Hydraphiles: design, synthesis and analysis of a family of synthetic, cation-conducting channels
George W. Gokel*
Bioorganic Chemistry Program and Department of Molecular Biology
and Pharmacology, Washington University School of Medicine, 660 S. Euclid Ave., Campus Box 8103, St. Louis, MO 63110, USA.. E-mail: ggokel@molecool.wustl.edu
First published on UnassignedUnassigned24th December 1999
Abstract
The concept of channels has been with us more than a century. For half a century, biologists have studied the remarkable workings of protein and peptide channels that permit various cations and small molecules to pass through the phospholipid bilayer membrane. During the past decade, attempts have been made by chemists and biochemists to examine the action of channel compounds from the chemical point of view and to model their function using synthetic structures. What follows is a description of our own efforts to design, synthesize, and characterize a cation transporter that functions in a phospholipid bilayer.
George Gokel was born in New York City but moved as a child to Florida where he grew up. He studied chemistry at Tulane University in New Orleans and earned a doctorate in chemistry at the University of Southern California in Los Angeles. After post-doctoral work with Donald Cram at UCLA and a short stint at the DuPont Chemical Co., Dr Gokel began his academic career. He has held positions in chemistry departments at the Pennsylvania State University, the University of Maryland, and the University of Miami. He is currently Professor in the Department of Molecular Biology and Pharmacology and Director of the Bioorganic Chemistry Program at the Washington University School of Medicine in St. Louis. |
Introduction
During the second half of the 20th century, there have been three important trends in organic chemistry. By the 1950s, the study of physical organic chemistry had moved to the forefront of the science. The study of steric and electronic effects and their influence on mechanism was at center stage. The refinement of physical organic principles provided the critical underlayment for the systematic development of synthetic methodology and strategy. Synthetic chemistry was built on the dual foundations of imagination and physical organic chemistry and reached ascendancy during the 1970s and 1980s. The importance of these two areas continues to be profound. Our understanding of mechanism and our ability to synthesize essentially whatever we can envision has spurred the organic chemist’s imagination into supramolecular chemistry, particularly into the realms of bioorganic chemistry and materials development. Both of these areas face a similar challenge. In short, it is to design a compound that has a desired property or function without knowing precisely how such function is controlled. We have referred to the effort to design compounds having specific functions rather than specific structures as ‘property-directed synthesis’.1 This article is about our efforts to design compounds that perform as transmembrane channels2 in phospholipid bilayers.At the beginning of our effort to design functional synthetic channel compounds, we confronted a difficulty faced by everyone who attempts to mimic nature. The problem is to design a chemical compound that will function as the natural material does even though Nature’s mechanism or mode of action may be imperfectly understood. This problem was compounded in the case of channel models because many in the biological community viewed proteins as the only authentic channels. Even peptides that exhibited channel-like function were regarded by some as intriguing but marginally useful.
Design strategy
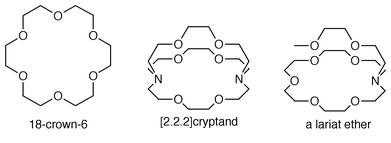
The basic issue that must be considered in the design of a cation-conducting channel is how to get the cation from one side of a bilayer membrane to the other. Organic chemists have dealt with the issue of transporting cations across various membranes by designing, preparing, and using a variety of carrier molecules. These carriers function by complexing a cation at one surface of a membrane, carrying it ‘ferry-boat style’ across the non-polar or insulator regime of the membrane, and then releasing it at the opposite membrane surface. Crown ethers have proved to be particularly successful in transporting cations across bulk membranes. In this context, many combinations of macrocycles, salts, and solvents have been studied.3Our early work with macrocycles led us to confront an interesting problem. The cation complexation constant is given by KS (usually as the decadic logarithm, i.e. log KS). The equilibrium constant is determined by the rates at which complexation and decomplexation occur, i.e.KS = k1/k−1 = kc/kd = kcomplex/kdecomplex. Simple crown ethers4 such as 18-crown-6 show fast binding and release kinetics as required for successful carrier transport but cation binding selectivity is relatively poor. The cryptands are strong binders that show excellent cation selectivity but their binding and release kinetics are poor. We thus developed the family of compounds we named ‘lariat ethers’5 that could achieve the three-dimensional binding arrangement characteristic of cryptands and that also would exhibit good binding dynamics.6
The use of compounds that combine structural features thought to be important with flexibility (and therefore adaptability) was a cornerstone of our channel design philosophy. What were the critical structural features? The first consideration was whether or not the channel would span the bilayer. This is a particularly intriguing question because in 1989, when the original design work was underway, the thickness of a bilayer was at least subject to interpretation if not unclear. Actually, there are three identifiable regions within the bilayer as shown in Fig. 1. These are the insulating regime or ‘hydrocarbon slab’, the polar headgroups, and the midpolar regime. The overall thickness of the membrane will depend upon the identities of the headgroups and the fatty acids.7
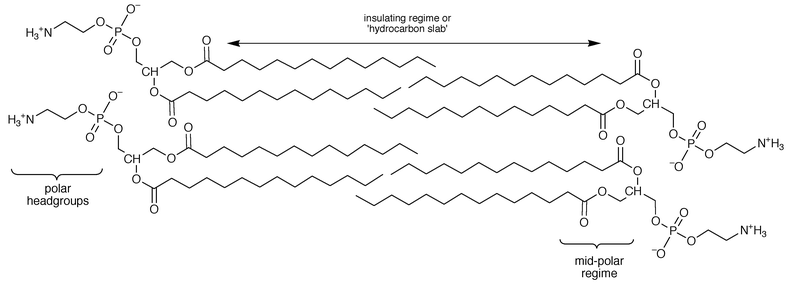 | ||
| Fig. 1 | ||
Chemists generally consider the ‘membrane thickness’ to be the entire width of the bilayer. This is known from X-ray structures of liposomes to be about 40 Å or more.8 Biologists often regard the thickness of a membrane to be 30–35 Å since this is the value obtained from electrophysiological measurements that corresponds to the insulator regime.9 The two values are different but both are correct in their context. The question of what, exactly, requires to be spanned by a channel model compound clearly remains. Should the channel’s length be 30 or 50 Å?10 Is the ‘correct’ length somewhere in between?
This issue illustrates a fundamental problem in modeling biological function. We may choose a span of either 30 or 40 Å to incorporate into our design. Assume we choose 40 Å, complete the design and synthesis, and then assess transport activity. If no cation transport is observed, does that mean that the length is wrong or that some other design feature is inappropriate? The length may be changed to 35 Å. If no cation transport occurs, should lengths of 30, 45 Å, etc. be tried? No variation in length will make the molecule function if some critical feature different from span is ill-designed. Combinatorial approaches could lead to optimization of this length but only after a functional design is in hand.
Several variables can immediately be recognized as bearing on channel function. These include the presence of donor groups, the ‘relays’, headgroups, and the conceptual models for the channel. Each of these variables has aspects that must be considered in the design of a synthetic channel. The consideration must, in the channel case, be done without having an adequate picture of how the wonderful and complex proteins actually work.
Donor groups
It seems reasonable that donor groups such as O, N or S must be present in a channel compound or how would the channel interact with a cation? In the design of the channel, one must consider which donor groups to incorporate. Do we wish only a few donors to be present or should they be numerous? Perhaps the decision about numbers will be influenced by whether the donors are strong or weak. The strength or weakness of a donor group depends on the cation with which it interacts. For example, sulfur (thioether) is a good donor for Zn2+ but less effective for K+.Ether oxygens, like water, are good donor groups for alkali metals. In that case, what sort of scaffolding should be used to organize the donor groups? Should the donor groups be incorporated into a macrocycle? 18-Crown-6 is selective for K+ but will a channel incorporating 18-crown-6 also be selective for this cation? Indeed, can we think about cation selectivity in channels in the same way we conceive of complexation?11 Let us consider 18-crown-6 and its ability to complex Na+ and K+. In aqueous solution, where binding constants are low, 18-crown-6 is selective for K+ over Na+. The respective binding constants are: KS (Na+) = 6.5; KS (K+) = 118.12 This translates to an 18-fold selectivity for K+ over Na+. As noted above, KS = kcomplex/krelease = k1/k−1. The binding rates are known for these two cases and they differ by 2-fold: k1 (Na+) = 2.2 × 108 M−1; k1 (K+) = 4.4 × 108 M−1. The selectivity therefore lies principally in the ≡10-fold difference in cation release rates: k−1 (Na+) = 3.4 × 107 s−1; k-1 (K+) = 3.7 × 106 s−1.
The reaction rates are important because a channel is a dynamic structure. The selectivity of a crown ether or cryptand is determined by what cation is bound relative to another. Which cations are transported rather than retained must define the selectivity of a channel. Thus, the binding selectivity of 18-crown-6 for K+ over Na+ may mean that a channel incorporating this macrocycle would pass Na+ and thus show transport selectivity for it rather than for K+. When we observed above that ‘sulfur (thioether) is a good donor for Zn2+ but less effective for K+’, the context was binding rather than ‘permitting’ the cation to pass by. Thioether might be a ‘good’ channel donor group for an alkali metal or alkaline earth metal ion in the sense that it permits K+ or Ca2+ but restricts Zn2+.
Headgroups
An amphiphile is a compound having two different affinities or ‘philicities’. The amphiphile sodium dodecyl sulfate has a polar sulfonic acid that constitutes the ‘head’ and a 12-carbon span that comprises the ‘tail’. It seems reasonable that a channel would be an amphiphile since it must insert into a bilayer membrane that is constituted of phospholipid amphiphiles. If a single molecule spans the channel, it must be a twin-headed amphiphile.In a channel molecule, the headgroup is required to play a second role: it must serve as, or lead to, a cation entry portal. One point of a membrane is, after all, to prevent salts from getting into or out of a cell. If the channel is to function, it must facilitate the entry and exit of cations (anions, small molecules etc.) without disrupting the membrane structure. It must create a controlled orifice in both the intra- and extra-vesicular surfaces of the bilayer.
There are two obvious challenges in the design of headgroups for a synthetic channel. First, where should the headgroup be placed relative to membrane elements? One possibility is at the membrane surface but an alternative is in the midpolar regime, which is the gateway to the hydrocarbon slab. Second, how polar should the headgroup be? If it is fully charged, should the field be positive or negative? It seems reasonable that a negatively charged headgroup would attract a cation and a cationic headgroup would favor an anion but this is more intuition than knowledge. Information about headgroup preferences might be gleaned from the specifics of protein channel structures. Although the amino acid sequences of many protein channels are known, the three dimensional structures of few have been established.
For most proteins, transmembrane segments are identified by subjecting the amino acid sequence to hydropathy analysis.13 Typically, a computer program examines the entire length of the protein searching for sequences of amino acids that are hydrophobic. A transmembrane segment is about 20 amino acids if it is α-helical and it is about 10 amino acids if it is a β-sheet. Assuming an α-helical transmembrane segment (the common situation), the program looks for a sequence that is expected to partition into a low polarity medium. It is interesting to note that in an α-helix, each amino acid spans about 1.5 Å. Thus, a 20 amino acid sequence translates to a 30 Å span—the estimated thickness of the insulating regime. From this discussion, the problem is apparent. If one doesn’t know the exact structure of the protein, it is hard to guess whether the polar residues, if any, are on the membrane surface or in the midpolar regime. In a synthetic channel, should there possibly be ‘headgroups’ in both positions? If one is unsure of the headgroup position(s), then guessing whether the environment is positively or negatively charged is obviously a challenge.
Water14 is a ubiquitous element in biology and certainly present in bilayer membranes as well as in many proteins. What role will water play in transport? It seems reasonable to think that cations will be only partly desolvated as they pass through the membrane. Complete desolvation is a high-energy process and it is hard to see why all of the water would be stripped from a cation on the periplasmic side of the membrane when it must be rehydrated on the cytoplasmic surface. If water is attached to a cation, how does this affect transport? Must we consider the larger size of the fully hydrated cation rather than its crystallographic diameter? Will discrete molecules of water remain attached to individual ions or will there be exchange with the environment? How does the presence of water affect the choice of donor groups? Amidst all of these variables, one thing that seems clear is that it will be difficult for any positively charged species to traverse a 30 Å, nonpolar span without some interim stabilization.
Our original design for a cation-conducting channel is shown in Fig. 2. The questions posed above were dealt with as follows. Diaza-18-crown-6 polyethers were selected to serve as both headgroups and entry portals. It was known15 that the crowns could bind both Na+ (log KS = 2.99)16 and K+ (log KS = 3.80). It was also known from the early work of Kuwamura17 and of Okahara18 that alkyl-substituted crown ethers formed micellar aggregates when sonicated in aqueous suspension. We demonstrated that twin-tailed diazacrowns could form stable liposomes, suggesting that the crown would be effective as a head group.19
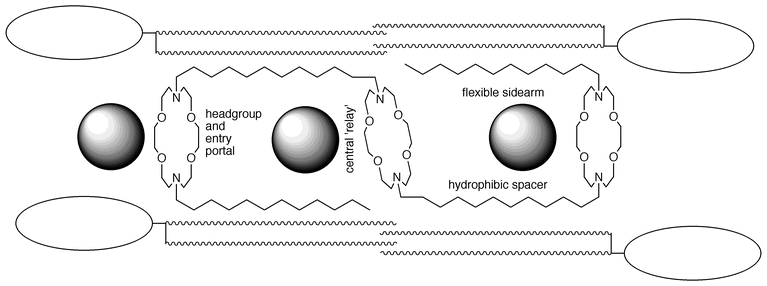 | ||
| Fig. 2 | ||
The two distal macrocycles were expected to serve both as headgroups and as entry portals. A K+ cation (ionic diameter ≡2.7 Å) can pass through the center of the macrocycle as can Na+ (≡2 Å). The central macrocycle was also expected to serve as a portal. Its role was predicted to be as a ‘relay station’ for the transient cation. The polar interaction of the crown with the transient cation at the least polar locale within the bilayer (the midplane) was expected to provide transient stabilization so the transmembrane journey could be completed. It was unknown at the time of the design how this might be accomplished within a protein channel.20
Dodecyl groups were chosen to be the hydrocarbon spacer chains and sidearms. The notion was that the two covalently attached chains would define the channel’s overall length while the flexible sidearms organized along the lipid axis to provide the other ‘wall’ of the channel. A C–C bond is about 1 Å in the linear sense. The dodecyl chain is therefore just under 14 Å. This provides a span of ≡28 Å plus the thickness of the macrocycle. Such a span was expected to cover the insulator regime of the bilayer. The arbitrary decision was made to locate the channel’s headgroup in the bilayer’s midpolar regime rather than on the membrane surface.
Diaza-18-crown-6 groups were chosen as the macrocycles because invertable nitrogen imparts flexibility to the system and obviates the problem of stereoisomerism. The protonation state of the nitrogen atoms within the bilayer is unknown at the time of this writing. It is worth noting that the schematic of the channel and four phospholipid monomers shows the latter with headgroups proportional to the lipid chain lengths.
Alternative designs
The development of synthetic channel models has been considered in a number of groups.21 In some cases, compounds were designed de novo and in others the structures of the products of certain reactions suggested that they might possess channel activity. On occasion, the transport of Na+, K+ or Ca2+ was not studied but the assessment of transport efficacy was limited to H+ or Co2+. Thus, the references cited represent a great range of approaches, designs, structural types and success.Synthesis of channel 1
The first channel in the tris(macrocycle) family, designated C12<N18N>C12<N18N>C12<N18N >C12 in a shorthand we developed some years ago,22 presented the interesting problem of being nearly, but not quite, symmetrical. Of course, there is two-fold symmetry through the central macrocycle. The distal crowns, however, are attached to dodecyl chains on either side but are not symmetrical. Our best current synthetic approach23 is accomplished as follows.In the first step, diaza-18-crown-6 is monoalkylated by 1-bromododecane to give C12<N18N> (Scheme 1). This, in turn, is treated with 1,12-dibromododecane to give C12<N18N>C12Br. Use of the latter to dialkylate diaza-18-crown-6 affords channel 1 (1). This approach was the model for the more than 30 members of this novel structural family now in hand.
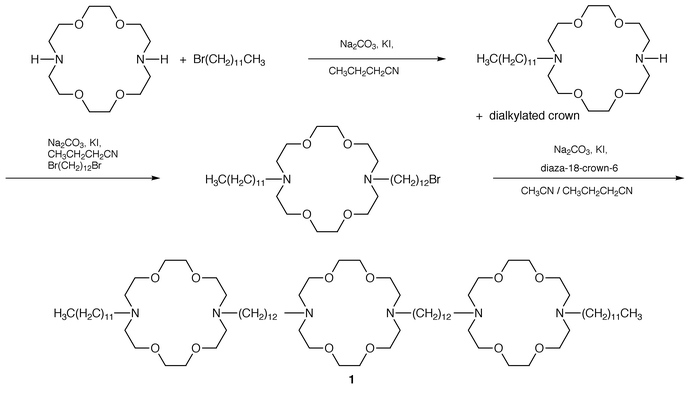 | ||
| Scheme 1 | ||
Assessment of ionophoretic activity
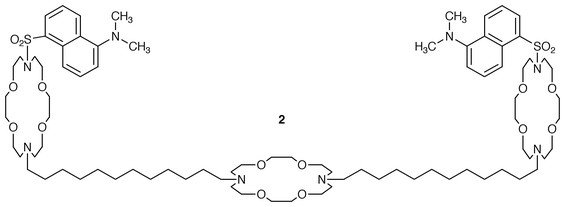
Three methods were used to assess the efficacy of the synthetic cation transporters: fluorescence, 23Na NMR and planar bilayer conductance. The fluorescence technique24 was used for determining proton flux in a few compounds and only at an early stage of the study. Planar bilayer methods are discussed below. The bulk of the quantitative measurements were accomplished by using a dynamic NMR method. In short, phospholipid liposomes (vesicles) are created in the presence of NaCl. 23Na NMR shows a single line for Na+(inside) and Na+ (outside). When Dy3+, a shift reagent, is added to the external solution, the chemical shift of the external Na+ changes. When an ionophore is added to the bilayer, internal and external Na+ may equilibrate and the exchange rate constant may be determined from the concentration dependence of the linewidth change: K = 1/τ = π(Δν1−Δν0).25Multiple experiments at concentration ranges from 0–20 μM are required to determine the rate constant for a single transporter. The experiments were therefore done in tandem with a standard of known activity. In the early work, this standard was the naturally occurring, channel-forming peptide gramicidin.26 Gramicidin is an excellent pore-former that sometimes functions even when experimental conditions are not properly maintained. Thus, the failure of a synthetic channel-former to transport Na+ might occur due to poor experimental conditions rather than lack of efficacy and gramicidin might function despite the experimental problems. We have thus adopted Dn<N18N>C12<N18N>C12<N18N>Dn (Dn = dimethylaminonaphthylsulfonyl or dansyl) as our experimental standard. The ‘dansyl channel’ transports cations very reproducibly but fails when experimental conditions are not properly maintained. We have also modified the [Na+] from 100 to 250 mM which gives better entrapment and more reproducible experimental results.
Using the 23Na NMR method, we found channel 1 transported Na+ across a phospholipid bilayer at a rate about 27% of that observed for gramicidin. The exchange rate observed for gramicidin is ≡175 s−1 so channel 1 is transporting cations on the millisecond time scale. We were also able to correlate the transport rate with a number of structural variations although the details are beyond the scope of this review. It was interesting to note, however, that when terminal macrocycles were altered from C12<N18N>≡ to <18N>≡ (aza-18-crown-6), Na+ transport activity was lost. Replacement of the sidearm by benzyl, substituted benzyl, naphthyl, dansyl and others led to differences in transport rates but most sidearm changes afforded functional channels.
Naming the family of compounds
Many of the early channels were tris(macrocycle)s and we referred to these compounds as such. As structural variations led to the removal of one or more macrocycles, the name was no longer appropriate. We considered the name ‘hydraphile’ as a possibility because of its association with the two-headed monster slain by Hercules. The dictionary27 provided additional inspiration in two other definitions. A hydra is ‘any of several small freshwater polyps of the genus Hydra and related genera, having a naked cylindrical body and an oral opening surrounded by tentacles’. Clearly the shape and tentacles were highly suggestive. An additional definition added to the appropriateness of the name: ‘A persistent or multifaceted problem that cannot be eradicated by a single effort’.Control experiments
The fact that sodium flux was observed in the presence of 1 was very encouraging but not conclusive. It could mean that all of the design concepts were as originally conceived. It is always nice to have one’s ideas proven successful. Still, the fact of sodium transport was permissive rather than conclusive. It was possible, for example, that the tris(macrocycle) functioned simply by detergent action. To assess this possibility, the tris(macrocycle) ionophore was replaced by either Triton X-100, a neutral detergent, or sodium dodecyl sulfate, an anionic detergent. The concentration range in the 23Na NMR experiment was expanded from the typical 0–20 μM by ten-fold to 0–200 μM but no cation flux (line broadening) could be detected in either case.21It was possible that the tris(macrocycle)s were unusually active carrier molecules rather than pore-formers. A conventional concentric tube apparatus was used to assess carrier transport through a bulk CHCl3 membrane in a group of 10 compounds.28 In this experiment, a beaker is charged with CHCl3 and water. A glass tube is then suspended in the beaker through the upper water layer and into the CHCl3. The outer, upper aqueous ring is thus separated from the inner core of water. A NaX salt can be carried through the CHCl3 bulk membrane from inner core to outer ring. On the atomic scale, a distance of ≡107 Å must be traversed so the channel mechanism is precluded. The transport rates observed in this experiment (relative to valinomycin) were compared to those obtained for the same compounds in the 23Na NMR/bilayer experiment (relative to gramicidin). In short, the data showed no discernible correlation. This does not prove the channel mechanism but clearly discounts carrier transport within the bilayer.29 These findings comport with the observation that fragments of the channel such as C12<N18N>C12<N18N>C12 or <18N>C12<N18N>C12<N18> and known carriers such as PhCH2<N18N>CH2Ph were not sufficiently active to show transport when assessed by the NMR method.
It is interesting to note that addition of the tris(macrocycle)s to the CHCl3 concentric tube system led to a dramatic increase in hydration of that solvent.27 No further work was undertaken to resolve this issue because it was tangential to the main thrust of the effort.
It was possible that the rate differences observed for structurally related channels might be due only to variations in the extent of membrane penetration. Octanol–water partition coefficients30 were determined for several substituted crown ethers and the experimentally determined values were compared to data calculated by the Hint module of Sybyl.27 Agreement between experiment and calculation was good. The data showed that the tris(macrocycle)s favored octanol (i.e. the membrane) by >1010 up to as much as 1030. Although the rate differential was not due to differences in partitioning, a minor kinetic effect was observed. When the ionophore was added to the preformed suspension of liposomes and then analyzed immediately, the plots of 1/τ vs. [ionophore] showed curvature. If NMR analysis was delayed for an hour, the lines were essentially straight. Likewise, if the vesicles were formed in the presence of the ionophore (direct incorporation) linear data were obtained. Care was thus taken to permit equilibration of the sample system.
The channel’s conformation
Changing the size of the central macrocycle diminished the transport rate but did not preclude it. Substituting the central macrocycle by an O(CH2CH2O)3 chain again impeded but did not prohibit sodium transport. We concluded that the central macrocycle in R<N18N>C12<N18N>C12<N18N>R was parallel to the lipid axis rather than parallel to the other two macrorings. Thus, we inferred that the cation passed by but not through the central macrocycle. This conformation is illustrated in Fig. 3.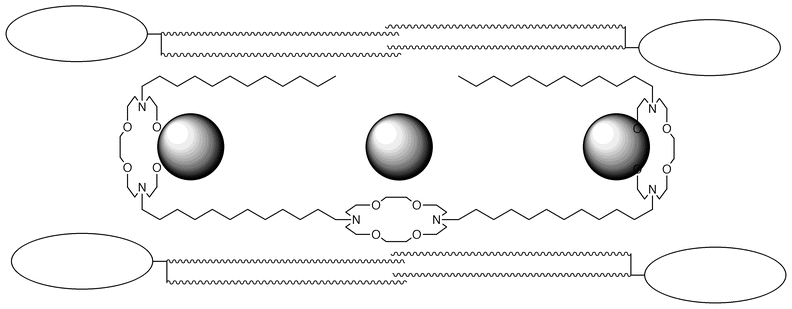 | ||
| Fig. 3 | ||
Assessment of optimal distances
The tris(macrocyclic) channels were designed to function in a phospholipid bilayer but membrane dimensions and the placement of a channel within it are elusive. An attempt was therefore made to experimentally determine the optimal length of the channel. This was done by varying the length of the covalent, hydrophobic spacers in PhCH2<N18N>C12<N18N>C12<N1 8N>CH2Ph. It was assumed that the overall conformation would remain similar as the chain length was varied by 2 methylenes on either side of the central macrocycle. The starting point for this exercise is indicated on the graph (Fig. 4) by an arrow. It was anticipated that incremental lengthening of a flexible assembly would lead to some reduction in efficacy as less favorable conformations were adopted. As chain length diminished, it was expected that a point would be reached at which the structure was simply too small to span the bilayer. Note that each change of 2 methylene units in the spacer is an overall change of 4 methylenes or ≡4 Å in span. Shortening the chain by 4 Å or lengthening by 8 Å drops the transport rate to about half. Shortening by 8 Å leads to an inactive ionophore. Note that by ‘inactive’ we mean that no transport activity can be detected by the 23Na NMR method.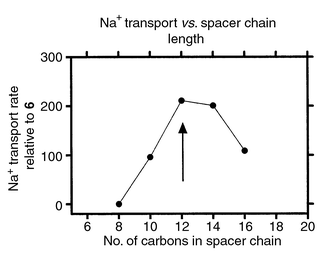 | ||
| Fig. 4 | ||
Assessment of the conformation and location of the channel within the bilayer
The synthetic tris(macrocycle) channel compounds can readily be modified to incorporate various structural probes. In particular, fluorescent headgroups can be included as an integral part of the structure. In biochemical studies, for example, the indolyl residue of tryptophan is often used as a fluorescent probe. Fluorescent dansyl residues were incorporated into the channel as headgroups: Dn<N18N>C12<N18N>C12<N18N>Dn, 2. The fluorescence spectrum was determined in a variety of solvents from nonpolar to polar as well as in a phospholipid bilayer. Note that lipids were carefully screened to be sure that any fluorescent impurities were absent. The fluorescence maxima (λmax) are shown in Fig. 5, plotted as a function of solvent polarity (the Reichardt parameter, ET).31 A dashed line indicates the fluorescence maximum determined for the dansyl channel. The polarity experienced by the dansyl group is between that of ethanol and methanol—about what would be expected for the glyceryl ester regime of a phospholipid. In any event, the dansyl environment of the channel is significantly more polar than would be expected were it embedded in the membrane’s ‘hydrocarbon slab.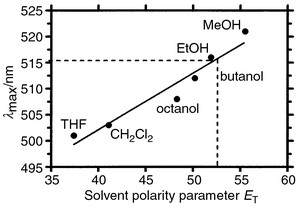 | ||
| Fig. 5 | ||
Fluorescence depth quenching
The heterocyclic ‘doxyl’ group quenches fluorescence by virtue of its unpaired electron spin. By using doxyl-substituted phospholipids, it is possible to estimate how far from the bilayer’s midplane is the dansyl ‘headgroup’. 7-Doxyl- and 12-doxyl-palmitoyl-substituted phosphatidylcholines were used along with the dansyl channel to estimate headgroup separation.32 Application of the appropriate equations33 gives a value for the headgroup separation of 28 Å (i.e. the distance of the headgroup from the bilayer’s midplane is 14 Å). If the hydrocarbon slab is approximately 30 Å thick and the dansyl groups are ≡6 Å, whether measured laterally or transversely, one concludes that the channel’s termini are in or near the midpolar region created by the glyceryl ester residues. This comports with the position estimated from dansyl fluorescence.Experiments designed to address headgroup issues
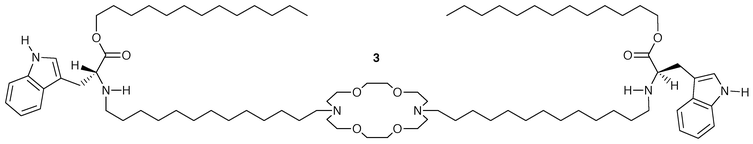
It was assumed that the conformation illustrated in Fig. 1 required the distal macrocycles to function both as headgroups and entry portals. An effort was made to confirm experimentally the ability of diaza-macrocycles to serve as amphiphile headgroups. It was found that a range of 2-armed diaza-18-crown-6 derivatives could form stable liposomes when sonicated in aqueous suspension.34 In a molecule such as C18<N18N>C18, the octadecyl chains can function only as hydrophobic tails so the macrocycles must comprise the headgroups. Successful formation of stable liposomes from R<N18N>R clearly implies the efficacy of the crown as a headgroup. Evidence on the interaction of the headgroup with cations is discussed below.The frequent observation of the rare amino acid tryptophan at the boundary margins of putative transmembrane segments of proteins suggested that it might play some important role in channel formation. In separate work, we demonstrated that indole, the sidechain of tryptophan, could function as a headgroup for the formation of stable liposomes.35 Clearly, tryptophan cannot function as an entry portal for cations in the same sense that crowns do. We prepared 3, which incorporated the essential channel elements shown in Fig. 1, but lacked the cation entry portal. Compound 3 showed no cation transport ability as judged by any of the analytical methods attempted.
H-bond-induced blockage of the headgroup
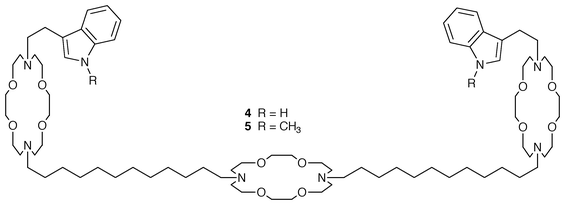
Because of our35 and others’33,36 speculation that tryptophan and/or its indole residue could play an anchoring role in phospholipid bilayer membranes, we prepared a tris(macrocycle) terminated in the indolylmethyl residue, i.e. InCH2CH2<N18N>C12<N18N>C 12<N18N>CH2CH2In 4. Although structurally similar to the highly active benzyl chan- nel (PhCH2<N18N>C12<N18N>C12<N 18N>CH2Ph), the indolyl channel showed no ability to transport Na+.37 Both CPK models and Monte Carlo simulations showed that a hydrogen bond between the indole NH and a macroring oxygen atom could form. An infrared band, attributable to H-bond formation, did not alter its position during 100-fold dilution. This suggests that the H-bond is intramolecular.This result, although inferential, is clearly important. It implies that weak H-bond interactions can block the channel. This, in turn, implies that the conformation in Fig. 2 is correct or why would occlusion of the entry portal block the channel? Replacement of the indolyl residue by a methylindolyl, i.e. replacement of the NH group by NCH3, gave a compound, 5, that was fully active as a channel.
Application of the Hammett equation
If a cation enters the channel by going through or passing by the distal macrocycle(s), it should experience the stereoelectronics of that group. We prepared three channel compounds of the type PhCH2<N18N>C12<N18N>C12<N1 8N>CH2Ph in which the aromatic ring of the benzyl group was para-substituted. The substituents were H (shown), 4-methoxy and 4-nitro. A straight-line relationship was observed.38 Admittedly, the graph (Fig. 6) involves only three points but the difficulty of synthesis and analysis will be apparent.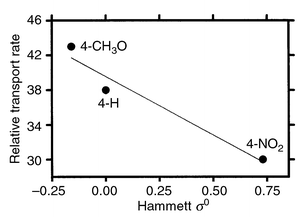 | ||
| Fig. 6 | ||
The critical results are as follows. First, r2 for the three-point line is 0.95—a respectable value. Second, the slope of the line is negative as expected for the interaction of a cation with a neutral host. The slope is shallower than observed for complexation of cations by dibenzyldiaza-18-crown-6 derivatives.39 This is expected for a transient interaction. A third point, not apparent from the graph, is that when relative transport rates obtained in the concentric tube experiment (see above) were plotted vs. Hammett ς0, the straight-line ‘correlation’ had a zero slope and r2 = 0.4.
Changes in headgroup size
Only limited work has thus far been completed to assess the influence of headgroup size. Two channels were prepared for this study. In both cases, the terminal residue (‘flexible sidearm’ in Fig. 1) was fluorobenzyl. The distal macrocycles were either 15- or 18-membered. When cation transport was assessed by using the NMR method, transport of Na+ by the 15-membered ring channel was found to be about 60% of that determined for the compound having 18-membered distal macrocycles.40The aggregation state of the channel
The availability of both the dansyl (2) and N-methylindolyl (5) channels provided an opportunity to assess the aggregation state of the channel. We found that the N-methylindolyl-sidearmed channel absorbed light at 283 nm and fluoresced at 343 nm—the wavelength at which the dansyl-terminated channel absorbs. An experiment was undertaken in which the amount of channel was held constant and the mole fractions of 2 and 5 were varied from 0→1 and 1→0, respectively. A logarithmic plot of the fluorescence ratio as a function of mole fraction gave a line with a slope of 1.12. The slope of this line has been interpreted to be the aggregation state. Thus, at least for these two compounds, the channel operates, within experimental error, as a monomer.4123Na NMR transport results
The 23Na NMR experiment as it is used to assess transport in a bilayer membrane was described above. A number of structural variations have been incorporated into channel 1 and the relative transport abilities of these compounds have been measured. Selected results are shown in Table 1 using the shorthand described above in which <N18N> represents 4,13-diaza-18-crown-6.| Sidearm | Headgroup | Spacer | Center | krel |
|---|---|---|---|---|
| Dodecyl (C12H25) | <N18N> | C12H24 | <N18N> | 27 |
| Benzyl (CH2C6H5) | <N18N> | C12H24 | O–2,6–C10H6–O | <2 |
| Benzyl | <N18N> | C12H24 | <N18N> | 39 |
| Dansyl | <N18N> | C12H24 | <N18N> | 24 |
| Dansyl | <N18N> | C12H24 | <N15N> | 19 |
| 4-Fluorobenzyl | <N18N> | C12H24 | <N18N> | 26 |
| 2-(3-Indolyl)ethyl | <N18N> | C12H24 | <N18N> | <2 |
| 2-(N-Methyl-3-indolyl)- ethyl | <N18N> | C12H24 | <N18N> | 23 |
| None | <18N> | C12H24 | <N18N> | <2 |
Patch clamping results
The ability of various synthetic and peptidic compounds to function as cation-conducting channels may be demonstrated by a technique called ‘planar bilayer conductance’, or PBC. In this technique, a phospholipid bilayer is formed in a pinhole separating two salt phases. When the membrane is formed over the pinhole, it turns dark and the system is sometimes referred to as a black lipid membrane. Using electrodes and a patch clamp amplifier, the electrical response of a bilayer membrane imbued with a transporter may be observed. A membrane is normally insulating so increases in the electrical response are interpreted as the passage of ions through it.We conducted a number of experiments that confirmed ion transport. Our most recent PBC results are for a calixarene-based channel.42 The patch clamp or PBC technique is wonderfully sensitive but it is a difficult method to master and to reproduce. In some cases, supposedly identical samples show high and no channel activity. In other cases, the membrane itself collapses and terminates the study before sufficient data have been acquired. We have thus preferred the NMR method; it is cumbersome but, so far at least, it has proved to be reliable.
Conclusions
Clearly many channel models are possible. Our basic design is effective and its modular design is proving useful to assess individual structural issues. In work that is yet to be published, we have explored covalent attachment of the sidearms and variations in the central relay unit. Cation/anion selectivity, ion selectivity in general, and rectification all remain important challenges in this emerging area.Acknowledgements
I warmly thank the many co-workers whose names are on the cited references who have been intimately involved in this project. I appreciate their hard work, their clever ideas, and their keen insight and criticism. Support of this work by grants from NIH (GM 36262), NATO and NSF (CHE-9805840) is gratefully acknowledged.References
- G. W. Gokel, J. C. Medina and C. Li, Synlett, 1991, 677 CrossRef CAS.
- W. D. Stein, Channels, Carriers, and Pumps, Academic Press, New York, 1990; Search PubMed; B. Hille, Ionic Channels of Excitable Membranes, 2nd edn., Sinauer Press, Sunderland, MA, 1992; Search PubMed; D. J. Aidley and P. R. Stanfield, Ion Channels: Molecules in Action, Cambridge University Press, Cambridge, 1996. Search PubMed.
- B. A. Moyer, Complexation and Transport, in Molecular Recognition: Receptors for Cationic Guests, vol. 1, Comprehensive Supramolecular Chemistry, ed. G. W. Gokel, Elsevier, Oxford, 1996, pp. 377–416; Search PubMed; T. Araki and H. Tsukube, Liquid Membranes: Chemical Applications, CRC Press, Boca Raton, 1990. Search PubMed.
- G. W. Gokel, Crown Ethers and Cryptands, in Monographs in Supramolecular Chemistry, ed. J. F. Stoddart, The Royal Society of Chemistry, London, England, vol. 3, 1991, p. 190. Search PubMed.
- The name lariat ether was suggested by the CPK models that resembled a looped rope. Moreover, the mode of complexation was envisioned as a ‘rope and tie’ combination of macroring and sidearm. When the first manuscript was submitted to this august Journal, it was accepted but an editor deleted the name lariat because he or she felt that the name was not officially sanctioned nomenclature. We felt the name was an important element in conceptualizing these compounds. We therefore withdrew the paper with the intention of submitting it elsewhere. The editor relented and the report appeared in due course: G. W. Gokel, D. M. Dishong and C. J. Diamond, J. Chem. Soc., Chem. Commun., 1980, 1053..
- G. W. Gokel and J. E. Trafton, Cation Binding by Lariat Ethers, in Cation Binding by Macrocycles, ed. G. W. Gokel and Y. Inoue, Marcel Dekker, New York, 1990, pp. 253-310; Search PubMed; G. W. Gokel, Lariat Ethers, in Inclusion Phenomena, ed. J. L. Atwood, E. Davies and D. D. MacNicol, Oxford University Press, 1991, vol. 4, pp. 287–328; Search PubMed; G. W. Gokel, Chem. Soc. Rev., 1992, 21, 39; Search PubMed; G. W. Gokel and O. F. Schall, Lariat Ethers, in Comprehensive Supramolecular Chemistry, ed. G. W. Gokel, Elsevier, Oxford, 1996, pp. 97–152. Search PubMed.
- M. C. Wiener and S. H. White, Biophys. J., 1992, 61, 434 Search PubMed.
- Small unilamellar vesicles (SUV) formed by sonicating egg phosphatidylcholine lipids show internal and external radii of 65 and 105 Å respectively, suggesting a membrane thickness of 40 Å [P. Yeagle, The Membranes of Cells, Academic Press, New York, 1987, p. 47]. For a review of lipid packing in the solid state, see H. Hauser and G. Poupart, Lipid Structure, in ref. 9..
- The Structure of Biological Membranes, ed. P. Yeagle, CRC Press, Boca Raton, 1992. Search PubMed.
- R. P. Rand and V. A. Parsegian, The Forces between Interacting Bilayer Membranes and the Hydration of Phospholipid Assemblies, in The Structure of Biological Membranes, ed. P. Yeagle, CRC Press, Boca Raton, 1992, pp. 251–306. Search PubMed.
- J. S. Bradshaw, R. M. Izatt, A. V. Bordunov, C. Y. Zhu and J. K. Hathaway, Crown Ethers, in Molecular Recognition: Receptors for Cationic Guests, ed. G. W. Gokel, Elsevier, Oxford, 1996, vol. 1, pp. 35–95. Search PubMed.
- G. W. Liesegang, A. Vasquez, N. Purdie and E. M. Eyring, J. Am. Chem. Soc., 1977, 3240 CrossRef CAS.
- T. E. Creighton, Proteins: Structures and Molecular Properties, 2nd edn., Freeman, New York, 1993, p. 154. Search PubMed.
- F. Franks, Water, Royal Society of Chemistry, Cambridge, 1984, p. 69. Search PubMed.
- K. A. Arnold, J. C. Hernandez, C. Li, J. V. Mallen, A. Nakano, O. F. Schall, J. E. Trafton, M. Tsesarskaja, B. D. White and G. W. Gokel, Supramol. Chem., 1995, 5, 45 Search PubMed.
- Values are for N,N′-bis(n-dodecyl)-4,13-diaza-18-crown-6 in anhydrous CH3OH at 25 °C..
- T. Kuwamura and T. Kawachi, Yukagaku, 1979, 28, 195(Chem. Abstr.197990206248 Search PubMed; T. Kuwamura, M. Akimaru, H. L. Takahashi and M. Arai, Kenkyu Hokoku-Asahi Garasu Kogyo Gijutsu Shoreikai, 1979, 35, 45(Chem. Abstr.19819561394q Search PubMed; T. Kuwamura and S. Yoshida, Nippon Kagaku Kaishi, 1980, 427(Chem. Abstr.19809328168e Search PubMed.
- M. Okahara, P. L. Kuo, S. Yamamura and I. Ikeda, J. Chem. Soc., Chem. Commun., 1980, 586; Search PubMed; I. Ikeda, K. Iwaisako, Y. Nakatsuji and M. Okahara, Yukagaku, 1986, 35, 1001(Chem. Abstr.198710686714). Search PubMed.
- L. E. Echegoyen, J. C. Hernandez, A. Kaifer, G. W. Gokel and L. Echegoyen, J. Chem. Soc., Chem. Commun., 1988, 836 RSC; S. L. De Wall, K. Wang, D. L. Berger, S. Watanabe, J. C. Hernandez and G. W. Gokel, J. Org. Chem., 1997, 62, 6784 CrossRef CAS.
- B. Roux and R. MacKinnon, Science, 1999, 285, 100 CrossRef CAS.
- I. Tabushi, Y. Kuroda and K. Yokota, Tetrahedron Lett., 1982, 23, 4601 CrossRef CAS; F. M. Menger, D. S. Davis, R. A. Persichetti and J. J. Lee, J. Am. Chem. Soc., 1990, 112, 2451 CrossRef CAS; Y. Kobuke, K. Ueda and M. Sokabe, J. Am. Chem. Soc., 1992, 114, 7618 CrossRef CAS; J. G. Neevel and R. Nolte, Tetrahedron Lett., 1984, 25, 2263 CrossRef CAS; R. J. M. Nolte, A. J. M. Beijnen, J. G. Neevel, J. W. Zwikker, A. J. Verkley and W. Drenth, Isr. J. Chem., 1984, 24, 297 Search PubMed; U. F. Kragten, M. F. M. Roks and R. J. M. Nolte, J. Chem. Soc., Chem. Commun., 1985, 1275 RSC; L. Jullien and J. M. Lehn, Tetrahedron Lett., 1988, 29, 3803 CrossRef CAS; L. Jullien and J. M. Lehn, J. Inclusion Phenom., 1992, 12, 55 CAS; J. Canceill, L. Jullien, L. Lacombe and J. M. Lehn, Helv. Chim. Acta, 1992, 75, 791 CrossRef CAS; M. Pregel, L. Jullien and J. M. Lehn, Angew. Chem., Int. Ed. Engl., 1992, 31, 1637 CrossRef; M. Pregel, L. Jullien, J. Canceill, L. Lacombe and J. M. Lehn, J. Chem. Soc., Perkin Trans. 2, 1995, 417 RSC; V. E. Carmichael, P. Dutton, T. Fyles, T. James, J. Swan and M. Zojaji, J. Am. Chem. Soc., 1989, 111, 767 CrossRef CAS; T. Fyles, T. James and K. Kaye, Can. J. Chem., 1990, 68, 976 CAS; T. Fyles, K. Kaye, T. James and D. Smiley, Tetrahedron Lett., 1990, 1233 CrossRef CAS; K. Kaye and T. Fyles, J. Am. Chem. Soc., 1993, 115, 12 315 CrossRef; T. Fyles, T. James, A. Pryhitka and M. Zojaji, J. Org. Chem., 1993, 58, 7456 CrossRef CAS; M. R. Ghadiri, J. R. Granja and L. K. Buehler, Nature, 1994, 369, 301 CrossRef CAS; N. Khazanovich, J. R. Granja, D. E. McRee, R. A. Milligan and M. R. Ghadiri, J. Am. Chem. Soc., 1994, 116, 6011 CrossRef CAS; E. Stadler, P. Dedek, K. Yamashita and S. Regen, J. Am. Chem. Soc., 1994, 116, 6677 CrossRef CAS; N. Voyer and M. Robitaille, J. Am. Chem. Soc., 1995, 117, 6599 CrossRef CAS; Y. Tanaka, Y. Kobuke and M. Sokabe, Angew. Chem., Int. Ed. Engl., 1995, 34, 693 CrossRef CAS; G. Deng, M. Merritt, K. Yamashita, V. Janout, A. Sadownik and S. L. Regen, J. Am. Chem. Soc., 1996, 118, 3308; A. Matsubara, K. Asami, A. Akagi and N. Nishino, Chem. Commun., 1996, 2069 RSC; D. Seebach, A. Brunner, H. M. Buerger, R. N. Reusch and L. L. Bramble, Helv. Chim. Acta, 1996, 79, 507 CrossRef CAS; H. Wagner, K. Harms, U. Koert, S. Meder and G. Boheim, Angew. Chem., Int. Ed. Engl., 1996, 35, 2643 CrossRef CAS; S. Matile, J. Am. Chem. Soc., 1997, 119, 8726 CrossRef; L. A. Weiss, N. Sakai, B. Ghebremariam, C. Ni and S. Matile, J. Am. Chem. Soc., 1997, 119, 12 142 CrossRef CAS; J.-C. Meillon and N. Voyer, Angew. Chem., Int Ed. Engl., 1997, 36, 967 CrossRef CAS; A. D. Pechulis, R. J. Thompson, J. P. Fojtik, H. M. Schwartz, C. A. Lisek and L. L. Frye, Bioorg. Med. Chem., 1997, 5, 1893 CrossRef CAS; L. Chen, N. Sakai, S. T. Moshiri and S. Matile, Tetrahedron Lett., 1998, 39, 3627 CrossRef CAS; T. D. Clark, L. K. Buehler and M. R. Ghadiri, J. Am. Chem. Soc., 1998, 120, 651 CrossRef CAS; C. Ni and S. Matile, Chem. Commun., 1998, 755 RSC; R. N. Reusch and H. L. Sadoff, Proc. Natl. Acad. Sci. USA, 1988, 85, 4176 CAS.
- J. C. Hernandez, J. E. Trafton and G. W. Gokel, Tetrahedron Lett., 1991, 269.
- O. Murillo, S. Watanabe, A. Nakano and G. W. Gokel, J. Am. Chem. Soc., 1995, 117, 7665 CrossRef CAS.
- K. Kano and J. H. Fendler, Biochim. Biophys. Acta, 1978, 509, 289 CAS.
- F. G. Riddell and M. K. Hayer, Biochem. Biophys. Acta, 1985, 817, 313 Search PubMed; F. G. Riddell and S. J. Tompsett, Biochim. Biophys. Acta, 1990, 1990, 193.
- D. A. Doyle and B. A. Wallace, Biomembranes, 1996, 6, 327; Search PubMed; B. A. Wallace, J. Struct. Biol., 1998, 121, 123 CrossRef CAS.
- American Heritage Dictionary, 3rd edn., Houghton-Mifflin, 1992. Search PubMed.
- The apparatus is described in detail in ref. 20..
- O. Murillo, I. Suzuki, E. Abel, C. L. Murray, E. S. Meadows, T. Jin and G. W. Gokel, J. Am. Chem. Soc., 1997, 119, 5540 CrossRef CAS.
- Lipophilicity in Drug Action and Toxicology, ed. V. Pliska, B. Testa and H. v. d. Waterbeemd, in Methods and Principles in Medicinal Chemistry, ed. R. Mannhold, H. Kubinyi and H. Timmerman, VCH, Weinheim, 1996, vol. 4. Search PubMed.
- C. Reichardt, Chem. Rev., 1994, 94, 2319 CrossRef CAS.
- E. Abel, G. E. M. Maguire, E. S. Meadows, O. Murillo, T. Jin and G. W. Gokel, J. Am. Chem. Soc., 1997, 119, 9061 CrossRef CAS.
- K. Kachel, E. Ascuncion-Punzalan and E. London, Biochemistry, 1995, 34, 15 475 CrossRef CAS.
- S. L. De Wall, K. Wang, D. L. Berger, S. Watanabe, J. C. Hernandez and G. W. Gokel, J. Org. Chem., 1997, 62, 6784 CrossRef CAS.
- E. Abel, M. F. Fedders and G. W. Gokel, J. Am. Chem. Soc., 1995, 117, 1265 CrossRef CAS.
- M. Schiffer, C.–H. Chang and F. J. Stevens, Protein Eng., 1992, 5, 213 CrossRef CAS.
- O. Murillo, E. Abel, G. E. M. Maguire and G. W. Gokel, Chem. Commun., 1996, 2147 RSC.
- O. Murillo, I. Suzuki, E. Abel and G. W. Gokel, J. Am. Chem. Soc., 1996, 118, 7628 CrossRef CAS.
- D. A. Gustowski, V. J. Gatto, J. Mallen, L. Echegoyen and G. W. Gokel, J. Org. Chem., 1987, 52, 5172 CrossRef CAS.
- C. L. Murray, E. S. Meadows, O. Murillo and G. W. Gokel, J. Am. Chem. Soc., 1997, 119, 7887 CrossRef CAS.
- E. Abel, G. E. M. Maguire, O. Murillo, I. Suzuki and G. W. Gokel, J. Am. Chem. Soc., 1999, 121, 9043 CrossRef CAS.
- J. de Mendoza, F. Cuevas, P. Prados, E. S. Meadows and G. W. Gokel, Angew. Chem., Int. Ed., 1998, 37, 1534 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2000 |