagpur for funding. We are thankful to the Central Research Facility (CRF), IIT Kharagpur, for the instrumental facilities. S. S. thanks the University Grants Commission (UGC), New Delhi, for fellowship support. S. K. P. thanks SERB and the J. C. Bose National Fellowship, Government of India, for funding. S. S. and S. K. P. acknowledge NSM-Param Yukti for providing computational facilities. P. K. C. thanks the Department of Science and Technology, DST-FIST (SR/FST/CS-II027/2014), New Delhi, India, for providing funds for the ultrafast TA facility. S. M. thanks the MHRD (Govt. of India) for the Prime Minister's Research Fellowship.References
- J. M. Campos-Martin, G. Blanco-Brieva and J. L. G. Fierro, Angew. Chem., Int. Ed., 2006, 45, 6962–6984 CrossRef CAS
.
- N. Wang, S. Ma, P. Zuo, J. Duan and B. Hou, Adv. Sci., 2021, 8, 2100076 CrossRef CAS
.
- Y. Zhang, C. Pan, G. Bian, J. Xu, Y. Dong, Y. Zhang, Y. Lou, W. Liu and Y. Zhu, Nat. Energy, 2023, 8, 361–371 CrossRef CAS
.
- K. Jiang, J. Zhao and H. Wang, Adv. Funct. Mater., 2020, 30, 2003321 CrossRef CAS
.
- C. Krishnaraj, H. Sekhar Jena, L. Bourda, A. Laemont, P. Pachfule, J. Roeser, C. V. Chandran, S. Borgmans, S. M. J. Rogge, K. Leus, C. V. Stevens, J. A. Martens, V. Van Speybroeck, E. Breynaert, A. Thomas and P. Van Der Voort, J. Am. Chem. Soc., 2020, 142, 20107–20116 CrossRef CAS
.
- H. Hou, X. Zeng and X. Zhang, Angew. Chem., Int. Ed., 2020, 59, 17356–17376 CrossRef CAS PubMed
.
- J. García-Serna, T. Moreno, P. Biasi, M. J. Cocero, J.-P. Mikkola and T. O. Salmi, Green Chem., 2014, 16, 2320–2343 RSC
.
- S. Fukuzumi, Y.-M. Lee and W. Nam, Chem.–Eur. J., 2018, 24, 5016–5031 CrossRef CAS
.
- Y. Kofuji, Y. Isobe, Y. Shiraishi, H. Sakamoto, S. Tanaka, S. Ichikawa and T. Hirai, J. Am. Chem. Soc., 2016, 138, 10019–10025 CrossRef CAS
.
- F. Banerjee, P. D. Gupta, S. Roy and S. K. Samanta, ACS Appl. Mater. Interfaces, 2024, 16, 58689–58702 CrossRef CAS PubMed
.
- Y. Yang, B. Cheng, J. Yu, L. Wang and W. Ho, Nano Res., 2021, 1–9 Search PubMed
.
- S. Zhao, X. Zhao, H. Zhang, J. Li and Y. Zhu, Nano Energy, 2017, 35, 405–414 CrossRef CAS
.
- Y. Shiraishi, T. Takii, T. Hagi, S. Mori, Y. Kofuji, Y. Kitagawa, S. Tanaka, S. Ichikawa and T. Hirai, Nat. Mater., 2019, 18, 985–993 CrossRef CAS
.
- V. Nian, Appl. Energy, 2016, 179, 1069–1080 CrossRef
.
- A. Modak, A. Ghosh, A. R. Mankar, A. Pandey, M. Selvaraj, K. K. Pant, B. Chowdhury and A. Bhaumik, ACS Sustainable Chem. Eng., 2021, 9, 12431–12460 CrossRef CAS
.
- M. Cai, Y. Zhang, Y. Zhao, Q. Liu, Y. Li and G. Li, J. Mater. Chem. A, 2020, 8, 20386–20392 RSC
.
- H.-K. Lai, Y.-Z. Chou, M.-H. Lee and K.-Y. A. Lin, Chem. Eng. J., 2018, 332, 717–726 CrossRef CAS
.
- Y. Guo and J. Chen, ChemPlusChem, 2015, 80, 1760–1768 CrossRef CAS
.
- P. Das, J. Roeser and A. Thomas, Angew. Chem., Int. Ed., 2023, 62, e202304349 CrossRef CAS
.
- I. Krivtsov, A. Vazirani, D. Mitoraj and R. Beranek, ChemCatChem, 2023, 15, e202201215 CrossRef CAS
.
- S. Wei, S. Chang, H. Li, Z. Fang, L. Zhu and Y. Xu, Green Chem., 2024, 26, 6382–6403 RSC
.
- Y. Shiraishi, M. Matsumoto, S. Ichikawa, S. Tanaka and T. Hirai, J. Am. Chem. Soc., 2021, 143, 12590–12599 CrossRef CAS PubMed
.
- X. Chen, W. Zhang, L. Zhang, L. Feng, C. Zhang, J. Jiang, T. Yan and H. Wang, J. Mater. Chem. A, 2020, 8, 18816–18825 RSC
.
- Y. Guo, X. Yang, R. Sun, X. Hu, C. Shu, X. Yang, H. Gao, X. Wang and B. Tan, Small, 2024, 20, 2403743 CrossRef CAS
.
- Q. Ma, X. Zhang, L. Ji and S. Liao, Polym. Chem., 2019, 10, 6662–6668 RSC
.
- D. S. Kundu, J. Schmidt, C. Bleschke, A. Thomas and S. Blechert, Angew. Chem., Int. Ed., 2012, 51, 5456–5459 CrossRef CAS
.
- J. Wei, X. Zhang, Y. Zhao and R. Li, Macromol. Chem. Phys., 2013, 214, 2232–2238 CrossRef CAS
.
- S. Sau, F. Banerjee and S. K. Samanta, ACS Appl. Nano Mater., 2023, 6, 11679–11688 CrossRef CAS
.
- F. Banerjee, S. Bera, T. Nath and S. K. Samanta, Nanoscale, 2024, 16, 11999–12006 RSC
.
- Y. Zhang, Q. Wang, R. Li, Z. Lou and Y. Li, Crystals, 2020, 10, 159 CrossRef CAS
.
- S. Sau, S. Karmakar, F. Banerjee and S. K. Samanta, Nanoscale, 2024, 16, 21096–21105 RSC
.
- S. Bera, S. Sau, N. Kumar and S. K. Samanta, ACS Appl. Nano Mater., 2024, 7, 529–538 CrossRef CAS
.
- S. Bera, F. Banerjee and S. K. Samanta, ACS Appl. Nano Mater., 2023, 6, 15002–15011 CrossRef CAS
.
- Y. Zhang, M. Zhang, Z. Han, S. Huang, D. Yuan and W. Su, ACS Catal., 2021, 11, 1008–1013 CrossRef CAS
.
- R. Singuru, K. Dhanalaxmi, S. C. Shit, B. M. Reddy and J. Mondal, ChemCatChem, 2017, 9, 2550–2564 CrossRef CAS
.
- J. Chen, H. Li, M. Zhong and Q. Yang, Green Chem., 2016, 18, 6493–6500 RSC
.
- S. Rat, A. Chavez-Sanchez, M. Jerigová, D. Cruz and M. Antonietti, ACS Appl. Polym. Mater., 2021, 3, 2588–2597 CrossRef CAS
.
- J. Jeromenok and J. Weber, Langmuir, 2013, 29, 12982–12989 CrossRef CAS PubMed
.
- J. Rouquerol, D. Avnir, C. W. Fairbridge, D. H. Everett, J. M. Haynes, N. Pernicone, J. D. F. Ramsay, K. S. W. Sing and K. K. Unger, Pure Appl. Chem., 1994, 66, 1739–1758 CrossRef CAS
.
- H. Xu, X. Li, H. Hao, X. Dong, W. Sheng and X. Lang, Appl. Catal., B, 2020, 285, 119796 CrossRef
.
- S. Manna, K. Maity, F. Banerjee, S. Sau and S. K. Samanta, Chem. Commun., 2025, 61, 9670–9673 RSC
.
- Y.-X. Tan, Y.-P. He, D. Yuan and J. Zhang, Appl. Catal., B, 2018, 221, 664–669 CrossRef CAS
.
- Z. Chen, D. Yao, C. Chu and S. Mao, Chem. Eng. J., 2023, 451, 138489 CrossRef CAS
.
- Y. Kondo, Y. Kuwahara, K. Mori and H. Yamashita, Chem, 2022, 8, 2924–2938 CAS
.
- J.-Y. Yue, Y.-T. Wang, X.-L. Ding, Y.-F. Fan, L.-P. Song, P. Yang, Y. Ma and B. Tang, Mater. Chem. Front., 2022, 6, 3748–3754 RSC
.
- H. H. Willard and P. Young, J. Am. Chem. Soc., 1933, 55, 3260–3269 CrossRef CAS
.
- C. Chu, D. Yao, Z. Chen, X. Liu, Q. Huang, Q. Li and S. Mao, Small, 2023, 19, 2303796 CrossRef CAS
.
- Q. Wu, J. Cao, X. Wang, Y. Liu, Y. Zhao, H. Wang, Y. Liu, H. Huang, F. Liao, M. Shao and Z. Kang, Nat. Commun., 2021, 12, 483 CrossRef CAS
.
- Y.-X. Ye, J. Pan, Y. Shen, M. Shen, H. Yan, J. He, X. Yang, F. Zhu, J. Xu, J. He and G. Ouyang, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2115666118 CrossRef CAS
.
- B. C. Moon, B. Bayarkhuu, K. A. I. Zhang, D. K. Lee and J. Byun, Energy Environ. Sci., 2022, 15, 5082–5092 RSC
.
- J. Liu, Y. Zou, B. Jin, K. Zhang and J. H. Park, ACS Energy Lett., 2019, 4, 3018–3027 CrossRef CAS
.
- Y. Hong, Y. Cho, E. M. Go, P. Sharma, H. Cho, B. Lee, S. M. Lee, S. O. Park, M. Ko, S. K. Kwak, C. Yang and J.-W. Jang, Chem. Eng. J., 2021, 418, 129346 CrossRef CAS
.
- Q. Tian, X.-K. Zeng, C. Zhao, L.-Y. Jing, X.-W. Zhang and J. Liu, Adv. Funct. Mater., 2023, 33, 2213173 CrossRef CAS
.
- X. Li, G. Zhang, N. Li, Q. Xu, H. Li, J. Lu and D. Chen, Adv. Funct. Mater., 2024, 34, 2316773 CrossRef CAS
.
- Y.-Y. Yang, H. Guo, D.-W. Huang, L. Li, H.-Y. Liu, L. Sui, Q. Wu, J.-J. Zhu, L. Zhang and C.-G. Niu, Chem. Eng. J., 2024, 479, 147863 CrossRef CAS
.
- T. Yang, D. Zhang, A. Kong, Y. Zou, L. Yuan, C. Liu, S. Luo, G. Wei and C. Yu, Angew. Chem., Int. Ed., 2024, 63, e202404077 CrossRef CAS
.
- X. Chang, J. Yang, D. Han, B. Zhang, X. Xiang and J. He, Catalysts, 2018, 8, 147 CrossRef
.
- K. Maity, S. Sau, F. Banerjee and S. K. Samanta, ACS Appl. Mater. Interfaces, 2024, 16, 50834–50845 CrossRef CAS
.
- J. Cheng, W. Wang, J. Zhang, S. Wan, B. Cheng, J. Yu and S. Cao, Angew. Chem., Int. Ed., 2024, 63, e202406310 CrossRef CAS
.
- H. Wang, S. Jiang, W. Shao, X. Zhang, S. Chen, X. Sun, Q. Zhang, Y. Luo and Y. Xie, J. Am. Chem. Soc., 2018, 140, 3474–3480 CrossRef CAS PubMed
.
- A. Yamakata, T.-a. Ishibashi and H. Onishi, J. Phys. Chem. B, 2001, 105, 7258–7262 CrossRef CAS
.
- X. Fang, Q. Shang, Y. Wang, L. Jiao, T. Yao, Y. Li, Q. Zhang, Y. Luo and H.-L. Jiang, Adv. Mater., 2018, 30, 1705112 CrossRef
.
- Y. Hou, F. Liu, C. Nie, Z. Li and M. Tong, Environ. Sci. Technol., 2023, 57, 11675–11686 CrossRef CAS PubMed
.
- L. Ran, Z. Li, B. Ran, J. Cao, Y. Zhao, T. Shao, Y. Song, M. K. H. Leung, L. Sun and J. Hou, J. Am. Chem. Soc., 2022, 144, 17097–17109 CrossRef CAS PubMed
.
Footnotes |
| † Electronic supplementary information (ESI) available: Additional materials and methods with instrument details, 1H and 13C NMR and FT-IR spectra for monomers, TEM images, XPS, cyclic voltammograms, EPR, qualitative tests for H2O2, electrochemical data, theoretical analysis including the structures of all the intermediates, energy profile diagrams in bare water, recycling study, and comparison for H2O2 generation rate with literature reports. See DOI: https://doi.org/10.1039/d5ta03188e |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2025 |