The origin of unidirectional charge separation in photosynthetic reaction centers: nonadiabatic quantum dynamics of exciton and charge in pigment–protein complexes†
Abstract
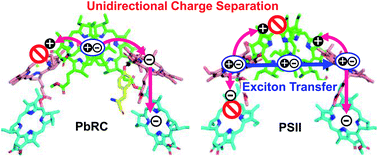
Exciton charge separation in photosynthetic reaction centers from purple bacteria (PbRC) and photosystem II (PSII) occurs exclusively along one of the two pseudo-symmetric branches (active branch) of pigment–protein complexes. The microscopic origin of unidirectional charge separation in photosynthesis remains controversial. Here we elucidate the essential factors leading to unidirectional charge separation in PbRC and PSII, using nonadiabatic quantum dynamics calculations in conjunction with time-dependent density functional theory (TDDFT) with the quantum mechanics/molecular mechanics/polarizable continuum model (QM/MM/PCM) method. This approach accounts for energetics, electronic coupling, and vibronic coupling of the pigment excited states under electrostatic interactions and polarization of whole protein environments. The calculated time constants of charge separation along the active branches of PbRC and PSII are similar to those observed in time-resolved spectroscopic experiments. In PbRC, Tyr-M210 near the accessary bacteriochlorophyll reduces the energy of the intermediate state and drastically accelerates charge separation overcoming the electron–hole interaction. Remarkably, even though both the active and inactive branches in PSII can accept excitons from light-harvesting complexes, charge separation in the inactive branch is prevented by a weak electronic coupling due to symmetry-breaking of the chlorophyll configurations. The exciton in the inactive branch in PSII can be transferred to the active branch via direct and indirect pathways. Subsequently, the ultrafast electron transfer to pheophytin in the active branch prevents exciton back transfer to the inactive branch, thereby achieving unidirectional charge separation.
- This article is part of the themed collection: Editor’s Choice – Graeme Day


 Please wait while we load your content...
Please wait while we load your content...