Chemical activation of boron nitride fibers for improved cationic dye removal performance†
Abstract
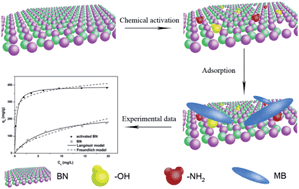
Adsorption represents an efficient and economical approach for water purification and substantial research is being performed to develop effective sorbent materials. Porous boron nitride (BN) composed of light elements is considered as a promising candidate for pollution treatment due to its unique polarity of B–N bonds, high specific surface area, numerous structural defects, chemical stability, and oxidation resistance. However, the adsorption performance based on porous BN is hindered by either few activated sites or a low degree of crystallinity. In this work, we have developed a simple chemical method to activate pre-obtained well-crystallized porous BN fibers in acid solution. The successful chemical activation has been identified by FTIR spectra and zeta potential measurements. Benefiting from these advantageous features, the activated BN fibers with high stability exhibited enhanced cationic dye removal performance compared to the un-activated ones. The effects of pH value, contact time, temperature and adsorbent amount on the methylene blue (MB) adsorption properties were analyzed. The adsorption equilibrium data were interpreted in terms of Langmuir and Freundlich models and the results showed that Langmuir isotherms best represent the adsorption system. The maximum adsorbed amount for MB was as high as 392.2 mg g−1 at pH 8.0 and 30 °C. The adsorption rate was sharply enhanced after chemical activation. The excellent reusability of the activated BN was also confirmed. It is shown that chemical activation plays a key role in enhanced dye adsorption performance. Therefore, our developed chemical activation method for porous BN fibers opens the door toward the practical application of activated BN for drinking water purification.

 Please wait while we load your content...
Please wait while we load your content...