Effect of olefin and aromatics on thiophene adsorption desulfurization over modified NiY zeolites by metal Pd
Abstract
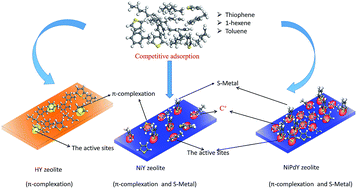
NiY and NiPdY adsorbents were prepared by incipient wetness impregnation and characterized by X-ray diffraction (XRD), N2 adsorption–desorption isotherms, scanning electron microscopy (SEM), and Fourier transform infrared spectroscopy (FT-IR). The effects of olefin and aromatics on desulfurization over NiY and NiPdY adsorbents were studied; moreover, the adsorption mechanisms of different compounds, including thiophene, olefin, and aromatics, were investigated by in situ FT-IR. The results showed that thiophene, olefin, and aromatics were adsorbed on HY through π complexation, which will lead to strong competitive adsorption. Significantly, after modifying HY zeolite by metal Ni, the adsorption desulfurization rate could be improved from 57.0% to 65.7% in the olefin–thiophene system (M-II) and from 21.8% to 73.1% in the aromatic–thiophene system (M-III) because S–metal bonds were formed directly when thiophene once touched the Ni surface. The breakthrough sulfur capacity of NiY was 0.68 mg g−1 in M-II and 0.88 mg g−1 in M-III. However, introduced Ni also increased the number of Brönsted acid sites, which could result in protonated reactions of thiophene and olefin and lead to blockage of pores. The metal Pd was chosen to further modify the acidity and pore structure of NiY. The results showed that not only was the adsorption desulfurization rate improved further from 65.7% to 85.8% in M-II and from 73.1% to 87.0% in M-III, but breakthrough sulfur capacity was also enhanced from 0.68 to 1.64 mg g−1 in M-II from 0.88 to 2.01 mg g−1 in M-III.

 Please wait while we load your content...
Please wait while we load your content...