CO2 reduction at low overpotential on Cu electrodes in the presence of impurities at the subsurface
Abstract
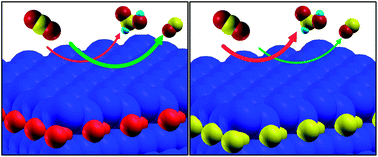
We report a theoretical analysis of the Cu electrode, modified by O impurities at the subsurface, concerning the electrochemical activity of the CO2 reduction reaction and selectivity between CO and HCOOH evolution. On the basis of quantum mechanical simulations, we propose a novel Cu electrode modified by C impurities at the subsurface for CO2 electroreduction, which has a better catalytic selectivity.

 Please wait while we load your content...
Please wait while we load your content...